International Journal of Blood Research and Disorders
The Levels of PAI-1, Urokinase uPA, Thrombomodulin and TFPI in Essential Thrombocythemia
Oktay Bilgir1*, Ferda Bilgir2, Mehmet Calan3 and Giray Bozkaya4
1Department of Hematology, Izmir Bozyaka Training and Research Hospital, Bozyaka, Izmir, Turkey
2Department of Allergy & Immunology, Katip Celebi University, Medical School, Ataturk Training and Research Hospital, Izmir, Turkey
3Department of Biochemistry, Izmir Bozyaka Training and Research Hospital, Bozyaka, Izmir, Turkey
4Department of Internal Medicine, Izmir Bozyaka Training and Research Hospital, Bozyaka, Izmir, Turkey
*Corresponding author:
Oktay Bilgir, Department of Hematology, Izmir Bozyaka Training and Research Hospital, Bozyaka, Izmir, Turkey, Tel: +902322505050/5242, Fax: +902322614444, E-mail:
oktaybilgir@gmail.com
Int J Blood Res Disord, IJBRD-2-016, (Volume 2, Issue 2), Research Article; ISSN: 2469-5696
Received: July 21, 2015 | Accepted: September 02, 2015 | Published: September 05, 2015
Citation: Bilgir O, Bilgir F, Calan M, Bozkaya G (2015) The Levels of PAI-1, Urokinase uPA, Thrombomodulin and TFPI in Essential Thrombocythemia. Int J Blood Res Disord 2:016. 10.23937/2469-5696/1410016
Copyright: © 2015 Bilgir O, et al. This is an open-access article distributed under the terms ofthe Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Thromboembolic attacks are frequent in essential thrombocythemia (ET) cases, and the causes of thromboembolism include many acquired causes in addition to some genetic factors like JAK 2. This study is intended to evaluate the levels of some coagulation parameters such as PAI-1, uPA, TM, TFPI and vWF (von Willebrant Factor) on thromboembolic attacks.
Materials and Methods: The study group consisted of 30 essential thrombocythemia patients with a mean age of 59.5 ± 9.9 and 30 healthy controls with a mean age of 56.2 ± 6.1. Plasma PAI, uPA, TM, TFPI and vWF levels were determined by ELISA method according to manufacturer's instructions.
Results: There was a significant difference in PAI-1, uPA, TM, and TFPI levels between the patient and control groups (p < 0.05). However, no statistically significant difference was obtained in vWF levels between two groups (p > 0.05).
Conclusions: High levels of PAI-1, uPA, TM and TFPI indicate that some coagulation parameters are activated, and we assume that this may cause thromboembolic attacks regardless of genetic factors.
Keywords
Essential thrombocythemia, Plasminogen activator inhibitor-1, Urokinase plasminogen activator, Thrombomodulin, Tissue factor pathway inhibitor
Introduction
Generally, ET is a myeloproliferative disorder characterized by persistent peripheral thrombocytosis and abnormal megakaryocytic proliferation in the bone marrow [1]. ET is a rare cause of thrombocytosis and may be associated with both venous and arterial thrombosis in 10-75% of cases, depending largely on the study and whether vasomotor symptoms are included in thrombotic manifestations [2-4]. Thrombotic events may be observed in both microvascular and macrovascular arterial circulation in patients with ET [3].
In general, risk factors for thrombosis in patients with ET include: increased age, a prior thrombotic episode, and the presence of risk factors for atherosclerotic heart disease [5]. Several authors agree that there does not appear to be a linear relationship between platelet counts and thrombosis [2,6], but others attribute both the development of thrombosis and hemorrhage to an increasing platelet count [1]. Furthermore, there is no consistent evidence that the in vitro assessment of platelet aggregation will identify patients at risk for thrombosis [3]. It is therefore not surprising that the task of appropriately adding those patients with ET who are at increased risk for thrombotic events poses both a clinical and laboratory challenge. As a result a detailed understanding the thrombotic mechanisms becomes difficult owing to the possible multifactorial nature of thrombosis in these patients.
It is well recognized that plasminogen activator inhibitor (PAI-1) acts as the primary physiological inhibitor of plasminogen activation in vivo, and is associated with a variety of thrombotic conditions when elevated, including myocardial infarction and deep venous thrombosis. Elevated PAI-1 concentration was found to be a reliable predictor of coronary events in patients with angina pectoris or previous myocardial infarctions [7]. Moreover, PAI-1 appears to be upregulated in obesity, atherosclerosis, hypertension, glucose intolerance, insulin resistance and malignancy [8]. Although not often reported, an association between PAI-1 elevations and thrombogenesis has also been a feature in patients with ET [6,9].
Material and Methods
Patients
30 patients diagnosed with ET and 30 healthy persons as control were enrolled in this study. The ET diagnoses were made as per the diagnostic criteria suggested by the Polycythemia Vera Study Group [10]. Age averages of both groups were similar (59.5 ± 9.9 vs. 56.2 ± 6.1, p: 0.121) (Table 1). No symptoms of any acute infection or inflammatory disease were seen in the ET cases and healthy control group. None of the ET cases or healthy controls was on oral contraceptives or hormone replacement therapy during their blood sample collections. All researches were approved by the local ethics committee. The procedure used was in compliance with the 1975 Helsinki Declaration as revised in 2000, and their blood samples were collected after their informed consents were taken.
Laboratory analyses
Venous fasting blood samples were collected from an antecubital vein in evacuated tubes with 3.8% trisodium citrate as anticoagulant (Vacuette, Greiner Bio-One, Austria). After centrifugation, the plasma samples were aliquated, frozen and stored at -80℃ for PAI-1, uPA, thrombomodulin (TM), TFPI and vWF analysis. Repeated freezing and thawing processes were avoided. Plasma PAI (Bender MedSystems Inc, Vienna, Austria), uPA, TM, TFPI and vWF (American Diagnostica Inc., Stamford, CT, USA) levels were determined by ELISA method according to manufacturer's instructions.
Statistical analyses
All statistical analyses were performed using the Statistical Package for Social Sciences (SPSS, version 11.0 for Windows, Chicago, Ill, USA). Data were expressed as mean ± standard deviation (SD). A "p" value < 0.05 was accepted as significant. Kolmogorov-Smirnov test was performed to assess the normality of the variables. T-test and Mann-Whitney U tests were used to compare the variables. According to Kolmogorov-Smirnov test, the distribution of TFPI and vWF were normal (p > 0.05) whereas the distribution of PAI, uPA and TM were not normal. Normally distributed variables were compared with t-test whereas other parameters were compared with Mann-Whitney U test.
![]()
Table 1: Levels and comparison of parameters in patients and controls (mean ± SD).
View Table 1
Results
The study group consisted of 30 essential thrombocytosis patients with a mean age of 59.5 ± 9.9 and 30 healthy controls with a mean age of 56.2 ± 6.1 (Table 1). Mean PAI-1, uPA, TM, TFPI and vWF levels of groups are presented in table 1.
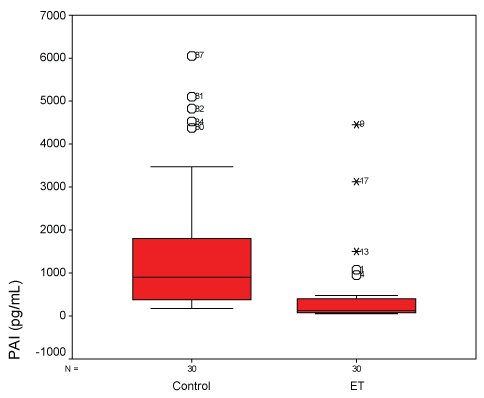
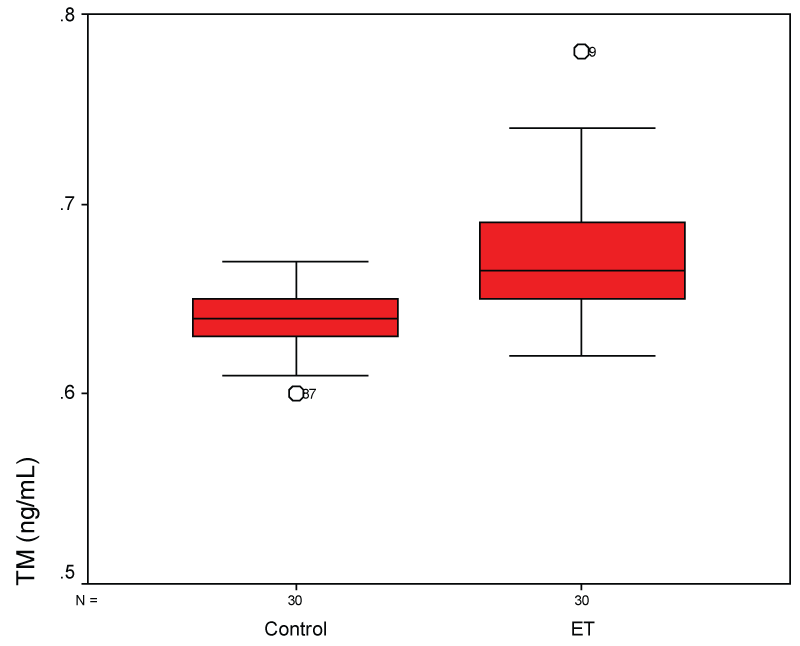
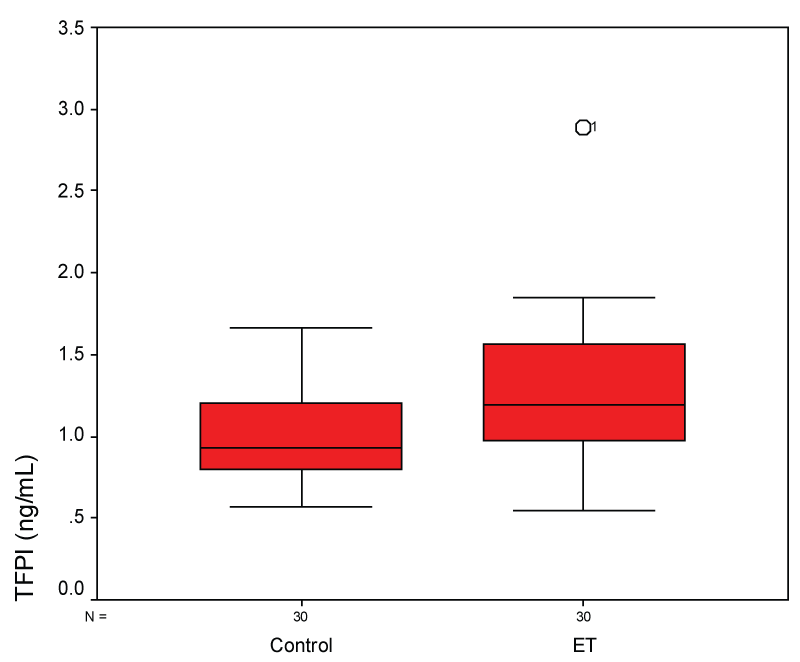
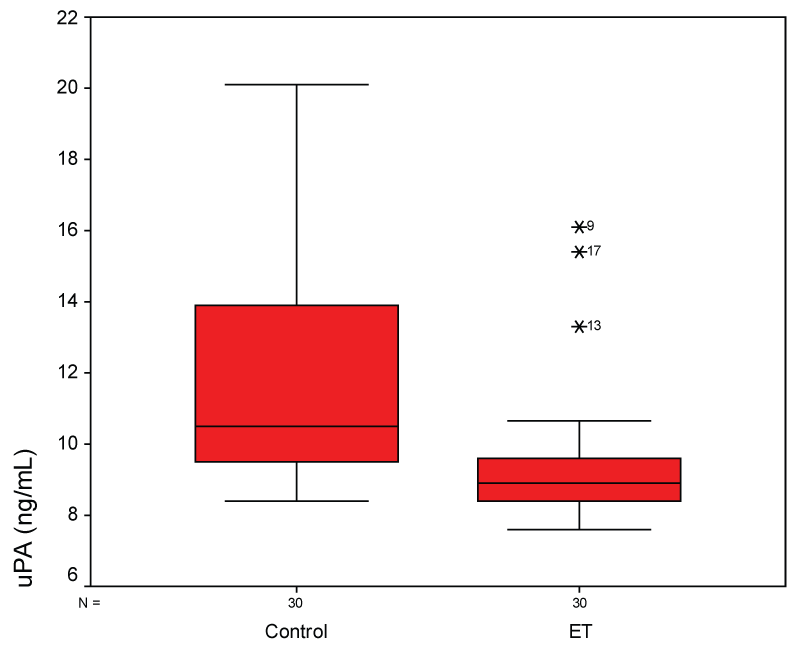
There was a significant difference in PAI-1 (Figure 1), TM (Figure 2), TFPI (Figure 3), and uPA (Figure 4) levels between patient and control groups (p < 0.05). However, no statistically significant difference was obtained in vWF levels between two groups (p > 0.05).
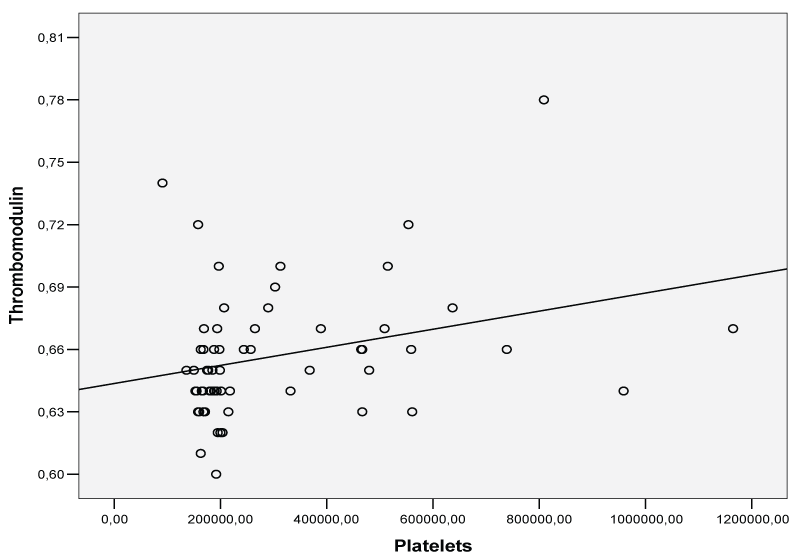
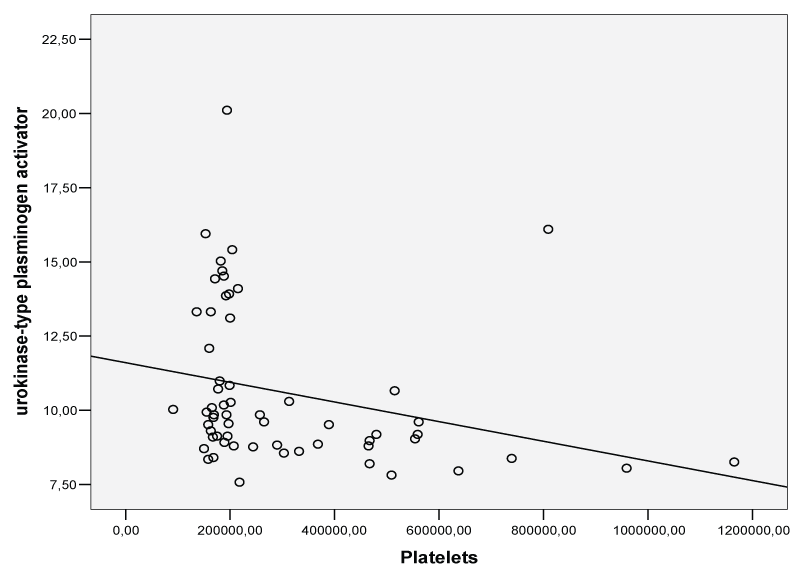
Pearson's correlation analysis revealed that platelet count was correlated with thrombomodulin (Figure 5; r = 0.30, p = 0.020), TFPI (Figure 6; r = 0.30, p = 0.019) and negatively with uPA (Figure 7; r = - 0.27, p = 0.036) in the whole study group.
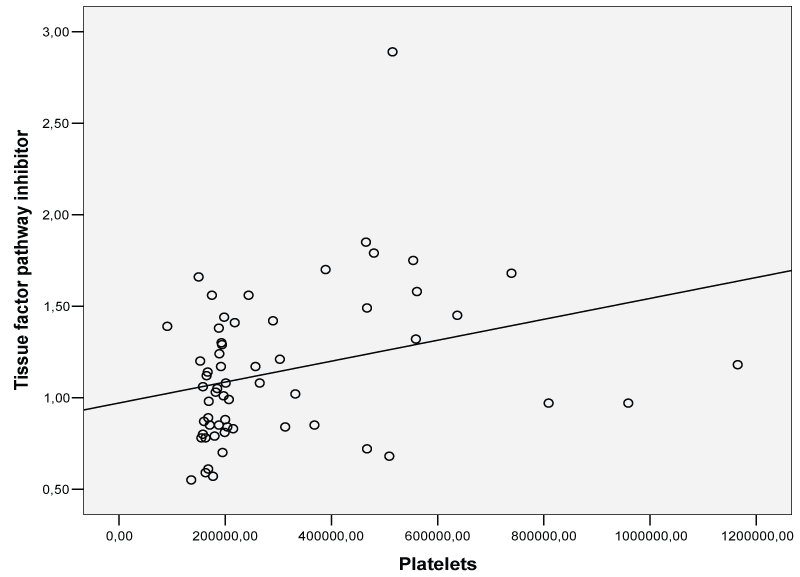
.
Figure 6: Platelet count was correlated with Tissue factor pathway inhibitor in the study group.
View Figure 6
Discussion
Plasminogen activator inhibitor-1 (PAI-1), a member of the serine proteinase inhibitor (serpin) family, is produced in the liver and endothelial cells and is expressed in thrombocytes, liver cells, monocytes, and smooth muscle cells [11]. Most active proportion of PAI-1 is found primarily in the liver and secondarily in the spleen [11]. A small portion of PAI-1 is found in the brain and myocardium [12]. Normal human blood contains independent pools of plasma and platelet PAI-1 as well, in varying amounts and activity [10]. Although found in small quantities (7%), it is the plasma PAI-1 that shows the highest activity. Platelets, in contrast, account for the remaining 93% of blood PAI-1, with only 3-5% activity [13]. PAI-1 is affected by gender, age, obesity, plasma concentrations of tumor necrosis factor (TNF-alpha), interleukin-1(IL-1), tumor growth factor (TGF)-β, lipopolysaccharides, phorbol esters, glucocorticoid hormones, VLDL, and insulin [14]. Possessing a circadian rhythm, PAI-1 shares similarities with hormones and other cytokines associated with thromboembolic events (including myocardial infarctions), with peak activity in the early morning followed by rapid tapering [15]. Amongst other features, PAI-1 is known to be an acute phase reactant. Increased plasma PAI-1 activity is found during acute events such as myocardial infarction, postoperative period, endotoxemia, and septicemia [12]. In fact, PAI-1 levels have been shown to be a good prognostic factor for fatal outcomes in septicemia because PAI-1 is increased up to fivefold in response to endotoxin, lipopolysaccharides, and IL-1 elevations [12].
The PAI-1 molecule reacts rapidly with tissue plasminogen activator (tPA) and uPA, forming very stable, stoichiometric 1:1 complexes, as is typical for serpins [15]. However, PAI-1 lacks cysteines (and hence disulfide bonds) unlike other serpins but contains multiple methionines. The detection of PAI-1 mRNA and PAI-1 antigen in megakaryocytes suggests that PAI-1 may be deposited into alpha granules during the maturation of these cells. Studies have shown that approximately 4000 molecules of PAI-1 per platelet are localized in the alpha granules [16]. On activation, platelets will secrete both PAI-1 and alpha 2-antiplasmin, inhibiting plasmin formation and activity, respectively [17]. Most of the platelet PAI-1 appears to be in the latent form, a phenomenon that may be associated with the stabilizing effects of vitronection [18]. It has been suggested that the latent form of PAI-1 may be responsible for the resistance to thrombolytic therapy by platelet-rich thrombi [19]. However, the presence of a large storage pool of latent PAI-1 in platelets also raises the possibility that mechanisms may exist for the activation of this form of PAI-1 in vivo.
Although found in low concentrations in the blood, PAI-1, is primarily responsible for in vivo inhibition of both tissue type (tPA) and urokinase-type (uPA) plasminogen activators. Under the influence of both genetic and environmental factors, PAI-1 elevation has been associated with the promotion of both venous and arterial thrombus formation in a multitude of medical conditions [20]. PAI-1 has been positively correlated with severe hypertension, myocardial disease, deep venous thrombosis, malignancies, including myeloproliferative diseases, disorders of glucose and lipid metabolism, type II diabetes mellitus, polycystic ovarian syndrome, and is influenced by both alcohol consumption and acute infection [20-22]. Although the association between arterial thrombosis and myeloproliferative disorders has been previously established, the relation between ET, PAI-1 elevations, and arterial thrombogenesis has been only sporadically mentioned in the literature [23,24].
PAI-1 is expressed within invasive foci of multiple types of human cancer [25]. It was reported that PAI-1 is not produced by epithelial cancer cells but by the stromal cells contained within various tumors. High levels of PAI-1 have been found within the endothelial cells of small vessels in breast, gastric, liver, pancreas, lung, ovary, skin, brain, and colon carcinomas. High PAI-1 levels in certain tumors may predict poorer outcome. Additionally, a strong correlation between the number of tumor-positive axillary lymph nodes, particularly in breast cancer, and increased levels of PAI-1 has been made [25]. This association may have significant prognostic and therapeutic implications and may serve as supportive evidence for PAI-1 surveillance in cancer patients at risk for thromboembolic events.
Tissue factor pathway inhibitor (TFPI) is an endothelial membrane-associated protein [26]. TFPI co-localizes with tissue factor and macrophages in atherosclerotic plaques, where it may reduce thrombotic and inflammatory potential [27]. In vascular injury models, TFPI infusion or local gene delivery had favorable effects on thrombosis and restenosis [28], while TFPI deficiency in atherosclerosis-prone mice was associated with more atherosclerosis, more plaque tissue factor activity and decreased time to occlusive thrombosis with vascular injury [29]. TFPI circulates primarily bound to lipoproteins with a smaller free component; both can be measured with commercial assays [30]. Both free and total TFPI concentration may be higher with acute or chronic vascular disease [31]. Not all studies agree, however [32]. Epidemiologic studies have reported conflicting results concerning correlation of TFPI with vascular risk factors [33]. However, TFPI is essentially the stoichiometric inhibitor of extrinsic pathway (FVIIa-tissue factor) of coagulation. The complex that it forms through binding with FXa rapidly inactivates the FVIIa-tissue factor. It should be considered that this increase seen in the TFPI level is caused by the co-activation of the tissue factor with FVIIa.
As we know, Thrombomodulin (TM) binds with α-thrombin, and neutralizes its procoagulant activity; and plays a cofactor role of α-thrombin in the activation of Protein C and TAFI (Thrombin Activatable Fibrinolysis Inhibitor), and has anticoagulant and antifibrolinytic function. Its increase in ET indirectly shows that α-thrombin is activated in this disease group.
Increase in the PAI-1 and uPA levels in ET cases indicates that coagulation is activated in addition to vascular injury. Although genetic factors such as JAK2 are shown to cause thromboembolism in ET, also vascular injury as well as activation of coagulation may be assumed to cause this.
uPA is one of the major extrinsic activators in the blood, and is effective in both vascular tissue and extravascular tissue. uPA is reported to have a significant role in the activation of matrix metalloproteinase, as well as degradation of extracellular matrix through its plasminogen activation [34,35]. Particularly, it has an amplifying effect on the formation speed of plasmin secondary to fibrin in the fibrinolytic process as does t-PA on vascular tissue. Although it is assumed that uPA is primarily effective in extracellular matrix degradation, as well as tissue repair and remodeling, the existence of its profibrinolytic features should not be ignored. Since it has showed negative correlation with the number of thrombocytes in our study, we consider that fibrinolysis occurs after coagulation in these cases, and the number of thrombocytes decrease after occurrence of this process.
Consequently, due to increase in the PAI-1, uPA, TM and TFPI values in ET, we assume that some of the coagulation parameters activated in this disease should be included in the factors that cause thromboembolism.
References
-
Colombi M, Radaelli F, Zocchi L, Maiolo AT (1991) Thrombotic and hemorrhagic complications in essential thrombocythemia.A retrospective study of 103 patients. Cancer 67: 2926-2930.
-
Fenaux P, Simon M, Caulier MT, Lai JL, Goudemand J, et al. (1990) Clinical course of essential thrombocythemia in 147 cases. Cancer 66: 549-556.
-
Cripe L, Hromas R (1998) Malignant disorders of megakaryocytes. Semin Hematol 35: 200-209.
-
Pósán E, Ujj G, Kiss A, Telek B, Rák K, et al. (1998) Reduced in vitro clot lysis and release of more active platelet PAI-1 in polycythemia vera and essential thrombocythemia. Thromb Res 90: 51-56.
-
Watson KV, Key N (1993) Vascular complications of essential thrombocythemia: A link to cardiovascular risk factors. Br J Haematol 83: 198-203.
-
Bellucci S, Tobelem G, Flandrin G (1986) Essential thombocythemia. Cancer 58: 2440-2447.
-
Juhan-Vague I, Pyke SD, Alessi MC, Jespersen J, Haverkate F, et al. (1996) Fibrinolytic factor and the risk of myocardial infarction or sudden death in patients with angina pectoris. Circulation 94: 2057-2063.
-
Bazzan M, Tamponi G, Gallo E, Stella S, Schinco PC, et al. (1993) Fibrinolytic imbalance in essential thrombocythemia: A role of platelets. Haemostasis 23: 38-44.
-
Cancelas JA, Garcia-Avello A, Garcia-Frade LJ (1994) High plasma level of plasminogen activator inhibitor 1 (PAI-1) in polycythemia vera and essential thrombocythemia are associated with thrombosis. Thromb Res 75: 513-520.
-
Loskutoff DJ, Smad F (1998) The adipocyte and hemostatic balance in obesity. Arterioscler Thromb Vasc Biol 18: 1-6.
-
Urdén G, Chmielewska J, Carlsson T, Wiman B (1987) Immunological relationship between PAI-1 from different sources. Thromb Haemost 57: 29.
-
Dawson S,Henney A (1992) The status of PAI-1 as a risk factor for arterial and thrombotic disease: A review. Atherosclerosis 95: 105-117.
-
Simpson AJ, Booth NA, Moore NR (1990) The platelet and plasma pools of plasminogen activator inhibitor (PAI-1) vary independently in disease. Br J Haematol 75: 543-548.
-
Takada A, Takada Y, Urano T (1994) The physiological aspects of fibrinolysis. Thromb Res 76: 1-31
-
Winman B (1995) Plasminogen activator inhibitor 1 (PAI-1) in plasma: Its role in thrombotic disease. Thromb Haemost 74: 71-76.
-
Konkle BA, Schick PK, He X, Liu RJ, Mazur EM (1993) Plasminogen activator inhibitor-1 mRNA is expressed in platelets and megakaryocytes and megakaryoblastic cell line CHRF-288. Arterioscler Thromb 13: 669-674.
-
Fay WP, Eitzman DT, Shapiro AD, Madison EL, Ginsburg D (1994) Platelets inhibit fibrinolysis in vitro by both plasminogen activator inhibitor-1-dependent and independent mechanisms. Blood 83: 351-356.
-
Lindahl TL, Sigurdarttir O,Winman B (1989) Stability of plasminogen activator-1 (PAI-1). ThrombHaemost 62: 748-751.
-
Nordenham A, Wiman B (1997) Plasminogen activator inhibitor (PAI-1) content in platelets from healthy individuals genotyped for the 4G/5G polymorphism in the PAI-1 gene. Scand J Clin Lab Invest 57: 453-461.
-
Glueck CJ, Fontaine RN, Gupta A, Alasmi M (1997) Myocardial infarction in a 35-year-old man with homocysteinemia, high plasminogen activator inhibitor activity, and resistance to activated protein C. Metabolism 46: 1470-1472.
-
Sartori MT, Wiman B, Vettore S, Dazzi F, Girolami A, et al. (1998) 4G/5G polymorphism of PAI-1 gene promoter and fibrinolytic capacity in patients with deep venous thrombosis. Thromb Haemost 80: 956-960.
-
Margaglione M, Cappucci G, d'Addedda M, Colaizzo D, Giuliani N, et al. (1998) PAI-1 plasma levels in a general population without clinical evidence of atherosclerosis: relation to environment and genetic determinants. Arterscler Thromb Vasc Biol 18: 562-567.
-
Eriksson P, Nilsson L, Karpe F, Hamsten A (1998) Very-low-density lipoprotein response element in the promoter region of the human plasminogen activator inhibitor-1 gene implicated in the impaired fibrinolysis of hypertriglyceridemia. Arterioscler Thromb Vasc Biol 18: 20-26.
-
Starnberg L, Lawrence D, Ny T (1988) The organization of the human plasminogen activator inhibitor-1 gene. Implications on the evolution of the serine-protease inhibitor family. Eur J Biochem 176: 609-616.
-
Pappot H, Gårdsvoll H, Rømer J, Pedersen AN, Grøndahl-Hansen J, et al. (1995) Plasminogen activator inhibitor type 1 in cancer: Therapeutic and prognostic implications. Biol Chem 378: 259-267.
-
Kato H (2002) Regulation of functions of vascular wall cells by tissue factor pathway inhibitor: basic and clinical aspects. Arterioscler Thromb Vasc Biol 22: 539-548.
-
Crawley J, Lupu F, Westmuckett AD, Severs NJ, Kakkar VV, et al. (2000) Expression, localization, and activity of tissue factor pathway inhibitor in normal and atherosclerotic human vessels. Arterioscler Thromb Vasc Biol 20: 1362-1373.
-
Yin X, Yutani C, Ikeda Y, Enjyoji K, Ishibashi-Ueda H, et al. (2002) Tissue factor pathway inhibitor gene delivery using HVJ-AVE liposomes markedly reduces restenosis in atherosclerotic arteries. Cardiovasc Res 56: 454-463.
-
Westrick RJ, Bodary PF, Xu Z, Shen YC, Broze GJ, et al. (2001) Deficiency of tissue factor pathway inhibitor promotes atherosclerosis and thrombosis in mice. Circulation 103: 3044-3046.
-
Morange PE, Renucci JF, Charles MA, Aillaud MF, Giraud F, et al. (2001) Plasma levels of free and total TFPI, relationship with cardiovascular risk factors and endothelial cell markers. Thromb Haemost 85: 999-1003.
-
Soejima H, Ogawa H, Yasue H, Kaikita K, Nishiyama K, et al. (1999) Heightened tissue factor associated with tissue factor pathway inhibitor and prognosis in patients with unstable angina. Circulation 99: 2908-2913.
-
Morange PE, Blankenberg S, Alessi MC, Bickel C, Rupprecht HJ, et al. (2007) Prognostic value of plasma tissue factor and tissue factor pathway inhibitor for cardiovascular death in patients with coronary artery disease: the AtheroGene study. J Thromb Haemost 5: 475-482.
-
Sakkinen PA, Cushman M, Psaty BM, Kuller LH, Bajaj SP, et al. (1998) Correlates of antithrombin, protein C, protein S, and TFPI in a healthy elderly cohort.Thromb Haemost 80: 134-139.
-
Jensen MK, Holten-Andersen MN, Riisbro R, de Nully Brown P, Larsen MB, et al. (2003) Elevated plasma levels TIMP-1 correlate with plasma suPAR/uPA in patients with chronic myeloproliferative disorders. Eur J Haematol 71: 377-384.
-
Jensen MK, Riisbro R, Holten-Andersen MN, de Nully Brown P, Junker P,et al. (2003) Collagen metabolism and enzymesof the urokinase plasminogen activator system in chronic myeloproliferative disorders: correlation between plasma-soluble urokinase plasminogen activator receptor and serum markers for collagen metabolism. Eur J Hematol 71: 276-282.