International Journal of Critical Care and Emergency Medicine
An Unstable Coronary Situation: A Case of Two Aborted Myocardial Infarctions in Three Days
Ephraim B Winzer* and Axel Linke
Department of Innere Medizin/Kardiologie, Herzzentrum Leipzig, Germany
*Corresponding author:
Ephraim B Winzer, Innere Medizin/Kardiologie, Herzzentrum Leipzig, Struempellstrasse 39, 04289 Leipzig, Germany, Tel: 0049-341-865252051, Fax: 0049-341-8651838, E-mail: bece@medizin.uni-leipzig.de
Int J Crit Care Emerg Med, IJCCEM-3-021, (Volume 3, Issue 1), Case Report; ISSN: 2474-3674
Received: July 10, 2016 | Accepted: January 23, 2017 | Published: January 25, 2017
Citation: Winzer EB, Linke A (2017) An Unstable Coronary Situation: A Case of Two Aborted Myocardial Infarctions in Three Days. Int J Crit Care Emerg Med 3:021. 10.23937/2474-3674/1510021
Copyright: © 2017 Winzer EB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
In patients with stable coronary artery disease percutaneous coronary intervention (PCI) effectively improves symptoms in the absence of any effect on prognosis. In contrast, in patients with acute coronary syndrome (ACS) PCI significantly reduces the risk of acute myocardial infarction and death as compared to conservative, noninvasive strategy.
In ACS patients with multivessel disease the interventionalist has to identify the culprit lesion responsible for symptoms. Contrast medium enhanced coronary angiography is the standard imaging technique for the evaluation of coronary vessels. In the absence of high grade stenosis, this technique often fails to detect vulnerable plaques.
In a 50-year old man with multivessel disease presenting with ACS, a high grade stenosis of the right coronary artery was identified as culprit lesion and treated with stent implantation, whereas low grade stenosis of the left anterior descending artery (LAD) and the first posterolateral branch (PLA1) remained invasively untreated. Underlying vulnerable plaques were missed by coronary angiography and were responsible for two further coronary events with aborted myocardial infarctions on day two and four after initial evaluation.
An intravascular imaging of the PLA1 and LAD during the first catheter that helps to differentiate between stable and unstable coronary lesions might have been indicated to detect unstable plaques guiding further interventions. Thereby, two subsequent aborted myocardial infarctions could have been avoided.
Keywords
Acute coronary syndrome, Culprit lesion, Vulnerable plaque, Multivessel disease, Intravascular imaging
Introduction
In patients with stable coronary artery disease percutaneous coronary intervention (PCI) effectively improves symptoms in the absence of any effect on prognosis [1]. In contrast, in patients with acute coronary syndrome (ACS) PCI significantly reduces risk of acute myocardial infarction and death as compared to conservative, noninvasive strategy.
In ACS patients with multivessel disease the benefit of a complete coronary revascularization approach compared to the strategy of a culprit-only PCI is still under debate [2,3]. The interventionalist has to identify the culprit lesion responsible for symptoms. Contrast medium enhanced coronary angiography is the standard imaging technique for the evaluation of coronary vessels, but recognition of vulnerable plaques without significant reduction in luminal diameter is challenging and sometimes impossible.
Case Report
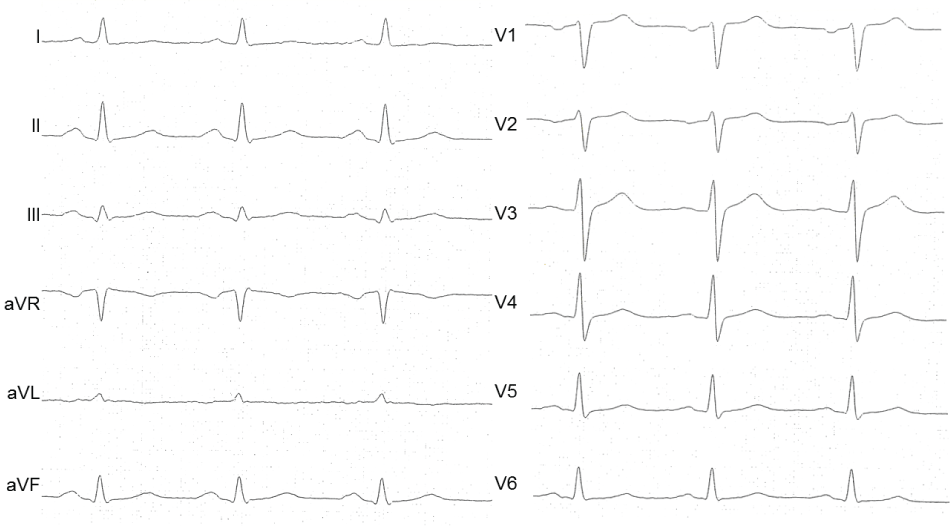
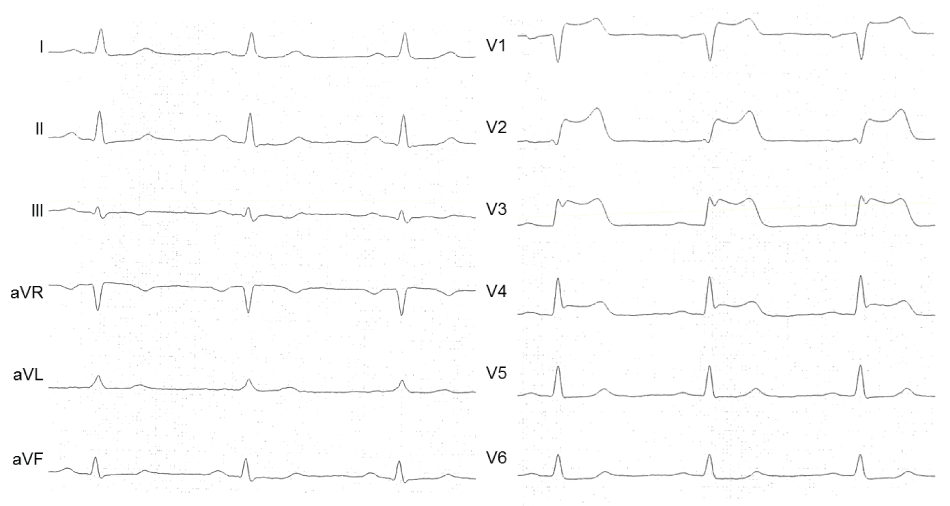
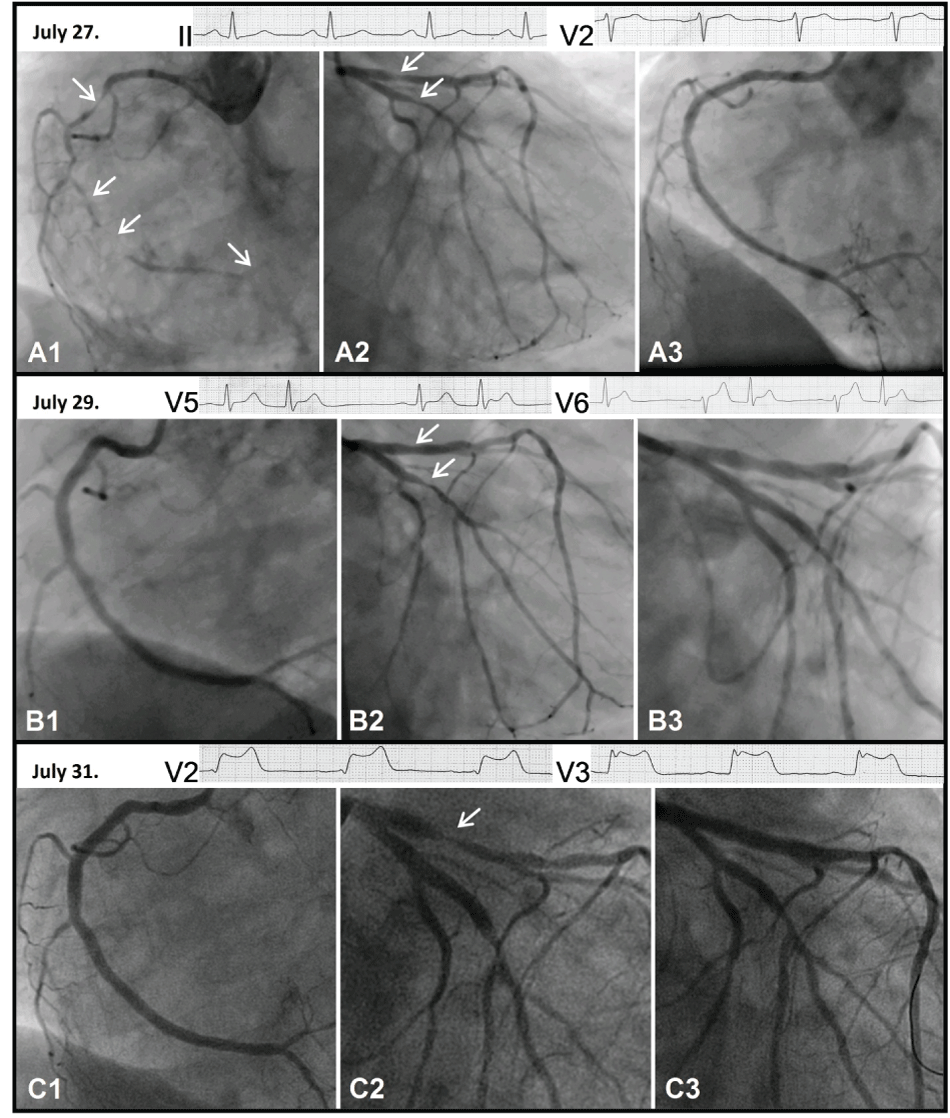
A 50-year-old man was admitted due to unstable angina. Recently a cardiac stress test indicated myocardial ischemia. In addition he reported symptoms of peripheral arterial occlusive disease and a prior nicotine abuse of 30 pack years, stopped 6 weeks ago. ECG (Figure 1), cardiac enzymes and echocardiography were normal at admission. An elevated C-reactive protein indicated an inflammatory response. He received aspirin, betablocker, adrenoceptor blocker, statin and clopidogrel. Coronary angiography at 15 hours after symptom onset revealed an occlusion of the right coronary artery (RCA) (Figure 2 A1), a 75% stenosis of the first posterolateral branch (PLA1) and a 50% stenosis of the left anterior descending artery (LAD) (Figure 2 A2). The RCA was recanalized and five drug eluting stents (DES) were implanted due to the long distance of the lesion (Figure 2 A3). The ECG remained unchanged (Figure 3).
.
Figure 2 A1: Right coronary artery, July 27.
Figure 2 A2: Left coronary artery, July 27.
Figure 2 A3: Right coronary artery after percutaneous coronary intervention, July 27.
Figure 2 A ECG II: Electrocardiogram lead II, July 27.
Figure 2 A ECG V2: Electrocardiogram lead V2, July 27.
Figure 2 B1: Right coronary artery, July 29.
Figure 2 B2: Left coronary artery, July 29.
Figure 2 B3: Left coronary artery after percutaneous coronary intervention, July 29.
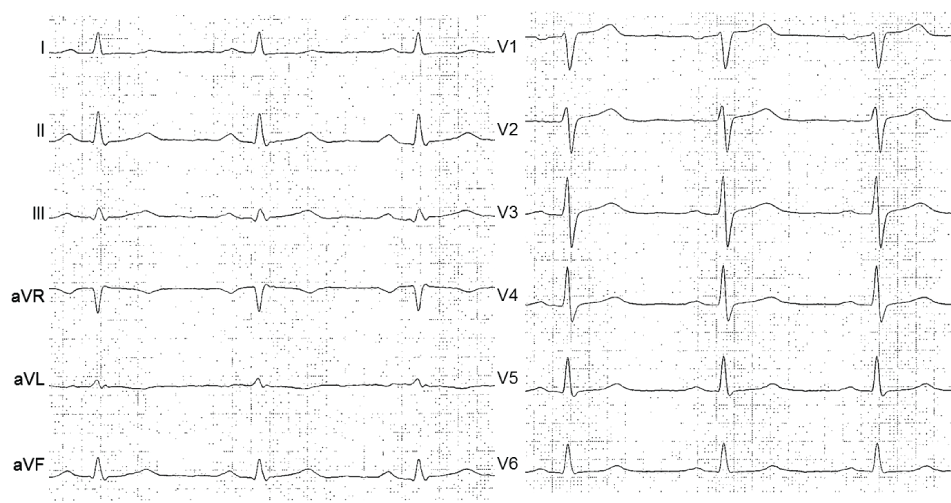
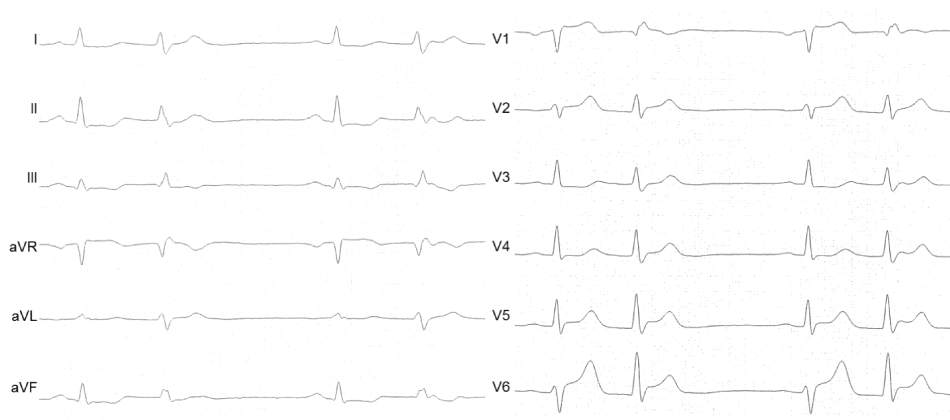
Figure 2 B ECG V5: Electrocardiogram lead V5, July 29.
Figure 2 B ECG V6: Electrocardiogram lead V6, July 29.
Figure 2 C1: Right coronary artery, July 31.
Figure 2 C2: Left coronary artery, July 31.
Figure 2 C3: Left coronary artery after percutaneous coronary intervention, July 31.
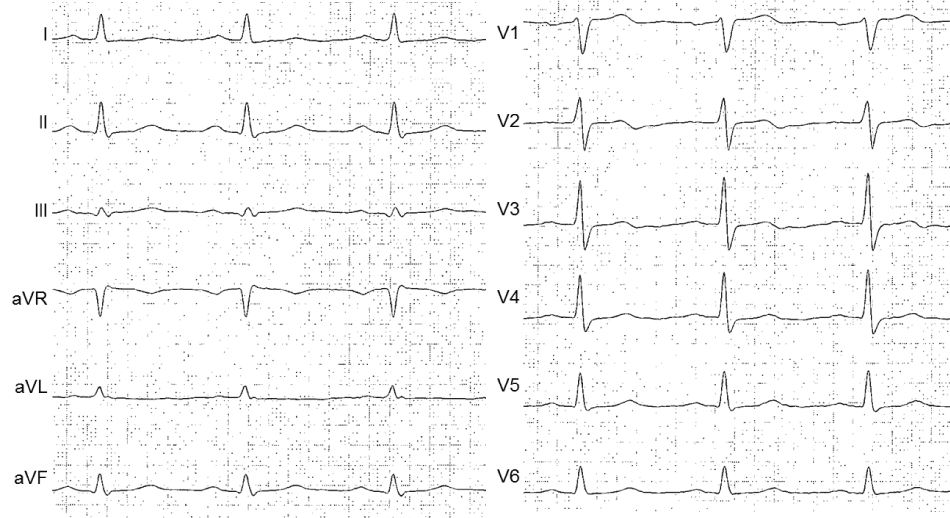
Figure 2 C ECG V2: Electrocardiogram lead V2, July 31.
Figure 2 C ECG V3: Electrocardiogram lead V3, July 31.
View Figure 2
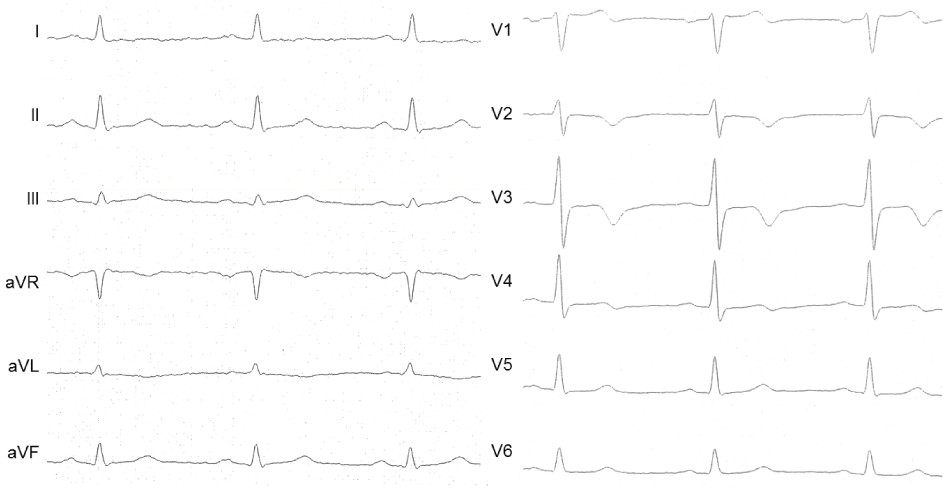
Two days later the patient complained about acute chest pain. The ECG showed ST-segment elevations in leads V5 and V6 (Figure 4). Repeated angiography ruled out an acute stent thrombosis (Figure 2 B1) but now revealed a high grade stenosis of the PLA1 (Figure 2 B2). One DES was implanted (Figure 2 B3), resulting in a complete resolution of the ST-segment elevations (Figure 5). This episode was not associated with a rise in cardiac enzymes.
The patient was discharged the next day and readmitted 48 hours later with recurrent angina and ST-segment elevations in leads V1-V4 (Figure 6). Angiography showed good results in stented vessels (Figure 2 C1 and Figure 2 C2) and subtotal occlusion of the LAD. Coronary spasm of the LAD was ruled out by intracoronary administration of 0.2 mg of Nitroglycerin. One DES was implanted (Figure 2 C3), again instantly normalizing the ECG (Figure 7) in the absence of a troponin release. No further cardiac events occurred during the following year and the patient remained free of symptoms.
Discussion
In this case vulnerable plaques in PLA1 and LAD initially escaped from recognition in coronary angiography since they were invisible. In the following days these plaques caused two distinct severe and life threatening cardiac events despite optimal medical treatment.
Even though q-waves were absent in the inferior leads of the ECG, the angiogram did not show relevant collateral blood flow to the occluded RCA and the recanalization of the RCA with a 0.014-inch angioplasty guidewire succeeded without major difficulty the occlusion of the RCA might have been chronic and therefore, the judgement of the interventionalist that the occluded RCA represents the culprit lesion might have been wrong. The unstable plaques of LAD and PLA1 resulted only in low grade coronary stenosis responsible for the insensitivity of coronary angiography. However, there are diagnostic tools available that help us to understand the coronary situation in multivessel disease and ACS and support clinical decision making.
In stable coronary artery disease additional evaluation of coronary lesions by fractional flow reserve (FFR) can identify their hemodynamic relevance. FFR-guided coronary intervention of those lesions that cause myocardial ischemia is associated with improved prognosis in the long term, driven by a reduction in the need of urgent revascularization [4]. However, in our case FFR of the stenotic lesions of LAD and PLA1- even if angiographically mild - might have uncovered significantly severe stenosis causing a reduction in poststenotic perfusion pressure.
In contrast, intravascular ultrasound with virtual histology (VH-IVUS) helps to differentiate between stable coronary lesions with higher amount of dense calcium and unstable lesions characterized by thin cap fibroatheroma (TCFA) and non-calcified or mixed deposits with higher amount of necrotic core. Especially the presence of plaque rupture and thrombus due to the contact of platelets with the highly thrombogenic necrotic core are associated with coronary events since thrombus formation leads to significant reduction of minimal lumen area and coronary obstruction [5]. Several studies demonstrated that the use of IVUS as additional diagnostic tool is feasible in patients presenting with ACS [6]. Kim, et al. reported a significantly reduced critical event free rate in coronary artery disease patients having IVUS-detected vulnerable plaques in non-target vessels [7]. This finding could be confirmed in the large PROSPECT study. About 50% of recurrent cardiovascular events in patients who initially presented with ACS were related to angiographically mild non-culprit lesions, most of them TCFA [8]. However, not every vulnerable plaque causes cardiovascular events and even ruptured plaques frequently remain asymptomatic [9]. IVUS imaging is limited by low special resolution and does not allow the direct measurement of fibrous cap thickness, which is < 65 mm in TCFA. Therefore, the surrogate of a confluent necrotic core of ≥ 10% in direct contact with the lumen and a plaque burden of ≥ 40% was used in most VH-IVUS studies to define TCFA [10]. Compared to the gold standard of histologic assessment VH-IVUS was shown to have a diagnostic accuracy of 74-82% for the detection of TCFA [11].
With 10-20 μm the image resolution is much higher with optical coherence tomography (OCT) allowing estimation of cap thickness. Additionally, in a comparison of both methods in patients with acute myocardial infarction the incidence of plaque rupture, fibrous cap erosion and intracoronary thrombus observed by OCT was significantly higher compared to IVUS [12]. On the other hand this technique is limited by its penetration of less than 2 mm and its inability to detect a large and deep lipid core, a pathologic feature of plaque prone to rupture.
Recently, Yonetsu, et al. performed a three vessel imaging study with OCT in 38 patients presenting in ACS. In 17 patients the culprit lesion was identified as a ruptured plaque, whereas in 21 patients the plaque found at the culprit site was non-ruptured with intact fibrous cap. Interestingly, patients with ACS caused by plaque rupture were found to have increased pancoronary vulnerability in non-culprit plaques indicating that plaque rupture causes a temporal biochemical milieu that may predispose to widespread plaque destabilization [13]. The same group found in a prospective cohort of ACS patients that those with plaque rupture or massive thrombus at the culprit lesion had a higher adverse cardiac event rate during a follow-up of 1.5 years compared to patients with intact fibrous cap at the culprit site. Therefore, the presence of plaque rupture at the culprit lesion might identify a group of ACS patients at very high risk for future events with the need for aggressive systemic or local treatment [14]. However, prospective clinical studies that tested the impact of pre-procedural use of intravascular imaging on intervention strategy and clinical outcome are lacking. The Preventive Implantation of Bioreserbable Vascular Scaffold on Stenosis with Functionally Insignificant Vulnerable Plaque (PREVENT) trial, is planned to solve the dilemma regarding how to treat functionally insignificant vulnerable plaques. A total of 1600 patients will be randomized to either optimal medical therapy of functionally insignificant vulnerable plaques (FFR ≥ 0.08) or implantation of bioresorbable vascular scaffolds. In this trial IVUS, OCT and near-infrared spectroscopy are used for the classification of vulnerable plaque with two or more of the following criteria: (i) minimal lumen area of < 4 mm2, (ii) plaque burden of > 70% by IVUS, (iii) large lipid-rich plaque on near-infrared spectroscopy, or (iv) TCFA defined by OCT or VH-IVUS. (NCT02316886)
This case clearly demonstrates that ACS is associated with pancoronary vulnerability and high risk for future events even in the short term. Intravascular imaging of the PLA and LAD during the first catheter might have been indicated to detect these unstable plaques. Image-guided further interventions in the PLA1 and LAD could have been avoided two aborted myocardial infarctions in three days.
Disclosures
The authors have no conflicts of interest to declare.
The authors followed the ClinMed author guidelines.
Supplementary Video
Supplementary File
References
-
Sedlis SP, Hartigan PM, Teo KK, Maron DJ, Spertus JA, et al. (2015) Effect of PCI on Long-Term Survival in Patients with Stable Ischemic Heart Disease. N Engl J Med 373: 1937-1946.
-
Qiao Y, Li W, Mohamed S, Nie S, Du X, et al. (2015) A comparison of multivessel and culprit vessel percutaneous coronary intervention in non-ST-segment elevation acute coronary syndrome patients with multivessel disease: a meta-analysis. EuroIntervention 11: 525-532.
-
Sardella G, Lucisano L, Garbo R, Pennacchi M, Cavallo E, et al. (2016) Single-Staged Compared With Multi-Staged PCI in Multivessel NSTEMI Patients: The SMILE Trial. J Am Coll Cardiol 67: 264-272.
-
De BB, Pijls NH, Kalesan B, Emanuele Barbato, Pim AL Tonino, et al. (2012) Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med 367: 991-1001.
-
Fujii K, Kobayashi Y, Mintz GS, Takebayashi H, Dangas G, et al. (2003) Intravascular ultrasound assessment of ulcerated ruptured plaques: a comparison of culprit and nonculprit lesions of patients with acute coronary syndromes and lesions in patients without acute coronary syndromes. Circulation 108: 2473-2478.
-
Choi SY, Mintz GS (2010) What have we learned about plaque rupture in acute coronary syndromes? Curr Cardiol Rep12: 338-343.
-
Kim SH, Hong MK, Park DW, Lee SW, Kim YH, et al. (2009) Impact of plaque characteristics analyzed by intravascular ultrasound on long-term clinical outcomes. Am J Cardiol 103: 1221-1226.
-
Stone GW, Maehara A, Lansky AJ, de Bruyne B, Cristea E, et al. (2011) A prospective natural-history study of coronary atherosclerosis. N Engl J Med 364: 226-235.
-
Maehara A, Mintz GS, Bui AB, Castagna MT, Canos D, et al. (2002) Morphologic and angiographic features of coronary plaque rupture detected by intravascular ultrasound. J Am Coll Cardiol 40: 904-910.
-
Garcia-Garcia HM, Gonzalo N, Regar E, Serruys PW (2009) Virtual histology and optical coherence tomography: from research to a broad clinical application. Heart 95: 1362-1374.
-
Obaid DR, Calvert PA, Gopalan D, Parker RA, Hoole SP, et al. (2013) Atherosclerotic plaque composition and classification identified by coronary computed tomography: assessment of computed tomography-generated plaque maps compared with virtual histology intravascular ultrasound and histology. Circ Cardiovasc Imaging 6: 655-664.
-
Kubo T, Imanishi T, Takarada S, Kuroi A, Ueno S, et al. (2007) Assessment of culprit lesion morphology in acute myocardial infarction: ability of optical coherence tomography compared with intravascular ultrasound and coronary angioscopy. J Am Coll Cardiol 50: 933-939.
-
Vergallo R, Ren X, Yonetsu T, Kato K, Uemura S, et al. (2014) Pancoronary plaque vulnerability in patients with acute coronary syndrome and ruptured culprit plaque: a 3-vessel optical coherence tomography study. Am Heart J 167: 59-67.
-
Yonetsu T, Lee T, Murai T, Suzuki M, Matsumura A, et al. (2016) Plaque morphologies and the clinical prognosis of acute coronary syndrome caused by lesions with intact fibrous cap diagnosed by optical coherence tomography. Int J Cardiol 203: 766-774.