International Journal of Critical Care and Emergency Medicine
Cerebral Air Embolism in Commercial Flights: A Potentially Fatal Complication of Intrathoracic Lesions
Beatriz Oyanguren*#, Araceli Alonso-Cánovas#, Alicia de Felipe and Jaime Masjuán
Department of Neurology, Hospital Universitario Ramón y Cajal, IRYCIS (Health Research Institute), Universidad de Alcalá, Madrid, Spain
#Beatriz Oyanguren and Araceli Alonso-Cánovas are joint first authors as they contributed equally to this manuscript.
*Corresponding author:
Beatriz Oyanguren Rodeño, Department of Neurology, Hospital Universitario Ramón y Cajal. Carretera de Colmenar km 9,100, 28034, Madrid, Spain, Tel: +3491-3368-000, Fax: +3491-336-9016, E-mail: beaoyan@hotmail.com
Int J Crit Care Emerg Med, IJCCEM-3-022, (Volume 3, Issue 1), Case Report; ISSN: 2474-3674
Received: July 10, 2016 | Accepted: February 14, 2017 | Published: February 18, 2017
Citation: Oyanguren B, Alonso-Cánovas A, de Felipe A, Masjuán J (2017) Cerebral Air Embolism in Commercial Flights: A Potentially Fatal Complication of Intrathoracic Lesions. Int J Crit Care Emerg Med 3:022. 10.23937/2474-3674/1510022
Copyright: © 2017 Oyanguren B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Cerebral air embolism (CAE) may exceptionally occur in patients with thoracic lesions during air travelling.
Methods: We present 5 new cases of CAE during flights and review precedent cases.
Results: Five patients with CAE were admitted to our Emergency Department (A-E). All experienced loss of consciousness (LOC). Chest radiograph showed thoracic bullae. Brain computed tomography (CT) showed subarachnoid bubbles (A), intraparenchymal bubbles (B), global hypoperfusion (C), or no abnormalities (D, E). With supportive treatment, four survived, while B died. Including ours, 18 cases of CAE during commercial flights have been reported. LOC and dyspnea were common. Thoracic cysts (9) and bullae (9) were found. Ten died. Five had experienced episodes suggesting CAE during previous flights. In patients with complete recovery, infarction, edema and intraparenchymal bubbles were absent in basal neuroimaging.
Conclusions: CAE may occur in patients with thoracic cysts while air travelling, causing typically neurological and respiratory symptoms early in the flight.
Keywords
Cerebral air embolism, Air travelling, Hyperbaric oxygen therapy, Thoracic cyst, Bullae
Abbreviations
CAE: Cerebral Air Embolism; LOC: Loss of Consciousness; CT: Computed Tomography; mRS: modified Rankin Scale; HBO: Hyperbaric Oxygen Therapy; MRI: Magnetic Resonance Image; COPD: Chronic obstructive pulmonary disease ; ED: Emergency Department ; DVT: Deep Venous Thrombosis; F: Female; M: Male; CHD: Coronary Heart Disease; AF: Atrial Fibrillation; APS: Antiphospholypid Syndrome; TIA: Transient Ischemic Attack; HTN: Hypertension; OB: Obesity; GCS: Glasgow Coma Scale; ECG: Electrocardiogram; TTE: Transthoracic Echocardiogram; NIHSS: National Institute of Health Stroke Scale; rTPA: Recombinant Tissue Plasminogen Activator
Introduction
Medical emergencies have been estimated to occur in 1 of every 604 flights. Particularly neurological problems linked to air traffic have been reported to be very frequent, specially strokes and seizures, which account for 2% and 5.8% of in-flight medical emergencies respectively [1,2].
Cerebral air embolism (CAE) is relatively common in diving and invasive procedures, but CAE linked to air traffic is considered exceptional: in the last three decades only thirteen cases have been reported. While flying, thoracic cysts or bullae may expand with the relative decrease in pressure inside the cabin (Boyle-Mariotte's law). Microrupture of these lesions may cause air bubbles to embolize to the systemic circulation [3,4].
Our tertiary university hospital is the reference centre for the international airport of the city. Eighteen per cent of the referrals from the airport are neurological problems [1]. We analyzed our five new cases of CAE during commercial flights as well as previously reported cases (Table 1) [5-17]. Patients' data are anonymously presented, following our institution's research committee's recommendations.
![]()
Table 1: Clinical features of CAE cases during commercial flights reported in scientific literature.
View Table 1
Cases Presentation
Case A
A 62-year-old cigarette smoker woman, with history of a transient ischemic attack unrelated to flying, experienced sudden dyspnea and LOC within 60 minutes of a flight. On admission, she was unconscious (Glasgow Coma Scale, GCS, 11), with forced oculocephalic deviation to the right, right central facial palsy, an unintelligible speech, and had a generalized tonic-clonic seizure. Brain CT scan, performed on admission to our Emergency Department (ED), 3 hours after symptoms' onset, showed three subarachnoid bubbles without parenchymal abnormalities, and chest radiograph a large thoracic bulla of 8 cm. Electrocardiogram (ECG) and transthoracic echocardiogram (TTE, including right to left shunt screening) were normal. A relative contraindication for Hyperbaric Oxygen Therapy (HBO) because of the presence of bullae was considered, and intravenous phenytoin and mechanical ventilation were initiated. A Magnetic Resonance Image (MRI) performed twenty-four hours later was unremarkable. After four days her condition improved; she was discharged without sequelae, with the recommendation of avoiding further air travelling.
Case B
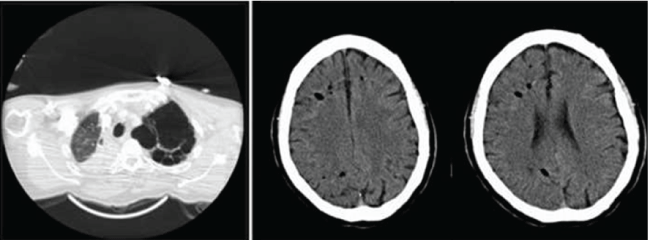
A 47-year-old man, cigarette smoker with emphysematous chronic obstructive pulmonary disease (COPD), hypertension, hypercholesterolemia, and obesity (OB), experienced sudden dyspnea and progressive LOC in the second hour of a flight. On admission, he was unconscious (GCS 3), with low oxygen saturation, and had several generalized tonic-clonic seizures. ECG and TTE were normal. Chest CT yielded several apical bullae on the left lung apex and complete atelectasis of the right lung (Figure 1a). Admission brain CT scan, performed three hours after the onset of symptoms, showed multiple intraparenchymal air bubbles in both cerebral hemispheres (Figure 1b and Figure 1c). A relative contraindication for HBO because of the presence of COPD was considered, and supportive treatment with intravenous phenytoin and mechanical ventilation were initiated. Cranial MRI performed twenty-four hours later showed large ischemic areas in both cerebral hemispheres and cerebellum, as well as generalized edema. Progressive deterioration of his neurologic condition and infectious complications led to decease in the sixth week.
.
Figure 1: Radiologic findings in case B. Plain thoracic CT showing emphysematous bullae on the left lung apex (1a) and plain brain CT showing air bubbles on both cerebral hemispheres (1b, 1c).
View Figure 1
Case C
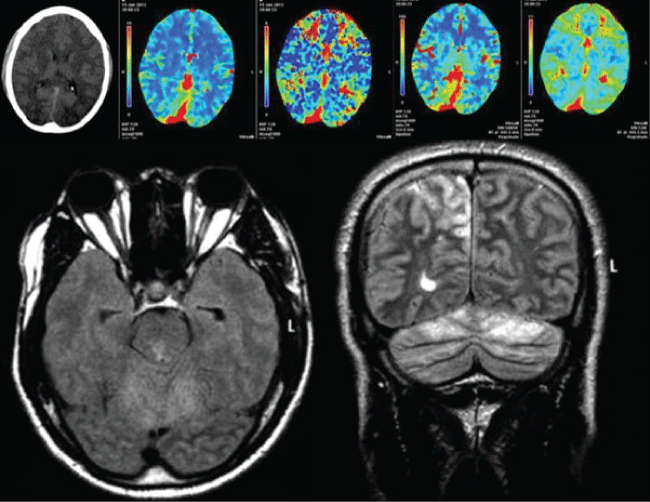
A 33-year-old non smoker man had been diagnosed with an emphysematous bulla on the left lung fifteen years ago. Screening for alpha-1antitripsine deficiency was negative and the patient had refused surgery. He had experienced three episodes of dizziness, headache and chest pain early in the course of previous flights. In the current episode, he experienced chest pain, headache, and transient LOC in the first hour of a flight. He was drowsy but responsive, and referred loss of vision in both eyes. On admission, visual examination yielded absence of mirror tracking with preserved response of pupils to light, and temperature was high at 37.5 °C. Chest X ray showed left lung pneumothorax. A plain brain CT scan (six hours after symptoms' onset) was unremarkable. He presented generalized tonic-clonic seizures, followed by severe agitation, which led to sedation, treatment with phenytoin, mechanical ventilation and placement of a thoracic tube. Chest CT scan showed multilobar pneumonia of the right lung and massive left lung pneumothorax. A multimodal CT (eighteen hours after symptom onset) disclosed diffuse brain edema and generalized hypoperfusion, with normal angiography. MRI performed on the 3rd day of admission showed bilateral hyperintense lesions in T2 and FLAIR sequences in parietal and occipital lobes, cerebellum, pons and medulla (Figure 2). These abnormalities were consistent with endothelial damage due to posterior circulation diffuse cerebral air embolism. HBO was contraindicated because of the presence of pneumothorax. After fifteen days he gradually improved. Control MRI performed one month later showed near resolution of the abnormalities, with two foci of abnormal signal in subcortical right parietal lobe and left cerebellar hemisphere. He was discharged with mild ataxia of the left upper limb, on antiepileptic therapy, and advised not to travel by plane until his left pulmonary bulla was surgically treated.
.
Figure 2: Radiologic findings in case C. Brain CT scan performed 18 hours from the onset showing diffuse edema on both cerebral hemispheres (2a) multimodal CT scan, (2b) cerebral blood volume, (2c) time to peak, (2d) cerebral blood flow, (2e) mean transit time, showed preserved volume with reduced cerebral blood flow and increased transit time, interpreted as global hypoperfusion, FLAIR (2f) and T2 (2g) sequences of initial cranial MRI showing bilateral hyperintensities in right parietal lobe, mesencephalon and cerebellar hemispheres.
View Figure 2
Case D
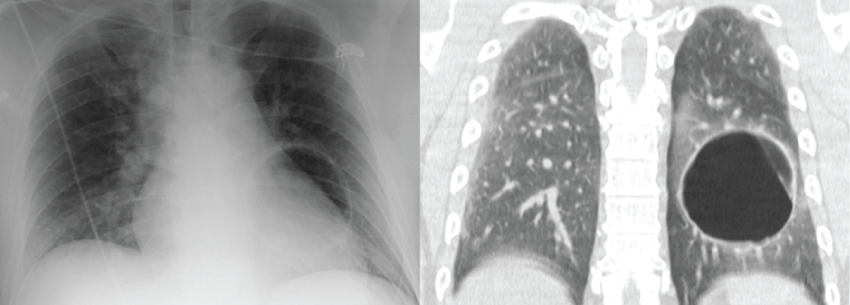
A 45-year-old non smoker man, diagnosed of a small left lung cyst ten years ago, experienced LOC, visual loss and right hemiparesis during a commercial flight two hours after takeoff. On admission, one hour later, he was drowsy but responsive, disorientated, with incoherent speech. He had right hemiparesis (3+1 in the National Institute of Health Stroke Scale -NIHSS-) and referred loss of vision in both eyes. Temperature was 36 °C. Chest radiograph showed a left lung cavity (Figure 3a). Plain brain CT scan and CT angiogram of circle of Willis (3.5 hours after symptoms' onset) were both unremarkable. He received intravenous recombinant tissue plasminogen activator (rTPA) upon the initial diagnosis of posterior territory acute stroke, and was then admitted to the Stroke Unit. Along the following twenty-four hours, level of consciousness, orientation, speech, strength and vision improved. Thoracic CT scan showed a giant bulla on the left lung (Figure 3b). Diffusion-weighted MRI of the brain (twenty-four hours after symptom onset) showed small acute infarctions in multiple locations: left frontal subcortical semioval centre, bilateral cortical parasagital prerrolandic areas and bilateral occipital cortex. Gadolinium-enhanced MRI also showed diffuse patchy frontal, parietal and occipital leptomeningeal enhancement. Troponin T levels were 2.1 ng/ml on admission (normal values < 0.5 ng/ml) and normalized within the following 6 hours. EKG and TTE were normal and there were no clinical symptoms of heart disease or deep venous thrombosis (DVT). EEG showed unspecific diffuse encephalopathy and CSF analysis was unremarkable. He was discharged four days later, asymptomatic. Thoracic surgery was organized as an outpatient and he was advised not to travel by plane until his bulla was surgically treated.
.
Figure 3: Radiologic findings in case D. Chest radiograph showed a left lung cavity (3a) and plain thoracic CT showed a giant bulla on the left lung (3b).
View Figure 3
Case E
A 37-year-old male experienced LOC and seizures during a commercial flight soon after takeoff. On admission, he was drowsy but able to follow commands, and had left hemiparesis. Temperature was 36 °C. Chest radiograph showed a right lung cavity. Plain brain CT scan (performed four hours after the onset of symptoms) was unremarkable. He was admitted to the Intensive Care Unit due to LOC, where he also suffered several tonic clonic seizures. He received antiepileptic treatment with intravenous valproic acid and phenytoin, with good response. Thoracic CT scan showed a cyst on the right lung. MRI of the brain, performed fourty-eight hours after symptom onset, showed an acute infarction on the left cerebellum. EEG and CSF analysis were both unremarkable. On the third day of admission he was transferred to the Department of Neurology where he experienced good recovery. He was discharged eight days later, asymptomatic. Surgery of the cyst was arranged as an outpatient, advising not to travel by plane until his lung lesion was removed.
Discussion
CAE is increasingly recognized as an infrequent but serious complication of commercial flights. With our five cases, the number of patients reported rises to eighteen [5-17] (Table 1). Middle age travelers (mean age 53.6 years, range 19-71), most of them without known pulmonary disease, were affected.
Onset of symptoms early in the flight was common. There was LOC in all patients, seizures in nine, and focal neurological signs in eight (hemiparesis, aphasia, vision loss), as well as accompanying respiratory symptoms (dyspnea, chest pain) in nine. In 8 cases (6-9, 12, 13, 16, D), patients experienced embolic phenomena in other locations, mainly coronary heart disease.
Except for four cases with normal basal neuroimaging [7, Case C, D, E], CT and MRI scans showed bubbles (either within the parenchyma or the subarachnoid space), edema and/or infarctions. Bubbles were only seen in early performed CT scans, and may be missed in late CT scans, as in our cases C-E and other cases previously reported [5-7,15]. Ten patients died and two remained moderate-severely disabled. Six patients had a complete or near complete recovery: in all of them infarctions, edema and intraparenchymal bubbles were absent on admission CT neuroimaging [Case A, C, D, E, 7, 17]. The presence of these abnormalities in early performed neuroimaging may thus be an unfavorable prognostic factor. Subarachnoid bubbles seen in early performed CT scans, however, were not related with bad prognosis [Case A, 17]. On the other hand, absence of intraparenchymal bubbles alone did not guarantee a good prognosis [5,6,15], since the presence of bubbles may also be related with the timing of initial neuroimaging.
Interestingly, multimodal CT in one of our patients (Case C), and also in a previously reported patient [12], showed global hypoperfusion without angiographic abnormalities. Furthermore, the MRI of Case D showed small acute strokes in multiple cerebral locations, without any arterial obstruction, as well as diffuse leptomeningeal enhancement. All these findings are probably related to diffuse endothelial damage.
Hyperbaric Oxygen (HBO) therapy was applied only in five cases [7,10,12,13,15]. In our patients, a relative (Cases A, B, D, E) or absolute (Case C) contraindication for HBO, were the reasons for withholding this therapy. In the remaining, the reasons were unavailability [6], worsening of the patient's condition [8,9,14], diagnosis delay [16], and were not reported in four [5,11,17]. HBO is considered beneficial in air embolism. However, its most feared complications include rupture of air-filled cavities due to changes in pressure, pneumothorax and systemic air embolism. HBO may then be rejected in presence of thoracic cysts and severe lung disease. Thus, given CAE in patients with thoracic bullae during air travelling is a rare condition, it lacks evidence of HBO's efficacy and security in this specific setting [18,19].
It is also noteworthy that five patients [Case C, 8, 10, 12, 15] had experienced neurological problems (LOC, stroke, seizures, syncope, headache) in previous flights, and six patients had been previously diagnosed with thoracic lesions [Case C, D, 6, 8, 10, 15]. Three patients did not survive the latter event of CAE [8,10,12] and one remained severely impaired [15].
This underscores the importance of ruling out CAE in passengers with neurological problems on board, especially when the onset is early in the flight and LOC and respiratory symptoms occur.
The demonstration of thoracic cysts or bullae, as well as the presence of bubbles, edema and infarctions in neuroimaging, help confirm diagnosis. Although the outcome is generally poor, the absence of edema and intraparenchymal abnormalities in basal neuroimaging may indicate a better prognosis.
As thoracic cystic lesions bear an uncertain risk for CAE, it may be advisable to discourage individuals with known thoracic cysts or bullae from air traveling, at least until these lesions are surgically treated.
Acknowledgements
Both Beatriz Oyanguren Rodeño and Araceli Alonso Cánovas had full access to all of the data in the manuscript and take responsibility for the integrity and accuracy of the data. All authors contributed substantially to the work to take responsibility for the content and have read and approved the final version of the manuscript.
Conflict of Interest
This study was not industry-sponsored. The authors have no conflicts of interest or disclosures to report.
References
-
Alonso-Cánovas A, de Felipe-Mimbrera A, González-Valcárcel J, García-Barragán N, Corral I, et al. (2011) Neurology at the airport. J Neurol Neurosurg Psychiatry 82: 981-985.
-
Nable JV, Tupe CL, Gehle BD, Brady WJ (2015) In-flight medical emergencies during commercial travel. N Engl J Med 373: 939-945.
-
Muth CM, Shank ES (2000) Gas embolism. N Engl J Med 342: 476-482.
-
Heckmann JG, Lang CJ, Kindler K, Huk W, Erbguth FJ, et al (2000) Neurologic manifestations of cerebral air embolism as a complication of central venous catheterization. Crit Care Med 28: 1621-1625.
-
Neidhart P, Suter PM (1985) Pulmonary bulla and sudden death in a young airplane passenger. Intensive Care Med 11: 45-47.
-
Zaugg M, Kaplan V, Widmer U, Baumann PC, Russi EW (1998) Fatal air embolism in an airplane passenger with a giant intrapulmonary bronchogenic cyst. Am J Respir Crit Care Med 157: 1686-1689.
-
Closon M, Vivier E, Breynaert C, Duperret S, Branche P, et al. (2004) Air embolism during an aircraft flight in a passenger with a pulmonary cyst: a favorable outcome with hyperbaric therapy. Anesthesiology 101: 539-542.
-
Almeida FA, Desouza BX, Meyer T, Gregory S, Greenspon L (2006) Intrapulmonary bronchogenic cyst and cerebral gas embolism in an aircraft flight passenger. Chest 130: 575-577.
-
Almeida NJS, Schuller D (2007) Cerebral air embolism resulting from an airplane flight. J Resp Dis 28: 304-308.
-
Edwardson M, Wurth D, Lacy JM, Fink J, Becker K (2008) Cerebral air embolism resulting in fatal stroke in an airplane passenger with a pulmonary bronchogenic cyst. Neurocrit Care 10: 218-221.
-
Salameh J (2010) A 62-year-old woman with cerebral artery air embolism during commercial air travel. Neurologist 16: 136-137.
-
Arnaiz J, Marco de Lucas E, Piedra T, Arnaiz Garcia ME, Patel AD, et al. (2011) In-flight seizures and fatal air embolism: the importance of a chest radiograph. Arch Neurol 68: 661-664.
-
Machicado JD, Davogustto G, Burgeois S, Kaldis P, Jani PP, et al. (2013) Fatal Air Embolism: A rare complication of bronchogenic cysts in an airplane passenger. Am J Respir Crit Care Med 188: 249-250.
-
Yeung JT, Ma JK, Mak YF, Lam VS (2013) Fatal cerebral air embolism related to an air flight. Hong Kong Med J 19: 352-353.
-
Desgranges FP, Cour M, Hernu R, Delafosse B, Argaud L (2013) Cerebral air embolism during an aircraft flight in a passenger with an air-filled lung cavity associated with remote lung surgery. J Thorac Cardiovasc Surg 146: e18-20.
-
Saliou G, Leblanc PE, Cauquil C, Theaudin M, Ract C, et al. (2014) Sudden Loss of Consciousness during a Flight. Cerebrovasc Dis 37: 470-471.
-
Gudmundsdottir JF, Geirsson A, Hannesson P, Gudbjartsson T (2015) Major ischaemic stroke caused by an air embolism from a ruptured giant pulmonary bulla. BMJ Case Rep.
-
Grim PS, Gottlieb LJ, Boddie A, Batson E (1990) Hyperbaric oxygen therapy. JAMA 263: 2216-2220.
-
Blanc P, Boussuges A, Henriette K, Sainty JM, Deleflie M (2002) Iatrogenic cerebral air embolism: importance of an early hyperbaric oxygenation. Intensive Care Med 28: 559-563.





