Journal of Family Medicine and Disease Prevention
Family Resource and Health Perspective of Children with Turner Syndrome: A Model Testing
Jih Yuan Chen1*, Mei Chyn Chao2, Sheng Yi Yang3, Yea Hui Xiao4 and Ying Hui Lin4
1School of Nursing, Kaohsiung Medical University, Taiwan
2Department of Pediatrics, Kaohsiung Medical University, Taiwan
3Department of Psychology, Kaohsiung Medical University, Taiwan
4Department of Nursing, Kaohsiung Medical University Hospital, Taiwan
*Corresponding author: Jih Yuan Chen, Associate Professor, College of Nursing, Kaohsiung Medical University, Kaohsiung, 100 Shih Chuan 1st Rd, Kaohsiung, ROC 80761, Taiwan, E-mail: jiyuch@kmu.edu.tw
J Fam Med Dis Prev, JFMDP-2-030, (Volume 2, Issue 2), Quantitative Research Article; ISSN: 2469-5793
Received: February 10, 2016 | Accepted: April 05, 2016 | Published: April 08, 2016
Citation: Chen JY, Chao MC, Yang SY, Xiao YH, Lin YH (2016) Family Resource and Health Perspective of Children with Turner Syndrome: A Model Testing. J Fam Med Dis Prev 2:030. 10.23937/2469-5793/1510030
Copyright: © 2016 Chen JY, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The objectives of this study were to elucidate family interaction and psychophysical health among families with members who have Turner Syndrome (TS) through structural equation modeling (SEM), and to investigate differences among patients and their fathers and mothers related to family support, functioning, and hardiness, and mental and physical health. Subjects were recruited from a genetic disease section of a pediatric clinic in a southern Taiwan medical center. There were 59 subjects aged 23 to 61 years from 36 families. The age range of 10 TS subjects was 23 to 36. All subjects were given the self-report questionnaires and cross-sectional analyses were performed. Subjects completed the Duke Health Profile (DH), Family Support (FS), Family Hardiness Index (FHI), and Family Assessment Device-General Function (FAD) tools. Parental responses to family scales indicated significantly greater anxiety, depression, and anxiety-depression, but better family functioning than among children with TS. However, children with TS experienced a lower level of general health, mental health, family hardiness, commitment, and control than mothers or fathers. SEM tests revealed model fitness regarding family support and family hardiness that affected Duke Health scores; the marital variable was found to affect family support and family functioning (χ2 = 29.03, df = 39, P = 0.88, SRMR = 0.05, RMSEA = 0.00). Family support functioned as a mediator in the relationship between family hardiness and family health and functioning. Family support is an important factor in explaining family health and function in families of children with TS. The results indicate families' needs to promote mutual communication and supportive interactions to address psychological distress and maintain family health and function.
Keywords
Family functioning, Duke Health Profile, Family support, Turner Syndrome
Turner Syndrome (TS) is a partial or complete absence of one X chromosome and as the most common sex-chromosome abnormality, affecting 1/2000-2,500 live- female births per year globally [1,2]. Females with TS (45, X karyotype) are characterized by short stature with growth velocity less than the 10th percentile for age and sex hormone deficiency leading to pubertal delay and infertility [3]. They may also display typical physical features, such as low hairline, low-set ears, a small mandible, a webbed neck [4,5], bicuspid aortic valve (30%) and aortic coarctation (12%), and partial anomalous pulmonary connection [6,7]; and tend to have higher BMIs and waist-to-hip ratios, a lower degree of physical activity, lower pulse rate, and lower density lipoprotein cholesterol levels, and higher levels of triglycerides. Other common features include edema of the hands or feet, liver abnormality, frequent otitis media, and hypertension [4,8,9]. Moreover, these girls exhibit a high incidence of lymphedema, osteoporosis and fractures, structural malformations of the kidney, autoimmune thyroiditis, diabetes mellitus, and inflammatory bowel disease [4,10-12]. Girls with TS also have higher risks for scoliosis and kyphosis than the general population [13], with 33% of girls with TS developing lordosis, 28% developing kyphosis [14]. In addition, 30% or more eventually develop hypothyroidism [15].
Davenport (2010) [16] recommended health screening tests at specific ages for TS problems-related to strabismus, hearing loss, and autoimmune disease; and proposed educating parents to be able to address concerns regarding growth, cardiovascular, and gonadal deficiencies, and learning disabilities. Parents and practitioners will need to consider whether to initiate growth hormone therapy if growth failure occurs. In prior studies, young girls with TS who did not received growth hormone treatment that had a huge impact on possible outcomes (e.g., normal height, breast development, and health-related quality of life) [17], and did not satisfy with height and breast development was a negative influence on health related quality of life scales [18], final height was seven cm greater in those who had growth hormone treatment than those who did not, but skeletal maturity was not accelerated [19]. Impact of growth hormone supplementation on adult height in turner syndrome: results of the Canadian randomized controlled trial. Baxter et al. (2007) [18] simply reported these findings from this study for their cochranes review article. As well as higher self-esteem, lower anxiety, and better social and emotional function had than those who did not [16,17,20].
Parents may keep secrets about health-related conditions among family members and may have difficultly communicating with their daughters regarding the infertility associated with TS. Other debilitating conditions, such as learning disability, attention deficit disorder, or severe social isolation are also in children with TS, all of which can create additional stress for parents. The many abnormalities associated with TS can interfere with parent-child bonding, make parents feel deeply uncomfortable, and lead to parent-child conflict [8]. Sutton et al. (2006) [2] indicated that parents of TS daughters may experience overload, role changes, limitation of social activity, and economic and emotional stress as the result of problems arising from caring for them. Parental distress may diminish family support, decrease family hardiness, and damage health, family function.
Hardiness has been shown to influence the ability adapt effectively to the demands of care to maintain family function coping in families of children with TS [21,22]. For example, previous work has indicated that family hardiness and family support for caregivers of children with ADHD may have a positive effect on family members' health and family function [23]. Similarly, Chen (2008) [24] found that family hardiness positively mediated the association between age at diagnosis and family function in families of children with Duchene muscular dystrophy (DMD). Family hardiness and family support indirectly influenced family general function as a mediator by means of its association with marital relationship [23]. These studies, however, did not address which components of family hardiness and family support or other resources most predict family health or family function in the face of disabling illness in a child (Figure 1).
The main aim of the current study was to investigate how factors related to family resources including family hardiness, family support, family income, and marital status influenced general health and function in families of girls with TS. Differential influences of these factors were examined for the families with affected children. Finally, structural equation modeling (SEM) was used to determine interactions between family resource variables and psychophysical health and family function.
Methods
Sample
The Kaohsiung Medical University Hospital Institutional Review Board approved the study. Potential subjects were informed of the study by the physician in the genetic diseases section of a pediatric clinic in a southern Taiwan medical center. Selection criteria included families of girls diagnosed with karyotype-proven TS. Follow-up interviews or telephone calls who ascertain if the families is still interested to solicit participation. Interested parents and TS patients who were over 20 years were gave or sent packets including a description of the study, informed consent form, and the measurement tools. Written informed consent was obtained from all subjects and returned at the time of interview in the clinic or by mail in an envelope separate from that used for the completed measures. The data derived from 59 subjects (aged 23 to 61 years, mean age: 42.24 years, SD: 8.9 years) in 36 of families of children with TS (aged 1.5 to 36 years, n = 24 aged ≤ 20 years, mean age: 16.5 years, SD: 8.8 years), included mothers (N = 30, aged 33 to 57 years, mean age: 43.4 years, SD: 6.67 years), fathers (N = 19, aged 33 to 61 years, mean age: 47.0 years, SD: 7.23 years), and patients (n = 10, aged 23 - 36 years, mean age: 29.7 years, SD: 6.1 years).
Data Collection
Data were collected related to subjective family resource variables (hardiness, support, and family characteristics) and subjective outcome variables (family health and function). We will identify which family resource variables directly and indirectly influence family general health and family general function. All measurement tools were translated from English and previously validated in other research including back-translated in the standard manner to check meaning in Taiwan.
Indicators of subjective family resources
Family resources refer to the family's capabilities and strengths to resist crises and achieve harmony and balance [23]. The components of family resources in this study included family hardiness, family support, and family characteristics, such as marital status, monthly income, employment, and location of residence, all of which may create energy within the family to overcome adversity and conflict [6].
Family hardiness: Family hardiness reflects the energy resources that promote family adjustment and adaptation by family members working together to minimize the negative effects of demands and stressors [25]. Family hardiness was measured using the Family Hardiness Index (FHI), a 20-items scale that addresses components of commitment-8 items, challenge-six items, and control-6 items [26]. The response alternatives were scored on a scale from 0 to 3 (0 = false, 1= mostly false, 2 = mostly true, 3 = true) yielding a total scores range of 0-60. Higher scores indicated greater levels of perceived hardiness in the family. The Cronbach's alpha for this study was .88 and the internal consistency reliability results for the subscales were 0.82, 0.71, and 0.68 respectively.
Family support: Family support was defined as a family member's satisfaction with the family's responsiveness to and caring about his or her personal needs. This study employed the Family Caring Index (Family APGAR) to measure perceived family support along five components: adaptation, partnership, growth, affection, and resolve [10]. This was measured by the Family APGAR (FAPGAR) [27] which included 5 items using a 3-point rating scale ( 0 = hardly ever, 1 = some of time, or 2 = almost always) with a summative score ranging from 0 to 10, with higher scores indicating higher levels of perceived family support. The FAPGAR demonstrated excellent reliability in our sample (Cronbach's alpha: 0.88).
Family characteristics: Socio-demographic information about age (coded subject's age < 30 years = 1, 31-50 years = 2, > = 51 years = 3; coded participant patient child's age < 12 years = 4, 12-18 years = 3, 19-24 years = 2, 25-30 years = 1, > 31 years = 0), gender (coded male =1, female = 0), nationality (coded Taiwanese = 0, non-Taiwanese = 1), marital status (coded married = 1, non-married = 0), education (coded graduate = 5, bachelor's = 4, high school = 3, and junior high school = 2, below junior high school = 1), occupation (coded professional = 2, technical/staff = 1, labor/household/ none = 0), location of residence (coded rural = 0, urban = 1, city/town = 2), family income (coded > NT$ 50,000 = 2, NT$ 30,000~50,000 = 1, < NT$ 30,000 = 0), family structure (coded nuclear = 1 or non-nuclear = 0) and development (with preschool child/school child coded = 0, with adolescent child coded = 1, and with adult child coded = 2), spent TV pleasure time (coded every day less than 3 hours = 2, 4-7 hours = 1, and more than 8 hours = 0), and family power structure including decision maker (coded father = 3, mother = 2, parent together = 1, other else = 0), submit idea for problem-solving (coded father = 3, mother = 2, parent together = 1, other else = 0), and power to propose idea or plan (coded directly propose = 2, indirectly propose = 1, silence/other else = 0) was obtained from registration records and verified during interviews or self-reported.
Indicators of subjective outcome variables
Family health: Family health comprises the well-being of family members, including physical, psychological, social, and emotional health. These elements are promoted by essential protective and recovery factors, such as maintaining a sense of security, receiving essential information about TS, and so on. Family health was measured using the Duke Health Profile (DH)-17- item scale [28] with three-simple response options worded differently for various subscales. For example, some item responses were 2 = "yes, describing me exactly" 1 = "describe me somewhat" and 0 = "no, does not describe me at all". Others were rated in terms of the extent of difficulty experienced in an area (2 = none, 1 = some, 0 = a lot), or frequency of experiences during a 1-week period (2 = a lot, 1 = some, 0 = none, or others 2 = none, 1 = 1- 4 days, 0 = 5 - 7 days). Multi-item mean scores were transformed from the response scale of 0, 1, or 2 to 0, 50, or 100. The tool addresses five positive health subscales (physical, mental, and social health, and self-esteem (five items each); and perceived health (one item). Higher scores on these subscales indicate better perceived health. The instrument also includes five negative health subscales (pain and disability (one item each); anxiety (six items), depression (five items), and anxiety-depression (seven items). Some items on these subscales were reverse scored and contributed to several subscales. Higher scores corresponded to lower perceptions of health. Reliability of the overall Duke scale was good (Cronbach's alpha: 0.85), and the internal consistency reliability results for the subscales ranged from 0.64 to 0.83.
Family function: Family function refers to the outcome of a family's ability to utilize family resources or other sources of support, high levels of family health and family function have been shown to provide resilience that produces family energy for overcoming conflict [25]. Family function was measured using the Family Assessment Device of General Function (FAD-GF) [29,30], which comprises 12 items with 4 response options - from "strongly agree" to "agree" to "disagree" to "strongly disagree". Half of the items are reversed scored, and total possible responses ranging from 1 to 4 (total scores were divided by numbers of items). Higher scores indicate the family member's perception of greater difficulty overall family general functioning (A score of 2.00 or above indicates problematic family functioning) [29]. The internal consistency reliability result was 0.88.
Statistical Analysis
Data analysis was performed using descriptive correlations, means (SD, 95% CI), frequencies, and regression using SPSS 19.0. Structural equation modeling (SEM) was conducted to test a fixed model among the relationships of observed and unobserved variables with AMOS 19.0 [31]. The measurement model constructs the relations between latent (endogenous) variables (family hardiness and family support) that cannot be observed directly, and measured (exogenous) variables that can be observed directly and are measurable. Figure 1 shows measurement model A that is constructed from individual perceptions of the variables of adaptation, partnership, growth, affection, and resolve; measurement model B is constructed from variables related to control, challenge, and commitment; measurement model C is constructed from family support and family hardiness to general family function; measurement model D is constructed from family support and family hardiness to general health; measurement model E is constructed from family member's marital status or monthly income to family support and family hardiness; measurement model F is constructed from family member's marital status or monthly income to general family function and general health; measurement model G is constructed from family member's marital status or monthly income to family support and family hardiness, and then, effected on general health or family general function [32].
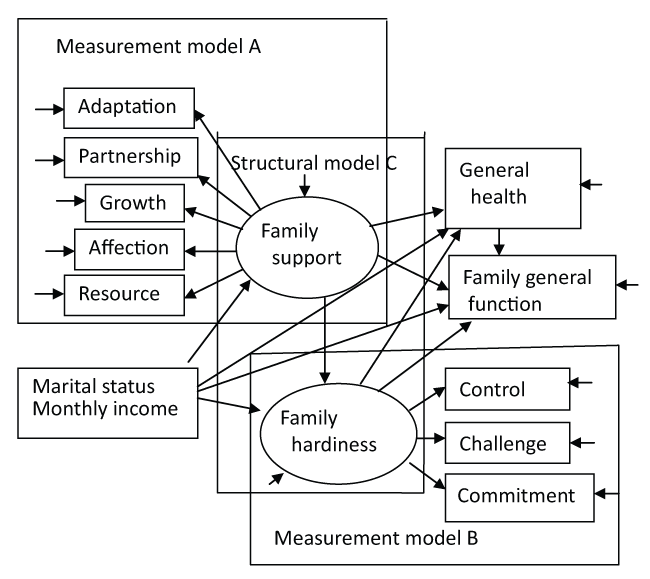
.
Figure 1: An expanded model of the regressive dependencies between latent variables (family support and family hardiness) illustrating the distinction between the structural model and the measurement models.
View Figure 1
In AMOS, multivariate outliers were determined using the Mahalanobis distance statistic (Mahalanobis d2) that presented the squared distance from centroid. The critical ratio of multivariate kurtosis of 2.24 and the coefficient of multivariate kurtosis of 9.84, the absolute values of univariate kurtosis and skewness for all variables are small than 1 (exhibit significant skewness and/or kurtosis using a benchmark of ± 2.0) that reaches a multivariate normal distribution [33,34]. Mahalanobis d2 presented the squared distance from the centroid with no value of p2 smaller than 0.05, d2/59 = 25.61 (CMIN = 30.09, P = 0.82).
Results
Less than 32% of the 59 parents were unemployed (Table 1). There were 59 subjects in the study, including 49 parents (30 mothers and 19 fathers) and 10 adult TS patients. Subjects were mainly female (67.8% female, 32.2% male). The majority of the subjects were Taiwanese (78 %), married (76 %), level of education had an associate's degree or higher (61%), while 28.8% completed high school; and 42.4% reported monthly income that was less than NT$ 60,000 (Table 1). The return rate for completed questionnaires was 69.5%.
Table 1 shows family characteristics of the sample. Most families (83%) reported living in a city and exhibited a nuclear family structure (80%). More than half (64%) of family development stage were school or adolescent. Less than half of the families reported their primary decision maker (41%) and problem-solver (39%) in the families were fathers, 88% of subjects directly power to proposed ideas within their families, and 61% had an associate's degree or higher education.
![]()
Table 1: Characteristics of the subjects and families (N = 59).
View Table 1
Cronbach's alphas for the four major measurements in the study were reliable as shown in Table 2. Mean self-reported the Duke Health Profile, the FAPGAR, the FAD-General Function, and the Family Hardiness scores, and their subscales as reported by male parents, female parents, and patients with TS are presented in Table 2. One-way ANOVA results comparing differences among three groups of subjects including male parents, female parents, and patients. Table 2 indicated that the responses of patients with TS to health scales perceived significantly lower levels of anxiety, depression, and anxiety-depression than their male or female parents. Conversely, patients with TS experienced lower levels of perceived general health, mental health, family hardiness, commitment, and control than their parents (female or male). In addition, patients with TS exhibited significantly greater family general function scores than either group of parents.
![]()
Table 2: Descriptive Data for Predictor and Outcome Variables, and compare with differences among Three Groups of the Subjects (N = 59).
View Table 2
Based on Pearson's correlations among patients' characteristics variables and dependent variables, Duke Health scores were significant and positively correlated with family support, FAD general function, and family hardiness (including all three components of commitment, challenge, and control) (Table 3). Family support score was significantly and positively correlated with FAD general function and family hardiness, including all three components. FHI scores were significantly and positively correlated at 0.01 with FAD general function scores (with lower scores indicating better function). All of the above dependent variables' scores were significantly positively correlated with marital status, and monthly income. Participant's age, education, employment, and location of resilience were not significantly correlated with any of the above dependent variables.
![]()
Table 3: The Correlation Cross Table of DH, FAD, FS, Marital Status, and Their Subscales (N = 59).
View Table 3
Regression permitted us to establish prediction equations in which independent variables were based on dependent variables using simple linear regression. Table 4 shows that family hardiness and family support were predictive factors for family general function (r = 0.80, r-squared change = 0.06). Predictive factors for general health were monthly income and family hardiness (r = 0.62, r-squared change = 0.15). General health and family general function were predictive factors for family hardiness (r = 0.76, r-squared change = 0.06) and family general function was predictor for family support (r = 0.75, r-squared change = 0.17).
![]()
Table 4: Predictors of Family General Function, Duke Health Profile, Family Hardiness and Family Support in Families of Children with Chromosomal Abnormality (N = 59).
View Table 4
As shown unstandardized and standardized estimates in Figure 2, the SEM test revealed (Arbuckle, 2008) that family support and family hardiness significantly directly affected general health (r = 0.20) and family general function (r = -0.46); while family support indirectly affected family general function through general health or family hardiness. In addition, family hardiness indirectly affected family general function through general health, or family support and general health. Marital status was found to directly affect family support (r = -0.35), family hardiness (r = -0.22), or general health (r = -0.16), while indirectly affecting family hardiness through family support and indirectly affecting general health through family hardiness or family support. The results indicate that a good fit model was obtained (χ2 = 30.09, df = 38, P = 0.82, SRMR = 0.05, GFI = 0.92, NFI = 0.92, RFI = 0.88, CFI = 1.0, RMSEA = 0.00).
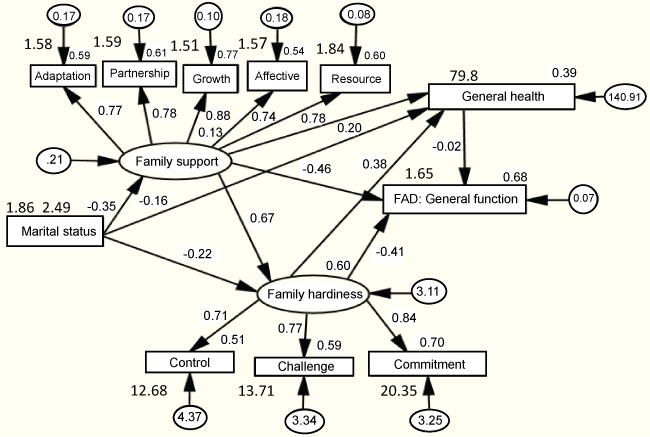
.
Figure 2: Predictors on FAD general function from family characteristics by family hardiness and family support (Chi-square = 30.09, df = 38, NFI = 0.92, RFI = 0.88, CFI = 1.0, RMSEA = 0.00), standardized estimates & unstandardized estimates.
View Figure 2
Discussion and Conclusions
The objectives of this study were to elucidate family interactions among families whose members have Turner syndrome. The researchers tested families' general health, family support, family hardiness, and family general function regarding to the child with TS. The results reinforce all of the measurements exhibited high internal reliability and constructive validity. Comparisons were made to determine whether differences existed in these experiences among three groups of subjects. The lack of significant differences in scores on family general function, family hardiness, family support, and general health between male and female parents is consistent with previous research indicating no parental gender differences [35,36]. The results reinforce the need to address the tendency toward parental anxiety, depression, and anxiety-depression among both fathers and mothers of girls with TS that consists with [37] confirming of more parental anxiety and depression and higher stress in parents of children with ADHD. The findings support the assertion that parental stress is associated with more family conflict, greater distress, and less social support [38,39]. Professionals who with these families can provide information about TS and organizational support programs that assist families to care for themselves and to promote their general health and to increase supportive family interactions.
The patients with TS self-reported higher scores related to family general function and lower scores for family hardiness and family support than parents. The results might suggest health care professionals to provide TS girls' to recognize their condition's managements and health promotion as well as to support them to adapt their family general function. All of the patients with TS were adults who would enter into society and interact with others. Having relatively poor mental health may make such interaction difficult, and health care professionals can assist these patients to develop stronger self-images and improve their mental health.
Plant and Sanders (2007) [40] reported that parents' examination of care-giving responsibilities had a mediating effect on the relationship between the child's disability and parental stress. Chen and Clark (2007, 2010) [6,41] found that earlier detected the disease influenced the parents' health of children with DMD. However, in this study, we did not explore the patients with TS conditions and diagnosis at age as factors to affect the outcome indicators. In the future, socio-demographic characteristics by themselves did emerge as independent variables of particular relevance to families outcomes.
Preliminary analysis indicated that marital status and monthly income of subjects were significantly correlated with family support, family hardiness, family general function, and general health. Monthly income was shown to influence family general function and general health. Health professionals can assist families in this area by promoting access to and suggesting use of economical supplies. No other subjects' characteristics were associated with the dependent variables (outcome indicators). These findings were dissimilar to Davenport's (2006) [42], who reported that females perceived family functioning to be better than did males and age to be linearly related to perceived family dysfunction. However, in this study, only marital status influenced subjects' perceptions of family support, family hardiness, family general function, and general health. Marital status also functioned as a mediator in relationships between family support and general health, family support and family hardiness, and general health and family general function. All subjects with TS were young adults and not married that may have some regretful feelings in the family to influence atmosphere. Health professionals can provide attachment relationship training programs by doing natural health or family health promotion that families can involve in the situation and get coherence.
The large effect for subjects' perceptions of family hardiness in the prediction of general health and family general function was consistent with studies showing that family support functioned as a mediator in the relationship between family hardiness and family health, and between family hardiness and family general function. Moreover, the relationship may suggestion that health professionals design family support programs to strengthen family hardiness for families. Family members support through health promotion, psychological and spiritual care, financial support and convenient community services can lead to better parental health and contribute to overall family health and adaptation [41]. This model is recommended for nursing interventions in order to help parents better understand their children's needs and the impact of transitions, to decrease their psychological distress, and to increase mutual communication and supportive interactions.
The current study used a cross-sectional research design; therefore, it was impossible to confirm inferences about causations. Especially, a key limitation of the sample size was that the increased factor relationship was associated with 78 subjects in the minimum acceptable sample size for the CFA (Wolf, Harrington, Clark, & Miller, 2013) [43]. However, Sideridis, et al. (2014) [44] indicated that sample sizes of 50 subjects suffice to accurately estimate measurement and structural paths associated with minimal bias when relying on the IFI, CFI, and NNFI.
Contribution
This study contributes to the growing body of literature suggesting that family support and family hardiness influence family general function and general health when families are faced with disabling diseases in their families. This and previous studies reinforce the need for nursing interventions to promote family hardiness and support can help maintain parental health and promote family adaptation in the face of a child with TS addressing family interaction issues. The findings of this study clearly indicate that nursing interventions as described could/would provide key support for family hardiness and family support.
Acknowledgement
We would like to thank of Dr. Yong-Yuan Chang, children with Tuner Syndrome and their parents for their contributions to the completion of this research. Worked was funded by grant KMUH-N10005, KMU2014024.
Ethical Statement
The Kaohsiung Medical University Hospital Institutional Review Board approved the study (KMUH-IRB-20140058).
References
-
Stochholm K, Juul S, Juel K, Naeraa RW, Gravholt CH (2006) Prevalence, incidence, diagnostic delay, and mortality in Turner syndrome. J Clin Endocrinol Metab 91: 3897-3902.
-
Sutton EJ, Young J, McInerney-Leo A, Bondy CA, Gollust SE, et al. (2006) Truth-telling and Turner Syndrome: the importance of diagnostic disclosure. J Pediatr 148: 102-107.
-
Morgan T (2007) Turner syndrome: diagnosis and management. Am Fam Physician 76: 405-410.
-
Bondy CA, Turner Syndrome Study Group (2007) Care of girls and women with Turner syndrome: a guideline of the Turner Syndrome Study Group. J Clin Endocrinol Metab 92: 10-25.
-
Gonzalez L, Witchel SF (2012) The patient with Turner syndrome: puberty and medical management concerns. Fertil Steril 98: 780-786.
-
Chen JY, Clark MJ (2007) Family function in families of children with Duchenne muscular dystrophy. Fam Community Health 30: 296-304.
-
Hong DS, Dunkin B, Reiss AL (2011) Psychosocial functioning and social cognitive processing in girls with Turner syndrome. J Dev Behav Pediatr 32: 512-520.
-
Amundson E, Boman UW, Barrenäs ML, Bryman I, Landin-Wilhelmsen K (2010) Impact of growth hormone therapy on quality of life in adults with turner syndrome. J Clin Endocrinol Metab 95: 1355-1359.
-
McCarthy K, Bondy CA (2008) Turner syndrome in childhood and adolescence. Expert Rev Endocrinol Metab 3: 771-775.
-
Goldacre MJ, Seminog OO (2014) Turner syndrome and autoimmune diseases: record-linkage study. Arch Dis Child 99: 71-73.
-
JørgensenKT, Rostgaard K, Bache I, Biggar RJ, Nielsen NM, et al. (2010) Autoimmune diseases in women with Turner's syndrome. Arthritis Rheum 62: 658-666.
-
Sybert VP, McCauley E (2004) Turner's syndrome. N Engl J Med 351: 1227-1238.
-
Kim JY, Rosenfeld SR, Keyak JH (2001) Increased prevalence of scoliosis in Turner syndrome. J Pediatr Orthop 21: 765-766.
-
Olszewska E, Wisniewski A, Madej M, Trzcinska D, Tabor P, et al. (2010) [Posture in Turner syndrome patients]. Pediatr Endocrinol Diabetes Metab 16: 189-195.
-
Gravholt CH (2004) Epidemiological, endocrine and metabolic features in Turner syndrome. Eur J Endocrinol 151: 657-687.
-
Davenport ML (2010) Approach to the patient with Turner syndrome. J Clin Endocrinol Metab 95: 1487-1495.
-
Bannink EM, Raat H, Mulder PG, de Muinck Keizer-Schrama SM (2006) Quality of life after growth hormone therapy and induced puberty in women with Turner syndrome. J Pediatr 148: 95-101.
-
Baxter L, Bryant J, Cave CB, Milne R (2007) Recombinant growth hormone for children and adolescents with Turner syndrome. Cochrane Database Syst Rev CD003887
-
Stephure DK, Canadian Growth Hormone Advisory Committee (2005) Impact of growth hormone supplementation on adult height in turner syndrome: results of the Canadian randomized controlled trial. J Clin Endocrinol Metab 90: 3360-3366.
-
Kiliç BG, Ergür AT, Ocal G (2005) Depression, levels of anxiety and self-concept in girls with Turner's syndrome. J Pediatr Endocrinol Metab 18: 1111-1117.
-
Astedt-Kurki P, Tarkka MT, Rikala MR, Lehti K, Paavilainen E (2009) Further testing of a family nursing instrument (FAFHES). Int J Nurs Stud 46: 350-359.
-
Svavarsdottir EK, Rayens MK (2005) Hardiness in families of young children with asthma. J AdvNurs 50: 381-390.
-
Chen JY, Clark MJ, Chang YY, Liu YY, Chang CY (2014) Factors affecting perceptions of family function in caregivers of children with attention deficit hyperactivity disorders. J Nurs Res 22: 165-175.
-
Chen JY (2008) Mediators affecting family function in families of children with Duchenne muscular dystrophy. Kaohsiung J Med Sci 24: 514-522.
-
Chen JY, Instone SL (2008) Family resilience and functioning in child with DMD: Functioning and resilience in families with children with Duchenne muscular dystrophy Saarbrucken, Germany: VDM Verlag Dr. Muller Aktiengesellschaft.
-
McCubbin HI, Thompson AI, McCubbin MA (2001) Family measures: Stress, coping, and resiliency --Inventories for research and practice. Honolulu, Hawaii: Kamehameha Schools.
-
Smilkstein G, Ashworth C, Montano D (1982) Validity and reliability of the family APGAR as a test of family function. J Fam Pract 15: 303-311.
-
Parkerson GRJ (2002) User's guide for Duke Health Measures. Durham, NC: Department of Community and Family Medicine, Duke University Medical Center.
-
Epstein NB, Baldwin LM, Bishop DS (1983) The McMaster Family Assessment Device. Journal of Marital and Family Therapy 9: 171-180.
-
Van Pareren YK, Duivenvoorden HJ, Slijper FM, Koot HM, Drop SL, et al. (2005) Psychosocial functioning after discontinuation of long-term growth hormone treatment in girls with Turner syndrome. Horm Res 63: 238-244.
-
Byrne BM (2010) Structural Equation Modeling with AMOS: Basic concepts, applications, and programming.
-
Arbuckle JL (2008) AMOS 17.0 User’s guide.
-
Gao S, MokhtarianPL, Johnston RA (2008) Nonnormality of data in structural equation models. Transportation Research Record: Journal of the Transportation Research Board 2082: 116-124.
-
Walker DA (2010) A confirmatory factor analysis of the attitudes toward research scale. Multiple Linear Regression Viewpoints 36: 18-27.
-
Dyson LL (1997) Fathers and mothers of school-age children with developmental disabilities: parental stress, family functioning, and social support. Am J Ment Retard 102: 267-279.
-
Hagborg WJ (1989) A comparative study of parental stress among mothers and fathers of deaf school-age children. Journal of Community Psychology 17: 220-224.
-
Cussen A, Sciberras E, Ukoumunne OC, Efron D (2012) Relationship between symptoms of attention-deficit/hyperactivity disorder and family functioning: A community-based study. Eur J Pediatr 171: 271-280.
-
Kazak AE, Marvin RS (1984) Differences, difficulties and adaptation: Stress and social networks in families with a handicapped child. Family Relations 33: 67-77.
-
Reiter-Purtill J, Schorry EK, Lovell AM, Vannatta K, Gerhardt CA, et al. (2008) Parental distress, family functioning, and social support in families with and without a child with neurofibromatosis 1. J Pediatr Psychol 33: 422-434.
-
Plant KM, Sanders MR (2007) Predictors of care-giver stress in families of preschool-aged children with developmental disabilities. J Intellect Disabil Res 51: 109-124.
-
Chen JY, Clark MJ (2010) Family resources and parental health in families of children with Duchenne muscular dystrophy. J Nurs Res 18: 239-248.
-
Davenport ML (2006) Evidence for early initiation of growth hormone and transdermal estradiol therapies in girls with Turner syndrome. Growth Horm IGF Res 16 Suppl A: S91-97.
-
Wolf EJ, Harrington KM, Clark SL, Miller MW (2013) Sample Size Requirements for Structural Equation Models: An Evaluation of Power, Bias, and Solution Propriety. Educ Psychol Meas 76: 913-934.
-
Sideridis G, Simos P, Papanicolaou A, Fletcher J (2014) Using structural equation modeling to assess functional connectivity in the brain: Power and sample size considerations. Educ Psychol Meas 74: 733-758.





