International Journal of Allergy Medications
Valsartan/Hydrochlorothiazide Induced Pemphigus Foliaceus
Rosa Giménez-García* and MariaNuñez-Cabezón
Hospital Rio Hortega, Valladolid, Spain
*Corresponding author:
Dra Rosa Giménez- García, Calle Carabela 115, Boecillo, 47151 Valladolid, Spain, E-mail: rosagim@hotmail.com
Int J Aller Medcations, IJAM-2-012, (Volume 2, Issue 1), Case Report
Received: December 31, 2015: Accepted: March 25, 2016: Published: March 29, 2016
Citation: Giménez-García R, Nuñez-Cabezón M (2016) Valsartan/Hydrochlorothiazide Induced Pemphigus Foliaceus. Int J Aller Medications 2:012
Copyright: © 2016 Giménez-García R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The term pemphigus refers to a group of autoimmune intraepidermal blistering diseases of skin and mucous membranes in which there is loss of adhesion between keratinocytes, following disruption of desmosomes, as a consequence of autoantibody binding to various antigens on the cell surface. Drug-induced pemphigus mimics idiopathic pemphigus clinically, histopathologically and immunologically. Exogenous factors as burns, UV radiation and X-rays can be involved in the development of pemphigus. Other factors as drug intake (thiol drugs or SH drugs, phenol drugs and nonthiolnonphenol drugs) are involved in inducing or triggering pemphigus outbreak, frequently resembling pemphigus foliaceus (PF) [1-4]. Antihypertensives have been reported to cause pemphigus vulgaris or pemphigus foliaceus [5]. We report a case of valsartan/hydrochlorothiazide- induced pemphigus foliaceus.
Case Report
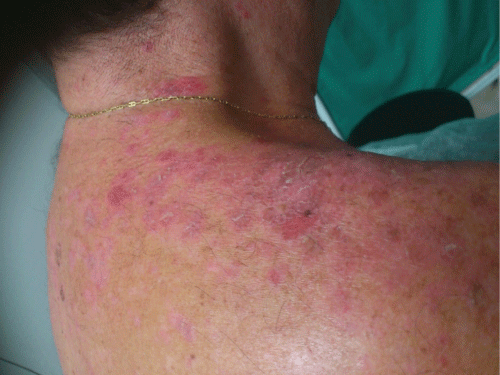
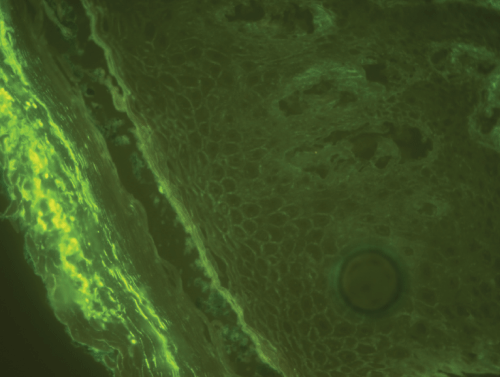
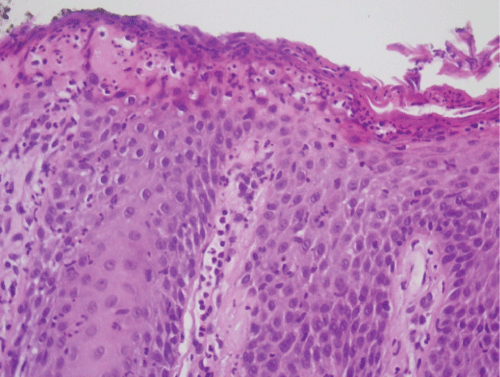
A 70-year-old man with personal history of NSAIDs allergy, asthma, endocarditis, hypercholesterolemia, hyperuricemia and hypertension presented to us with a skin eruption. He has not family history of autoimmune diseases. His antihypertensive medication regimen included enalapril that was stopped because of cough and he was started to be treated with oral valsartan 160 mg plus hydrochlorothiazide during the 12 month period before he presented to us with lesions on the scalp an itchy skin rash. Dermatological examination revealed erythematous, scaly crusted plaques and erosions over the face and on the chest, back and shoulders (Figure 1). Mucosal lesions were not observed. Skin biopsy showed eosinophilic spongiosis of the epidermis and subcorneal blistering with acantholysis at the upper epidermis (Figure 2). The histopathologic examination was consistent with PF. Direct immunofluorescence revealed intercellular staining for IgG and C3 at the upper epidermis (Figure 3). Laboratory investigations showed eosinophilia. Renal and liver functions test, serum levels of complements C3 C4 and antinuclear antibodies revealed no abnormalities. ELISAs detected antiDsg1 antibodies (index 8.55) but not anti Dsg-3.IFN gamma release test nor drug-induced lymphocyte stimulation test were not available. Pemphigus foliaceus was diagnosed. Treatment with oral prednisone 30 mg daily plus azathioprine 100 mg (as a glucocorticoid -sparing agent) was started and valsartan was discontinued, resulting in gradual improvement. Skin lesions recurred after valsartan was reintroduced by cardiologist 6 month later.
.
Figure 2: Subcorneal blister formation and acantholytic keratinocytes. Eosinophilic spongiosis of the epidermis.
View Figure 2
Discussion
Valsartan is an angiotensin II receptor blocker (ARB), a newer class of antihypertensives. Rarely some skin condition such as angioedema, urticarial, photosensitive rash, lichenoid eruption, pseudolymphomatous eruption and bullous pemphigoid, induced by valsartan have been reported [6-13]. ARBs or sartans, widely prescribed as a substitute of angiotensin-converting enzyme inhibitors(ACEi), were reported as possible triggers of PF in patients treated with candesartan and telmisartan, losartan and valsartan [5,14].
Pemphigus foliaceus is a rare autoimmune blistering disease in which the binding of autoantibodies to desmoglein 1 (Dsg1) results in acantholysis (loss of intercellular adhesion between keratinocytes) and development of subcorneal blisters. Clinical findings include superficial bullae, and more often erosive lesions leaving areas of crusting over an erythematous base, localized on the chest, back, shoulders, face and scalp. Nikolsky' sign (blistering of the skin as a response to slight rubbing or pressure) is usually positive. The diagnosis is based on clinical picture, histopathological findings and presence of autoantibodies detected by direct or indirect immunofluorescence [15]. The most common variant of drug-induced pemphigus is pemphigus foliaceus (PF) with anti desmoglein 1 auto antibodies, caused by thiol containing drugs [3] although pemphigus vulgaris (PV) has also been described. Associated drugs most commonly offenders are penicillamin, captopril and penicillins, and rarely pyritinol, thioproline, rifampicin, cephalosporins, gold, methimazole, levodopa, opioids phenobarbital, cytokins, piroxicam, aspirin, nifedipine, propranolol and angiotensin converting enzyme inhibitors other than captopril. Lesions may develop from 1 day to 6 months after administration of the culprit drug [16].
Yoshimura et al. [3] found that 8 of 17 patients with drug-induced pemphigus showed foliaceus -like appearance and responsible drugs were thiol-containing drugs in 16 (bucillamine, d-penicillamine, cetapril, thiopronine and captopril).
Feng et al. [17] studied 6 cases of drug induced pemphigus. Five out of 6 cases of drug induced pemphigus presented as pemphigus foliaceus, were sensitive to low doses of corticosteroids and had good prognosis.
Criteria for diagnosis of drug-induced pemphigus include a history of exacerbation while using the same drug in addition to blistering, acantholysis and positive direct IF on the skin showing cell surface staining of IgG, C3 deposition [17].
The in vitro IFN-gamma release from the patient's lymphocytes test has been shown to be of diagnostic value in drug-induced skin reactions [2]. 32-2B immunolabeling is useful for diagnosing drug-induced pemphigus and a normal pattern is an indicator of a good prognosis [18].
Most of patients go into remission upon withdrawal of the offending drug but some patients must be treated with systemic corticosteroids, other immunosuppressants or agents as antimalarial drugs, rituximab, mycophenolate mofetilor intravenous immunoglobulin.
Conclusion
Many drugs can induce pemphigus. A drug origin should be considered in every new patient with pemphigus and in every flare-up of the disease. The diagnosis of drug-induced pemphigus is challenging. The interval between the administration of drug and the onset of cutaneous manifestations is not well defined. The majority of the patients with drug-induced pemphigus show pemphigus foliaceus -like clinical and histopathological features with anti-Dsg 1 autoantibodies. A meticulous clinical history can detect the presence of facilitating or triggering factors. A prolonged latency period between the administration of the drug and the onset of the cutaneous manifestations may occur. Discontinuing the offending drug reduce the need for medical treatment.
References
-
Ruocco V, Ruocco E, Lo Schiavo A, Brunetti G, Guerrera LP, et al. (2013) Pemphigus: etiology, pathogenesis, and inducing or triggering factors: facts and controversies. Clin Dermatol 31: 374-381.
-
Brenner S, Goldberg I (2011) Drug-induced pemphigus. Clin Dermatol 29: 455-457.
-
Yoshimura K, Ishii N, Hamada T, Abe T, Ono F, et al. (2014) Clinical and immunological profiles in 17 Japanese patients with drug-induced pemphigus studied at Kurume University. Br J Dermatol 171: 544-553.
-
Landau M, Brenner S (1997) Histopathologic findings in drug-induced pemphigus. Am J Dermatopathol 19: 411-414.
-
Pietkiewicz P, Gornowicz-Porowska J, Bowszyc-Dmochowska M, Dmochowski M1 (2015) A retrospective study of antihypertensives in pemphigus: a still unchartered odyssey particularly between thiols, amides and phenols. Arch Med Sci 11: 1021-1027.
-
Kalra A, Cooley, Palaniswamy C, Kalra A, Zanotti-Cavazzoni SL (2012) Valsartan-induced angioedema in a patient on Angiotensin-Converting-Enzyme Inhibitor for years: Case report and literature review. Am J Ther 19: 189-192.
-
Irons BK, Kumar A (2003) Valsartan-induced angioedema. Ann Pharmacother 37: 1024-1027.
-
de la Serna Higuera C (2000) [Angioedema and urticarial reaction induced by valsartan]. Med Clin (Barc) 114: 599.
-
Martínez Alonso JC, Domínguez Ortega FJ, Méndez Alcalde J, Fuentes Gonzalo MJ (2003) Angioedema due to valsartan. Allergy 58: 367.
-
Frye CB, Pettigrew TJ (1998) Angioedema and photosensitive rash induced by valsartan. Pharmacotherapy 18: 866-868.
-
Gencoglan G, Ceylan C, Kazandi AC (2009) Linear lichenoid drug eruption induced by valsartan. Clin Exp Dermatol 34: e334-335.
-
Sawada Y, Yoshiki R, Kawakami C, Fukamachi S, Sugita K, et al. (2010) Valsartan-induced drug eruption followed by CD30+ pseudolymphomatous eruption. Acta Derm Venereol 90: 521-522.
-
Femiano F (2003) [Mucocutaneous bullous pemphigoid induced by valsartan. A clinical case]. Minerva Stomatol 52: 187-190.
-
Bae YI, Yun SJ, Lee SC, Park GT, Lee JB (2008) Pemphigus foliaceus induced by an angiotensin II receptor blocker. Clin Exp Dermatol 33: 721-723.
-
James KA, Culton DA, Diaz LA (2011) Diagnosis and clinical features of pemphigus foliaceus. Dermatol Clin 29: 405-412, viii.
-
Ahronowitz I, Fox L (2014) Severe drug-induced dermatoses. Semin Cutan Med Surg 33: 49-58.
-
Feng S, Zhou W, Zhang J, Jin P (2011) Analysis of 6 cases of drug-induced pemphigus. Eur J Dermatol 21: 696-699.
-
Maruani A, Machet MC, Carlotti A, Giraudeau B, Vaillant L, et al. (2008) Immunostaining with antibodies to desmoglein provides the diagnosis of drug-induced pemphigus and allows prediction of outcome. Am J Clin Pathol 130: 369-374.