Journal of Genetics and Genome Research
Transplantation of hBMSCs Loaded on Collagen Scaffolds for the Regeneration of Canine Tissue-Engineered Urethras
Jianjian Wu1#, Weisheng Jia1#, Wei Chen1, Feng Zhou1, Zhenqiang Fang1, Xianglin Hou2,3, Bing Chen2,3, Gang Ye1*, and Jianwu Dai2,3*
1Department of Urology, Xinqiao Hospital, Third Military Medical University, China
2Institute of Combined Injury, State Key Laboratory of Trauma, Burns and Combined Injury, Chongqing Engineering Research Center for Nanomedicine, College of Preventive Medicine, Third Military Medical University, China
3Key Laboratory of Molecular Developmental Biology, Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, China
#First authors
*Corresponding author:
Gang Ye, Department of Urology, Xinqiao Hospital, Third Military Medical University, Chongqing 400037, China, Tel/Fax: +86-023-68755683, E-mail: gangyeah@126.com
Jianwu Dai, Institute of Combined Injury, State Key Laboratory of Trauma, Burns and Combined Injury, Chongqing Engineering Research Center for Nanomedicine, College of Preventive Medicine, Third Military Medical University, 30 Gaotanyan Street, Shapingba District, Chongqing 400038, China; Key Laboratory of Molecular Developmental Biology, Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, 3 Nanyitiao, Zhongguancun, Beijing 100190, China, Tel/Fax: +86-010-82614426, E-mail: jwdai@genetics.ac.cn
J Genet Genome Res, JGGR-2-015, (Volume 2, Issue 1), Research Article; ISSN: 2378-3648
Received: April 23, 2015 | Accepted: June 19, 2015 | Published: June 21, 2015
Citation: Wu J, Jia W, Chen W, Zhou F, Fang Z, et al. (2015) Transplantation of hBMSCs Loaded on Collagen Scaffolds for the Regeneration of Canine Tissue-Engineered Urethras. J Genet Genome Res 2:015. 10.23937/2378-3648/1410015
Copyright: © 2015 Wu J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To estimate the function of human bone marrow derived mesenchymal stem cells (hBMSCs) loaded on collagen scaffolds in the regeneration of canine tissue-engineered urethras.
Methods: In this study, hBMSCs (1×106 hBMSCs per 1cm2 collagen scaffold) were loaded on tubular collagen scaffolds (5cm in length). A 5cm long urethra segment near the penis bone was excised surgically. A collagen scaffold seeded with hBMSCs was sutured to two ends of the host urethra for the urethral repair in the experimental group, whereas the unseeded scaffold was used in the control group. Retrograde urethrography was performed before the operation and at 6 months after the surgery. At 6 months post-surgery, the animals were sacrificed, and the reconstructed urethras were collected for histological observation. An anti-human nuclei monoclonal antibody was used for immunofluorescent staining to detect the human cells in the reconstructed urethras. The growth of the urothelium, smooth muscles and blood vessels were evaluated by immunofluorescent staining.
Results: The macroscopic observation and retrograde urethrography demonstrated that varying degree of urethra strictures were observed in all the animals of two groups. But the severer urethrostenosis usually appeared in the control group. Histologically, the scaffolds seeded with the hBMSCs formed a stratified urothelium layer that was similar to the appearance of the normal urothelium; however, in most areas, the unseeded scaffolds formed a discontinuous mucosa with a monolayer of urothelial cells. There were more smooth muscles and blood vessels growing than those in controls, although they were far fewer than normal. Positive cells were found in the experimental group with anti-human nuclei immunofluorescence staining.
Conclusion: In this study, hBMSCs loaded on collagen scaffolds were able to promote the regeneration of canine tissue-engineered urethras, predominantly for the urothelium.
Keywords
hBMSCs, Collagen, Urethral repair, Tissue engineering, Beagles
Introduction
Urethral injury is a common urological disease all over the world. The traditional method of treatment is the surgical reconstruction by using autologous tissues such as penile skin [1], buccal mucosa [2], lingual mucosa [3] and colonic mucosa [4]. Nonetheless, such application is restricted by various complications including strictures and urethral fistula [5], and the limited supply of autologous tissues [6]. In this case, another therapeutic method is needed.
The development of tissue-engineering technology has recently presented a novel method of repairing urethral injuries. A key factor of this technology is the use of seeding cells, which are required especially for a tubularized urethral replacement. The significance of seeding cells for urethral tissue engineering has attracted more attention gradually. Numerous studies have showed that the tubular scaffold seeded with cells can successfully repair the urethral defects [7,8]. Among varieties of cells, stem cells are considered as an appropriate resource of seeding cells. It is well known that they play an important role in tissue regeneration with the ability to self-renew [9]. Compared to other stem cells, BMSCs are not only less immunogenic but also less tumorigenic, and have fewer ethical problems [10,11]. Autologous BMSCs have been reported in some experimental researches for urethral reconstruction with small animals [12,13]. However, there is hardly any study about using hBMSCs for urethral repair in larger animals up to now. In the present study, we selected hBMSCs as seeding cells for urethral repair in a canine model to estimate their function.
Materials and Methods
Experimental animals
Adult male beagles (10.5-13.5kg, n=10) used in this study were purchased from the Experimental Animal Center of Xinqiao Hospital, Third Military Medical University, Chongqing, China. They were randomly divided into the experimental group (hBMSCs-collagen scaffolds) and control group (collagen scaffolds) of 5 subjects each.
Preparation of the collagen scaffolds and hBMSCs
The collagen scaffolds (collagen type I) [14] and hBMSCs were obtained from the Key Laboratory of Molecular Developmental Biology (Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, Beijing, PR China). The 5.0cm×3.0cm×0.2cm collagen membranes were sterilized by 12k Gy Co60 irradiation. The process of dealing with hBMSCs was approved by the Human Experimentation and Ethics Committee. Cell culture was done as previously described [15]. The hBMSCs were collected, counted, and analyzed by flow cytometry for phycoerythrin anti-human phycoerythrin-CD29, CD105, CD106, CD31, CD34, CD44, and HLA-DR expression (Becton Dickinson, Franklin Lakes, NJ) as previously described [16]. We prepared hBMSCs into cell suspension prior to injection.
Surgical procedure and postoperative management
All experiments were conducted with the approval of the Animal Experimentation Ethics Committee of the Third Military Medical University.
Firstly, beagles were anesthetized with pentobarbital sodium (30mg/kg). The urethra, which was near the penis bone, was exposed through a longitudinal perineal incision. A 5cm long urethra section was resected completely and used as a normal urethra for the histological analysis. The prepared collagen scaffold was tubulated by continuous suturing with 3-0 absorbable sutures, according to the diameter of the host urethra, and the tubular scaffold was impregnated with hBMSCs (1×106 hBMSCs per 1cm2 collagen scaffold). Subsequently, the cell-seeded scaffold was attached to the two ends of the host urethra with 3-0 absorbable sutures. During the surgery, a 12F catheter was retained to support the urethra and drain urine from the bladder. The skin and subcutaneous tissues were closed layer by layer. In the control group, the scaffold was injected with the culture medium; the other steps were identical to those previously described. Penicillin G sodium (5×104U/kg/day i.m.) was administered for 7 days postoperatively. The urethral catheter was removed 2 weeks post surgery.
Retrograde Urethrography
Retrograde urethrography was performed before the operation and at 6 months after the surgery. The beagles were anesthetized with pentobarbital sodium (30mg/kg), and placed on the examining table in the oblique position. A Foley catheter (8F) was inserted into the urethra, until the balloon was well within urethral orifice, and the balloon was inflated with 2ml sterilized distilled water. Contrast solution (Ioversol Injection, Jiangsu Hengrui Medicine Co., Ltd.) was gently injected via the catheter while the films were obtained.
Macroscopic observation and histological evaluation
The animals were sacrificed at 6 months postoperative. The urethras were removed and anatomized longitudinally for the macroscopic observation. One-half of the urethral tissues were fixed with 10% neutral buffered formalin for 24 hours. After being embedded in paraffin, the tissues were sectioned routinely into 2.5μm thick slices for HE-staining to observe the structure of the reconstructed urethras.
Immunofluorescence analysis
The remaining urethral tissues were immediately frozen in 5-μm thick sections. First, the sections were fixed with 4% paraformaldehyde at room temperature for 10 minutes. After washing with phosphate buffer saline (PBS), 3% hydrogen peroxide and 0.3% Triton X-100 (Sigma, America) were applied for 10 minutes and 20 minutes respectively. Subsequently, 10% goat serum was used to block the non-specific antigen for 20 minutes. Mouse monoclonal anti-human nuclei (Abcam, Britain) were added to the sections at a dilution ratio of 1:100. After incubation overnight at 4°C, Alexa Fluor® 488 goat anti-mouse IgG antibody (Abcam, Britain) was added to the sections for 30 minutes. The nuclei were counterstained with DAPI for 10 minutes.
The method as mentioned above was performed to detect urothelial cells by using mouse monoclonal [AE1/AE3+5D3] to pan cytokeratin (Abcam, Britain) at a dilution ratio of 1:100; smooth muscle cells, using mouse monoclonal [4A4] to alpha smooth muscle actin (Abcam, Britain) with at a dilution ratio of 1:200; and blood vessels, using rabbit polyclonal to von willebrand factor (Abcam, Britain) at a dilution ratio of 1:400. The sections stained by immunofluorescence were examined with a laser scanning confocal microscope (Leica TCS SP5, Germany) and quantitatively analyzed by Image-Pro Plus (IPP) software.
Statistical analysis
Statistical analysis was performed with Statistical Package for Social Science (SPSS) software. The mean fluorescence density of all subjects from each group was analyzed by the t-test for two independent samples. The data were represented as mean ± standard deviation (SD); P<0.05 was considered significantly different, and P>0.05 was considered to be not different.
Results
hBMSCs labeling
CD29, CD44, CD105, CD106 and fetal liver kinase-1 (Flk-1) were expressed by the hBMSCs. However, human leukocyte antigen-DR (HLA-DR), CD31, and CD34 were not expressed. The cells exhibited the capacity for multi-lineage differentiation, especially into adipocytes, osteoblasts and chondrocytes.
Postoperative observations
All beagles survived until they were euthanized. The operative incision healed within two weeks postoperatively. After catheter removal, all the five beagles of experimental group showed a mild difficulty in voiding, which did not have any influence on their survival. However, all the five beagles of control group suffered from varied degree of dysuria at different periods. Urinary fistula was observed in two of them, and cystostomy was operated on one beagle with serious dysuria.
Macroscopic observation and retrograde urethrography
Compared with the control group, the urethras in the experimental group were relatively soft and elastic. All the reconstructed urethras could form a complete cavity despite stenosis, and the mucosal surfaces were smooth and ruddy in the experimental group.Similar situation occurred only in two animals in the control group, but their mucosal surfaces appeared rough and pale. The reconstructed urethras in the remaining three beagles could not form a complete cavity with two suffering from obvious stenosis and one suffering from even atresia (Figure 1).
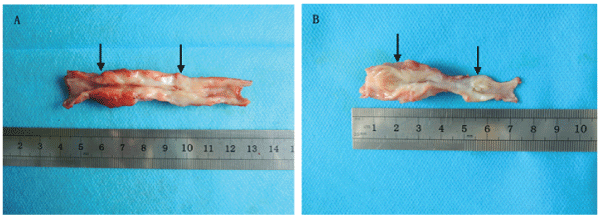
Figure 1: The macroscopic observation of the reconstructed urethras at 6 months postoperative. (A) Experimental group, (B) Control group. The area between the two black arrows indicates the repaired urethra.
View Figure 1
Retrograde urethrography indicated that varying degree of urethra strictures were observed in all the animals of two groups. But the severer urethrostenosis usually appeared in the control group (Figure 2).
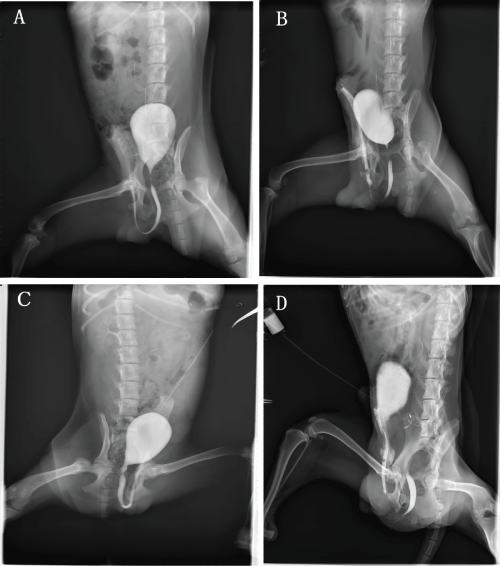
Figure 2: Retrograde urethrography before urethral reconstruction (A, C) and at 6 months postoperatively (B, D) in the experimental group (A, B) and the control group (C, D).
View Figure 2
Hematoxylin and eosin staining
The results of the HE staining revealed that the reconstructed urethras in the experimental group had formed stratified urothelium layers that were similar in appearance to the normal urothelium. The reconstructed urethras in the control group formed a discontinuous mucosa with a monolayer of urothelial cells in most areas, and the arrangement of cells was irregular. The submucosa was primarily covered with fibrous tissues, though a small number of smooth muscles and blood vessels were observed in both groups (Figure 3).
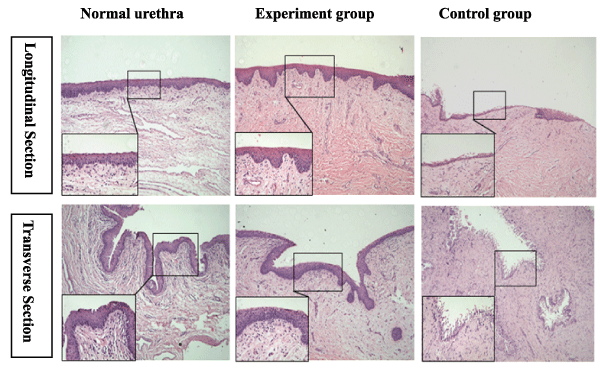
Figure 3: The comparative H&E staining of a normal urethra and the reconstructed urethra in the experimental group and the control group at 6 months postoperative. Reduced from ×100,400.
View Figure 3
Immunofluorescent staining to detect human cells
The immunofluorescence examination with anti-human nuclei staining showed that a small number of positive cells were found in the experimental group, but not found in the control group. These cells were predominantly distributed in the submucosa and were rarely found on the mucosal surfaces, which suggested that hBMSCs were able to survive in beagles and play a role in the regeneration of urethras (Figure 4).
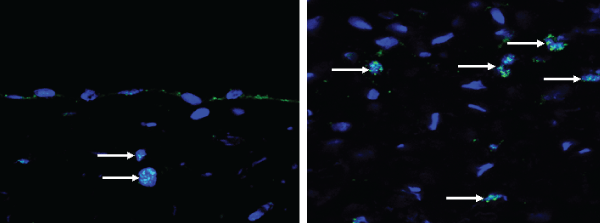
Figure 4: The fluorescence expression of human nuclei in the reconstructed urethras (longitudinal section); the white arrows indicate the positive cells in the experimental group. Reduced from ×800.
View Figure 4
Immunofluorescence analysis of urethral tissue elements
Stratified urothelium was clearly confirmed by anti-pancytokeratin AE1/AE3 staining in the experimental group. In contrast, single layer urothelium was confirmed in the control group. The statistical results suggested that the mean fluorescence density of the epithelial cells (0.204 ± 0.009) in the experimental group was significantly higher than that (0.067 ± 0.009, P<0.05) in the control group; however, there was no difference in comparison with the normal urethras (0.209 ± 0.006, P>0.05). The smooth muscles and blood vessels were not significantly confirmed in either group by the anti-alpha smooth muscle actin and anti-von Willebrand factor staining. Although the mean fluorescence density of the smooth muscles (0.063 ± 0.005) or blood vessels (0.036 ± 0.004) in the experimental group was higher than that (smooth muscles: 0.040 ± 0.005; blood vessels: 0.024 ± 0.004, P<0.05) in the control group, it was far lower than that (smooth muscles: 0.147 ± 0.006; blood vessels: 0.127 ± 0.007, P<0.05) in the normal urethras (Figure5,6).
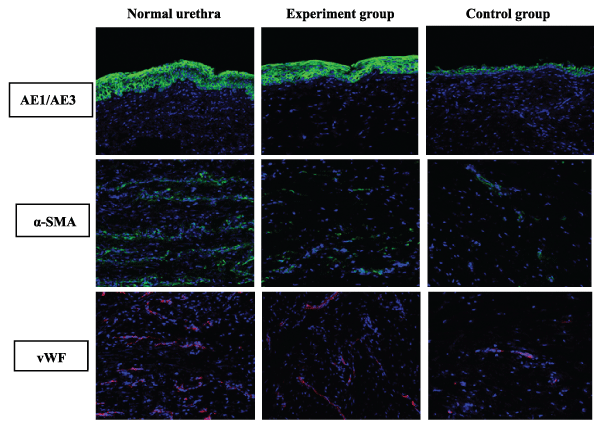
Figure 5: The comparative immunofluorescence staining of a normal urethra and the reconstructed urethra in the experimental group and the control group at 6 months postoperative by the anti-AE1/AE3, α-SMA and vWF staining (longitudinal section). Reduced from ×400.
View Figure 5
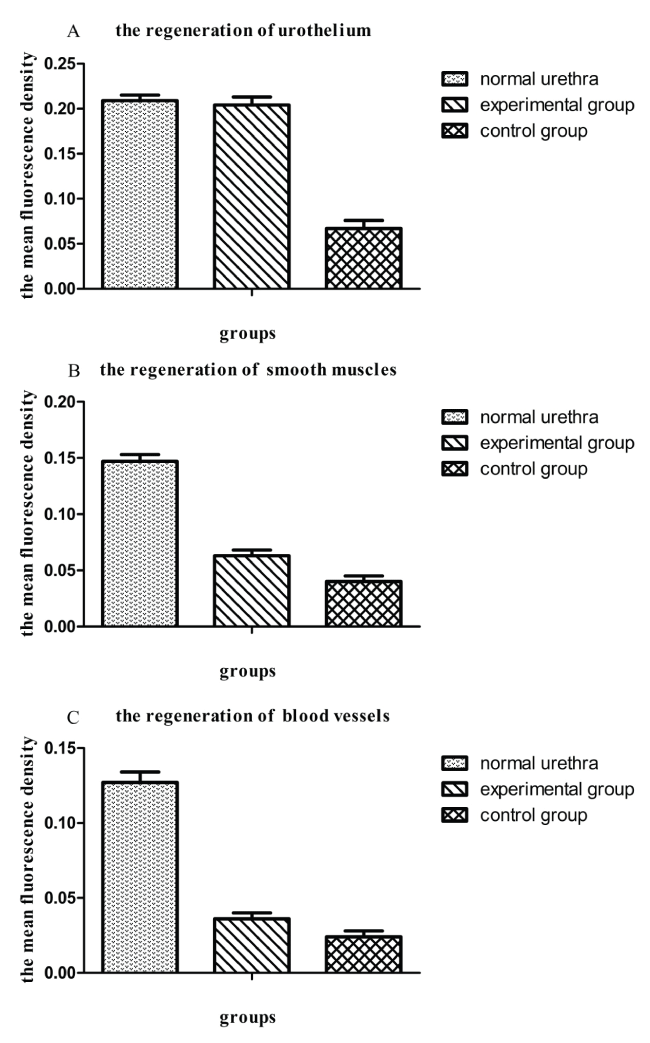
Figure 6: The statistical analysis of the mean fluorescence among the normal urethra, the experimental group and the control group. A: The regeneration of urothelium; B: The regeneration of smooth muscles; C: The regeneration of blood vessels. The data were represented as mean ± standard deviation (SD); P<0.05 was considered significantly different, and P>0.05 was considered to be not different.
View Figure 6
Discussion
In recent years, tissue engineering technology has become widespread for urethral reconstruction in animals. But for extensive urethral defects, the results of simple scaffold-only repairing are not satisfactory [17]. In these cases, cell-seeded scaffolds are needed for tissue regeneration [18]. Various cells including bone marrow mesenchymal stem cells [12,13], urothelial cells [19], adipose tissue-derived stem cells [20], and oral keratinocytes [21] have been used for urethral regeneration in many studies.
Of these cells, hBMSCs have shown great clinical promise. The relatively wide range of differentiation, with a high proliferation rate and relative ease of isolation, of hBMSCs, ensure that they are an ideal source of cells for regenerative medicine [22]. Furthermore, these cells display properties of immunoregulatory and anti-inflammatory [23]. Because of their ability to secrete cytokines and growth factors, they have the potential to reduce scarring and inflammation and stimulate the regeneration of tissues [24]. In this study, hBMSCs were selected as seeding cells to evaluate how human cells interact with canine tissue-engineered urethras and as a prerequisite for clinical stem cell therapy.
Typically, the seeded grafts were applied in vivo after being incubated for 7 days in vitro [7]. According to the clinically relevant ethics rules, adult stem cells are not allowed for any specific treatment in vitro before transferring into humans [25]. Our previous work has showed that collagen-based scaffolds could provide an appropriate microenvironment to support cell attachment and proliferation [14]. To approach clinical conditions, the cells in our study were directly injected into the collagen scaffold instead of three-dimensional culture in vitro. After 6 months, human nuclei were found in the reconstructed urethras, which demonstrated that hBMSCs could be attached to the scaffold and contribute to the formation of tissue-engineered urethras.
For this study, the problem of immunological response in hBMSCs applied to canines should be considered. However, hBMSCs are characterized by low immunogenicity. These cells express low levels of major histocompatibility complex-I (MHC-I) molecules on their cell surfaces, and do not express MHC-II molecules and co-stimulatory molecules, including CD80, CD86, or CD40 [26]. Furthermore, hBMSCs plays an active role in inhibiting T-cell proliferation [27]. Actually, immunological rejection rarely occurs when hBMSCs are used for xenotransplantation. The expression of human nuclei indirectly confirmed that hBMSCs were able to survive in canines.
The results showed that hBMSCs could promote urethral tissue growth, especially for the urothelium regeneration. In comparison, the cell-seeded scaffolds developed a layer of stratified epithelium similar to the normal. Nevertheless, few positive cells were found in the epithelium by the anti-human nuclei staining. The secretory function rather than the differentiation function is suggested to play an important role in epithelium regeneration. Epithelium growth is important for constructing a tissue-engineered urethra because the epithelial cell lining could serve as a barrier to urinary invasion and prevention of physical friction after transplantation [28]. A complete epithelial layer may protect the reconstructed urethra against atresia [29]. In addition, smooth muscles and blood vessels are two other significant elements in rebuilding a tissue-engineered urethra. Histological examination indicated that there were more smooth muscles and blood vessels growing than those in controls, although they were far fewer than normal. We also found some human cells were distributed in the submucosa which probably suggested hBMSCs aided in the formation of blood vessels and smooth muscles by promoting the migration of progenitor cells of the host to the cell-seeded scaffold. A relatively thicker muscle layer may prevent the cell-seeded urethra from collapsing and adhesion [18]. As another important factor, blood supply can provide nutrients and oxygen to the urethral tissue [30]. It was reported that hBMSCs could promote vascularization by releasing growth factors including VEGF, β-FGF and angiopoietin [31]. The growth advantage of epithelium in the cell-seeded constructs may relate to the better blood supply.
In our study, hBMSCs were considered a promotion of urethral tissue regeneration. However, all the repaired urethras existed varying degrees of stenosis which could influence the normal function; one limitation might be the low strength of scaffold materials. Another possible cause of the stenosis is that the reconstructed urethra did not form a complete cavernous body, but mostly covered with fibrous tissues outside the mucous membrane. The cavernous body of urethra which can ensure the urethra more elastic to avoid scar contraction, is thought to be significant in the process of urethral regeneration [32].
In conclusion, hBMSCs loaded on collagen scaffolds can potentially promote the regeneration of canine tissue-engineered urethras, predominantly for the urothelium. However, the functional recovery is not satisfactory with the development of stenosis. Further research about the exact mechanism of stenosis and the effective solution is needed before this approach can be put into clinical practice. In addition, to ensure the long-term fate of these cells following transplantation, considerable research still needs to be performed.
Acknowledgements
The present study was funded by Strategic Priority Research Program of the Chinese Academy of Sciences. Grant number: XDA01030000.
References
-
McAninch JW, Morey AF (1998) Penile circular fasciocutaneous skin flap in 1-stage reconstruction of complex anterior urethral strictures. J Urol 159: 1209-1213.
-
Duckett JW, Coplen D, Ewalt D, Baskin LS (1995) Buccal mucosal urethral replacement. J Urol 153: 1660-1663.
-
Song LJ, Xu YM, Hu XY, Zhang HZ (2008) Urethral substitution using autologous lingual mucosal grafts: an experimental study. BJU Int 101: 739-743.
-
Xu Y, Qiao Y, Sa Y, Zhang H, Zhang X, et al. (2002) An experimental study of colonic mucosal graft for urethral reconstruction. Chin Med J (Engl) 115: 1163-1165.
-
Barbagli G, Guazzoni G, Lazzeri M (2008) One-stage bulbar urethroplasty: retrospective analysis of the results in 375 patients. Eur Urol 53: 828-833.
-
Venn SN, Mundy AR (1998) Urethroplasty for balanitis xerotica obliterans. Br J Urol 81: 735-737.
-
De Filippo RE, Yoo JJ, Atala A (2002) Urethral replacement using cell seeded tubularized collagen matrices. J Urol 168: 1789-1792.
-
Raya-Rivera A, Esquiliano DR, Yoo JJ, Lopez-Bayghen E, Soker S, et al. (2011) Tissue-engineered autologous urethras for patients who need reconstruction: an observational study. Lancet 377: 1175-1182.
-
Yu RN, Estrada CR (2010) Stem cells: a review and implications for urology. Urology 75: 664-670.
-
Doi D, Morizane A, Kikuchi T, Onoe H, Hayashi T, et al. (2012) Prolonged maturation culture favors a reduction in the tumorigenicity and the dopaminergic function of human ESC-derived neural cells in a primate model of Parkinson's disease. Stem Cells 30: 935-945.
-
Otto WR, Wright NA (2011) Mesenchymal stem cells: from experiment to clinic. Fibrogenesis Tissue Repair 4: 20.
-
Wang K, Guan Y, Liu Y, Zhu M, Li T, et al. (2012) Fibrin glue with autogenic bone marrow mesenchymal stem cells for urethral injury repair in rabbit model. Tissue Eng Part A 18: 2507-2517.
-
Li CL, Liao WB, Yang SX, Song C, Li YW, et al. (2013) Urethral reconstruction using bone marrow mesenchymal stem cell- and smooth muscle cell-seeded bladder acellular matrix. Transplant Proc 45: 3402-3407.
-
Shi C, Li Q, Zhao Y, Chen W, Chen B, et al. (2011) Stem-cell-capturing collagen scaffold promotes cardiac tissue regeneration. Biomaterials 32: 2508-2515.
-
Han Q, Sun Z, Liu L, Chen B, Cao Y, et al. (2007) Impairment in immuno-modulatory function of Flk1(+)CD31(-)CD34(-) MSCs from MDS-RA patients. Leuk Res 31: 1469-1478.
-
Bao X, Wei J, Feng M, Lu S, Li G, et al. (2011) Transplantation of human bone marrow-derived mesenchymal stem cells promotes behavioral recovery and endogenous neurogenesis after cerebral ischemia in rats. Brain Res 1367: 103-113.
-
Chapple C, Osman N, MacNeil S (2013) Developing tissue-engineered solutions for the treatment of extensive urethral strictures. Eur Urol 63: 539-541.
-
Orabi H, AbouShwareb T, Zhang Y, Yoo JJ, Atala A (2013) Cell-seeded tubularized scaffolds for reconstruction of long urethral defects: a preclinical study. Eur Urol 63: 531-538.
-
Xie M, Song L, Wang J, Fan S, Zhang Y, et al. (2013) Evaluation of stretched electrospun silk fibroin matrices seeded with urothelial cells for urethra reconstruction. J Surg Res 184: 774-781.
-
Zhao Z, Yu H, Xiao F, Wang X, Yang S, et al. (2012) Differentiation of adipose-derived stem cells promotes regeneration of smooth muscle for ureteral tissue engineering. J Surg Res 178: 55-62.
-
Li C, Xu YM, Song LJ, Fu Q, Cui L, et al. (2008) Urethral reconstruction using oral keratinocyte seeded bladder acellular matrix grafts. J Urol 180: 1538-1542.
-
Becker C, Jakse G (2007) Stem cells for regeneration of urological structures. Eur Urol 51: 1217-1228.
-
Oh JY, Kim MK, Shin MS, Lee HJ, Ko JH, et al. (2008) The anti-inflammatory and anti-angiogenic role of mesenchymal stem cells in corneal wound healing following chemical injury. Stem Cells 26: 1047-1055.
-
Ankrum J, Karp JM (2010) Mesenchymal stem cell therapy: Two steps forward, one step back. Trends Mol Med 16: 203-209.
-
Department of Ethics, Chinese National Human Genome Center in Shanghai (2014) Recommendations of ethical codes for human adult stem cells clinical trials. Chinese Medical Ethic 27: 191-194.
-
Si YL, Zhao YL, Hao HJ, Fu XB, Han WD (2011) MSCs: Biological characteristics, clinical applications and their outstanding concerns. Ageing Res Rev 10: 93-103.
-
Tse WT, Pendleton JD, Beyer WM, Egalka MC, Guinan EC (2003) Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation 75: 389-397.
-
Mikami H, Kuwahara G, Nakamura N, Yamato M, Tanaka M, et al. (2012) Two-layer tissue engineered urethra using oral epithelial and muscle derived cells. J Urol 187: 1882-1889.
-
Dorin RP, Pohl HG, De Filippo RE, Yoo JJ, Atala A (2008) Tubularized urethral replacement with unseeded matrices: what is the maximum distance for normal tissue regeneration? World J Urol 26: 323-326.
-
Heitz M, Pottek T, Schreiter F (1998) [Anatomy and blood supply of the penis and urethra]. Urologe A 37: 2-7.
-
Kaigler D, Krebsbach PH, Polverini PJ, Mooney DJ (2003) Role of vascular endothelial growth factor in bone marrow stromal cell modulation of endothelial cells. Tissue Eng 9: 95-103.
-
Song L, Xu Y, Fu Q, Li C, Zhu W (2011) An experimental study of nonwoven polyglycolic acid membrane for urethral reconstruction. J Mod Urol 16: 293-295.





