International Journal of Cancer and Clinical Research
Breast Cancer in Surgical Site: Unusual Presentation of an Ectopic Milk Line Malignancy
Zsolt Varga1, Miklós Török2, Éva Sebő 3, Judit Tóth4 and Dezső Tóth1*
1Department of General Surgery, Kenézy Teaching Hospital, Hungary
2Kenézy Breast Center, Kenézy Teaching Hospital, Hungary
3Department of Pathology, Kenézy Teaching Hospital, Hungary
4Department of Oncology, Kenézy Teaching Hospital, Hungary
*Corresponding author:
Dezső Tóth, Department of General Surgery, Kenézy Teaching Hospital, 2-26 Bartók Street, Debrecen 4043, Hungary, Tel: +36-30-9388-867, Fax:+36-52-511-797, E-mail: detoth@gmail.com
Int J Cancer Clin Res, IJCCR-3-042, (Volume 3, Issue 1), Case Report; ISSN: 2378-3419
Received: November 11, 2015 | Accepted: January 29, 2016 | Published: January 30, 2016
Citation: Varga Z, Török M, Sebő É, Tóth J, Tóth D (2016) Breast Cancer in Surgical Site: Unusual Presentation of an Ectopic Milk Line Malignancy. Int J Cancer Clin Res 3:042. 10.23937/2378-3419/3/1/1042
Copyright: © 2016 Varga Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Primary ectopic breast cancer is a rare entity, which is mistaken for other benign skin or soft tissue lesions, easily.
A 56-year old woman with history of laparoscopic cholecystectomy was referred to outpatient clinic 3 years after the operation with a non-healing reddish lump in the surgical scar. The lesion was situated at the site of the right subcostal port, in the medioclavicular line, below the ribcage. Local excision was performed and histological evaluation revealed poorly differentiated adenocarcinoma, which was thought to be metastatic. After multiple recurrences, appearance of an axillary lymph node metastasis and persistent search without any sign of other tumor, it had to be considered as a possibility, if there is no distant source of metastases, rather than a the lesion is a primary cancer. Relying on its location, behavior and result of immunohistochemistry, multidisciplinary team identified the lesion as triple-negative ectopic breast cancer. We performed a radical excision extended to the peritoneum, including partial, 3 cm long resection of the eleventh rib and a level II. Right-sided axillary block dissection was done simultaneously. The abdominal wall was reconstructed with component separation technique combining polypropylene and polytetrafluoroethylene meshes. The multidisciplinary team proposed a treatment for triple-negative ectopic milk-line breast cancer, containing 4 cycles of TAC chemotherapy and fractionated 50 Gray dose of local radiotherapy. This adjuvant oncological treatment is in progress.
This entity should be considered in differential diagnosis in cases of adenocarcinomas close to normal breasts and in the line of embryonic mammary ridges. So, we can avoid a significant delay in treatment and we can offer a better prognosis for the patient.
Keywords
Breast cancer, Ectopic breast, Aberrant breast tissue, Milk line, Surgery
Introduction
The ectopic breast tissue is a heterogeneous, but relatively common condition. The exact incidence is unknown; it is estimated from 0.22-6% depends on the investigated ethnic group, and more common among women [1]. It consists of both supernumerary breasts and aberrant breast tissue. Development of supernumerary breasts is a well-known process during the fourth to fifth week of gestation from the mammary ridges, or “milk lines”, resulting as a failure of physiological regression [2]. Consequently they may appear from the axilla to the groin. Supernumerary breasts or so-called polymastia may present with wide range of appearance from patch of hair to complete breast with nipple-areola complex and glandular tissue. Kajava et al. classified this range into eight categories [3].
Aberrant breast tissue is an island of ectopic breast tissue close to the normal breast, most commonly in the axilla. In contrast with supernumerary breasts, it doesn't communicate with the overlying skin and has no organized secretory system [4]. It is hypothesized that these islands are diverticuli of the ipsilateral breast, even though no histological connection has been described so far [4].
It could have the same pathological changes as normal breast, including malignancies. While primary breast cancer of ectopic breast tissue is rather rare, several cases have been reported in the literature [4,5]. The most common type of ectopic breast tissue cancer is an invasive lobular carcinoma (79%) [4].
We present a case, with an atypical presentation of ectopic milk line malignancy caused a diagnostic difficulties and a significant delay in treatment.
Case Report
A 56-year old woman with a history of hypertension and laparoscopic cholecystectomy (LC) was referred to outpatient clinic 3 years after the operation with a non-healing reddish lump in the surgical scar by her family physician. Preceding her admission a 2-weeks long conservative treatment was applied without any sign of relief.
According to her medical records, LC was performed in a local hospital owing to symptomatic gallstone disease in elective setting. The procedure was uncomplicated; the patient was discharged after an uneventful postoperative period. The histopathologic examination of the removed gallbladder confirmed signs of chronic inflammation.
The non-healing scar was situated at the site of the right subcostal port, in the medioclavicular line, below the ribcage. It was described as an oval shaped, mildly hyperaemic and oozing, firm, slightly mobile papule, which was 3 × 1 cm large. As reported by the patient, the size of the nodule has slowly increased and its color changed from white to reddish in months. She had no family history of breast cancer or any other malignancies. This finding was most consistent with foreign body granuloma, keloid scar and soft tissue tumor.
Local excision was performed in the previous hospital. The histological evaluation of the specimen revealed poorly differentiated adenocarcinoma, which was thought to be metastatic. Excision margins were not assessable owing to thermal injury.
The thorough search for the primary tumor had started, but none of the diagnostic modalities, including meticulous physical examination, chest X-ray, abdominal ultrasonography (US), upper and lower gastrointestinal endoscopy, and positron emission tomography with computed tomography (PET-CT) scan could find the primary tumor. A detailed pathological revision of the formerly removed gallbladder was performed without any further abnormalities.
In the forthcoming two years the lesion had slowly recurred and was re-excised two more times, supplemented by 50 Gray of local radiotherapy. During the continuous follow-up a repeated PET-scan revealed pathologic uptake in the right axilla. The subsequent mammography and US examination proved a pathological axillary lymph node, without any other findings in either breast, supra- or infraclavicular regions.
Fine-needle aspiration biopsy (FNAB) and core biopsy (CB) was taken from the affected lymph node. The harvested cells were severely atypical with large vesicular nucleus, large central nucleolus, pale eosinophilic cytoplasm with well-defined edges. The expression of cytokeratin 7 (CK7) pan-cytokeratin (pan-CK) and S100 protein markers were assessed by immunohistochemistry as positive, cytokeratin 20 (CK20) marker was positive on some tumor cells, breast carcinoma - associated glycoprotein 225 (BCA225) was partially positive, pale estrogen receptor (ER) and progesterone receptor (PR) positivity were seen on some tumor cells. The gross cystic disease fluid protein-15 (GCDFP-15), thyroid transcription factor 1 (TTF1), carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA19-9), human melanoma black 45 (HMB45), leukocyte common antigen (LCA) markers were negative. The results of immunohistochemistry staining are summarized in Table 1.
![]()
Table 1: Results of immunohistochemistry staining's from different samples.
View Table 1
At this point the patient was referred to our hospital for multidisciplinary team (MDT) decision-making, concerning treatment strategy.
After reviewing the patient's medical records, localization and behavior of the tumor, MDT agreed as the abdominal lesion not a metastasis, but presumably a primary cancer. In the light of immunohistochemistry, two possible malignancies came into question: eccrine sweat gland adenocarcinoma and invasive ductal extramammary breast cancer. The immunophenotype was not specific to either entity, but the exact localization and tumor behavior made the latter more probable.
After the necessary precautions and informed consent of the patient, we performed a radical excision extended to the peritoneum, including partial, 3 cm long resection of the eleventh rib and a level II. Right sided axillary block dissection was done simultaneously. The abdominal wall was reconstructed with component separation technique combining polypropylene and polytetrafluoroethylene meshes.
The 18 × 9 × 3.5 cm large specimen contained a 21 × 17 mm and a second, 12 × 12 mm sized tumor mass, exulcerating the skin and infiltrating the abdominal muscles without peritoneal invasion. Every surgical margin was free of tumor for more than 15 mm in depth. Only one of the removed 14 lymph nodes was involved with metastatic adenocarcinoma cells.
The histological evaluation of both specimens was consistent with the result of previous FNAB and CB, except for S100 marker with ER and PR negativity. Cerb-B2 score was 0, the proliferating tumor cells were forming aberrant glands. (Table 1, Figure 1).
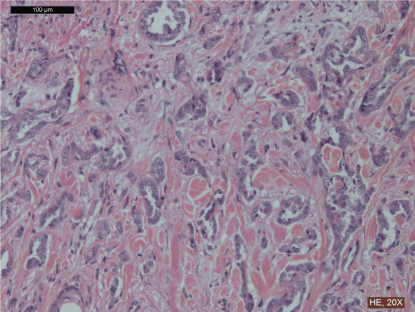
.
Figure 1: Histological slide of specimen after radical excision of the tumor, haematoxylin and eosin staining, 20x magnification. The proliferating tumor cells have large vesicular nuclei, large central nucleolus and forming aberrant glands.
View Figure 1
In the early postoperative period a partial, superficial abdominal wound dehiscence developed, which healed after regular wound care in secondary intention (Figure 2).
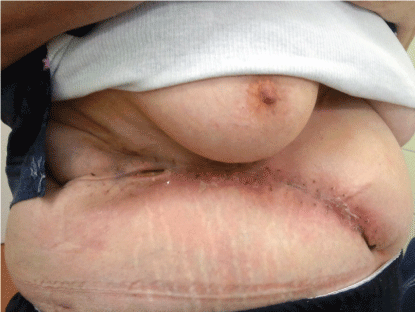
.
Figure 2: Surgical site, 4 weeks after the operation. A partial, superficial wound dehiscence healed after regular wound care in secondary intention.
View Figure 2
Given the patient history, macro- and microscopic finding the MDT proposed treatment for triple-negative ectopic milk-line breast cancer, containing 4 cycles of TAC chemotherapy and fractionated 50 Gray dose of radiotherapy. This adjuvant oncological treatment is in progress at the primary institute.
Discussion
Primary ectopic breast cancer is a rare entity, which is mistaken for other benign skin or soft tissue lesions, easily. Biopsy and wide local excision often clarifies the etiology, but sometimes further evaluation needed.
In cases of histologically proven adenocarcinoma close to either breast needs to be distinguished from metastatic breast cancer and sweat gland tumors.
The presence of normal, ectopic breast tissue close to the tumor would have strengthened the diagnosis, although ectopic breast cancer without any surrounding normal breast parenchyma is a known and published phenomenon [5].
The clinical significance of the presence of the tumor at the exact surgical site was unknown. It could be a simple coincidence, although the effect of chronic inflammation on carcinogenesis is generally known. We did not find any data in the literature according to occurrence of ectopic breast malignancies and traumatic events.
Determining whether our patient's lesion actually derived from ectopic breast cancer tissue or from malignant transformation of a sweat gland with ductal differentiation (ductal eccrine adenocarcinoma), had been challenging [6]. Both morphologic pattern and immunohistochemistrical profile can be consistent with either condition, but not characteristic for any of them [7]. In our opinion, the typical localization of the tumor and biological behavior made the breast tissue origin more likely. Ectopic breast cancer should be considered in cases when malignant breast tissue is found in proximity of normal breast and along the milk line. At the time of initial MDT presentation in our institute, the first line treatment strategy was in to excision of the primary tumor and right sided axillary lymph node dissection, regardless the exact histological diagnosis. Extirpation of the tumor was achieved with radical excision extending the level of peritoneum and partial resection of eleventh rib. Infiltration of the abdominal muscles confirmed a locally invasive cancer.
Diagnosis of ectopic breast cancer is often delayed as consequence of rareness and atypical presentation, usually happens only after a surgical excision. Mean delay to diagnosis is 40.5 months [8], resulting to more advanced stage and poorer prognosis.
Being a type of rare tumors, no standard treatment exists; usually it is treated as similar phenotypical and staged normal breast cancer. Ipsilateral mastectomy has no role, adjuvant chemotherapy and irradiation shown to be beneficial [9].
In summary, we represented the difficulties of diagnostic and treatment modalities of ectopic breast cancers. It should be considered in differential diagnosis in cases of adenocarcinomas close to normal breasts and in the line of embryonic mammary ridges so that we can avoid a significant delay in treatment and can offer a better prognosis for the patient.
Informed Consent
Informed consent was obtained from the patient for publication of this case report and any accompanying images.
Conflict of Interest
The authors declare that they have no conflict of interest.
References
-
Velanovich V (1995) Ectopic breast tissue, supernumerary breasts, and supernumerary nipples. South Med J 88: 903-906.
-
Keith L Moore, TVN Persaud, Mark G Torchia (2011) The Developing Human: Clinically Oriented Embryology, 9th edition.
-
Kajava Y. (1915) The proportion of supernumerary nipples in the Finnish population. Duodecim 1: 143-170.
-
Marshall MB, Moynihan JJ, Frost A, Evans SR (1994) Ectopic breast cancer: case report and literature review. Surg Oncol 3: 295-304.
-
Toman N, Buschmann A, Muehlberger T (2008) Ectopic breast cancer without breast parenchyma. Breast 17: 3-4.
-
McLean SR, Shousha S, Francis N, Lim A, Eccles S, et al. (2007) Metastatic ductal eccrine adenocarcinoma masquerading as an invasive ductal carcinoma of the male breast. J Cutan Pathol 34: 934-938.
-
Bindra A, Bhuva V, Jasani J, Chauhan S, Shukla R, et al. (2012). Ductal Eccrine Carcinoma - A Sweat Gland Carcinoma With Ductular Differentiation - A Case Report. International Journal of Biological Medical Research 3: 1862-1864.
-
Visconti G, Eltahir Y, Van Ginkel RJ, Bart J, Werker PM (2011). Approach and management of primary ectopic breast carcinoma in the axilla: Where are we? A comprehensive historical literature review. J Plast Reconstr Aesthet Surg 64: e1-e11.





