International Journal of Cancer and Clinical Research
New Enzyme-Targeting Radiosensitizer (KORTUC) Containing Hydrogen Peroxide & Sodium Hyaluronate for Intra-Tumoral Injection Using Mice Transplanted with SCC VII Tumor
Ryo Akima1, Yasuhiro Ogawa2*, Shiho Morita-Tokuhiro1, Akira Tsuzuki1, Shin Yaogawa1, Shinji Kariya1, Norihiko Hamada1, Akihito Nishioka1, Shinichiro Masunaga3 and Koji Ono3
1Department of Diagnostic Radiology & Radiation Oncology, Medical School, Kochi University, Nankoku, Japan
2Hyogo Prefectural Kakogawa Medical Center, Kakogawa, Japan
3Particle Radiation Biology, Department of Radiation Life and Medical Science, Research Reactor Institute, Kyoto University, Kumatori, Japan
*Corresponding author:
Yasuhiro Ogawa, Hyogo Prefectural Kakogawa Medical Center, Kakogawa, Hyogo 675-8555, Japan, Tel: +81-79-497-7001; Fax: +81-79-438-3757, E-mail: ogaway@kochi-u.ac.jp
Int J Cancer Clin Res, IJCCR-3-048, (Volume 3, Issue 2), Original Article; ISSN: 2378-3419
Received: February 21, 2016 | Accepted: March 18, 2016 | Published: March 21, 2016
Citation: Akima R, Ogawa Y, Morita-Tokuhiro S, Tsuzuki A, Yaogawa S, et al. (2016) New Enzyme-Targeting Radiosensitizer (KORTUC) Containing Hydrogen Peroxide & Sodium Hyaluronate for Intra-Tumoral Injection Using Mice Transplanted with SCC VII Tumor. Int J Cancer Clin Res 3:048. 10.23937/2378-3419/3/2/1048
Copyright: © 2016 Akima R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The therapeutic effect of radiotherapy using linear accelerators (Linac) for relatively large tumors of more than several centimeters in diameter is reduced to one-third due to a large number of hypoxic tumor cells and a significant amount of anti-oxidative enzymes, such as many kinds of peroxidases and catalase. To overcome tumor hypoxia and abundant peroxidase activities, an injection of hydrogen peroxide into tumor tissue is considered to be a most effective method. However, this is considered difficult because 3% w/v hydrogen peroxide solution (Oxydol) is an antiseptic agent officially permitted only for skin or superficial lesions. Injection into an affected lesion may result in hydrogen peroxide soaking into a body cavity and/or blood vessel, possibly having a risk of causing an intra-arterial oxygen embolism. In our previous study, we aimed to identify the most effective combination of drugs which slows the pace of degradation of hydrogen peroxide in order to relieve local pain at the injection site and preserve high intratumoral partial oxygen pressure. In that study, sodium hyaluronate-hydrogen peroxide was identified as the most effective combination of drugs for preserving high intratumoral partial oxygen pressure for 24 h following intratumoral injection with the agent. Based on those results, the antitumor effect of the new radiosensitizer (KORTUC) containing hydrogen peroxide & sodium hyaluronate was examined using transplanted SCC VII tumors into female C3H/He mice prior to irradiation in this study. The inhibition of tumor growth was significantly greater in the sodium hyaluronate-hydrogen peroxide + irradiation group than in the other groups, in particular to the PBS-hydrogen peroxide + irradiation group. In conclusion, the addition of sodium hyaluronate to hydrogen peroxide produced a significant radiosensitizing effect.
Keywords
Hydrogen peroxide; Radiosensitizer; Sodium hyaluronate, Irradiation; KORTUC; Tumor hypoxia; Radiotherapy
Introduction
It is well known that a low oxygen environment decreases the effect of radiation therapy. We have developed a new enzyme-targeted radiosensitization treatment named KORTUC I using a hydrogen peroxide solution (Oxydol)-soaked gauze bolus for superficially exposed & unresectable neoplasms [1], based on our experimental results demonstrating hydrogen peroxide is a strong radiosensitizer [2-4]. When hydrogen peroxide is injected into tumor tissue, anti-oxidative enzymes such as peroxidase/catalase are inactivated. At the same time, oxygen molecules are generated from degradation of hydrogen peroxide by the catalytic reaction of peroxidases/catalase, and tumor hypoxia can be re-oxygenated. However, the duration of high oxygen concentration in the tumor tissue following the intra-tumoral injection of hydrogen peroxide is thought to be relatively short, because hydrogen peroxide diffuses rapidly into surrounding tissues and micro-vessels.
Therefore, our previous study aimed to identify the best combination of drugs containing hydrogen peroxide in terms of both relieving pain at the site of injection and preserving high intra-tumoral oxygen concentration. In the study, it was concluded that the most suitable combination of drugs for preserving high intra-tumoral oxygen concentration was sodium hyaluronate & hydrogen peroxide [5].
Since hydrogen peroxide is an irritant that may cause severe adverse effects, we needed to develop a new radiosensitizer that is safe for injection, effectively preserving intra-tumoral oxygen concentration while avoiding irritation of the local tissue. This was achieved through the addition of sodium hyaluronate, which makes the solution more viscous and slows degradation of hydrogen peroxide, allowing long-acting radiosensitization of the tumor tissue. In this study, the effect of radiosensitization treatment with intratumoral injection of a mixture of hydrogen peroxide and sodium hyaluronate was studied on transplanted SCC VII tumors of female C3H/He mice following radiotherapy.
Materials and Methods
Animals
The experiments were performed using C3H/He mice (7 weeks old, female, approximately 20 g) at the Institute for Laboratory Animal Research of the Medical School, Kochi University. Humidity and air temperature were held constant, and the animals were fed with standard diet pellets and water ad libitum. The care and treatment of mice were in accordance with institutional guidelines and were also performed according to animal ethical committee rules.
Tumor
On the first day of the experiment, approximately 5 × 104 SCC VII tumor cells were inoculated subcutaneously into the right lower thigh.
Medicine and group
For the experiment, mice were allocated to one of six groups (n = 8 in each group) according to the solution to be injected and with or without irradiation.
a) PBS alone (control)
b) PBS containing 0.5% w/v hydrogen peroxide (PBS-hydrogen peroxide)
c) 0.83% w/v sodium hyaluronate containing 0.5% w/v hydrogen peroxide (hyaluronate-hydrogen peroxide)
d) PBS alone with radiation (30 Gy single dose of 6 MeV electron beam)
e) PBS-hydrogen peroxide with radiation
f) Hyaluronate-hydrogen peroxide with radiation
The volume of intra-tumoral injection of the each agent was 0.25 ml.
Irradiation
When each tumor grew to approximately 10 mm in diameter, radiation therapy of 30 Gy single dose of 6 MeV electron beam (Linac: EXL-20TP, Mitsubishi Electric, Tokyo, Japan) following injection of 0.25 ml of the combination of drugs mentioned above was performed. For irradiation, mice were anesthetized with 0.1 ml/mouse of diluted pentobarbital sodium given as an intraperitoneal injection, and they were fixed on an apparatus specially developed for local irradiation of mice (Figure 1) [6,7].

.
Figure 1: Apparatus for local irradiation of mice.
The bottom and side plates of the apparatus are made of acryl plate. Mice are fixed in the radiation field (15 cm × 15 cm) and shielded whole body, except the tumor burden paws, by 4.5 mm thick copper plates. A bolus of 1.0 cm thickness was used for the corrections of dose distribution.
View Figure 1
Evaluation of the anti-tumor effect
During the 60-day observation period, we measured changes in tumor volume using calipers and calculated the survival rate in each group. The tumor volume was calculated using the following approximate expression.
Where V is tumor volume, W is length of the minor axis, and L is length of the major axis.
Results
Survival period and 60 day survival rate
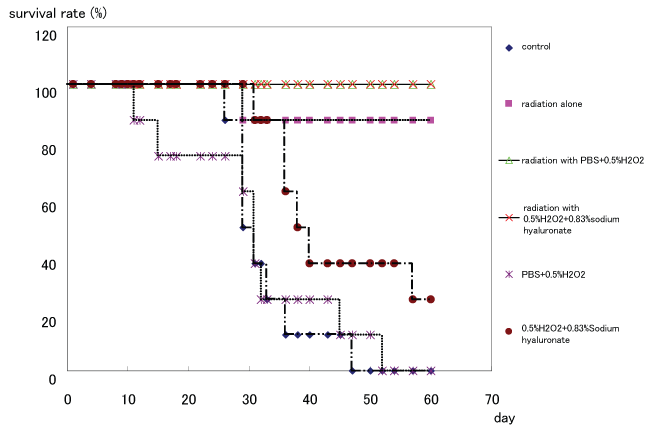
The 60-day survival rates of each group (control group, 0.5% hydrogen peroxide diluted with PBS group, 0.5% hydrogen peroxide + 0.83% sodium hyaluronate group, irradiation alone group, irradiation with 0.5% hydrogen peroxide diluted with PBS group, and irradiation with 0.5% hydrogen peroxide + 0.83% sodium hyaluronate group) were 0, 0, 25, 87.5, 100, and 100%, respectively (Figure 2). The median survival periods of each group were 27.5, 29.0, 37.0, 60.0, 60.0, and 60.0 days, respectively.
.
Figure 2: Survival curve for each group.
The 60-day survival rates of each group are 0, 0, 25, 87.5, 100, and 100%, respectively. The median survival periods of each group are 27.5, 29.0, 37.0, 60.0, 60.0, and 60.0 days, respectively. The survival period and the survival rate do not differ significantly between the irradiation with 0.5 % hydrogen peroxide + 0.83% sodium hyaluronate group and the other groups with irradiation.
View Figure 2
Antitumor effect
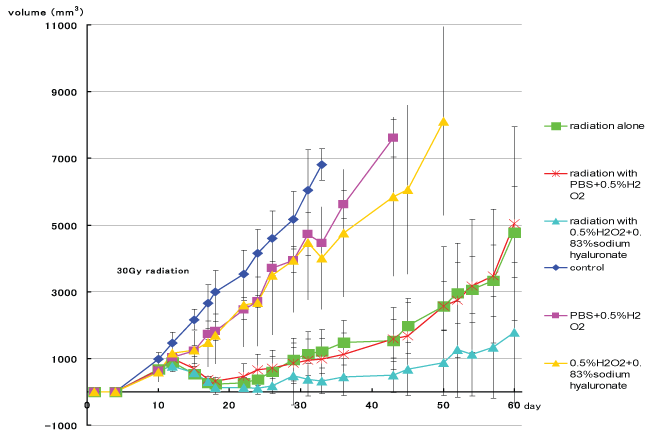
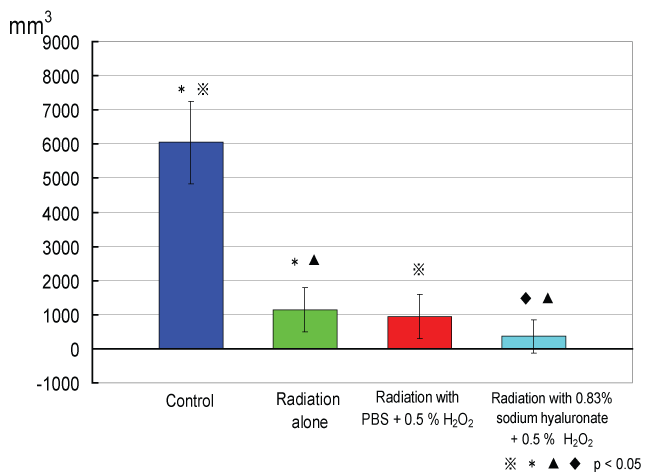
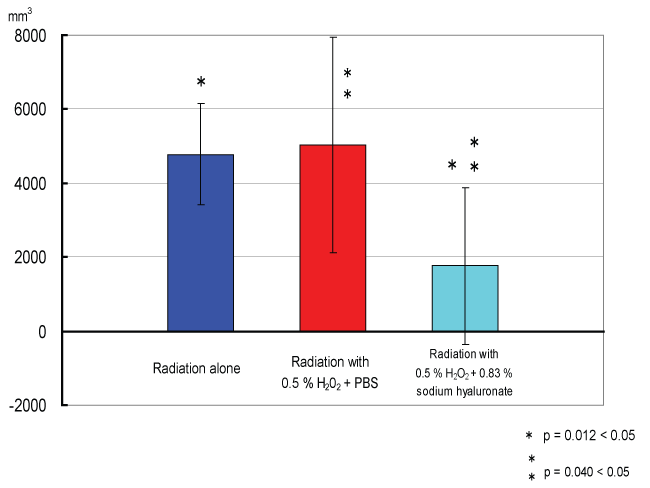
Tumor growth curves of each group are shown in figure 3. On day 31, tumor volume differed significantly between the irradiation groups and the non-irradiation groups (p< 0.05, t-test) (Figure 4). Figure 5 shows tumor volume of each irradiation group on day 60. The inhibition of tumor growth in the irradiation with 0.5% w/v hydrogen peroxide + 0.83% w/v sodium hyaluronate group was significantly greater compared to the other groups, in particular to the irradiation with 0.5% w/v hydrogen peroxide diluted with PBS group. Moreover, in some of the mice in the irradiation with 0.5% w/v hydrogen peroxide + 0.83% w/v sodium hyaluronate group, the tumor disappeared from naked-eye view, and a near complete response was shown on day 20 and day 30.
.
Figure 4: Tumor volumes of the control and irradiation groups on day 31.
On day 31, tumor volume differed significantly between the irradiation groups and the non-irradiation groups (p < 0.05, t-test).
View Figure 4
.
Figure 5: Tumor volumes of each irradiation group on day 60.
The inhibition of tumor growth in the hyaluronate-hydrogen peroxide irradiation group was significantly greater compared to the other groups, in particular to the PBS-hydrogen peroxide-irradiation group.
View Figure 5
Discussion
With X-ray and electron beam therapy using Linac, radical reactions are considered to make a large contribution to the therapeutic effect [8]. As tumors increase, tumor hypoxia and increased peroxidase activities are brought about mainly inside the tumor. Therefore, local control of the tumor becomes difficult in terms of tumor hypoxia and rich peroxidase activities. Therefore, to overcome these situations, we developed KORTUC (Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas), which resolves tumor hypoxia and inactivates peroxidase activities at the same time by degradation of hydrogen peroxide and catalytic reactions of peroxidase. Furthermore, with KORTUC I, which uses hydrogen peroxide-soaked gauze during irradiation, we obtained a remarkable local therapeutic effect for a low LET (linear energy transfer)-radiation-resistant tumor exposed to the skin surface [1]. Moreover, with KORTUC II, we obtained a prominent therapeutic response for soft tissue malignancies and advanced breast cancer [9].
For intratumoral injection of the agent, sodium hyaluronate is added to hydrogen peroxide to make the KORTUC radiosensitizer to relieve pain and maintain oxygen partial pressure in the tumor tissue by the viscosity of sodium hyaluronate [5]. To demonstrate the efficacy of adding sodium hyaluronate to hydrogen peroxide, in vivo experiments were performed using a mouse tumor system.
In this experiment, the irradiation group after local injection of 0.25 ml hyaluronic acid-0.5% w/v H2O2 showed a difference compared with other irradiation groups in both the survival period and the survival rate. The tumor growth controlling effect was significant compared with other groups, especially the irradiation group, after local injection of 0.25 ml PBS and 0.5% w/v H2O2. Moreover, in some mice in the groups, the tumor disappeared to the naked eye, and a remarkable therapeutic effect of near complete response was shown. This shows that adding the hyaluronic acid to hydrogen peroxide demonstrated a more radiosensitizing effect. The radiosensitizing effect is thought to be due to the pharmacokinetics of hydrogen peroxide and sodium hyaluronate. Sodium hyaluronate is a natural polymer compound by β-glucuronic acid and N-acetylglucosamine, and is one of the proteoglycans. The molecular formula of the hyaluronic acid is (C14H20NNaO11)n, and the pH is 6.8-7.8. It has a weight-average molecular weight of about 600,000 to 1.2 million, and it is colorless and transparent, sticky, and scentless. It becomes the architectural component of extracellular matrix in vivo, and it distributed into the joint cavity. Water retentivity is good, water is taken into the extracellular matrix, and it works at loosening the matrix, etc. Moreover, the half-life in the joint cavity is about 20 hours, and the concentration level in blood reaches its highest value in about 48 hours, after which it decreases gradually. Sodium hyaluronate is metabolized in the liver, and the majority is excreted in the breath, while part is excreted into the urine and feces.
Based on the characteristics mentioned above, we considered the reason that a significant difference was shown in the therapeutic effect by adding sodium hyaluronate to hydrogen peroxide was as follows. To begin with, it is thought that the sensitizer is distributed comparatively uniformly in the tumor tissues by the high viscosity of sodium hyaluronate, comparing with the case without sodium hyaluronate. When sodium hyaluronate is not added, it can be thought that the hydrogen peroxide is rapidly degraded into water and oxygen by intrinsic peroxidase, distribution of hydrogen peroxide in the tumor tissues becomes uneven, and re-oxygenation in hypoxic tumor tissue cannot be fully obtained.
However, by adding sodium hyaluronate, the sensitizer KORTUC can be distributed evenly in the hypoxic tumor tissue, which is thought to lead to the experimental results seen. Second, it is thought that the maintenance of the effect on the oxygen partial pressure depends on the absorption and metabolic delay due to high viscosity of sodium hyaluronate.
Therefore, it was experimentally shown that the therapeutic effects as a radiosensitizer were significantly increased by adding sodium hyaluronate to hydrogen peroxide. In addition, the remarkable therapeutic effects of the mixture of sodium hyaluronate and hydrogen peroxide have recently been elucidated in our clinical studies [9-23].
Conclusions
In this experimental study, the addition of sodium hyaluronate to hydrogen peroxide produced a better radiosensitizing effect than the use of hydrogen peroxide alone for intratumoral injection. In fact, we have already shown remarkable therapeutic effects of this treatment clinically in our recent papers [9-23].
Using the KORTUC method, we are now developing an advanced hepatocellular carcinoma treatment (KORTUC III), intraoperative irradiation therapy for locally advanced pancreas cancer (KORTUC IV), and locally advanced renal cell carcinoma treatment (KORTUC V). We are also trying to improve the treatment by developing a new long-acting radiosensitizer for once-weekly intratumoral injection (New KORTUC) [24].
Acknowledgments
This work was partially supported by a grant-in-aid for Scientific Research from the Japanese Ministry of Education, Cultures, Sports, Science and Technology (21591610, 25461916).
Author Contributions: Ryo Akima, Yasuhiro Ogawa, Norihiko Hamada, and Akihito Nishioka contributed to the study concept, experimental study design, data analysis and manuscript preparation. Yasuhiro Ogawa also contributed to injection of the KORTUC agent. Shiho Morita-Tokuhiro, Akira Tsuzuki, Shin Yaogawa, and Shinji Kariya contributed to irradiation of mice. Shinichiro Masunaga and Koji Ono contributed to providing SCC VII tumor cells.
Conflicts of Interest
The authors declare no conflict of interest.
References
-
Ogawa Y, Ue H, Tsuzuki K, Tadokoro M, Miyatake K, et al. (2008) New radiosensitization treatment (KORTUC I) using hydrogen peroxide solution-soaked gauze bolus for unresectable and superficially exposed neoplasms. Oncol Rep 19: 1389-1394.
-
Ogawa Y, Takahashi T, Kobayashi T, Kariya S, Nishioka A, et al. (2003) Mechanism of apoptotic resistance of human osteosarcoma cell line, HS-Os-1, against irradiation. Int J Mol Med 12: 453-458.
-
Ogawa Y, Takahashi T, Kobayashi T, Kariya S, Nishioka A, et al. (2003) Apoptotic-resistance of the human osteosarcoma cell line HS-Os-1 to irradiation is converted to apoptotic-susceptibility by hydrogen peroxide: a potent role of hydrogen peroxide as a new radiosensitizer. Int J Mol Med 12: 845-850.
-
Ogawa Y, Takahashi T, Kobayashi T, Kariya S, Nishioka A, et al. (2004) Immunocytochemical characteristics of human osteosarcoma cell line HS-Os-1: Possible implication in apoptotic resistance against irradiation. Int J Mol Med 14: 397-403.
-
Tokuhiro S, Ogawa Y, Tsuzuki K, Akima R, Ue H, et al. (2010) Development of a novel enzyme-targeting radiosensitizer (KORTUC) containing hydrogen peroxide for intratumoral injection for patients with low linear energy transfer (LET)-radioresistant neoplasms. Oncol Lett 1: 1025-1028.
-
Ogawa Y, Imanaka K, Ashida C, Takashima H, Imajo Y, et al. (1983) Active specific immunotherapy using the immune reaction of a low-dose irradiated tumor tissue. Int J Radiat Oncol Biol Phys 9: 533-537.
-
Ogawa Y, Goodman GB, Chaplin DJ, Grulkey W, Lam GK (1990) Combination therapy of pions and SPG (Sonifilan, Schizophyllan), a biological response modifier for mouse tumor systems. Int J Radiat Oncol Biol Phys 18: 1415-1420.
-
Ogawa Y (2016) Paradigm shift in radiation biology/radiation oncology-Exploitation of the "H₂O₂ Effect" for radiotherapy using low-LET (linear energy transfer) radiation such as X-rays and high-energy electrons. Cancers (Basel) 8:28.
-
Ogawa Y, Kubota K, Ue H, Kataoka Y, Tadokoro M, et al. (2009) Phase I study of a new radiosensitizer containing hydrogen peroxide and sodium hyaluronate for topical tumor injection: A new enzyme-targeting radiosensitization treatment, Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas, Type II (KORTUC II). Int J Oncol 34: 609-618.
-
Ogawa Y, Kubota K, Aoyama N, Yamanishi T, Kariya S, et al. (2015) Non-surgical breast-conserving treatment (KORTUC-BCT) using a new radiosensitization method (KORTUC II) for patients with stage I or II breast cancer. Cancers (Basel) 7: 2277-2289.
-
Yaogawa S, Ogawa Y, Morita-Tokuhiro S, Tsuzuki A, Akima R, et al. (2016) Serial assessment of therapeutic response to a new radiosensitization treatment, Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas, type II (KORTUC II), in patients with stage I/II breast cancer using breast contrast-enhanced magnetic resonance imaging. Cancers (Basal) 8: 1.
-
Tsuzuki A, Ogawa Y, Kubota K, Tokuhiro S, Akima R, et al. (2011) Evaluation of changes in tumor shadows and microcalcifications on mammography following KORTUC II, a new radiosensitization treatment without any surgical procedure for elderly patients with stage I and II breast cancer. Cancers (Basel) 3: 3496-3505.
-
Ogawa Y, Kubota K, Ue H, Tadokoro M, Matsui R, et al. (2011) Safety and effectiveness of a new enzyme-targeting radiosensitization treatment (KORTUC II) for intratumoral injection for low-LET radioresistant tumors. Int J Oncol 39: 553-560.
-
Ogawa Y, Kubota K, Ue H, Nishioka A, Kariya S, et al. (2007) Development and clinical application of a new radiosensitizer containing hydrogen peroxide and hyaluronic acid sodium for topical tumor injection - a new enzyme-targeting radiosensitization treatment, KORTUC II (Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas, Type II). Strahlenther Onkol 183: 100-101.
-
Hitomi J, Kubota K, Ogawa Y, Hamada N, Murata Y, et al. (2010) Non-surgical therapy and radiologic assessment of stage I breast cancer treatment with novel enzyme-targeting radiosensitization: Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas, type II (KORTUC II). Exp Ther Med 5: 769-775.
-
Miyatake K, Kubota K, Ogawa Y, Hamada N, Murata Y, et al. (2010) Non-surgical care for locally advanced breast cancer: radiologically assessed therapeutic outcome of a new enzyme-targeting radiosensitization treatment, Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas, Type II (KORTUC II) with systemic chemotherapy. Oncol Rep 24: 1161-1168.
-
Shimbo T, Yoshikawa N, Yoshioka H, Tanaka Y, Yoshida K, et al. (2013) Three cases of unresectable locally advanced breast cancer treated with local injection of the new radiosensitization (KORTUC). Rinsho Hoshasen 58: 1881-1886.
-
Aoyama N, Ogawa Y, Kubota K, Ohgi K, Kataoka Y, et al. (2013) Therapeutic response to a new enzyme-targeting radiosensitization treatment (KORTUC-SC) for patients with recurrent breast cancer. J Cancer Res Ther 1: 215-219.
-
Aoyama N, Ogawa Y, Kubota K, Miyatake K, Tadokoro M, et al. (2016) Therapeutic response to a new enzyme-targeting radiosensitization treatment (Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas) in patients with recurrent breast cancer. Oncol Lett (in print).
-
Aoyama N, Ogawa Y, Yasuoka M, Iwasa H, Miyatake K, et al. (2016) Therapeutic response to a new enzyme-targeting radiosensitization treatment (KORTUC II) for residual lesions in patients with Stage IV primary breast cancer following induction chemotherapy with epirubicin + cyclophosphamide and/or taxane. Oncol Lett. (in print).
-
Aoyama N, Ogawa Y, Nishimori M, Ohgi K, Iwasa H, et al. (2016) Therapeutic results of a new enzyme-targeting radiosensitization treatment (Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas II, KORTUC II) in patients with Stage I primary breast cancer. Oncol Lett. (in print).
-
Nishioka A, Ogawa Y, Miyatake K, Tadokoro M, Nogami M, et al. (2014) Safety and efficacy of image-guided enzyme-targeting radiosensitization and intraoperative radiotherapy for locally advanced unresectable pancreatic cancer.Oncol Lett 8: 404-408.
-
Obata S, Yamanishi M, Katsumi S, Ohta Y, Kan T, et al. (2015) A new treatment strategy for advanced rectal cancer: enzyme-targeting and radio-sensitization treatment, so-called KORTUC (Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas). Nagasaki Igakkai Zasshi 90: 135-145.
-
Morita-Tokuhiro S, Ogawa Y, Yokota N, Tsuzuki A, Oda H, et al. (2016) Development of a novel enzyme-targeting radiosensitizer (New KORTUC) using a gelatin-based hydrogel instead of a sodium hyaluronate. Cancers 8: 10.