Journal of Otolaryngology and Rhinology
Surgery Guidelines for Barrier Membranes in Guided Bone Regeneration (GBR)
Rispoli L1*, Fontana F1, Beretta M1, Poggio CE1,2* and Maiorana C1
1Implant Center for Edentulism and Jawbone Atrophies, University of Milan, School of Dentistry, Maxillofacial Surgery and Dentistry UOC, Italy
2Division of Prosthodontics, Eastman Institute for Oral Health, School of Medicine and Dentistry, University of Rochester, USA
*Corresponding author:
Rispoli L, Implant Center for Edentulism and Jawbone Atrophies, University of Milan, School of Dentistry, Maxillofacial Surgery and Dentistry UOC, Fondazione IRCCS Ca' Granda, Milan, MI, Italy, E-mail: rispolisurg@hotmail.com
Poggio CE, Division of Prosthodontics, Eastman Institute for Oral Health, School of Medicine and Dentistry, University of Rochester, Rochester, NY 14620, USA, E-mail: carlo_poggio@urmc.rochester.edu
J Otolaryngol Rhinol,
JOR-1-008, (Volume 1, Issue 2),
Review Article
Received: October 10, 2015: Accepted: November 13, 2015: Published: November 15, 2015
Citation: Rispoli L, Fontana F, Beretta M, Poggio CE, Maiorana C (2015) Surgery Guidelines for Barrier Membranes in Guided Bone Regeneration (GBR). J Otolaryngol Rhinol 1:008
Copyright: © 2015 Rispoli L, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: To analyze the biological principles of guided bone regeneration and to provide the clinician with information regarding the use of different membranes.
Materials and Methods: The choosing criteria among different membranes and the clinical options are presented. Finally, the surgical technique is described in details.
Results and Conclusions: Guided bone regeneration is one of the several surgical techniques that have been introduced in the last two decades for bone regeneration prior or at the time of implant placement. The rationale of this technique is the positioning of a barrier membrane between the bone and the connective tissue to create a secluded space and to enhance the proliferation of bone forming cells.
Keywords
Bone regeneration, Barrier membranes, Non resorbable membranes, Resorbable/absorbable membranes, Bone defect, Bone grafting, Jawbone atrophy, Alveolar ridge augmentation, Ridge morphology, Dental Implants
Introduction
Since Branemark et al. introduced the concept of osseointegration, bone volume is considered a prerequisite for successful implant treatment [1]. Inadequate bone quantity in cases of large bone defects secondary to trauma, tumor resection, advanced periodontitis, infection and/or iatrogenic injury represents a great challenge for an ideal implant placement.
In the last two decades several surgical techniques were introduced to overcome horizontal and/or vertical bone lack in implant sites in either a non-staged or staged approach. One technique of ridge augmentation is the guided bone regeneration (GBR) that uses barrier membranes, in order to prevent the invasion of surrounding ephithelial and fibrous tissue. Regeneration by GBR depends on the slower migration rate of osteogenic cells to the bone defect [2,3]. The underlying concept of GBR was first introduced more than 50 years ago, when cellulose acetate membranes were tested in the orthopedic field, for radial bone regeneration, rib and femoral bone defects [4,5]. Later, a series of animal studies provided evidence to show GBR concept can predictably facilitate healing of critical-sized bone defects or atrophic alveolar ridges prior to implant insertion [6,7]. Melchers hypothesis of selected cell repopulation formed the basis of the biological principle of guided tissue regeneration (GTR) [8]. These concept further evolved to a more compartmentalized principle of guided bone regeneration (GBR) developed by Nyman, Lindhe, Karring and Gottlow in the 1980s [2,3,9,10]. This introduced the importance of suitable environment for osteogenesis by excluding ephitelial and connective tissue cells from bone defects during the healing phase of the periodontal regeneration, faciliting the colonization of ligament cells, to form new connective tissue and alveolar bone.
These concepts were later applied to bone regeneration and experimental studies [11] demonstrated that membrane can predictably faciltate bone regeneration by enhancing angiogenesis and osteoprogenitor cells proliferation. In the 1990s, thanks to many clinical trials [12-15], principles of GBR were applied to the atrophic jaws.
Biological Bases of Guided Bone Regeneration (GBR)
Bone regeneration with membranes focused on three main biological principles: 1. Cell occlusiveness 2. Wound stabilization 3. Space making and maintenance.
Cell occlusiviness
A physical barrier is necessary to prevent the proliferation of competing soft tissue cells from the mucosa into the defects. Occlusivity is therefore closely linked to membrane porosity; a larger pore size will allow faster-growing cells to overpopulate the defect and inhibit the infiltation and activity of bone forming cells. A large pore size will decrease the resulting surface area of the material which could limit the initial cell adhesion onto the membrane. On the other hand, cell migration is limited if pores are too small enhancing, collagen deposition, formation of avascular tissue and infiltration as they are inadeguate for capillaries penetration.
Wound stabilization
Tissue integration stabilizes the healing wound process via blood clot differentiation and prevents connective tissue integration into the defect.
Space making and maintenance
Adequate stiffness stability of the membrane is required to avoid collapse within the defect at critical stages of the healing process allowing tissue differentiation by direct or appositional bone formation. It has been observed that bone formation is enhanced when resorbable membrane is tightly attached and immobilized to the bone surface [16].
From a biological point of view, the pattern of guided bone regeneration has a similar sequence to the alveolar bone healing after tooth extraction. Several experimetal studies have identified the pattern of bone formation after GBR procedures [17,18]. The first phase is characterized by osteoid tissue deposition within the defect after the formation of blood clot; the space created below the membrane is organized in a matrix of newly formed dense connective tissue. This stage is completed only if the stability is sufficient to avoid micromovements which have a inhibitory effect on bone formation. This connective tissue provided the mechanical properties necessary to allow the growth of capillaries during the angiogenesis that precedes the bone formation. According to Schenk et al. at 2 months is possible to observe a network of small blood vessels followed by woven bone deposition which initially consisting of osteoid matrix, followed by rapid mineralization [18]. The osteogenesis starts in close contact with bony defect walls, where a randomly oriented scaffold of delicate fibrous trabeculae is present, and progesses toward the center of the defect. The surface of the newly formed trabeculae are lined by a continuous layer of osteoblasts, and ongoing bone deposition further increase their diameter. The intertrabecular spaces surrouded by a well vascularized tissue and well organized connective fibers initially delimitate by osteoid matrix are integrated in the bone tissue. The result of this bone formation was a primary spongework. During this phase both structure and quality of newly formed woven matrix is mainly the result of bone maturation from randomly oriented trabeculae, whereas the secondary spongiosa, results from remodeling, consisting of substitution by resorption, followed by formation of parallel fibered and lamellar bone.
Barrier Membranes
A wide range of barrier materials has been used in GBR procedures, including polytetrafluoroethylene (PTFE), expanded PTFE (e-PTFE), high density-PTFE (d-PTFE), titanium reinforced ePTFE, titanium mesh, poly(lactic acid) (PLA), poly(glycolic acid)(PGA), poly(-caprolactone)(PCL), poly(urethane), poly(lactic acid-polysiloxane hybrid), native and/or synthetic collagen, freeze-dried dura mater; freeze-dried fascia lata allografts, pericardium and cortical foil.
The barrier membrane is expected to fullfill the following design criteria, as described by Scantlebury [19].
• biocompatibility (without inflammation or immunological response)
• tissue integration
• clinical manageability
• space-making ability and maintenance
• cell-occlusiveness
Membranes are divided into two main groups: non-resorbable and resorbable.
Non-resorbable membranes
Non-resorbable membranes include polytetrafluoroethylene (PTFE) and titanium mesh. In critical size defects non-resorbable devices have better abilities to achieve successful regeneration for their stiffness, controlled time of barrier effect and lack of resorption process. The main limitations of non-resorbable is the need for an additional surgery for their removal and wound dehiscence because of incomplete coverage during healing. With the development of resorbable materials and the increasing evidence of their effectiveness the use of ePTFE and titanium mesh has become limited to specific indications.
Expanded-PTFE (e-PTFE) high density -PTFE (d- PTFE) membranes: Polytetrafluoroethylene (PTFE) membranes can be divided into three microsctrures types: expanded polytetrafluoroethylene (e-PTFE, Teflon), high denisty-PTFE (d-PTFE) and titanium reiforced e-PTFE. Their use in GBR procedures has been extensively tested in clinical and experimental trials. They have superior space-maintaining properties and capacity for cell occlusion than resorbable membranes. The PTFE matrix confers cellular selectivity and allows the passage of liquids. [13,14,18,20-23]. E-PTFE membranes have been the main protagonists for bone guided regeneration in the early 1990s to cover dehiscences or fenestration bony defects around implants [24-27]; to preserve and regenerated bone around implants placed in fresh extraction sockets [14,28-30]; for vertical and/or horizontal ridge regeneration [15,22,31] and to protect the bony window during sinus lift procedures [32]. Currently their use is limited to vertical and/or horizontal critical size defects in combination with bone graft or resorbable membranes for optimal bone width and height in GBR. Early exposure of e-PTFE membranes, because of larger pore size, constitutes a not infrequent observation that implies the need for premature removal with negative outcomes on bone regeneration [33]. It was shown that for high denisty-PTFE (d-PTFE) membranes, the risk of bacterial colonization is less than of e-PTFE even after exposure. The primary soft tissue closure is not required because of this high density and submicrom pore sizes thus enhancing vertical and/or horizontal bone regeneration and soft tissue healing [34-37].
Titanium reiforced e-PTFE d-PTFE membranes (TR e-PTFE ) (TR d-PTFE ): The reinforcement of PTFE membranes with flexible titanium frameworks allows the membrane to be shaped to fit a variety of defects and provides additional stability in supracrestal bone defects and large dehiscence around dental implants and superior preservation of the regenerated ridge during healing period [12]. Extra stabilization with tacks/miniscrews or titanium mesh often required to more predictable regenerative results [38].
Resorbable membranes
Resorbable membranes offer many advantages over non-resorbable devices: there is no need for removal, they present improved soft tissue healing, and they have a better cost-benefit ratio and lower risk of additional morbidity and complications [39]. Stiff resorbable membranes promote a similar degree of bone formation as a non-resorbable membranes [40,41]. The main disadvantages are the lack space making ability and the unpredictable degree of degradation. Early loss of barrier function directly affects bone formation especially in case of premature exposures and/or implant stability [42-47]. Bioresorbable materials belong to the group of natural or synthetic polymers (aliphatic polyesters and collagen).
Polylactide (PLA) and polyglycolide (PGA) membranes: Polylactide PLA and Polyglycolide PGA are synthetic products made of aliphatic polyesters available for clinical use in maxillofacial and dental surgery during GBR procedures [43,44]. They are degradated, through hydrolysis, respectively, to lactic and glycolic acid. These two substances are then metabolized to CO2 and H2O by the Krebs cycle. Some experimental and clinical studies have shown the effectiveness of polymeric membranes for the treatment of dehiscences and fenestrations around implants, especially in combination with autologous bone graft [43,44]. However, some authors found that, once exposed PLA/PGA membranes started to resorb almost instantly reducing the function barrier time and space-making ability of the membrane [42]. It has been hypothesized that the process of hydrolytic degradation, typical of these membranes, is associated with the resorption of a part of the regenerated bone [45].
Collagen membranes: Collagen is widely used for the fabrication of resorbable membranes, since it constitutes up to 50% of the proteins of the human body. It is degraded and disposed through enzymatic reactions resembling those involved in normal tissue turnover. Resorbable collagen membranes are primarily produced with bovine and porcine collagen.
The choice of collagen, compared to other synthetically derived products, is due to a combination of optimal biodegradation over time (absorbed through catabolic process), high angiogenesis, fibroblasts chemotaxis and semi-permeability, clot stabilization and favorable tissue integration [46].
The high tissue integration has been particularly observed for type I and type III porcine collagen membranes. The main shortcoming of collagen is its rapid biodegradation by enzymatic activities of macrophages, by polymorphonuclear leukocytes and some periodontal bacteria. The collagen membranes have an estimated time of resorption of between 4 and 8 weeks, resulting in a very limited barrier effect and space-making capacity [47]. To overcome this limitation, these should be used in combination with autologous bone grafts or xenogenic bone substitutes to retain the original position of the membrane itself and not collapse in the defect. Several techniques have been used to extend the collagen barrier effect, referred to as "cross-linking" [48,49]. Animal studies have shown that the degradation of the cross-linked membranes takes longer (from 4 weeks up to six months) than the not cross-linked [47,50]. Cross-linking of bovine and porcine-derived collagen types I and III was associated with prolonged biodegradation, decreased tissue integration and vascularization and foreign body reactions [47]. Increasing the density of cross links between collagen molecules has a negative effect on barrier biocompatibility and inhibits cells attachment and proliferation of PDL fibroblast and osteoblast [47,51-54]. Furthermore prolonged biodegradation seems to be associated with a host-tissue response, impaired transmembrane vascularization and tissue dehiscence [46,47].
Pericardium membranes: The pericardium membranes in comparison to collagen membranes have shown an effective crosslinking, suggesting prolonged resorption with the properties of native collagen. These have a resorption time of 2 to 6 months according to the site, biomechamnical load on the graft, age and health and metabolic status. Several in vitro and in vivo studies were performed to evaluate the effectiveness of decellularized pericardium membranes in enhancing bone augmentation [55-57]. Xenogenic pericardium is commonly derived from bovine and porcine sources. The native pericardium structure has three layers largely comprised a network of collagen and elastic fibres embedded in an amorphous matrix with a porous surface for cellular attachment and proliferation, but also in sufficient density for soft tissue exclusion. It's a cellularity, reliable consistency, suture retention and handling properties promoting the use of bovine pericardium in guided tissue regeneration and bone regeneration procedures. Pericardium from bovine sources has higher collagen content than the porcine pericardial tissue. In a pilot study has been shown a considerable cell proliferation significantly superior to that observed with porcine collagen types I and III (biodegradation within 4-8 weeks) and resorption of porcine pericardium within 8-12 weeks [58]. Porcine pericardium membrane proved in an experimental study to regenerate bone around implants placed in fresh extraction sockets and a complete regeneration for horizontal defects around implants compared to porcine collagen resorbable membrane [59,60]. Human pericardium as GBR membrane showed experimentally excellent mechanical properties enhancing bone regeneration [61]. More clinical trial is required to prove their efficiency in GBR.
Cortical foil membranes: Soft cortical bone porcine membranes were used in a split-mouth design study for alveolar ridge preservation procedure. The randomized-controlled trial revealed a significant reduction of bone resorption combining the use of porcine derived intrasocket xenograft with cortical membrane and a predictable maintenance of the horizontal and vertical ridge dimensions [62].
A recent approach for lateral GBR in posterior maxilla combining a resorbable barrier made from porcine cortical lamina, facing the inside of the defect, with a collagen membrane facing the outside, is presented in a case series with the advantages of space-maintain properties and long lasting barrier effect of the cortical layer and improved tissue integration of the collagen membrane. Histologic analysis at 6 months indicated a complete vascularization and substitution of the cortical bone lamina by newly formed connective tissue [63].
Clinical Applications
Since every membranes offers both advantages and disadvantages a membrane should be selected based on the biomaterial properties as well as the treatment requirements [64,65]. Resorbable membranes have been developed to avoid the need for surgical removal. In situations where the bone defect margins are well maintained by the membranes, favorable results have been reported [66]. Overall, their advantages are:
• single step procedure;
• patient's lower stress and morbidity;
• low risk of complications;
• better cost-benefit ratio;
• Allow to raise a split thickness flap at 2-stage implant surgery.
However, non resorbable membranes were found to have superior space-maintaining properties in critical size defects. [64,65]. This increased efficiency is also due to:
• better occlusiviness;
• ability to control the barrier effect (healing time 6-9 months);
• lack of a resorption process, which could negatively affect bone formation
The choice of the type of membrane to be used depends substantially from the characteristics of the bone defect. Mechanical durability, handling properties, resorption time can be adjusted to the clinical cases by changing composition, thickness, porosity size and manufacturing procedure. A precise classification of defects bone is difficult to perform because of the complexity and variety of anatomical situations that may arise. However, to provide directions for a correct treatment planning, peri-implant defects can be grouped into:
• fenestrations;
• dehiscence;
• horizontal defects;
• vertical defects or overbony
Fenestrations
Implant fenestrations are the clinical conditions that occur when the middle or apical (buccal or lingual) surface of the implant is exposed. It is a typical consequence of implant placement corresponding to an alveolar ridges concavity at the most apical portion, or to residual defects after granulomas or cysts extraction or in cases where, for prosthetic reasons, the position of the implant follow a different angle from that of the ridge. In these defects only a small implant portion (< 2 mm) is shown in vestibular/lingual plate.
The component of corono-apical fenestration can be long almost the implant (except the most coronal part, which must be intact). The extent of regeneration is absolutely limited and is performed contextually to the implant placement [67].
The use of GBR for the management of fenestrations has been proposed by several authors [40]. It was suggested the use of e-PTFE membranes, but more recently, for the reasons given above, it has been completely replaced by handling shaped resorbable membranes.
Dehiscences
Implant dehiscences are clinical conditions that occur when a part of the implant (also including the coronal portion) remains exposed from the bony ridge. They are frequently observed in cases of tiny alveolar ridges, or in the case of post-extraction implants with no buccal or lingual cortical bone. The dehiscence may be simple, when interest a small portion of the implants (vestibular or palatal), or, as the classification of periodontal bone defects, at 1, 2, 3 or 4 walls dehiscence.
Simple dehiscence: Most of the implant is inserted in the bone crest. While the extension in the apico-coronal axis may also correspond to the entire length of the implant, the size of dehiscence in the buccal-palatal axis is limited to < 2 mm. If left untreated it leads to a reduction of the implant integration and promote peri-implant soft tissue inflammation. The treatment plan consists in the use of a resorbable membrane in association with some chips of autogenous bone or bone substitute [39,40,67-70].
1 wall dehiscence: Occur when more half the diameter of the implant (> 2 mm) is exposed along buccal-palatal axis. Independently to the extent of the apical-coronal component, these clinical conditions can be effectively defined as real horizontal defects. The implants is stabilized in the most apical part of crest, but the amount of bone to be horizontally regenerated is considerable (at least 3-4 mm). The treatment plan goes through careful analysis of the characteristics of the defect.
It is a 1 wall defect, therefore is reduced the bone surface from which the osteogenic cells can proliferate. It is not a contenitive defect, therefore requires the use of a membrane to maintain a space for osteoprogenitors cells. The Tr-PTFE membrane associated with a bone graft is the option of choice, required in this cases acting as a scaffold for osteoconduction and as a source of osteogneic and osteoinductive substances for lamellar bone formation.
2, 3 or 4 walls dehiscence: are grouped into the same clinical condition in which at least two walls are present at the exposed implant. These defects are usually contenitive and they well support the bone graft with the membrane. Literature data largely confirm the use of GBR with a resorbable membrane for the management of such defects [39,40,67-70]. The choice in the treatment plan turns therefore towards the clinical use of a resorbable membrane in combination to a particulate bone graft.
Horizontal defects
Insufficient horizontal bone precludes the successful outcome of an ideal implant placement. The concept of GBR for the reconstruction of the alveolar ridge defect prior to implant placement has been developed in an effort to "guide" bone reconstruction treatment. One of the main problems in such situations is the lack of a delimitate and contenitive defect. The horizontal defects are flat and large and the membrane, if not properly supported, tends to collapse against the buccal plate of the edentulous alveolar ridge. Structural stability of the membrane optimize the healing potential by creating a space for blood clot stabilization and ingrowth and support to the tissue during bone regeneration.
The necessary surgical technique requires the use of a barrier with high "space making" and stability capacity and, therefore, a titanium-reinforced membrane (Tr-PTFE). The titanium framework of these devices it can be bent in order to fit the membrane to the receiving site defining the size and morphology of the bone regeneration.
Additional bone graft, in association, has been suggested to increase the regenerative potential of GBR [22,71,72] as the use of support fixing-screws to maintain stability of the membrane. A graft healing time of about 6-9 months is necessary before the insertion of the implants. This technique was developed from clinical and experimental studies conducted by Buser and co-workers [22] and since 1996 e-PTFE membranes have been widely used associated with autologous bone grafts for predictable lateral augmentation of the alveolar ridges.
Vertical defects
Vertical ridge deficiency constitutes one of the more difficult clinical situations for an ideal implant placement due to the presence of adjacent residual anatomical structures as inferior alveolar nerve and maxillary sinus. This deficit creates also difficulties especially in aesthetic area where the correct angulation of the implant and management of hard and soft tissues are treatment plan's mandatory requirements.
First experimental studies conducted by Dahlin et al. showed supracrestal bone regeneration by GBR [11]. The first clinical applications on humans are attributed to Simion et al. in 1994 [15].
In the last decade vertical ridge regeneration with Tr -PTFE-barrier membranes associated to a bone graft has been shown to be an effective and predictable procedure [71-73]. Autologous bone is a preferred material for grafting alone or a mixture of autologous bone and heterologous bone in 1: 1 ratio [68,71,73].
The success of the procedure largely depends on the surgeon's experience and scrupulous application of the surgical protocol. Implants can be placed in the same surgery time of the vertical ridge augmentation if there are at least residual 4-6 mm for implants stabilization (one-stage approach), otherwise they are placed 6-9 months after regenerations procedure (staged-approach) [71-74] (Figure 1, Figure 2, Figure 3, Figure 4 and Figure 5).
.
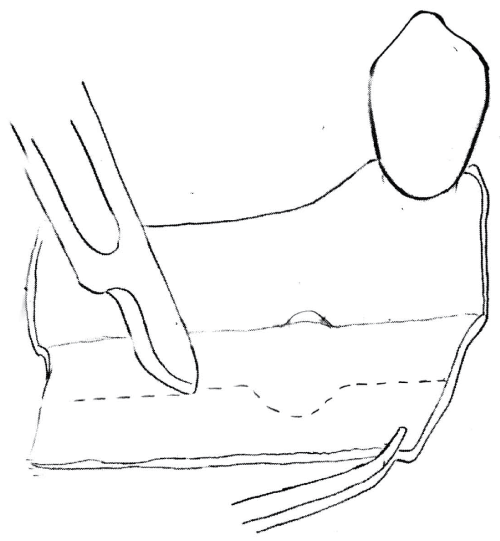
Figure 1: Titanium reinforzed e-PTFE membrane is fixed lingually with 2 mini-screws. Tend srews are positioned to define the amount of vertical regeneration. Cortical plate is perfored is perfored to ensure blood clot formation and mechanical interlocking of the graft.
View Figure 1
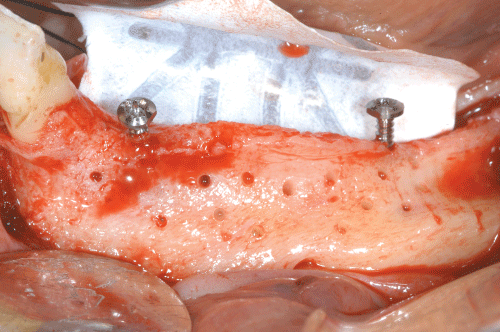
.
Figure 2: A mixture consisting of autologous bone and bone substitute in 1:1 ratio is packed into the defect. The membrane is pulled and fixed buccaly with 2 mini-screws.
View Figure 2
Surgical Technique
The procedure of GBR with a non-resorbable membranes is technically demanding and requires an excellent knowledge of the surgical technique. The use of a resorbable membrane is instead far more simple, quick and intuitive. Clinical steps are similar to those of previous technique. The following detailed description of GBR technique using non- resorbable membranes is explanatory as well for resorbable ones.
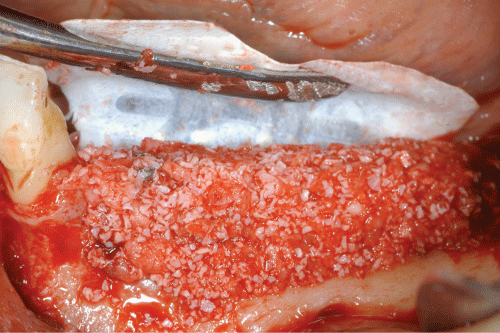
.
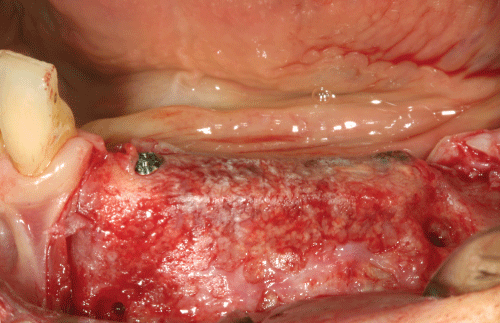
Figure 3: Membrane removal at 8 months. Buccal and Lingual full-thickness flaps are elevated to localize and remove the mini-srews.
View Figure 3
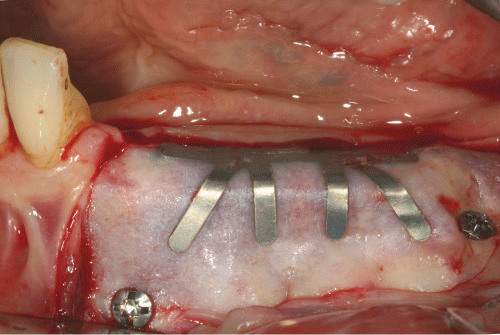
.
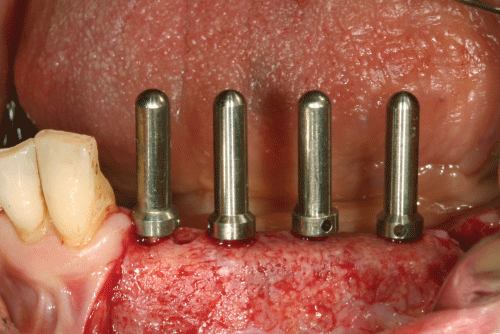
Figure 4: The underlying regenerated area with submerged clinical appearance of the tend mini-srews.
View Figure 4
Drug treatment
The antibiotic regimens recommended the intake of amoxicillin 750 mg + clavulanic acid 250 mg PO or Clindamycyn 600 mg PO, 2-3 times a day starting 1 day before surgery and continues for 6-7 days. Medications protocol includes 1 oral tab of ketoprofen 50 mg 1 hour before surgery and 1 infiltration of dexamethasone 4 mg after performing locoregional anesthesia. Few days before surgery is performed a session of oral hygiene and chlorhexidine 0.2% mouth wash rinses twice daily are prescribed until soft tissue closure (2 wks).
.
Figure 5: Prosthetic driven implants positioning in the regenerated ridge through a surgical guide.
View Figure 5
Flap design
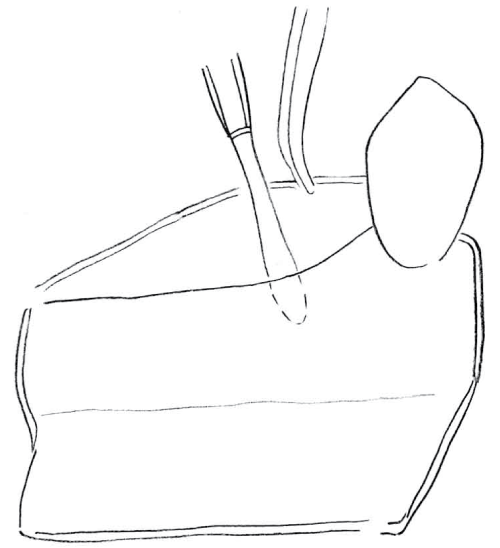
The meticulous recipient site preparation is one of the key points of the GBR. The surgery begins with full-thickness incision in the keratinized tissue of edentulous ridge. The buccal flap is then completed raised with two vertical incisions at mesial and distal end of the crestal incision (Figure 6). The buccal and palatal/lingual flaps accesses are raised to obtain a direct view of the defect. The bone surface is curetted to remove any residual connective and/or periosteum tissues. The buccal flap is then "released" to perform, at the end of the surgery, a suture without tension, a prerequisite essential to the success of the procedure. A continues periosteal incision, from mesial releasing incision to the distal, allows sliding flap several millimeters in coronal direction (Figure 7). If the procedure is performed in the lower, particularly care should be taken to avoid any damage to the mental nerve and vascular plexa of the floor of the mouth. The palatal flap is never released, while the lingual flap is mobilized and reflected beyond the mylohyoid insertion of the omohyoid in order to allow the coronal advancement of the flap (Figure 8). Care must be taken also to avoid soft tissue trauma and perforation that could lead to membrane exposure during the healing period.
.
Figure 8: The releasing incision on the lingual flap beyond the mylohyoid insertion.
View Figure 8
Membrane positioning
The membrane is shaped and trimmed to adapt to the residual alveolar ridge and to a predetermined volume to be augmented. It should not contact the periodontal tissue of the adjacent teeth and overlap the residual alveolar crest and the defect up to 3-4 mm and any interference during healing should be avoided. Support mini-screws ( 6 to 12 mm in length) are used to maintain vertical and horizontal space in cases of staged implant approach instead, in a one-stage approach, with residual height, at least of 6 mm, necessary for a primary stability, implants are left to protrude from cortical bone and acts as mini-screws supporting membrane. Later, the cortical plate is performed, with a fine bur, to ensure the bleeding necessary, from the marrow vascular spaces, to promote blood clot formation and provide mechanical interlocking of the graft. The e-PTFE membrane is first fixed to lingual or palatal plate by fixation mini-screws in the lower and/or tacks the upper. A particulate bone graft, at least 50% of autologous bone, is compacted into the bony defect and then covered with the membrane gently pulled buccally over the graft and adapted with same fixation devices at the mesial and distal buccal line angles to achieve optimal flap adaptation. Autologous bone normally is taken from mandibular ramus or body with bone scapers, piezoelectric devices or rarely with trephine burs (then particulated by a bone crusher).
Data from literature [64,70] suggest the use of a mixture consisting of autologous bone and bone substitute in 1:1 ratio. The rationale is to combine the osteogenetic and osteoinductive properties of the autograft with those osteoconductive of the bone substitute (eterologous, omologous, alloplastic) limiting the total bone quantity to be harvested resulting in minor trauma, stress, morbidity and complications to the patients.
Suturing
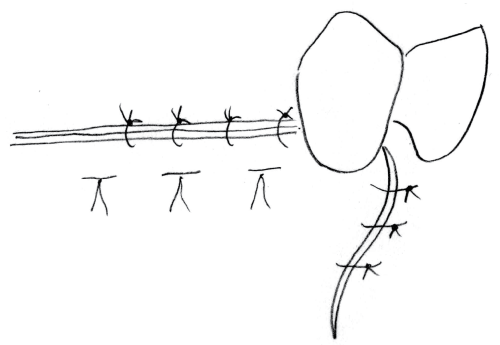
A correct suturing technique is essential for successful GBR surgery. First, sufficient flaps passivation and release are mandatory. They should overlap each other by at least 10 mm and beyond the grafted area by at least 5-6 mm for a free tension suture. Suturing consists in two different sutures: an internal horizontal mattress sutures to ensure proper flaps apposition (connective tissue surfaces facing each other by at least 3 mm) and single interrupted sutures between the horizontal mattress sutures for the vertical incisions (Figure 9). Sutures are removed 12-15 days following surgery. The use of an e-PTFE absorbable monofilament suture is recommended.
.
Figure 9: Suturing techique. Horizontal mattress suture and single interrupted sutures in between horizontal mattress sutures and on vertical incisions.
View Figure 9
Membrane removal
Membrane should remain in place and submerged for at least 6-9 months. After such time non- resorbable membrane can be removed to insert implants (staged approach) or healing abutments (one stage approach) by small crestal incision and 1 or 2 vertical incisions.
Conclusions
Guided Bone Regeneration GBR, with resorbable and non-resorbable membranes, is a well documented and literature supported technique. Resorbable membranes are widely used in clinical practice since their use lead to a minor mobility and surgical stress to the patient, low risk of exposure and complications and lowest cost. Non-resorbable membranes for their ability of space-making and the possibility to control the barrier effect over the time represent the choice materials for horizontal and vertical critical size defects. Adherence to proven surgical procedures protocols is a prerequisite for the successful use of these devices.
Some key factors for successful bone regeneration with membranes are identified as follow:
• Soft tissue healing by primary intention. Perfect closure of the wound is one of the basic requirements to avoid membrane exposure and therefore the failure of the regenerative procedure. This is achieved using a meticulous preparation of the recipient site and an appropriate suture technique. The buccal flap must be released, as described above, with the aim of increasing mobility and tension free suture.
• Membrane shaping and stabilization to the bone. Membrane must be adjusted to the surrounding bone tissue to prevent grafts invasions from non osteogenetic cells thus epithelial and connective cells. EPTFE membranes are stabilized with appropriate fixing devices, instead, resorbale membranes just lying on the grafted bone.
• Avoid membrane collapse. The creation and maintenance of adequate space are crucial for GBR. If not properly supported, membrane tends to collapse into the defect affecting the entire regeneration process. Some "space making" properties are missing with resorbable membranes, for this their use is indicated for contenitive defects and must be supported by bone graft. The non-resorbable membranes remain first choice devices for horizontal and vertical augmentation procedures with addition of bone grafts.
• Controlled healing time. A period of 6-9 months is considered sufficient to enhance bone formation and maturation. The resorbable membranes, compared to the non-resorbable ones, still have the important disadvantage of non controlled barrier effect over the time, so their use is limited to some defects.
• Adequate graft blood supply. Angiogenesis is mandatory for bone formation and maintenance. Cortical bone perforations around grafted area enhance bleeding and blood clot formation.
The authors state that the study presented was formed in accordance with the ethical standards established in the Declaration of Helsinki, and that the Informed consent was obtained from all participants prior to their enrollment in the study.
References
-
Branemark PI, Adell R, Albrektsson T, Lekholm U, Lundkvist S, et al. (1983) Osseointegrated titanium fixtures in the treatment of edentulousness. Biomaterials 4: 25-28.
-
Gottlow J, Nyman S, Karring T, Lindhe J (1984) New attachment formation as the result of controlled tissue regeneration. J Clin Periodontol 11: 494-503.
-
Karring T, Isidor F, Nyman S, Lindhe J (1985) New attachment formation on teeth with a reduced but healthy periodontal ligament. J Clin Periodontol 12: 51-60.
-
Hurley LA, Stinchfield FE, Bassett AL, Lyon WH (1959) The role of soft tissues in osteogenesis. An experimental study of canine spine fusions. J Bone Joint Surg Am 41A: 1243-1254.
-
Boyne PJ (1964) Regeneration of alveolar bone beneath cellulose acetate filter implants. J Dent Res 43: 827.
-
Boyne PJ, Mikels TE (1968) Restoration of alveolar ridges by intramandibular transposition osseous grafting. J Oral Surg 26: 569-576.
-
Boyne PJ (1969) Restoration of osseous defects in maxillofacial casualities. J Am Dent Assoc 78: 767-776.
-
Melcher AH (1976) On the repair potential of periodontal tissues. J Periodontol 47: 256-260.
-
Nyman S, Lindhe J, Karring T, Rylander H (1982) New attachment following surgical treatment of human periodontal disease. J Clin Periodontol 9: 290-296.
-
Nyman S, Lindhe J, Karring T (1989) Reattachment-new attachment. In: Lindhe J Textbook of Clinical Periodontology (2nd end) Copenhagen, Munksgaard, 450-476.
-
Dahlin C, Sennerby L, Lekholm U, Linde A, Nyman S (1989) Generation of new bone around titanium implants using a membrane technique: an experimental study in rabbits. Int J Oral Maxillofac Implants 4: 19-25.
-
Jovanovic SA, Nevins M (1995) Bone formation utilizing titanium-reinforced barrier membranes. Int J Periodontics Restorative Dent 15: 56-69.
-
Mellonig JT, Triplett RG (1993) Guided tissue regeneration and endosseous dental implants. Int J Periodontics Restorative Dent 13: 108-119.
-
Becker W, Becker BE (1990) Guided tissue regeneration for implants placed into extraction sockets and for implant dehiscences: surgical techniques and case report. Int J Periodontics Restorative Dent 10: 376-391.
-
Simion M, Trisi P, Piattelli A (1994) Vertical ridge augmentation using a membrane technique associated with osseointegrated implants. Int J Periodontics Restorative Dent 14: 496-511.
-
Amano Y, Ota M, Sekiguchi K, Shibukawa Y, Yamada S (2004) Evaluation of a poly-l-lactic acid membrane and membrane fixing pin for guided tissue regeneration on bone defects in dogs. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 97: 155-163.
-
Hammerle CH, Schmid J, Lang NP, Olah AJ (1995) Temporal dynamics of healing in rabbit cranial defects using guided bone regeneration. J Oral Maxillofac Surg 53: 167-174.
-
Schenk RK, Buser D, Hardwick WR, Dahlin C (1994) Healing pattern of bone regeneration in membrane-protected defects: a histologic study in the canine mandible. Int J Oral Maxillofac Implants 9: 13-29.
-
Scantlebury TV (1993) 1982-1992: a decade of technology development for guided tissue regeneration. J Periodontol 64: 1129-1137.
-
Aspenberg P, Goodman S, Toksvig-Larsen S, Ryd L, Albrektsson T (1992) Intermittent micromotion inhibits bone ingrowth. Titanium implants in rabbits. Acta Orthop Scand 63: 141-145.
-
Buser D, Dula K, Belser UC, Hirt HP, Berthold H (1995) Localized ridge augmentation using guided bone regeneration. II. Surgical procedure in the mandible. Int J Periodontics Restorative Dent 15: 10-29.
-
Buser D, Dula K, Lang NP, Nyman S (1996) Long-term stability of osseointegrated implants in bone regenerated with the membrane technique. 5-year results of a prospective study with 12 implants. Clin Oral Implants Res 7: 175-183.
-
Dahlin C, Gottlow J, Linde A, Nyman S (1990) Healing of maxillary and mandibular bone defects using a membrane technique. An experimental study in monkeys. Scand J Plast Reconstr Surg Hand Surg 24: 13-19.
-
Dahlin C, Lekholm U, Becker W, Becker B, Higuchi K, et al. (1995) Treatment of fenestration and dehiscence bone defects around oral implants using the guided tissue regeneration technique: a prospective multicenter study. Int J Oral Maxillofac Implants 10: 312-318.
-
Becker W, Dahlin C, Lekholm U, Bergstrom C, van Steenberghe D, et al. (1999) Five-year evaluation of implants placed at extraction and with dehiscences and fenestration defects augmented with ePTFE membranes: results from a prospective multicenter study. Clin Implant Dent Relat Res 1: 27-32.
-
Dahlin C, Andersson L, Linde A (1991) Bone augmentation at fenestrated implants by an osteopromotive membrane technique. A controlled clinical study. Clin Oral Implants Res 2: 159-165.
-
Jovanovic SA, Spiekermann H, Richter EJ (1992) Bone regeneration around titanium dental implants in dehisced defect sites: a clinical study. Int J Oral Maxillofac Implants 7: 233-245.
-
Becker W, Lynch SE, Lekholm U, Becker BE, Caffesse R, et al. (1992) A comparison of ePTFE membranes alone or in combination with platelet-derived growth factors and insuline-like growth factor-I or demineralized freeze-dried bone in promoting bone formation around immediate extraction socket implants. J Periodontol 63: 929-940.
-
Lazzara RJ (1989) Immediate implant placement into extraction sites: surgical and restorative advantages. Int J Periodontics Restorative Dent 9: 332-343.
-
Nyman S, Lang NP, Buser D, Bragger U (1990) Bone regeneration adjacent to titanium dental implants using guided tissue regeneration: a report of two cases. Int J Oral Maxillofac Implants 5: 9-14.
-
Nevins M, Mellonig JT (1992) Enhancement of the damaged edentulous ridge to receive dental implants: a combination of allograft and the GORE-TEX membrane. Int J Periodontics Restorative Dent 12: 96-111.
-
Tarnow DP, Wallace SS, Froum SJ, Rohrer MD, Cho SC (2000) Histologic and clinical comparison of bilateral sinus floor elevations with and without barrier membrane placement in 12 patients: Part 3 of an ongoing prospective study. Int J Periodontics Restorative Dent 20: 117-125.
-
Nowzari H, Slots J (1995) Microbiologic and clinical study of polytetrafluoroethylene membranes for guided bone regeneration around implants. Int J Oral Maxillofac Implants 10: 67-73.
-
Lee JY, Kim YK, Yun PY, Oh JS, Kim SG (2010) Guided bone regeneration using two types of non resorbable barrier membranes. J Korean Assoc Oral Maxillofac Surg 36: 275-279.
-
Hoffmann O, Bartee BK, Beaumont C, Kasaj A, Deli G, et al. (2008) Alveolar bone preservation in extraction sockets using non-resorbable dPTFE membranes: a retrospective non-randomized study. J Periodontol 79: 1355-1369.
-
Bartee BK, Carr JA (1995) Evaluation of a high-density polytetrafluoroethylene (n-PTFE) membrane as a barrier material to facilitate guided bone regeneration in the rat mandible. J Oral Implantol 21: 88-95.
-
Bartee BK (1995) The use of high-density polytetrafluoroethylene membrane to treat osseous defects: clinical reports. Implant Dent 4: 21-26.
-
Degidi M, Scarano A, Piattelli A (2003) Regeneration of the alveolar crest using titanium micromesh with autologous bone and a resorbable membrane. J Oral Implantol 29: 86-90.
-
Zitzmann NU, Naef R, Scharer P (1997) Resorbable versus nonresorbable membranes in combination with Bio-Oss for guided bone regeneration. Int J Oral Maxillofac Implants 12: 844-852.
-
Chiapasco M, Zaniboni M (2009) Clinical outcomes of GBR procedures to correct peri-implant dehiscences and fenestrations: a systematic review. Clin Oral Implants Res 20 Suppl 4: 113-123.
-
Imbronito AV, Todescan JH, Carvalho CV, Arana-Chavez VE (2002) Healing of alveolar bone in resorbable and non-resorbable membrane-protected defects. A histologic pilot study in dogs. Biomaterials 23: 4079-4086.
-
Simion M, Maglione M, Iamoni F, Scarano A, Piattelli A, et al. (1997) Bacterial penetration through Resolut membrane in vitro: an histologicsl and scanning electron microscopic study. Clin Oral Implants Res 8: 23-31.
-
Mayfield L, Nobreus N, Attstrom R, Linde A (1997) Guided bone regeneration in dental implant treatment using a bioabsorbable membrane. Clin Oral Implants Res 8: 10-17.
-
Lundgren D, Sennerby L, Falk H, Friberg B, Nyman S (1994) The use of a new bioresorbable barrier for guided bone regeneration in connection with implant installation. Case reports. Clin Oral Implants Res 5: 177-184.
-
Hurzeler MB, Quinones CR, Schupbach P (1997) Guided bone regeneration around dental implants in the atrophic alveolar ridge using a bioresorbable barrier. An experimental study in the monkey. Clin Oral Implants Res 8: 323-331.
-
Schwarz F, Rothamel D, Herten M, Wustefeld M, Sager M, et al. (2008) Immunohistochemical characterization of guided bone regeneration at a dehiscencetype defect using different barrier membranes: an experimental study in dogs. Clin Oral Implants Res 19: 402-415.
-
Rothamel D, Schwarz F, Sager M, Herten M, Sculean A, et al. (2005) Biodegradation of differently cross-linked collagen membranes: an experimental study in the rat. Clin Oral Implants Res 16: 369-378.
-
Bunyaratavej P, Wang HL (2001) Collagen membranes: a review. J Periodontol 72: 215-229.
-
Zahedi S, Legrand R, Brunel G, Albert A, Dewe W, et al. (1998) Evaluation of a diphenylphosphorylazide-crosslinked collagen membrane for guided bone regeneration in mandibular defects in rats. J Periodontol 69: 1238-1246.
-
Tal H, Kozlovsky A, Artzi Z, Nemcovsky CE, Moses O (2008) Long-term bio-degradation of cross-linked and non-cross-linked collagen barriers in human guided bone regeneration. Clin Oral Implants Res 19: 295-302.
-
Rothamel D, Schwarz F, Sculean A, Herten M, Scherbaum W, et al. (2004) Biocompatibility of various collagen membranes in cultures of human PDL fibroblasts and human osteoblast-like cells. Clin Oral Implants Res 15: 443-449.
-
Dimitriou R, Mataliotakis GI, Calori GM, Giannoudis PV (2012) The role of barrier membranes for guided bone regeneration and restoration of large bone defects: current experimental and clinical evidence. BMC Med 10: 81.
-
Steigmann M (2006) Pericardium membranes and xenograft particulate grafting material for horizontal alveolar ridge defects. Implant Dent 15: 186-191.
-
Sterio TW, Katancik JA, Blanchard SB, Xenoudi P, Mealey BL (2013) A prospective multicenter study of bovine pericardium membrane with cancellous particulate allograft for localized alveolar ridge augmentation. Int J Periodontics Restorative Dent 33: 499-507.
-
Stavropoulos A, Chiantella G, Costa D, Steigmann M, Windish P, et al. (2011) Clinical and Histologic evaluation of a granular bovine bone material used as an adjunct to GTR with a bioresorbable bovine pericardium collagen membrane in the treatment of intrabony defects. J Periodontol 82: 462-470.
-
Ahn YS, Kim SG, Kim CS, Oh JS, Lim SC (2012) Effect of guided bone regeneration with or without pericardium bioabsorbable membrane on bone formation. Oral Surg Oral Med Oral Pathol Oral Radiol 114: S126-131.
-
Bai M, Zhang T, Ling T, Zhou Z, Xie H, et al. (2014) Guided bone regeneration using acellular bovine pericardium in a rabbit mandibular model: in vitro and in vivo studies. J Periodontal Res 49: 499-507.
-
Rothamel D, Schwarz F, Fienitz T, Smeets R, Dreiseidler T, et al. (2012) Biocompatibility and biodegradation of a native porcine pericardium membrane: results of in vitro and in vivo examinations. Int J Oral Maxillofac Implants 27: 146-154.
-
Merli M, Moscatelli M, Mazzoni A, Merli M, Mariotti G, et al. (2013) Lateral bone augmentation applying different biomaterials. A clinical and histological evaluation of a case report. Z Zahnarztl Implantol 29: 70-79
-
Jimbo R, Marin C, Witek L, Suzuki M, Tovar N, et al. (2012) Bone morphometric evaluation around immediately placed implants covered with porcine-derived pericardium membrane: an experimental study in dogs. International J of Biomaterials.
-
Thomaidis V, Kazakos K, Lyras DN, Dimitrakopoulos I, Lazaridis N, et al. (2008) Comparative study of 5 different membranes for guided bone regeneration of rabbit mandibular defects beyond critical size. Med Sci Monit 14: 67-73.
-
Festa VM, Addabbo F, Laino L, Femiano F, Rullo R (2013) Porcine-Derived Xenograft Combined with a soft cortical membrane versus extractiom alone for implant site development: a clinical study in humans. Clin Implant dent Relat Res 15: 707-713.
-
Wachtel H, Fickl S, Hinze M, Bolz W, Thalmair T (2013) The bone lamina technique: a novel approach for lateral ridge augmentation--a case series. Int J Periodontics Restorative Dent 33: 491-497.
-
McAllister BS, Haghighat K (2007) Bone augmentation techniques. J Periodontol 78: 377-396.
-
Garg A (2011) Barrier membranes--materials review, Part I of II. Dent Implantol Update 22: 61-64.
-
Hammerle CH, Jung RE (2003) Bone augmentation by means of barrier membranes. Periodontol 2000 33: 36-53.
-
Hurzeler MB, Weng D (1996) A new technique to combine barrier removal at dehisced implant sites with a plastic periodontal procedure. Int J Periodontics Restorative Dent 16: 148-163.
-
Simion M, Misitano U, Gionso L, Salvato A (1997) Treatment of dehiscences and fenestrations around dental implants using resorbable and nonresorbable membranes associated with bone autografts: a comparative clinical study. Int J Oral Maxillofac Implants 12: 159-167.
-
Ito K, Nanba K, Murai S (1998) Effects of bioabsorbable and non-resorbable barrier membranes on bone augmentation in rabbit calvaria. J Periodontol 69: 1229-1237.
-
Dahlin C, Andersson L, Linde A (1991) Bone augmentation at fenestrated implants by an osteopromotive membrane technique. A controlled clinical study. Clin Oral Implants Res 2: 159-165.
-
Simion M, Jovanovic SA, Tinti C, Benfenati SP (2001) Long-term evaluation of osseointegrated implants inserted at the time or after vertical ridge augmentation. A retrospective study on 123 implants with 1-5 year follow-up. Clin Oral Implants Res 12: 35-45.
-
Fontana F, Santoro F, Maiorana C, Iezzi G, Piattelli A, et al. (2008) Clinical and histologic evaluation of allogeneic bone matrix versus autogenous bone chips associated with titaniumreinforced e-PTFE membrane for vertical ridge augmentation:a prospective pilot study. Int J Oral Maxillofac Implants 23: 1003-1012.
-
Simion M, Fontana F, Rasperini G, Maiorana C (2007) Vertical ridge augmentation by expanded polytetrafluoroethylene membrane and a combination of intraoral autogenous bone graft and deproteinized anorganic bovine bone (Bio Oss). Clin Oral Implants Res 18: 620-629.
-
Scarano A, Iezzi G, Quaranta A, Fiera E, Piattelli A (2006) I biomateriali in chirurgia odontostomatologica. Italian Oral Surgery 1: 11-23.