International Journal of Cancer and Clinical Research
A Randomized Double Blinded Study of Ganoderma Lucidum (Lingzhi) in Salvage Setting of Recurrent Gynecologic Cancer
Prapaporn Suprasert1*, Chatchawann Apichartpiyakul2, Choompone Sakonwasun2, Pimonphan Nitisuwanraksa3 and Rochana Phuackchantuck3
1Division of Gynecologic Oncology, Department of Obstetrics and Gynecology, Chiang Mai University, Thailand
2Department of Microbiology, Chiang Mai University, Thailand
3Research Administration Section, Chiang Mai University, Thailand
*Corresponding author:
Prapaporn Suprasert, Division of Gynecologic Oncology, Department of OB&GYN, Faculty of Medicine, Chiang Mai University, 110 Inthawarorod Road, Chiang Mai, Thailand, Tel: +66-53-945552-5, Fax: +66-53-894424, E-mail: psuprase@gmail.com
Int J Cancer Clin Res, IJCCR-2-021, (Volume 2, Issue 3), Research Article; ISSN: 2378-3419
Received: June 26, 2015 | Accepted: August 26, 2015 | Published: August 28, 2015
Citation: Suprasert P, Apichartpiyakul C, Sakonwasun C, Nitisuwanraksa P, Phuackchantuck R (2015) A Randomized Double Blinded Study of Ganoderma Lucidum
(Lingzhi) in Salvage Setting of Recurrent Gynecologic Cancer. Int J Cancer Clin Res 2:021. 10.23937/2378-3419/2/3/1021
Copyright: © 2015 Suprasert P, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Lingzhi is a widely used anti-cancer herbal medicine in Asian countries with limited knowledge of the efficacy especially in gynecologic cancer patients. This study was conducted to compare the efficacy of Lingzhi in the forms of water extract type and spore type with a placebo control in a salvage setting of gynecologic cancer treatment. Sixty gynecologic cancer patients who failed at least two regimens of chemotherapy were randomly divided equally to ingest 6000 mg/day of Lingzhi in form of water extract or 6000 mg/day of Lingzhi in the form of spore or placebo for 12 weeks. The patients were evaluated for toxicity, immunomodulation and quality of life every four weeks for five times. All three groups were similar in their basic clinical data. About half of the patients withdrew from the study with the major reason of rapid progression of disease. Finally, 11,8, and 9 patients were evaluated in water extract, spore and placebo arms, respectively. The best response in the present study was stable disease that achieved 38.1% in the water extract arm, 50% in the spore arm and none in the placebo arm (P = 0.06). The one-year overall survival was 63.6% in the water extract arm, 60% in the spore arm and 44% in the placebo arm (P = 0.217). The majority of hematologic and non hematologic effects, the mean immunomodulatory level and the quality of life were not significantly different for each arm in each visit. In conclusion, Lingzhi in either water extract or spore form seemed to control the disease in a salvage setting of gynecologic cancer patients without adverse effects.
Keywords
Ganoderma lucidum, Gynecologic cancer, Salvage treatment
Introduction
"Lingzhi" is the common name of Ganoderma lucidum, a well-known herbal medicine that has been widely used in Asian countries especially in China as health promotion for many years. It is a woody Basidiomycetes mushroom in the Ganodermataceae or Aphyllophorales family [1,2]. In addition, many previous publications identified the anti-cancer effects of Lingzhi in various cancers such as breast, colorectal and stomach cancer especially for in vitro studies [2-5]. The bioactive agents in Lingzhi consist of triterpenoids, polysaccharides, nucleotides, sterols, steroids, fatty acids, proteins/peptides and trace elements [4]. All these substances revealed many biological activities, including anti- tumor, immunomodulation, antiviral, antihepatitis, antioxidant, antihypertensive and antidiabetic processes that should be beneficial for treating cancer [4]. The two most widely used preparations of Lingzhi in the herb market were water extracts from the fruiting body and the spores. Both types were commonly used as anti- cancer drugs without conclusive evidence of benefits [1]. Furthermore, clinical studies on the effects of Lingzhi especially in gynecologic cancer patients were very limited. To identify the benefit of Lingzhi in this type of cancer, a randomized double blind study was conducted in our institute to compare the clinical outcomes, the toxicity, the immunomodulation and also the quality of life between Lingzhi given in the form of water extract, spores and placebo. Because of the initial trial of study, we decided to recruit only the recurrent gynecologic cancer patients who were in the salvage setting. Lingzhi in this study was planted in the Royal Project at Amphur Chiangdoa in the Chiang Mai Province located in the North of Thailand. The standard extraction process was prepared by the staff of the Faculty of Pharmacy, Chiang Mai University. This data will enhance the clinical data of the efficacy of Lingzhi in gynecologic cancer management.
Materials and Methods
A randomized double blind controlled trial was conducted in 2011 after approval by the local Ethics Committee to compare the efficacy of Lingzhi in the form of water extract and spore preparation with a placebo that was composed of a high dose of vitamin C in gynecologic cancer patients who had disease progression after receiving at least two chemotherapy regimens. All invited patients had an ECOG performance status score of less than two with normal bone marrow, hepatic and renal function and no clinical symptoms of intestinal obstruction. Finally, twenty patients were recruited in each group. After informed consent, the patients were randomly allocated to one of the three groups: water extract Lingzhi, spore Lingzhi and placebo. All the patients and the investigators were blinded. The patients received one type of the drug in powder form in similar packaging. The following amounts were present: Lingzhi in the form of body fruit extract 1,000 mg per pack, Lingzhi spores preparation 1,000 mg per pack and vitamin C 200 mg per pack. Participants were instructed to ingest one package with 200 ml of distilled water one hour before a meal twice a day on day one and two then to increase the dosage to two packages of drug ingested with 200 ml of distilled water before meals two times a day on day three and four. After that the dosage would be increased to three packages ingested in the same way for the remainder of the 12 weeks as long as there were no serious side effects. The patients were given appointments to check for toxicity along with a physical and pelvic examination and a self-report for evaluating their quality of life by using Thai-Modified Function Living Index Cancer Questionnaire every four weeks for five times. They were given the studied drug at visits one through three and had a CT-scan at the metastasis sites within one month before the start of the study and at visit five. The immunomodulatory tests consisting of serum CD4 -T cells, CD8 -T cells, Natural killer (NK) cell 9, and gamma interferon (IFN) were evaluated by blood tests at visits 1,2, 4 and 5. The toxicities were by evaluated by using the Common Terminology Criteria for Adverse Events version 3.0 and the responses were assessed by using the RECIST criteria [6,7]. The patients were withdrawn from the study when their disease progressed severely, had unacceptable toxicity, missed the follow up period for longer than two weeks or were unable to ingest the studied drug.
The clinical characteristics, the quality of life and the laboratory results of each group were noted and compared by using Chi-square, Fisher's Exact and one way ANOVA test. The overall survival was defined as the time between the month of the initial ingestion of the investigation drug and the month the patient died or the last contact. This survival time was estimated by the Kaplan-Meier method and compared among each group by using the log rank test. All statistical analysis was carried out using the SPSS for Windows program (Version 17.0, Chicago, II,USA). Statistical significance was set at P value less than 0.05.
Results
Twenty patients who met the inclusion criteria were enrolled in each arm. The clinical data was noted in Table 1. The mean age, mean body surface area, the initial diagnosis, the history of surgery or radiation, the underlying disease and the mean number of previous chemotherapy regimens were well balanced in each group. About half of all the studied patients were withdrawn from our study with most of them in the spore group. The reasons for withdrawal from the study were presented in Table 2. Nearly 70% of the reasons for withdrawal from the study were the prompt worsening of their performance status from rapid progression and need for hospitalization soon after starting the investigation drug. Only two cases withdrew due to the inability to ingest the investigated drug. One case in the water extract type could not tolerate the bitter taste. The other case in the placebo arm refused to ingest the investigation drug due to severe discomfort from marked abdominal distension.
![]()
Table 1: Clinical Data (N = 60)
View Table 1
![]()
Table 2: Withdrawal Reasons (N = 32)
View Table 2
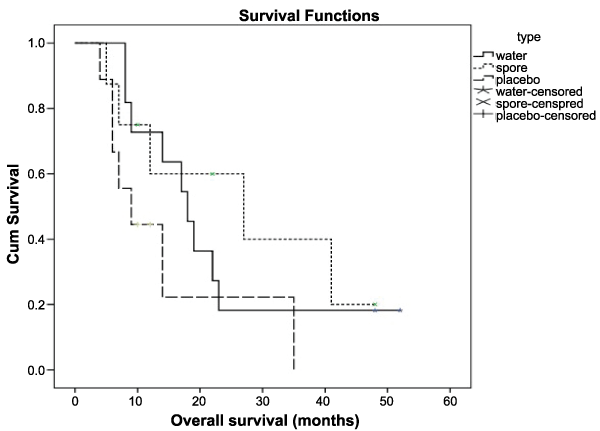
Finally, there were 11,8 and 5 patients who completed the protocol in the water extract, spore and placebo arms, respectively. Table 3 showed the outcome of seven patients who responded best with stable disease. Three patients ingested water extract and the other four patients ingested the spore type. All seven patients were initially diagnosed with ovarian cancer except one that had uterine cancer. The response rates were 42.9% in the water extract group and 50.0% in the spore arm group which represented 25.0% of all the studied patients. The different response rates were nearly significant with a P-value 0.06. With the median follow up of 13 months and a range of 4-52 months, the median overall survival was 17 months in the water extract arm, 22 months in the spore arm and nine months in the placebo arm. Both the response rate and median overall survival rates were not significantly different in each arm as shown in Table 3 and Figure 1.
.
Figure 1: The overall survival compared water extract, spore and placebo
At median FU 13.00 mo (4-52 months)
The median overall survival: water extract 17 mo, spore 22 mo, placebo 9 mo
1 year overall survival: water extract 63.6%, spore 60%, placebo 44.4%
P = 0.217
View Figure 1
![]()
Table 3: Outcomes of Each Arm
View Table 3
The details of each case except the latter two cases were recently published [8]. To complete the data of stable cases, the additional details of the two latest stable cases were briefly presented here. Both cases were diagnosed as ovarian cancer. One case was 59 years old with the major recurrence site at the vaginal stump. The histology was carcinosarcoma. She previously received four regimens of chemotherapy and one course of pelvic radiation. This woman entered the study after completing the last treatment at 13 months and received Lingzhi in form of water extract and died of her disease with an overall survival of 22 months. The other case was 59 years old who presented with pulmonary metastasis after resistance to three regimens of chemotherapy. Her final histology was transitional cell carcinoma. She was recruited to this study after finishing the last course of chemotherapy at 12 months. This second case was in the spore arm group and still alive with her disease until now after ten months.
.
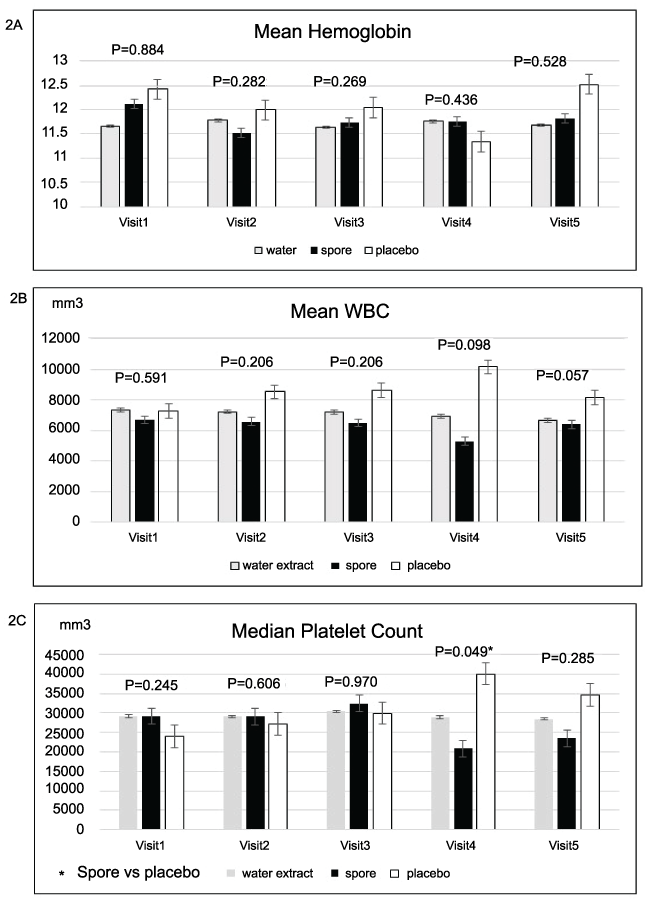
Figure 2: Mean complete blood count compared of 3 groups in each visit; no significant difference of mean Hb, WBC in each visit (figure IA, IB). However, mean platelet count of placebo group was significant higher than spore group in visit 4 (Figure IC)
View Figure 2
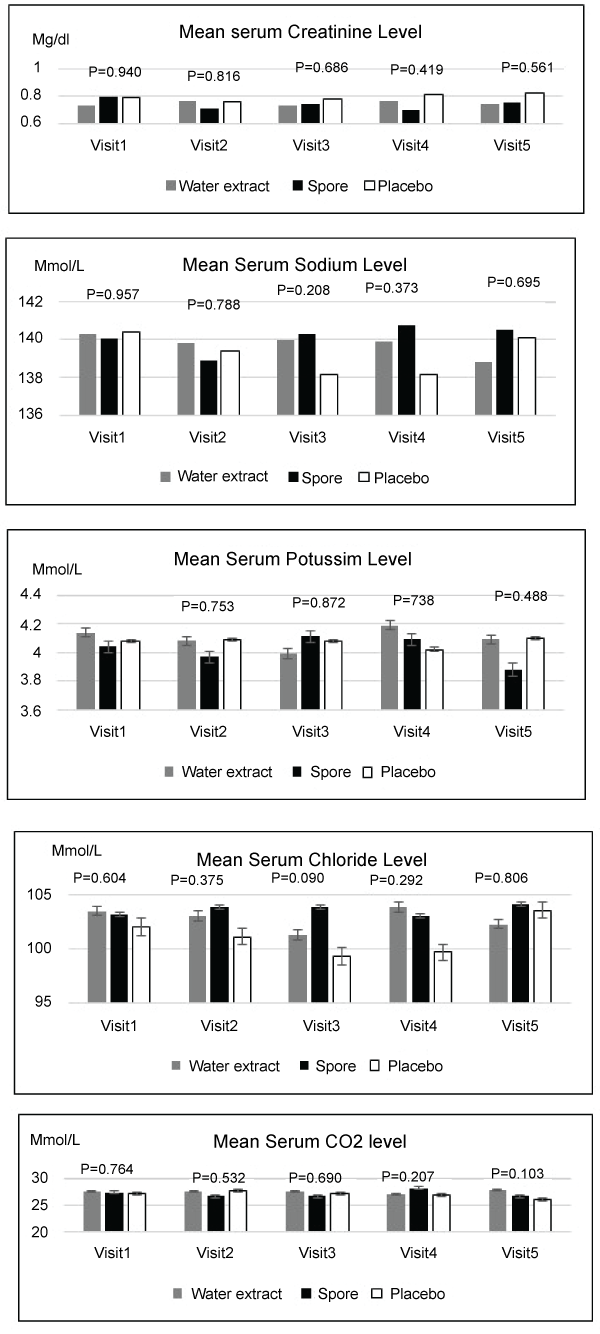
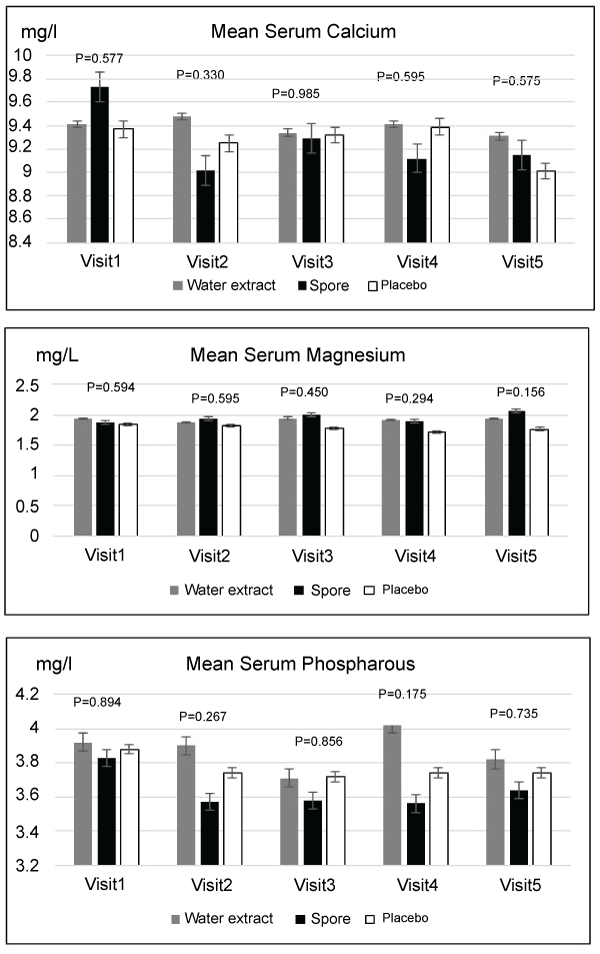
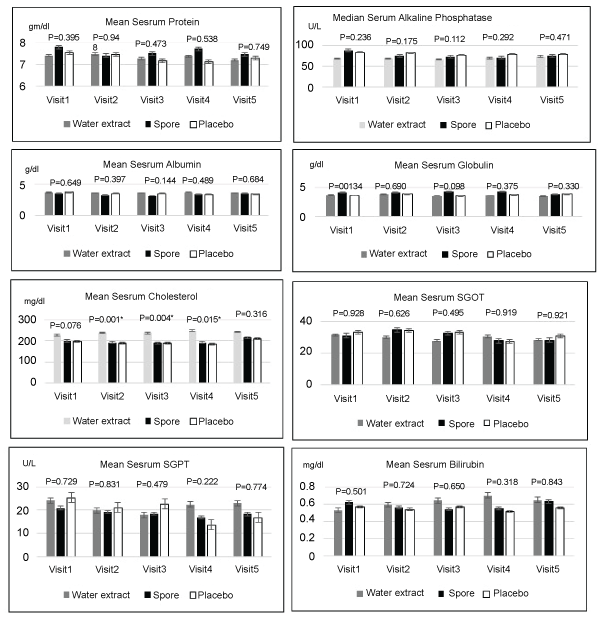
Regarding toxicity evaluated by blood test, mean levels of complete blood count, creatinine, electrolyte, calcium, magnesium, phosphorous and liver function tests were not significantly different among these three arms in each visit. Only the platelet count was significantly higher in the placebo group when compared to the spore group in visit four and the serum cholesterol was found to be significantly higher in the water extract group when compared to the spore and placebo arms. The details of these data were showed in Figures 2-5. Other side effects of the investigated drugs were not found except for one case in the water extract arm who felt that the investigated drug was too bitter to ingest.
.
Figure 4: Mean serum calcium, magnesium, phosphorous level compared of 3 groups in each visit (no significant difference)
View Figure 4
.
Figure 5: Mean serum liver function test compared of 3 groups in each visit; no significant difference in all except cholesterol that showed significant higher in water extract group when compared with placebo and spore group at visit 2,3,4 (Post Hoc)
View Figure 5
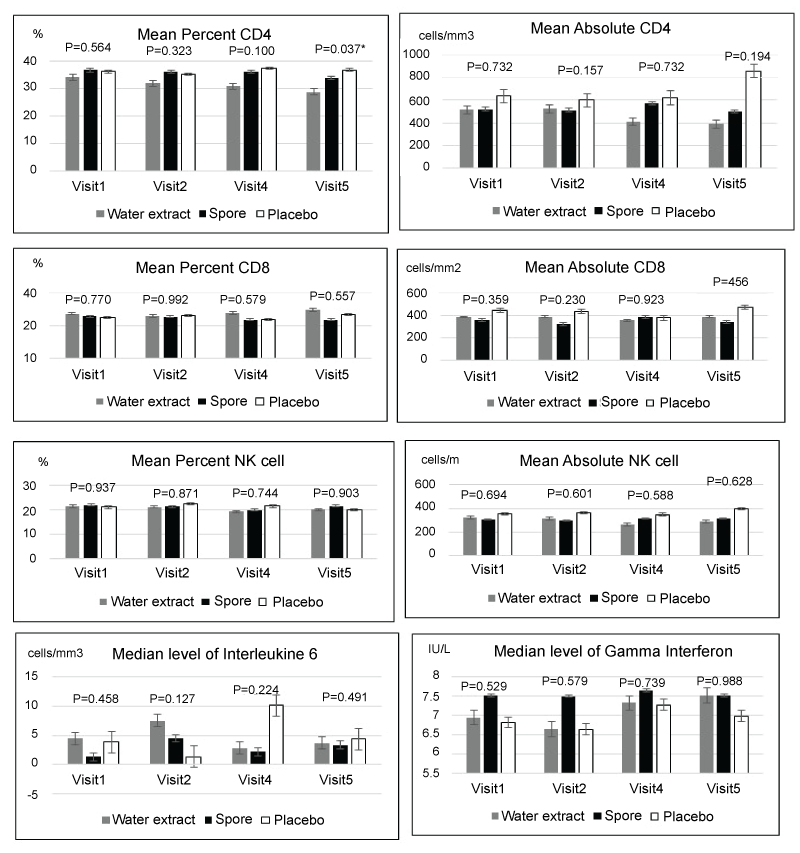
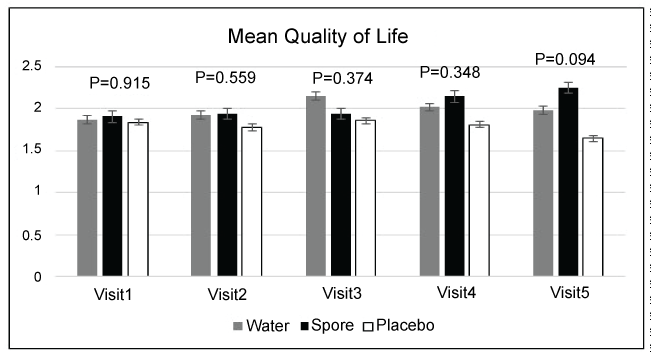
Concerning immunomodulation, the details were presented in Figure 6. The mean level of absolute CD4, percent CD8, absolute CD8, percent NK cell, absolute NK cell, the median level of interleukine 6 and gamma interferon were not significant among each group in each visit. Only the percent of CD4 was significantly higher in the placebo group when compared with water extract in visit 5. In addition the mean level of quality of life was also not significantly different among the three groups in each visit as shown in Figure 7.
.
Figure 6: Mean/median level of immune system compared of 3 groups in each visit; no significant difference in all except %CD 4 that showed significant higher in placebo group when compared water extract in visit 5
View Figure 6
.
Figure 7: Qualify of life of 3 groups in each visit with no significant difference
View Figure 7
Discussion
The salvage treatment of gynecologic cancer including chemotherapy, hormone, radiation and complementary therapy [9,10]. Lingzhi is one of the frequent Chinese herb using with this purpose. It was illustrated the immune modulation and anti-oxidation in animal model by inhibition of tumor growth and metastasis without causing adverse events [11,12]. The present study was the first randomized research regarding the clinical efficacy of Lingzhi in a setting of salvage treatment of gynecologic cancer patients. In the past, Zhao et al. [4] studied the effect of Lingzhi in spore form in a human ovarian surface epithelial cell line. They identified multiple antitumor effects such as decreasing tumor cell numbers, inhibiting colony formation/cell migration/spheloid formation, inducing cell cycle arrest at the G2/M phase and also enhancing cisplatin efficacy. Although our study included all types of gynecologic cancer patients, most of the studied patients were diagnosed with ovarian cancer. We found that all cases that response received Lingzhi either in the form of water extract or spores. Although the best response was only stability of disease, in the salvage setting, stable disease was also valuable. Furthermore, patients in both the water extract and spore arms tended to achieve a higher percentage of one-year survival and longer median overall survival than in the placebo arm. However, due to the low number of patients who completed five visits who could have a final outcome evaluated, clinical statistical significance was not reached.
The dosage of 3000 mg two times a day in this present study was the dosage that resembled a previously utilized dosage in other advanced stage cancer treatments that administered 1800 mg three times a day for 12 weeks [13,14]. We adjusted the dosage to two times a day for the reason of a greater convenience for ingesting the investigated drug.
Concerning toxicity of Lingzhi, both water extract and spore preparation did not have significant hematology and non-hematology adverse effects when compared to the placebo in all items except platelet count and cholesterol. The present study found the mean platelet count was highest in the placebo arm in visit 4. The higher stimulation of platelet formation in the placebo arm might be from the nature of tumor progression [15]. The high level of serum cholesterol in the arm of water extract that were significantly higher when compared with the placebo and spore group at visits 2,3,4 was similar to the data from Cochrane Database Systemic Review that showed no clinically significant reduction in total cholesterol with ingestion of Lingzhi [16]). Another Cochrane Database Systemic Review that specified only cancer treatment also not found major toxicity of Lingzhi from their review [17]. Thus, cancer patients who tolerated ingestion of Linzhi could be used it without definite contraindication.
Regarding the effect of immune system in the present study, this study could not see any significantly higher immune response in the arms of water extract and spores. These results were non-similar to the previous reports that found the significant increasing of many immune systems such as CD4, CD8, NK cells, etc. [13,14,18] . This might be from the different forms of Lingzhi and studied patients. However, it must be noted that during the ingested-Lingzhi visits, the immune system of our patients was slightly elevated when compared to the non-ingested visits. In addition, the quality of life of our patients also tended to improve in those patients who received Lingzhi. These results resembled the previous Cochrane Database Systemic Review result that found improvement in the quality of life in cancer patients who received Lingzhi [16].
The limitations of the present clinical trial were the low number of patients who completed the protocol in each arm. The major reasons for the withdrawal of patients were marked progression of the disease that often presented with marked ascites, bowel obstruction and ileus. Thus, most withdrawal patients could not ingest the investigated drug. However, the present clinical trial was conducted as randomized double blind trial with placebo control and studied many aspects including the response, the survival, the toxicity, the immune system and also the quality of life of Lingzhi in cancer patients. The result of the present study was reliable due to the study design.
Finally, Lingzhi in both forms, water extract and spore, when used as salvage treatment seem to control the disease and slightly improve the immune system in gynecologic cancer patients with minimal side effects that did not affect their quality of life. Due to these positive results, further research of the possibility of adding Lingzhi use with standard chemotherapy in gynecologic cancer patients must continue.
Acknowledgements
We wish to thank the Thai Traditional Medical Knowledge Fund, the Department for Development of Thai Traditional and Alternative Medicine in the Ministry of Public Health, Thailand for financial support for this project.
Conflict of Interest
We have no conflicts of interest.
References
-
Jin X, Ruiz Beguerie J, Sze DM, Chan GC (2012) Ganoderma lucidum (Reishi mushroom) for cancer treatment. Cochrane Database Syst Rev 6: CD007731.
-
Martínez-Montemayor MM, Acevedo RR, Otero-Franqui E, Cubano LA, Dharmawardhane SF (2011) Ganoderma lucidum (Reishi) inhibits cancer cell growth and expression of key molecules in inflammatory breast cancer. Nutr Cancer 63: 1085-1094.
-
Wu G, Qian Z, Guo J, Hu D, Bao J, et al. (2012) Ganoderma lucidum extract induces G1 cell cycle arrest, and apoptosis in human breast cancer cells. Am J Chin Med 40: 631-642.
-
Zhao S, Ye G, Fu G, Cheng JX, Yang BB, et al. (2011) Ganoderma lucidum exerts anti-tumor effects on ovarian cancer cells and enhances their sensitivity to cisplatin. Int J Oncol 38: 1319-1327.
-
Suarez-Arroyo IJ, Rosario-Acevedo R, Aguilar-Perez A, Clemente PL, Cubano LA, et al. (2013) Anti-tumor effects of Ganoderma lucidum (reishi) in inflammatory breast cancer in in vivo and in vitro models. PLoS One 8: e57431.
-
Cancer Therapy Evaluation Program (2006) Common Terminology Criteria for Adverse Events, Version 3.0.
-
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, et al. (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45: 228-247.
-
Suprasert P, Apichartpiyakul C, Sakonwasun C, Nitisuwanraksa P, Phuackchantuck R (2014) Clinical characteristics of gynecologic cancer patients who respond to salvage treatment with Lingzhi. Asian Pac J Cancer Prev 15: 4193-4196.
-
NCCN clinical practice guidelines in oncology (NCCN guidelines).
-
Chase DM, Gibson SJ, Sumner DA, Bea JW, Alberts DS (2014) Appropriate use of complementary and alternative medicine approaches in gynecologic cancers. Curr Treat Options Oncol 15: 14-26.
-
Kimura Y, Taniguchi M, Baba K (2002) Antitumor and antimetastatic effects on liver of triterpenoid fractions of Ganoderma lucidum: mechanism of action and isolation of an active substance. Anticancer Res 22: 3309-3318.
-
Weng CJ, Chau CF, Yen GC, Liao JW, Chen DH, et al. (2009) Inhibitory effects of ganoderma lucidum on tumorigenesis and metastasis of human hepatoma cells in cells and animal models. J Agric Food Chem 57: 5049-5057.
-
Gao Y, Zhou S, Jiang W, Huang M, Dai X (2003) Effects of ganopoly (a Ganoderma lucidum polysaccharide extract) on the immune functions in advanced-stage cancer patients. Immunol Invest 32: 201-215.
-
Gao YH, Dai XH, Chen GL,Ye J, Zhou S (2003) A randomized, placebo-controlled, multicenter study of Ganoderma lucidum (W.Curt.:Fr.) Lloyd (Aphyllophoromycetideae) polysaccharides (Ganopoly®) in patients with advanced lung cancer. Int J Med Mushr 5: 369-381.
-
Orellana R, Kato S, Erices R, Bravo ML, Gonzalez P, et al. (2015) Platelets enhance tissue factor protein and metastasis initiating cell markers, and act as chemo attractants increasing the migration of ovarian cancer cells. BMC Cancer 15: 290.
-
Klupp NL, Chang D, Hawke F, Kiat H, Cao H, et al. (2015) Ganoderma lucidum mushroom for the treatment of cardiovascular risk factors. Cochrane Database Syst Rev 2: CD007259.
-
Jin X, Ruiz Beguerie J, Sze DM, Chan GC (2012) Ganoderma lucidum (Reishi mushroom) for cancer treatment. Cochrane Database Syst Rev 6: CD007731.
-
Chen X, Hu ZP, Yang XX, Huang M, Gao Y, et al. (2006) Monitoring of immune responses to a herbal immuno-modulator in patients with advanced colorectal cancer. Int Immunopharmacol 6: 499-508.