International Journal of Critical Care and Emergency Medicine
Almost Total Airway Obstruction Due to Unidentified Massive Hemoptysis under Extracorporeal Membrane Oxygenation
Satoshi Kazuma1*, Yoshiki Masuda1, Hiroomi Tatsumi1, Kanako Takahashi1, Shinichiro Yoshida2, Maiko Honma1, Hitoshi Imaizumi2 and Michiaki Yamakage1
1Department of Intensive Care Medicine, Sapporo Medical University, Japan
2Department of Anesthesiology and Intensive Care Medicine, Tokyo Medical University, Japan
*Corresponding author:
Satoshi Kazuma, Department of Intensive Care Medicine, Sapporo Medical University, School of Medicine, South 1 West 16, Chuo-ku, Sapporo, Hokkaido 060-8543, Japan, Tel: 011-611-2111 (ext 3728), Email: sea_hawk_3104@yahoo.co.jp
Int J Crit Care Emerg Med, IJCCEM-2-012, (Volume 2, Issue 1), Case Report; ISSN: 2474-3674
Received: January 19, 2016 | Accepted: February 24, 2016 | Published: February 26, 2016
Citation: Kazuma S, Masuda Y, Tatsumi H, Takahashi K, Yoshida S, et al. (2016) Almost Total Airway Obstruction Due to Unidentified Massive Hemoptysis under Extracorporeal Membrane Oxygenation. Int J Crit Care Emerg Med 2:012. 10.23937/2474-3674/1510012
Copyright: © 2016 Kazuma S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
When life-threatening hemoptysis occurs, it is difficult to determine the focus of hemorrhage and to perform selective hemostasis in many cases. In this situation, it is difficult to maintain respiratory function due to mechanical obstruction by blood clots and blood aspiration.
Veno-venous extracorporeal membrane oxygenation (VV-ECMO) has been used in clinical practice for a number of years. It has been used successfully for the management of acute reversible pulmonary failure. Thus, VV-ECMO can be used for respiration in patients with life-threatening hemoptysis in which the bleeding focus cannot be identified, even if there is nearly total airway obstruction.
We report a successfully treated case of almost total airway obstruction due to unidentified massive hemoptysis under extracorporeal membrane oxygenation
Case Presentation
A 54-year-old female weighing 62 kg had hemoptysis with an unidentified focus 4 months before and had been followed in the Department of Respiratory Medicine. Angiographic embolization was performed for a right bronchial artery ramification suspected of being the bleeding focus by bronchofiberscopy, but there were few effects. Right middle lobectomy was performed to control the persistent hemoptysis, and tracheal extubation was performed on the second day of illness. Sustained hemoptysis developed again 8 hours after extubation. Tracheal intubation and mask ventilation were difficult due to trismus, her short neck and blood in her oral cavity, and she went into cardiopulmonary arrest within a few minutes. There was repetition of return of spontaneous circulation and cardiopulmonary arrest while trying to maintain the airway. Then veno-arterial extracorporeal membrane oxygenation (VA-ECMO) was performed via femoral vessels.
Tracheal intubation was successfully performed using Glidescope® (Verathon, Bothell, WA, United States). Blood pressure was 126/81 mmHg, heart rate was 104 beats/min, cardiac index was 3.6 L/min/m2 and SpO2 was 100% with support by VA-ECMO at a flow rate of 2.5 L/min. Lactate level just after cardiopulmonary arrest increased to 124 mg/dL. The airway was almost totally obstructed with old blood clots and new fresh blood, and complete removal of blood was difficult. Although oxygen delivery to the lower part of the body was stable, that to the upper body including cerebral perfusion might be decreased in a situation with hemodynamic stability and ventilatory impairment due to extremely low tidal ventilation. Thus, ECMO was switched from veno-arterial bypass to veno-venous bypass (VV-ECMO)(oxygenator, Biocube 4000®, Nipro Co., Osaka, Japan; centrifugal pump, Rotaflow Centrifugal Pump®, MAQUET, Rastatt, Germany; cannulas, NSH heparinized cannula®, TOYOBO, Osaka, Japan) via the internal jugular vein within 25 minutes after return of spontaneous circulation. Under VV-ECMO at a flow rate of 3.5 L/min (VV-ECMO settings: FIO2, 1.0; sweep flow, 4 L/min), results of blood gas analysis with the Jackson-Rees technique were as follows: pH, 7.24; PaO2, 188 mmHg; PaCO2, 41 mmHg; lactate, 95 mg/dL.
In consideration of post cardiac arrest syndrome, the temperature of blood sent by VV-ECMO was controlled at normothemia (bladder temperature controlled within 35-36°C) for neuroprotection [1]. Activation coagulation time (ACT) was managed by administration of heparin within 120-160 seconds, which is shorter than that in usual ECMO management for massive hemoptysis. The right inferior lobe, where retention of some blood was observed at the time of the first operation, was suspected of being a bleeding focus of the hemoptysis, and right inferior lobectomy was performed on the same day. In the surgery, blood clots occluding the trachea were removed by using a fogarty catheter, and tidal volume then recovered to about 350 ml.
Ventilation was stable under VV-ECMO support 2 hours after surgery, but outflows of new fresh blood gradually increased again from the nasal cavity, oral cavity, and tracheal tube. Tidal volume fell to about 100 ml, and SpO2 was less than 90%. Removal of blood clots and fresh blood by a bronchofiberscope and Fogarty catheter was difficult. In consideration of ventilator-induced lung injury, 25 mg/hr of rocuronium was continuously administered, and the ventilator setting was changed from assisted ventilation to adjustment ventilation. Red cell concentrates were administered with the goal of Hb of 15 g/dl for the purpose of increasing oxygen delivery. SaO2 was maintained at 75-80% by full support of VV-ECMO.
One hour later, ventilation was almost completely interrupted (Figure 1a). VV-ECMO was performed at a flow rate of 3.0-3.5 L/min to maintain oxygen saturation (SaO2) at 80% or more, and results of blood gas analysis showed pH of 7.30, PaO2 of 44 mmHg, PaCO2 of 60 mmHg, lactate of 24 mg/dL and SaO2 of 81% (FIO2of 0.6 or less and peak inspiratory pressure of 25 cm H2O or less). The patient’s condition was stable with blood pressure of 138/84 mmHg and heart rate of 104 beats/min, and transthoracic echocardiography revealed that cardiac output was sufficient. Serum lactate level did not increase and there was no organ failure.
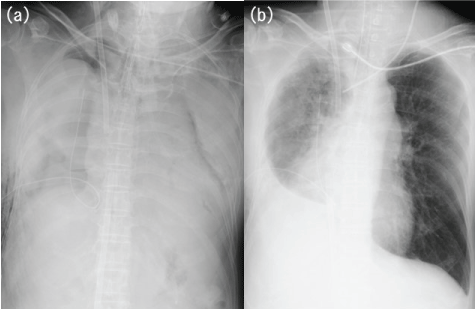
.
Figure 1: (a) Chest radiography showing almost total airway obstruction; (b) Just before VV-ECMO was discontinued.
View Figure 1
During VV-ECMO, blood gas analysis often showed an unexpected high oxygenation level (PaO2, 110 mmHg; SaO2, 99%). It was thought that liquid ventilation with blood that filled the airway occurred because the circulation and ventilation states did not change.
After the third day of illness, outflow of new blood from the tracheal tube was not seen. Blood clots were removed again using a bronchofiberscope, and tidal volume then gradually increased (Figure 2).
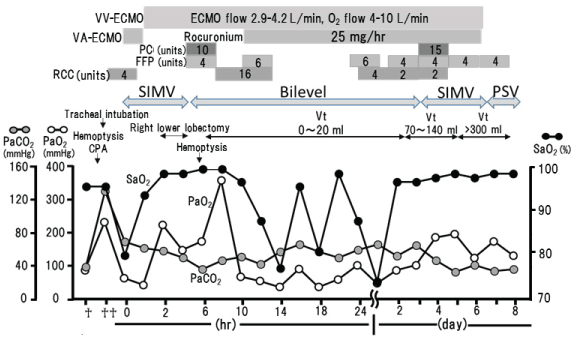
.
Figure 2: Clinical course on ICU admission. SIMV: synchronized intermittent mandatory ventilation; PSV: pressure support ventilation; Vt: tidal volume; RCC: red cell concentrate; FFP: fresh frozen plasma; PC: platelet concentration.
View Figure 2
On the seventh day of illness, results of blood gas analysis showed pH of 7.43, PaO2 of 150 mmHg, PaCO2 of 45 mmHg, and serum lactate of 11.0 mg/dl (FIO2, 0.45; PEEP, 5 cm H2O; minute volume, 7.9 L/min with no support by VV-ECMO), and VV-ECMO was therefore stopped (Figure 1b).
Tracheotomy was performed because disturbance of consciousness was prolonged, and she was transferred to a general ward on the 18th day of illness.
Five months later, there is no paralysis, and her consciousness state has recovered to a level of being able to obey commands. The Glasgow Coma Scale score was 10 (E3VTM6). Infectious diseases such as tuberculosis, collagen disease, bronchiectasis and neoplastic disease were negative from blood tests and bacteriologic culture and by image examinations. After performing lobectomy twice, pathologic examination of operatively extracted specimens revealed features of idiopathic pulmonary hemorrhage that required differential diagnosis of congestive vasculopathy due to pulmonary venous hypertension.
Discussion
The airway of the patient was almost totally obstructed by sustained massive hemoptysis and could not be ventilated, and respiratory function was completely dependent on VV-ECMO. There have been reports on preventive management by VV-ECMO for emergency cases, but there have been very few reports on respiratory management by VV-ECMO in a situation in which the airway was almost totally obstructed [2,3].
Although VA-ECMO was performed for cardiopulmonary arrest, it was changed to VV-ECMO after return of spontaneous circulation. During partial VA-ECMO, perfusate blood in the thoracic aorta mixes with left ventricular blood that has traversed the lungs. This is the reason why unsaturated blood that has traversed the non-functional lung might perfuse the cerebral tissue and cause hypoxic encephalopathy.
The blood sent by VV-ECMO is mixed with venous return (sending blood: right atrium blood = 3:1). As a result, if pulmonary function is almost totally obstructed as in this case, blood gas analysis would show PaO2 of 40 mmHg and SaO2 of 80% in theory. In fact, our patient showed SaO2 of 75-80%. If Hb is more than 15 g/dl and cardiac function is good, hypoxemia without a factor raising oxygen consumption such as infection can be permitted [4,5].
In general, anticoagulant management for thromboprophylaxis is necessary during ECMO. Both ACT and activated partial thromboplastin time (APTT) is managed by administration of heparin (ACT, 180-220 seconds; APTT, 1.5-2.5 fold of the normal level of APTT, respectively). However, anticoagulant management cannot be performed positively if there is active bleeding. ACT was managed modestly within 120-160 seconds in this case. Instead, thrombogenesis was prevented by management of the flow rate of ECMO at more than 3.0 L/min since clot formation in the circuit might be fatal.
After stopping off VV-ECMO, disturbance of consciousness was prolonged, but there was a tendency for recovery, and it is thought that cardiopulmonary arrest had a great influence.
During VV-ECMO, an inexplicable rise in SaO2 was seen repeatedly. It was thought that liquid ventilation might be effected. In general, liquid ventilation is performed with perfluorocarbon liquids that have low surface tension, and at atmospheric pressure, large amounts of oxygen and carbon dioxide dissolve in them [6]. It has been reported that liquid ventilation with a perfluorocarbon improved lung functions including respiratory distress syndrome [7], congenital diaphragmatic hernia [8], and adult respiratory distress syndrome [9]. It is thought that the nature of perfluorocarbon liquids is similar to that of erythrocytes containing hemoglobin. Oxygen might have been carried through blood that had filled the airway since the airway of the patient was almost totally obstructed by blood clots.
Competing of Interests
The authors declare that they have no competing interests. The authors have no affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the article.
References
-
Nielsen N, Wetterslev J, Cronberg T, Erlinge D, Gasche Y, et al. (2013) Targeted temperature management at 33°C versus 36°C after cardiac arrest. N Engl J Med 369: 2197-2206.
-
Park JM, Kim CW, Cho HM, Son BS, Kim do H (2014) Induced airway obstruction under extracorporeal membrane oxygenation during treatment of life-threatening massive hemoptysis due to severe blunt chest trauma. J Thorac Dis 6: E255-258.
-
Kim CW, Kim do H, Son BS, Cho JS, Kim YD, et al. (2015) The Feasibility of Extracorporeal Membrane Oxygenation in the Variant Airway Problems. Ann Thorac Cardiovasc Surg 21: 517-522.
-
Levy B, Taccone FS, Guarracino F (2015) Recent developments in the management of persistent hypoxemia under veno-venous ECMO. Intensive Care Med 41: 508-510.
-
Bartlett RH (2005) Physiology of ECLS. In: van Meurs K, ECMO: Extracorporeal Cardiopulmonary Support in Critical Care. (3rd edn), Extracorporeal Life Support Organization, 5-27.
-
Leach CL, Greenspan JS, Rubenstein SD, Shaffer TH, Wolfson MR, et al. (1996) Partial liquid ventilation with perflubron in premature infants with severe respiratory distress syndrome. The LiquiVent Study Group. N Engl J Med 335: 761-767.
-
Leach CL, Holm B, Morin FC 3rd, Fuhrman BP, Papo MC, et al. (1995) Partial liquid ventilation in premature lambs with respiratory distress syndrome: efficacy and compatibility with exogenous surfactant. J Pediatr 126: 412-420.
-
Major D, Cadenas M, Cloutier R, Fournier L, Wolfson MR, et al. (1995) Combined gas ventilation and perfluorochemical tracheal instillation as an alternative treatment for lethal congenital diaphragmatic hernia in lambs. J Pediatr Surg 30: 1178-1182.
-
Papo MC, Paczan PR, Fuhrman BP, Steinhorn DM, Hernan LJ, et al. (1996) Perfluorocarbon-associated gas exchange improves oxygenation, lung mechanics, and survival in a model of adult respiratory distress syndrome. Crit Care Med 24: 466-474.





