Obstetrics and Gynaecology Cases - Reviews
Growing Teratoma Syndrome Concurrent with Pregnancy: A Case Report and Literature Review
Aki Tanaka-Murakami*, Masakazu Abe, Yuka Kasamatsu, Nobuhiro Kado, Shiho Kuji, Nobutaka Takahashi, Munetaka Takekuma and Yasuyuki Hirashima
Division of Gynecology, Shizuoka Cancer Center Hospital, Japan
*Corresponding author: Aki Tanaka-Murakami, MD, PhD, Division of Gynecology, Shizuoka Cancer Center Hospital, 1007 Shimonagakubo, Nagaizumi-cho, Sunto-gun, Shizuoka 411-8777, Japan, Tel: 81-55-989-5222, Fax: 81-55-989-5783, E-mail: ak.tanaka@scchr.jp
Obstet Gynecol Cases Rev, OGCR-2-067, (Volume 2, Issue 8), Case Report and Literature Review; ISSN: 2377-9004
Received: November 05, 2015 | Accepted: November 27, 2015 | Published: November 30, 2015
Citation: Tanaka-Murakami A, Abe M, Kasamatsu Y, Kado N, Kuji S, et al. (2015) Growing Teratoma Syndrome Concurrent with Pregnancy: A Case Report and Literature Review. Obstet Gynecol Cases Rev 2:067. 10.23937/2377-9004/1410067
Copyright: © 2015 Tanaka-Murakami A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Growing teratoma syndrome (GTS) is a rare disease characterized by the appearance of benign metastatic lesions during or after chemotherapy for germ cell tumor. Complete surgical resection is important for the diagnosis and cure of GTS. We encountered a patient with a suspected GTS during her second pregnancy and childbirth; 4 years later, radical surgery confirmed the diagnosis. A tumor that was suspected to be GTS was observed in a 31 year old 2 para woman immediately after the completion of four cycles of bleomycin, etoposide, and cisplatin therapy after fertility-sparing surgery for stage IIIb ovarian immature teratoma. In accordance with the directives of the patient, the recommended radical surgery: total abdominal hysterectomy, left salpingo-oophorectomy, resection of the peritoneal dissemination, extended right hepatectomy, and lower anterior resection of the rectum, was performed after childbirth. Because ovarian germ cell tumors are characterized by young onset, fertility-sparing surgery is performed in most cases; in addition, the timing of tumor recurrence, onset of GTS, and pregnancy/childbirth may coincide. Total resection of the tumor affects the prognosis of GTS; therefore, minimally invasive surgery before tumor growth is advisable. For cases with concomitant pregnancy, simultaneous delivery and radical surgery may be possible with the cooperation of the gynecologic oncologist, obstetrician, and surgeon.
Key words
Growing teratoma syndrome, Ovary, Fertility-sparing surgery, Pregnancy
Introduction
Growing teratoma syndrome (GTS) is a rare disease characterized by the appearance of benign metastatic lesions during or after chemotherapy for germ cell tumor. Regardless of the source of metastasis, the level of tumor markers remains controlled after treatment, and the pathologic findings of the metastatic lesions show mature teratomas without malignant components. In 1982, Logothetis reported six cases that arose from germ cell malignancies (five testicular tumors and one ovarian tumor) and proposed the concept of GTS [1].
Among gynecological tumors, DiSaia et al. first reported cases that originated from an immature teratoma of the ovaries; they called this phenomenon chemotherapeutic retroconversion [2]. Amsalem et al. ultimately concluded that chemotherapeutic retroconversion and GTS are the same condition [3]. GTS originating from the ovaries most commonly presents with peritoneal dissemination similar to that in ovarian cancer [4]. Although the etiology of GTS has not been established, the two most cited theories are 1) chemotherapy destroys the immature components and only the mature components remain and 2) chemotherapy changes the cell kinetics from pluripotent malignant germ cells to mature benign teratomas [2,5,6].
In patients with suspected GTS, it is important to rule out the recurrence of malignant components, for which total tumor resection is required, for preventing growth and malignant transformation [7]. The reported rate of germ cell malignancy in ovarian tumors was low at approximately 4%, GTS in the ovarian tumor was reported a total of about 50 cases in Japan and overseas [4]. In a review of GTS cases, among which 7% were immature teratomas, the median age at onset of ovarian germ cell malignancy was 21 years (range, 5-38 years); the median period from the completion of postoperative chemotherapy to GTS onset was 8 months (range, 3-156 months) [8]. Therefore, in the timing of surgery for germ cell malignancy and GTS, the issue of fertility preservation arises. Cases of pregnancy and childbirth during remission after surgery for GTS have been reported [4,9]; however, no cases of pregnancy and delivery with concurrent GTS have been reported.
We encountered a patient with a stage 3b immature teratoma, who developed peritoneal dissemination immediately after adjuvant chemotherapy; GTS was strongly suspected. After the second pregnancy and childbirth, the disseminated lesions were completely resected 4 years after onset, and GTS was confirmed.
Case
The patient was a 30-year-old woman, gravida 2 para 2, with no pertinent medical or family history. Upon consultation with a local physician for a chief complaint of lower abdominal pain, a right ovarian tumor was diagnosed, and the patient was referred to our hospital.
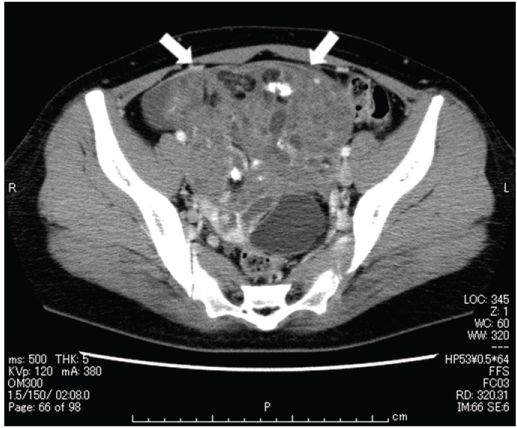
Internal examination and ultrasound imaging revealed a 16 cm solid tumor with good mobility in the right ovary. Computed tomography (CT) scan revealed fatty components and coarse calcification within the tumor, with the contrast enhancement of the solid part. The initial suspicion was of a malignant tumor, but there were no clear findings of metastasis (Figure 1). Magnetic resonance imaging (MRI) with T1-weighted imaging revealed a small, dotted, hyperintense signal that represented the fatty components of the tumor; for the solid part, isointense signals were seen on T1-weighted imaging, whereas hyperintense signals with contrast effect were seen on T2-weighted imaging. Measurement of serum tumor markers revealed the following levels: cancer antigen (CA)-125 at 120 U/mL, alpha-fetoprotein (AFP) at 329 ng/mL, and squamous cell carcinoma antigen (SCC) at 2.0 ng/mL. On the basis of these results, stage Ia immature teratoma of the right ovary was diagnosed, and surgery was scheduled.
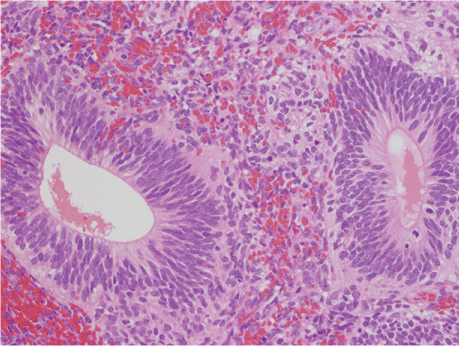
Upon making an incision into the abdomen, the right ovary was noted to have an infant's headsize; the capsule was ruptured and disseminated nodules of approximately 7 mm in size were observed in the greater omentum. The patient opted for fertility-sparing surgery; therefore, only the right adnexa and the affected greater omentum were resected. Pathological examination of the right ovarian tumor revealed mature components with the growth of glial tissue, choroid membrane tissue, bone, cartilage, tooth buds, colonic mucous membrane, ciliated columnar epithelium, and hair follicle. The mature components were mixed with immature components, including neural tube structures. Therefore, the pathological diagnosis was grade 1 immature teratoma. On the greater omentum, numerous disseminated lesions of varying sizes were found; there were immature components and implants that had differentiated into mature brain tissue (Figure 2). Peritoneal lavage cytology results were falsely positive, suspected the tumor components leakage. Therefore, advanced stage IIIb (pT3bNxM0) was diagnosed after surgery.
.
Figure 2: Histological feature of primary tumors.
Histological examination revealed mature components mixed with immature components such as neural tube structure. HE × 20
View Figure 2
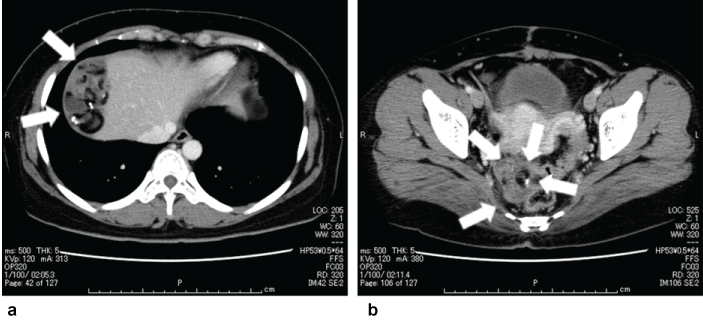
Following surgery, four cycles of BEP therapy (bleomycin 20 mg/m2 on days 2, 9, 16; etoposide 70 mg/m2 on days 1-5; and cisplatin 20 mg/m2 on days 1-5) were administered. After the completion of chemotherapy, a repeat CT scan revealed a new tumor with soft tissue calcification and fatty components in the right subphrenic space, paracolic gutter, and pouch of Douglas; recurrent peritoneal dissemination was suspected (Figure 3). Positron emission tomography (PET)-CT was inconclusive of a malignant etiology because of faint Fluorodeoxyglucose (18F; FDG) accumulation. Tumor markers that were elevated prior to treatment were within the normal range at 9 U/mL for CA-125, 3.2 ng/mL for AFP, and 1.0 ng/mL for SCC. On the basis of previous history, completion of chemotherapy and CT scan results, the level of tumor markers, GTS was strongly suspected. We recommended radical surgery, including total hysterectomy and left salpingo-oophorectomy, for definitive diagnosis and treatment.
.
Figure 3: CT findings of two sites of recurrent tumors after the adjuvant chemotherapy. The recurrent tumors are indicated by white arrows. (a) Right subphrenic space (b) Pouch of Douglas
View Figure 3
However, the patient strongly refused to undergo radical surgery. Thereafter, she was lost to follow-up and subsequently gave birth to her second child by caesarean section at another hospital.
.
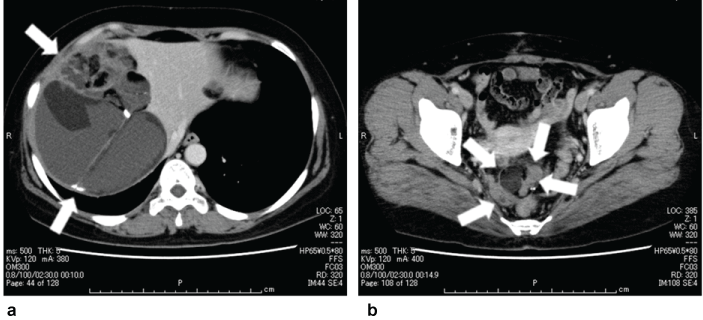
Figure 4: CT findings of two sites of recurrent tumors before the radical total resection.The recurrent tumors are indicated by white arrows. (a) Right subphrenic space (b) Pouch of Douglas
View Figure 4
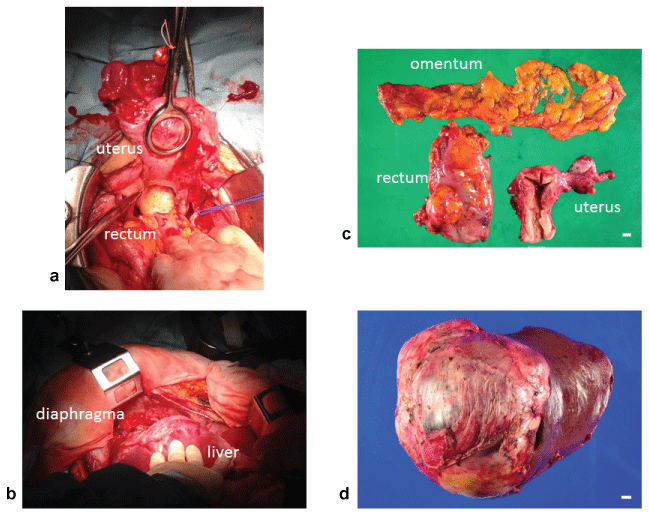
Four years and five months after the appearance of peritoneal dissemination, and following the delivery of her fourth child, the patient finally decided to undergo radical surgery and came to our hospital. CT scan at this time revealed the enlargement of all existing peritoneal dissemination, but there were no new lesions (Figure 4). She then underwent total abdominal hysterectomy, left salpingo-oophorectomy, resection of the peritoneal dissemination, extended right hepatectomy, and lower anterior resection of the rectum, with the complete removal of the lesions (Figure 5). The disseminated lesions between the right hepatic lobe and the diaphragm had a clear border, but strongly adhered to the diaphragm and invaded the liver. The adhesions were difficult to separate, the partial diaphragm and right hepatic lobes were resected to perform the total resection of the tumor. The total duration of the surgery was 13 h and 38 min, including 7 h of resection of the subdiaphragmatic dissemination; the total blood loss was 1,311 mL. The pathological diagnosis was mature teratoma without malignant findings, thus confirming the initial suspicion of GTS (Figure 6). One year and six months later, there were no signs of recurrence.
.
Figure 5: Laparotomy findings and operatively extracted specimens. (a) Pouch of Douglas (b) Right subphrenic space: separating form diaphragm (c) Uterus, left adnexa, greater omentum and dissemination on the rectum surface (d) Right lobe of the liver [c,d: scar bar 1 cm]
View Figure 5
.
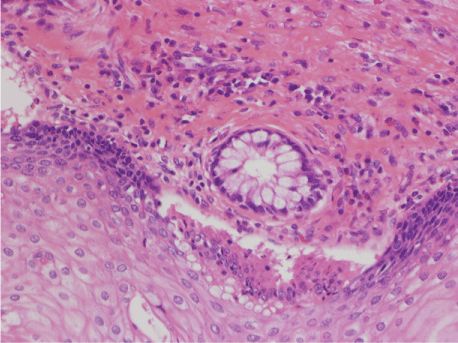
Figure 6: Histological feature of GTS tumors.
Mature teratoma without malignant findings composed of squamous epithelium and cilliated epithelial cell including goblet cell, cartilage tissue. HE × 20
View Figure 6
Discussion
The following two points are suggested during the review of this case.
Firstly, to the best of our knowledge, this is the first reported case in which a pregnant patient gave birth without perinatal complications despite concurrent GTS; in addition, she underwent complete tumor resection after childbirth. In the present case, the patient had her second pregnancy and delivery with concurrent GTS. Although cases of pregnancy and delivery during remission after GTS surgery have been reported [4,9], there are no reports of pregnancy and delivery with concurrent GTS. In some cases, GTS was managed by tumor resection alone and fertility was preserved [4]; on the other hand, some reports described that recurrence and malignant transformation are possible in GTS [4,10]. Therefore, as a general rule, radical total resection including internal genital organs resection in one surgical procedure is recommended for complete recovery [7,11]. However, germ cell malignancies often occur in young adults, and the usual initial choice is fertility-sparing surgery. When there is a high probability of GTS in patients who desire to preserve fertility, radical surgery may be delayed with caution until after pregnancy and childbirth, provided that the patient is stable and that she fully understands the risks.
Secondly, the observation period in this patient lasted for more than 4 years before radical resection. Although the tumor grew to some extent, there was no rapid progression or the appearance of new lesions. The degree of tumor growth differs according to each individual case [7], and there have been some cases in which the tumor did not change in size over extended periods of time [12]. With regard to tumor growth, the median growth rate in terms of tumor circumference was reported to be 0.7 cm per month; the median growth rate in terms of tumor volume was at 12.9 mL per month [7]. In the present case, the tumor grew slowly at a rate of 0.18 cm per month to approximately 111% of the size at the time of initial diagnosis. We hypothesize that this relatively slow and small tumor growth made a second pregnancy and childbirth possible in this patient. In patients who refuse radical surgery for fertility preservation, it may be important to thoroughly assess the extent to which the growth is predicted.
Andre et al. proposed three predictive factors of GTS onset, including 1) the presence of mature teratoma components in the primary germ celltumor; 2) incomplete resection during the initial surgery; and 3) the presence of metastatic lesions that are unresponsive to chemotherapy [13]. It has been reported that among patients with GTS arising from a primary ovarian lesion, patients with teratoma components in the primary lesion accounted for 62.5% [14]. Likewise, in the present case, mature teratoma components were present in the primary lesion, placing the patient at risk for GTS onset. Serial imaging evaluations have been recommended after every two courses of chemotherapy to prevent delayed diagnosis of GTS [7,13]. In the present case, if the CT scan had been performed after the second course of chemotherapy, we may have been able to detect the lesions when they were smaller and resectable by less invasive surgical methods. GTS is pathologically benign; however, unlike mature cystic teratomas of the ovaries, these lesions often have strong adhesions [15] that may require additional invasive surgical procedures, as demonstrated in the present case.
Chemotherapy and radiotherapy are considered to be beneficial adjuvant therapies for germ cell tumors [1,2,14]. However, their efficacy for GTS has not been reported, and surgery remains the gold standard for treatment [1,2,14,16]. To prevent regrowth or malignant transformation of the residual tumor after incomplete resection, it may be ideal for surgeons to aim for complete resection, accurate diagnosis, and appropriate surgical timing in a single procedure. In addition, thorough preparation with cooperation among the concerned members of the surgical team is important. Consensus on the timing of radical surgery for GTS has not been reached. The required surgical procedure becomes more invasive as a tumor grows; in corollary, tumor growth can make complete resection difficult. Therefore, it is advisable to perform surgery early [7,13]. Pressure symptoms and the invasion into adjacent organs caused by tumor growth may exacerbate the general condition and affect the ability of a patient to tolerate surgery. Although histologically benign, the tumor growth of GTS has been reported to cause deep vein thrombosis, pulmonary embolism, renal failure due to ureteral obstruction, intestinal necrosis due to mesenteric artery compression, and biliary obstruction, some of which are fatal [4,13]. On the other hand, if surgery is performed too early, benign lesions that are not grossly visible may be overlooked. Most GTS arising from ovarian germ cell tumors present with intraperitoneal dissemination that is similar to that in ovarian cancer [4]. However, unlike surgery for ovarian cancer, chemotherapy has uncertain effects for GTS, and total resection is required to achieve complete recovery. To achieve complete resection in one surgery, unless there is rapid growth or an emergency situation, imaging should be evaluated at regular intervals to ascertain all the specific sites of dissemination that need to be removed.
The incidence of GTS recurrence was reported to be 50%-83% after incomplete resection and 0%-4% after complete resection [7,13,14]. On the other hand, it has also been reported that the incidence of recurrence after the complete resection of GTS originating in the ovaries was 32% [4]. Furthermore, approximately 3%-5% of GTS lesions may undergo malignant transformation into sarcoma, squamous cell carcinoma, adenocarcinoma, or carcinoids [13,17,18].
In recent years, the usefulness of laparoscopic surgery for early-stage patients has been reported [9,18,19]. In fact, other reports have described the effectiveness of intraperitoneal endoscopic observation in detecting micro lesion [20,21]. It has also been reported that laparoscopy may be useful to avoid the risk of infertility [22]. However, the surgical procedure that is best for patients who wish to preserve fertility has not been established. Bentivegna et al. reported that of 38 GTS patients among 196 patients with immature teratoma of the ovaries, fertility-sparing surgery was performed in 20 patients and 5 patients were pregnant after the operation [4]. The surgical procedure for GTS should be similar to the tumor-debulking surgery for ovarian cancer. It has been suggested that according to the choice of the patient, fertility-sparing surgery may be performed as long as there are no apparent lesions in the uterus and ovaries. In patients who choose to undergo fertility-sparing surgery, regular ultrasonography, CT, and MRI are recommended for the early diagnosis of recurrence, which may be resected by minimally invasive methods.
For the diagnosis of GTS, the following three criteria should be fulfilled: 1) the appearance or growth of metastatic lesions during or after chemotherapy for germ cell tumor; 2) the normalization of tumor markers (i.e., AFP, human chorionic gonadotropin) that were initially elevated prior to treatment; and 3) the histopathology findings of mature teratoma without malignant cells on the surgically resected specimen [23]. Definitive diagnosis requires the pathological diagnosis of resected organs, but diagnostic imaging to predict GTS or the recurrence of malignant components is required prior to surgery. Although there are no imaging findings that clearly distinguish GTS from malignant recurrence, GTS has been characteristically depicted on CT as a high-density tumor with partial cystic changes, including calcification and fatty components, surrounded by a well-defined border and adjacent tissue but with no invasion [14,24,25]. In general, PET scanning is useful to distinguish benign tumors. However, it was thought to be of little use in differentiating GTS from the recurrence of malignant components [8,16]. Hariprasad et al. compared the PET and pathology findings of resected GTS specimens and reported that the area with FDG uptake on PET corresponded with benign brain tissue components, which have high glucose metabolism. Therefore, differentiating between GTS and the recurrence of malignant components on PET is considered difficult [26]. Nevertheless, PET may be useful for the detection of dissemination and the evaluation of any residual tumor [8,16,27,28]. In the present case, the faint FDG accumulation might be affected the presence of benign brain tissues. Although pathological examination of the completely resected tumor is required for definitive diagnosis, radiological imaging and tumor markers may provide clues on the most likely etiology.
There have been no effective treatment methods established for unresectable GTS. Some have reported that interferon [29-31], bevacizumab [32], and all-trans retinoic acid [33] exhibited some inhibitory effects on disease progression; however, the effect of these drugs was limited. Therefore, it is important to perform complete surgical resection.
The prognosis of completely resected GTS is considered to be good. Two large studies reported a 5-year survival rate of 89%-90% and a disease-free survival rate of 73% [7,13]. However, the late recurrence of GTS has also been reported. Bentivegna et al. reported that after a median observation period of 73 months (range, 3-263 months), 26% (10 cases) had initial recurrence after a median duration of 45 months (range, 2-132 months) [4]. The mean duration of GTS onset was reported to be 8 months; in prolonged cases, it extended to 8 years [34]. For cases at risk for recurrence, careful follow-up observation should be performed for 10 years after the disease onset [19,25,34,35]. In the present case, complete resection was successfully achieved by surgery after two childbirths within a span of 4 years; nevertheless, continued follow-up observation will be required for a long period.
Conclusion
Peritoneal dissemination with suspected GTS was diagnosed following first line therapy for stage IIIb immature teratoma. Although it is not recommended to go through pregnancy and childbirth with concurrent GTS, careful observation and delayed radical surgery may be possible for patients who choose to preserve fertility. However, we emphasize that the management of pregnant women with GTS requires a multidisciplinary team approach including gynecologic oncologists, obstetricians, and neonatologists in an institution that is capable of handling threatened premature labor and delivery that may be caused by the tumor growth during pregnancy. Furthermore, although GTS is benign, growth and invasion would make tumor resection extremely difficult; therefore, thorough examination and cooperation among surgeons is important.
References
-
Logothetis CJ, Samuels ML, Trindade A, Johnson DE (1982) The growing teratoma syndrome. Cancer 50: 1629-1635.
-
Di Saia PJ, Saltz A, Kagan AR, Morrow CP (1977) Chemotherapeutic retroconversion of immature teratoma of the ovary. below Obstet Gynecol 49: 346-350.
-
Amsalem H, Nadjari M, Prus D, Hiller N, Benshushan A (2004) Growing teratoma syndrome vs chemotherapeutic retroconversion: case report and review of the literature. Gynecol oncol 92: 357-360.
-
Bentivegna E, Azais H, Uzan C, Leary A, Pautier P, et al. (2015) Surgical Outcomes After Debulking Surgery for Intraabdominal Ovarian Growing Teratoma Syndrome: Analysis of 38 Cases. Ann surg oncol.
-
Merrin C, Baumgartner G, Wajsman Z (1975) Letter: Benign transformation of testicular carcinoma by chemotherapy. Lancet 1: 43-44.
-
Carr BI, Gilchrist KW, Carbone PP (1981) The variable transformation in metastases from testicular germ cell tumors: the need for selective biopsy. J Urol 126: 52-54.
-
Spiess PE, Kassouf W, Brown GA, Kamat AM, Liu P, et al. (2007) Surgical management of growing teratoma syndrome: the M. D. Anderson cancer center experience. J Urol 177: 1330-1334.
-
Kikawa S, Todo Y, Minobe S, Yamashiro K, Kato H, et al. (2011) Growing teratoma syndrome of the ovary: a case report with FDG-PET findings. J Obstet Gynaecol Res 37: 926-932.
-
Tzortzatos G, Sioutas A, Schedvins K (2009) Successful pregnancy after treatment for ovarian malignant teratoma with growing teratoma syndrome. Fertil Steril 91: 936.
-
Glass T, Cochrane DD, Rassekh SR, Goddard K, Hukin J. (2014) Growing teratoma syndrome in intracranial non-germinomatous germ cell tumors (iNGGCTs): a risk for secondary malignant transformation-a report of two cases. Child's nerv syst 30: 953-957.
-
Homs MY, Schreuder HW, Jonges GN, Witteveen PO (2013) Clinical and radiologic signs of relapsed ovarian germ cell tumor: tissue is the issue. Case Rep Obstet Gynecol.
-
Jeffery GM, Theaker JM, Lee AH, Blaquiere RM, Smart CJ, et al. (1991) The growing teratoma syndrome. Br J Urol 67: 195-202.
-
Andre F, Fizazi K, Culine S, Droz J, Taupin P, et al. (2000) The growing teratoma syndrome: results of therapy and long-term follow-up of 33 patients. Eur J Cancer 36: 1389-1394.
-
Tangjitgamol S, Manusirivithaya S, Leelahakorn S, Thawaramara T, Suekwatana P, et al. (2006) The growing teratoma syndrome: a case report and a review of the literature. Int J Gynecol Cancer Suppl 1: 384-390.
-
De Cuypere M, Martinez A, Kridelka F, Balague G, Maisongrosse V, et al. (2014) Disseminated ovarian Growing Teratoma Syndrome: a case -report highlighting surgical safety issues. Facts Views Vis ObGyn 6: 250-253.
-
Aide N, Comoz F, Sevin E (2007) Enlarging residual mass after treatment of a nonseminomatous germ cell tumor: growing teratoma syndrome or cancer recurrence? J Clin Oncol 25: 4494-4496.
-
Itani Y, Kawa M, Toyoda S, Yamagami K, Hiraoka K (2002) Growing teratoma syndrome after chemotherapy for a mixed germ cell tumor of the ovary. J Obstet Gynaecol Res 28: 166-171.
-
Shigeta N, Kobayashi E, Sawada K, Ueda Y, Yoshino K, et al. (2015) Laparoscopic excisional surgery for growing teratoma syndrome of the ovary: case report and literature review. J Minim Invasive Gynecol 22: 668-674.
-
Matsushita H, Arai K, Fukase M, Takayanagi T, Ikarashi H (2010) Growing teratoma syndrome of the ovary after fertility-sparing surgery and successful pregnancy. Gynecol Obstet Iinvest 61: 221-223.
-
Tasuku Mariya AN, Kanae Numata, Akari Kawamata, Takehito Nihei, Tsuyoshi Saito. Laparoscopic Resection of Growing Teratoma Syndrome Post Chemotherapy for an Immature Ovarian Terato (1):158-62.
-
Terumi Morita SM, Hirofumi Koike, Yukio Hirano, Yukiyoshi Sakaguchi, Takehumi Niguma (2014) A case of growing teratoma syndrome: laparoscopic resection of primary immature teratoma. Modern trends in obstetrics & gynecology 63: 95-99.
-
Practice Committee of the American Society for Reproductive Medicine in collaboration with the Society ofReproductive Surgeons (2013) Pathogenesis, consequences, and control of peritoneal adhesions in gynecologic surgery: a committee opinion. Fertil Steril 99: 1550-1555.
-
Umekawa T, Tabata T, Tanida K, Yoshimura K, Sagawa N (2005) Growing teratoma syndrome as an unusual cause of gliomatosis peritonei: a case report. Gynecol Oncol 99: 761-763.
-
Tongaonkar HB, Deshmane VH, Dalal AV, Kulkarni JN, Kamat MR (1994) Growing teratoma syndrome. J Surg Oncol 55: 56-60.
-
Nimkin K, Gupta P, McCauley R, Gilchrist BF, Lessin MS (2004) The growing teratoma syndrome. Pediatr Radiol 34: 259-262.
-
Hariprasad R, Kumar L, Janga D, Kumar S, Vijayaraghavan M (2008) Growing teratoma syndrome of ovary. Int J Clin Oncol 13: 83-87.
-
Han NY, Sung DJ, Park BJ, Kim MJ, Cho SB, et al. (2014) Imaging features of growing teratoma syndrome following a malignant ovarian germ cell tumor. J Comput Assist Tomogr 38: 551-557.
-
Oechsle K, Hartmann M, Brenner W, Venz S, Weissbach L, et al. (2008) [18F]Fluorodeoxyglucose positron emission tomography in nonseminomatous germ cell tumors after chemotherapy: the German multicenter positron emission tomography study group. J Clin Oncol 26: 5930-5935.
-
Rustin GJ, Kaye SB, Williams CJ, Newlands ES, Bagshawe KD, et al. (1984) Response of differentiated but not anaplastic teratoma to interferon. Br J Cancer 50: 611-616.
-
Kattan J, Droz JP, Culine S, Duvillard P, Thiellet A, et al. (1993) The growing teratoma syndrome: a woman with nonseminomatous germ cell tumor of the ovary. Gynecol Oncol 49: 395-399.
-
Tonkin KS, Rustin GJ, Wignall B, Paradinas F, Bennett M (1989) Successful treatment of patients in whom germ cell tumour masses enlarged on chemotherapy while their serum tumour markers decreased. Eur J Cancer Clin Oncol 25: 1739-1743.
-
Mego M, Reckova M, Sycova-Mila Z, Obertova J, Brozmanova K, et al. (2007) Bevacizumab in a growing teratoma syndrome. Case report. Ann Oncol 18: 962-963.
-
Veenstra CM, Vaughn DJ (2011) Third-line chemotherapy and novel agents for metastatic germ cell tumors. Hematol Oncol Clin North Am 25: 577-591, ix.
-
Djordjevic B, Euscher ED, Malpica A (2007) Growing teratoma syndrome of the ovary: review of literature and first report of a carcinoid tumor arising in a growing teratoma of the ovary. Am J Surg Pathol 31: 1913-1918.
-
Caldas C, Sitzmann J, Trimble CL, McGuire WP, 3rd (1992) Synchronous mature teratomas of the ovary and liver: a case presenting 11 years following chemotherapy for immature teratoma. Gynecol Oncol 47: 385-390.