International Journal of Cancer and Clinical Research
Fascin is Expressed in Basal-Liketype Triple Negative Breast Cancer Associated with High Malignant Potential in Japanese Women
Hiromichi Tsuchiya1, Akiko Sasaki1*, Yuko Tsunoda2, Masafumi Takimoto3, Terumasa Sawada4, Seigo Nakamura4, kentaro Iijima1 and Katsuji Oguchi1
1Department of Pharmacology, School of Medicine, Showa University, Japan
2Kameda Medical Center, Breast Center, Japan
3Department of Pathology, Showa University School of Medicine, Japan
4Department of Surgery, Division of Breast Surgery, Showa University School of Medicine, Japan
*Corresponding author:
Akiko Sasaki, Department of Pharmacology, School of Medicine, Showa University, 1, 3, 4) Hatanodai 1-5-8, Shinagawa-ku, Tokyo 142-8666, Japan, Tel: +81-3-3784-8125, E-mail: sakiko@med.showa-u.ac.jp
Int J Cancer Clin Res, IJCCR-2-035, (Volume 2, Issue 5), Original Article; ISSN: 2378-3419
Received: October 19, 2015 | Accepted: December 05, 2015 | Published: December 07, 2015
Citation: Tsuchiya H, Sasaki A, Tsunoda Y, Takimoto M, Sawada T, et al. (2015) Fascin is Expressed in Basal-Liketype Triple Negative Breast Cancer Associated with High Malignant Potential in Japanese Women. Int J Cancer Clin Res 2:035. 10.23937/2378-3419/2/5/1035
Copyright: © 2015 Tsuchiya H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
No molecular targeted drug has been developed for poor-prognostic triple-negative breast cancer (TNBC) because it has no hormone or human epidermal growth factor receptor 2 (HER2) sensitivity. In addition, TNBC has recently been sub classified into 6 types. In this study, we analyzed the expression of Fascin, an actin-binding protein associated with breast cancer, in 301 invasive breast cancer samples. Of these, 32 were identified as TNBC and were classified into the basal-like type and other non-basal-like subtypes. We also investigated the association of Fascin expression with clinic pathological factors. In the 301 breast cancer samples, the Fascin-positive rate was 56% (95% confidence interval [CI95%], 49-63) in luminal A, 14.3% ([CI95%], 0-29) in luminal B (HER2-negative), 52.9% ([CI95%], 36-70) in luminal B (HER2-positive), 31.3% ([CI95%], 15-47) in HER2-positive (non-luminal), and 81.3% ([CI95%], 68-95)in TNBC samples. In the 32 TNBC samples, the frequency of positivity for Fascin staining significantly increased for a Ki-67 index of 30% or above (P = 0.00013). The Fascin expression rate in the basal-like type of TNBC was significantly higher than that in the non-basal-like types (P = 0.0056). These findings showed the strong relationship between Fascin expression and the basal-like type of TNBC. It is expected to be expressed in Fascin-positive cells in proliferative basal-like type TNBC. The results suggest that Fascin plays a role as a marker of malignant potential in basal-like type TNBC and enhanced Fascin expression resulted in high cell growth in TNBC in Japanese women. Thus, Fascin may be a new candidate therapeutic target for TNBC in Japanese women.
Keywords
Fascin, Ki-67, Triple Negative Breast Cancer (TNBC), Basal-like type, Immunohistochemical staining
Introduction
Breast cancer is the most common cancer in women worldwide and is classified into various subtypes based on gene expression analysis. Triple-negative breast cancer (TNBC) is a subtype that does not express estrogen receptor (ER), progesterone receptor (PgR), and human epithelial growth factor receptor (HER2) [1]. TNBC accounts for 10-15% of breast cancer cases and has poor prognosis accompanied with distant metastasis in 30% of cases [2]. Owing to the lack of a specific target, hormone therapy and anti-HER2 treatments are ineffective, making the treatment of this breast cancer subtype difficult. Moreover, the rates of metastasis to visceral organs, as well that to the brain, and recurrence are high in TNBC and the prognosis is poor. Since TNBC is not sensitive to hormone therapy, no molecular target drug has been established, and the development of new therapy is strongly needed.
TNBC has recently been subclassified into 6 types: the basal-like (BL) types, i.e., BL1 and BL2, and the non-basal-like types, i.e., the immunomodulatory (IM), mesenchymal (M), mesenchymal stem like (MSL), and the luminal androgen receptor (LAR) types [3]. To establish molecular targeted therapy for each subtype, it is critical to identify the characteristics of and targets for the individual subtypes.
Fascin, a major actin filament-crosslinking protein encoded by the FSCN1 gene, increases cell motility by reorganizing the distribution and activity of the actin cytoskeleton. In tumor cells, Fascin tightly bundles actin filaments to form finger-like plasma membrane protrusions, invadopodia, which are associated with invasion. Fascin expression is upregulated in various epithelial malignancies, including high-grade breast carcinoma. Fascin is an actin-binding protein that localizes in tumor margins; it forms membrane protrusions while guiding the tip of the cell [4]. In addition, inverse correlations among Fascin expression, ER, and PgR [5] and the correlation of Fascin with poor-prognostic TNBC [6] have been reported. A recent study showed a highly significant correlation between Fascin expression and TNBC, especially in African-American women [7].
The aim of this study was to examine Fascin as a marker of high malignant potential in basal-like TNBC in Japanese women and investigate the relationship of Fascin expression with Ki-67 expression in different TNBC subtypes by performing immunohistochemical analysis.
Materials and Methods
Samples
Tumor specimens excised from 301 primary invasive ductal breast cancer patients at the department of surgery, division of breast surgery, Showa University School of Medicine, in 2011 were used. The use of samples was approved by the ethics committee of Showa University School of Medicine (application number: 1,374). In this study, patients with multifocal, metachronal, or bilateral breast cancers were excluded; the patients included had not received chemotherapy or hormone therapy before surgery.
Immunohistochemical staining
All samples were fixed in 10% formalin for 24 hours, and serial sections with 4 μm thickness were affixed to and dried on silane-coated slide glasses. After deparaffinization, preparations for Fascin staining were treated with 0.3% H2O2 for 10 minutes to remove endogenous peroxidase, followed by blocking with nonspecific blocking reagent (X0909, DAKO, Glostrup, Denmark) for 5 minutes. They were then reacted with the primary antibody, anti-Fascin antibody (1:100, DAKO), for 1 hour followed by reaction with Envision/HRP-labeled secondary antibody for 30 minutes. The color was developed using DAKO DAB Liquid System. For CK5/6 (1:100, DAKO), antigen activation by heat treatment was used before the reaction with primary antibody. EGFR was treated with proteinase K, which was followed by the use of the Envision method (diluted antibody, NICHIREI BIOSCIENCES Inc., Tokyo, Japan). For Ki-67, antigen activation treatment was applied before reaction with primary antibody (1:100, DAKO).
The intensity was scored according to the overall appearance as judged at different powers of magnification. The staining was estimated using the German immunoreactive score technique as follows [8]: slides were assessed for both the proportion of cells stained and staining intensity. Proportions were scored as follows: 0, no cell staining; 1, 1-25% stained cells; 2, 26-50% stained cells; 3, 50-75% stained cells; or 4, > 75% stained cells. The intensity was scored as follows: 0, no staining; 1, weak; 2, moderate; or 3, strong staining. The two scores were simplified into negative or positive. A final score > 3 was regarded as positive and < 3 was regarded as negative. The scoring was done manually by more than three individuals in a blinded manner.
Statistical analysis
Data were analyzed using the chi square test and P < 0.01 was regarded as significant.
Results
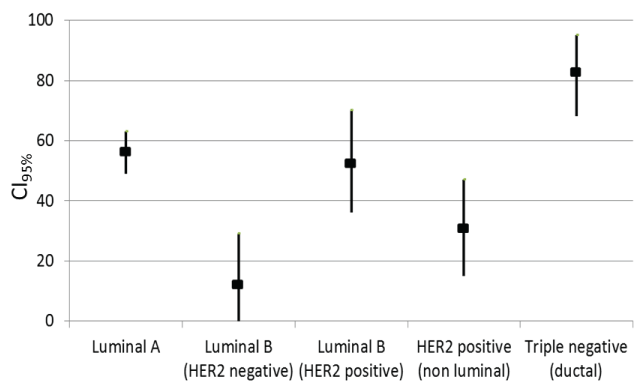
The Fascin-positive rates were 56% (confidence intervals [CI95%], 49-63), 14.3% ([CI95%], 0-29), 52.9% ([CI95%], 36-70), 31.3% ([CI95%], 15-47) and 81.3% ([CI95%], 68-95) in the luminal A, luminal B (HER2-negative), luminal B (HER2-positive), and HER2-positive non-luminal types and TNBC, respectively, showing that the rate was high in TNBC (Figure 1). Of the 32 TNBC patients, 14 and 12 patients younger than 50 years and 50 years old or older, respectively, were Fascin-positive. The results for the Fascin-positive and negative groups did not differ significantly by age (P = 0.17). For 16 and 10 patients, tumors with diameters lesser than 2 cm and 2 cm or larger, respectively, were Fascin-positive (P = 0.21). The Ki-67 indices were below 30% and 30% or higher in 1 and 25 Fascin-positive cases, respectively. The Fascin-positive rate was significantly higher in masses with a 30% or higher Ki-67 index (P = 0.00013). Regarding the indices of TNBC sub classification, CK5/6 was positive in 14 and 12 Fascin-positive cases, respectively. CK5/6 results did not significantly differ between the Fascin-positive and negative groups (P = 0.099). EGFR plus Fascin-positivity were seen in 19 cases. EGFR-negative plus Fascin-positive results were obtained in 7 cases. EGFR expression did not significantly differ between the Fascin-positive and negative groups (P = 0.27). The subtype was basal-like and non-basal-like in 23 and 3 Fascin-positive cases, respectively. The Fascin-positive rate was significantly higher in the basal-like subtype (P = 0.0056; Figure 2, Table 1).
.
Figure 1: The Fascin-positive rates were 56% (confidence intervals [CI95%], 49-63), 14.3% ([CI95%], 0-29), 52.9% ([CI95%], 36-70), 31.3% ([CI95%], 15-47) and 81.3% ([CI95%], 68-95) in the luminal A, B (HER2-negative), and B (HER2-positive) types, HER2-positive non-luminal type, and TNBC, respectively.
View Figure 1
![]()
Table 1: Associations of clinicopathology factors with Fascin expression of TNBC.
View Table 1
Discussion
Fascin-1 is a 55-kDa actin-binding protein, and its expression has been reported to be similar between embryogenesis and cancer histogenesis. It is also a tissue-specific protein expressed in nerves, follicular dendritic cells of lymphoid tissue, and cells of the basal lamina of stratified squamous epithelium from the embryogenesis to adult stages in humans [7]. It is used as a prognostic marker of oral squamous cell carcinoma [9] because its expression in invasive adenocarcinoma of the endocervix [10], melanocytes in the oral cavity, and highly malignant tumors of the uterus and human lung tissue has been confirmed. The possibility of Fascin-1 being a prognostic factor of breast cancer has been reported because it was expressed at a higher level in breast cancer stages T0-1, 2, and 3 than in the normal mammary gland, and the expression level was positively correlated with the survival rate [11]. Furthermore, it was reported that Fascin represents a potentially significant marker for targeted therapy, especially in African-American women [7]. However, to our knowledge, no such studies had been conducted on Fascin expression in Japanese women.
.
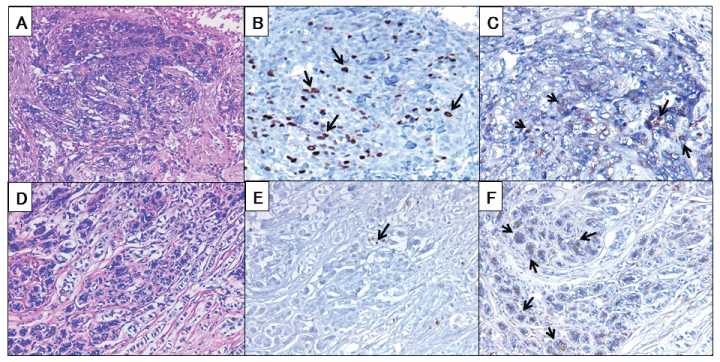
Figure 2: Immunohistochemical staining. (A) HE stain oftriple-negative breast cancer. (B) Ki-67 stain of triple-negative breast cancer. (C) Fascinstain oftriple-negative breast cancer. Fascin-positive in cytoplasm of tumor cells. (D) HE stain of luminal A. (E) Ki-67 stain of luminal A. (F) Fascin stain of luminal A. Fascin found in tumor cells shown by arrows (magnification × 200).
View Figure 2
Ki-67 is a protein present during all active phases of the cell cycle (G1, S, G2, and M) and is a cellular marker for proliferation. Ki-67 is strictly associated with cell proliferation. The Ki-67 index is used to determine high malignant potential of breast cancer in many institutions. The Ki-67 index evaluation varies across institutions and is not unified across settings [12]. In our 32 TNBC patients, the Fascin-positive rate was higher in basal-like type TNBC than the other subtypes, strongly suggesting that basal-like type TNBC and Fascin expression are associated. The Ki-67 index was significantly higher in the Fascin-positive TNBC, and it appeared that the enhanced Fascin expression was related with high cell growth activity in TNBC in Japanese women. Our current results indicate that Fascin expression is as useful as the Ki-67 index as an indicator of high malignant potential.
In 2011, Pietenpol et al. subclassified TNBC into 6 subtypes [13]. Of these, the BL1 and BL2 types account for 50% of all cases, and CK5/6 and EGFR are positive. We divided the patients into the basal-like (BL1, BL2) and non-basal-like (IM, M, MSL, LAR) types and investigated the association with Fascin expression. The Fascin-positive rate was higher in the basal-like than non-basal-like types. CK5/6 and EGFR were expressed at specifically high levels in the basal-like type. Basal-like type cell growth is very high, and cell cycle-related genes and DNA damage response genes (BRCA1 and BRCA2) are highly expressed [14,15]. Fascin, which binds to the actin cytoskeleton, is expressed during cell proliferation. Our results showed that Fascin was highly expressed in the basal-like type.
Macromolecular keratin CK5/6 (58 kDa, 56 kDa) is expressed in basal cells, mesotheliomas, and squamous cells in the active proliferation period. Analysis of the expression of one or more high molecular weight basal cytokeratins (CK5/6, CK14, and CK-17) and/or epidermal growth factor receptor (EGFR) is most commonly accepted as a method for identifying basal-like differentiation. EGFR is a receptor tyrosine kinase (RTK) of the ErbB family. Multiple signaling pathways, such as PI3K/AKT, mitogen-activated protein kinase (MAPK), and Wnt/β-catenin, are activated by EGFR to enhance proliferation, survival, invasion, and metastasis of cancer cells. Expression of EGFR is frequently associated with TNBC and has been viewed as a promising therapeutic target. Unfortunately, the therapeutic efficacy of EGFR-targeting agents has been disappointing in breast cancer, suggesting that other molecular drivers also contribute to the malignancy [14,15]. EGFR is expected to be expressed in Fascin-positive cells of proliferative basal-like type TNBC. EGFR is present on the cell membrane surface, expressed at a high level in cancer tissue, and is resistant to hormone therapy, chemotherapy, and radiotherapy, being related to poor prognosis and treatment resistance [16,17]. Based on these findings, it was clarified that EGFR is specifically expressed in Fascin-positive tissue of malignant basal-like type TNBC associated particularly with markedly proliferative tissue. This study suggested that enhanced Fascin expression was related to high cell growth activity of TNBC in Japanese women. It is expected to be expressed in Fascin-positive cells of proliferative basal-like type TNBC. Fascin may play a role as a marker of malignant potential in basal-like type TNBC and may be a new therapeutic candidate for TNBC in Japanese women.
References
-
Dey N, Smith BR, Leyland-Jones B (2012) Targeting basal-like breast cancers. Curr Drug Targets 13: 1510-1524.
-
Cleator S, Heller W, Coombes RC (2007) Triple-negative breast cancer: therapeutic options. Lancet Oncol 8: 235-244.
-
Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, et al. (2011) Identification of human triple-negative breast and preclinical models for selection of targeted therapies. J Clin Invest. 121: 2750-2767.
-
Grothey A, Hashizume R, Sahin A, McCrea PD (2000) Fascin, an actin-bundling protein associated with cell motility, is upregulated in hormone receptor negative breast cancer. Br J Cancer. 83: 870-873.
-
Nese N, Kandiloglu AR, Simsek G, Lekili M, Ozdamar A, et al. (2010) Comparison of the desmoplastic reaction and invading ability in invasive ductal carcinoma of the breast and prostatic adenocarcinoma based on the expression of heat shock protein 47 and fascin. Anal Quant Cytol Histol 32: 90-101.
-
Chen L, Yang S, Jakoncic J, J. Jillian Zhang, Xin-Yun Huang (2010) Migrastatin analogues target fascin to block tumourmetastasis. Natere. 15: 1062-1066.
-
Esnakula AK, Ricks-Santi L, Kwagyan J, Kanaan YM , DeWitty RL, et al. (2014) Strong association of fascin expression with triple negative breast cancer and basal-like phenotype in African-American women. J Clin Pathol 67: 153-160.
-
Remmele W, Schicketanz KH (1993) Immunohistochemical determination of estrogen and progesterone receptor content in human breast cancer. Computer-assisted image analysis (QIC score) vs subjective grading (IRS). Pathol Res Pract 189: 862-866.
-
Chen SF, Yang SF, Li JW, Nieh PC, Lin SY, et al. (2007) Expression of fascin in oral and oropharyngeal squamous cell carcinomas has prognostic significance - a tissue microarray study of 129 cases. Histopathology. 51: 173-183.
-
Stewart CJ, Crook M, Loi S (2012) Fascin expression in endocervical neoplasia: correlation with tumour morphology and growth pattern. J Clin Pathol 65: 213-217.
-
Zhang Y, Tong X (2010) Expression of the actin-binding proteins indicates that cofilin and fascin are related to breast tumour size. J Int Med Res. 38: 1042-1048.
-
Dowsett M, Nielsen TO, Ahern R, Bartlett J, Coombes RC, et al. (2011) Assessment of Ki67 in breast cancer: recommendations from the International Ki67 in Breast Cancer working group. J Natl Cancer Inst 16: 1656-1664.
-
Lehmann BD, Bauer JA, Ches X, Sanders ME, Chakravarthy AB, et al. (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest 121: 2750-2767.
-
Wiese DA, Thaiwong T, Yuzbasiyan-Gurkan V, Kiupel M (2013) Feline mammary basal-like adenocarcinomas: a potential model for human triple-negative breast cancer (TNBC) with basal-like subtype. BMC Cancer 13: 403.
-
Maeda T, Nakanishi Y, Hirotani Y, Fuchinoue F, Enomoto K, et al. (2015) Immunohistochemical co-expression status of cytokeratin 5/6, androgen receptor, and p53 as prognostic factors of adjuvant chemotherapy for triple negative breast cancer. Med Mol Morphol.
-
Nicholson RI, Gee JM, Harper ME (2001) EGFR and cancer prognosis. Eur J Cancer 37 Suppl 4: S9-15.
-
Cheang MC, Voduc D, Bajdik C, Leung S, McKinney S, et al. (2008) Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin Cancer Res 14: 1368-1376.





