International Journal of Cancer and Clinical Research
Cardiac Involvement in Carcinoid Syndrome: Role of Serial Standard and Contrast Echocardiography
Francesca Casadei, Antonella Moreo, Francesco Musca*, Paola Vallerio, Miriam Stucchi and Cristina Gannattasio
Cardiology IV, "A. De Gasperis" Department, Grande Ospedale Metropolitano Niguarda, Milan, Italy
*Corresponding author:
Francesca Casadei, Cardiology IV, "A. De Gasperis" Department, Grande Ospedale Metropolitano Niguarda, Milan 20159, Italy, E-mail: francesca.casadei@yahoo.it
Int J Cancer Clin Res, IJCCR-3-049, (Volume 3, Issue 2), Case Report; ISSN: 2378-3419
Received: February 01, 2016 | Accepted: March 21, 2016 | Published: March 23, 2016
Citation: Casadei F, Moreo A, Musca F, Vallerio P, Stucchi M, et al. (2016) Cardiac Involvement in Carcinoid Syndrome: Role of Serial Standard and Contrast Echocardiography. Int J Cancer Clin Res 3:049. 10.23937/2378-3419/3/2/1049
Copyright: © 2016 Casadei F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We report a case of a 52 year-old woman with cardiac carcinoid metastases who developed a moderate tricuspid regurgitation without the typical findings of carcinoid valve disease. Echocardiography allowed to define the mechanism of valve insufficiency. Moreover, standard and contrast echocardiography permitted characterization of the structural features of the mass, size measurement and monitoring growth during follow-up.
Keywords
Echocardiography, Cardiac metastasis, Carcinoid heart disease
Introduction
Neuroendocrine tumors are rare malignancies which arise from enterochromaffin cells in the gastrointestinal tract, bronchus and gonads. Growth progression is usually slow, over many years, symptoms arising only when tumor is large or has metastasized. Carcinoid tumors of the small-bowel may secrete a large quantity of vasoactive substances such as histamines, chromogranin A and serotonine which are metabolized by the liver. When liver metastasis occur, these vasoactive substances can reach the systemic circulation causing carcinoid syndrome characterized by flushing, diarrhea and bronchospasm. Carcinoid syndrome is rarely described in absence of liver metastases. Approximately 50% of patients with carcinoid syndrome develop carcinoid heart disease characterized by fibrotic plaque deposition on the endocardium of valve leaflets and cardiac chambers. Right-sided valvular dysfunction is the hall mark of carcinoid heart disease while left sided valves are relatively spared due to inactivation of serotonin in the lungs. Left sided valve involvement may occur in case of right to left shunt through a patent foramen ovale, in presence of bronchial neoplasia or in presence of high level of circulating serotonin [1].
Pericardial involvement has been described in form of pericardial metastasis [2] and carcinoid pericarditis [3] with pericardial effusion.
Another rare manifestation of carcinoid heart disease is represented by cardiac metastases. The most common primary tumor that metastasize to the heart are breast, lung and melanoma, where localization of neuroendocrine tumors are extremely rare. The overall incidence of myocardial carcinoid metastases among patients with metastatic carcinoid disease is about 2-4% [4,5] being even rarer in patients without liver metastasis with only few cases reported in literature [6,7].
Cardiac carcinoid metastases are usually asymptomatic with patients predominantly manifesting general cancer cachexia. Clinically cardiac metastases, depending on location and size, are generally characterized by heart failure, arrhythmias, ischemic symptoms or cardiac tamponade.
Carcinoid tumor infiltration of the heart can be the only cardiac manifestation of carcinoid heart disease. This underlines the importance of a complete echocardiographic evaluation in carcinoid syndrome to assess cardiac involvement and should include metastasis search, even in absence of carcinoid valve disease [8].
The largest cohort where the echocardiographic features of cardiac carcinoid metastasis were described, was taken from the Mayo Clinic [8], comprising 11 patients. In this study only tumors ≥ 1 cm could be detected by echocardiography and on echo evaluation metastatic carcinoid appeared as homogeneous, circumscribed, non infiltrating masses.
In addition to two-dimensional echocardiography, the usefulness of contrast echocardiography in early differentiation between malignant and benign masses has been reported [9]. Kirkpatrick et al. demonstrated the utility of echocardiographic contrast perfusion imaging to differentiate the neo-vascularization of malignancies from avascularity of thrombi or sparse vascularity of stromal tumors [10]. That study showed vascular tumors and malignancies having hyperenhancement compared to the adjacent myocardium, after contrast administration.
Here we present the case of a cardiac carcinoid metastasis with development of a tricuspid valve dysfunction without the typical findings of carcinoid valve disease. Standard and contrast echocardiography played a key role in diagnosis and follow-up.
Case Report
A 52-year-old woman with midgut carcinoid tumor underwent right hemicolectomy and lymphadenectomy and was treated with somatostatin analogue.
Ten years later the patient developed flushing and diarrhoea suggestive of carcinoid syndrome, confirmed by elevated values of urinary 5-hydroxyindoleacetic acid. Computed Tomography (CT) of the abdomen showed bowel anastomosis thickening indicating tumor recurrence. A 68Ga PET confirmed increased uptake at the bowel anastomosis and showed intense tracer uptake at the left breast and in the interventricular septum.
Clinically the patient was asymptomatic for cardiovascular symptoms. Electrocardiogram was negative for pathological findings, evidencing neither atrioventricular block nor intraventricular conduction delay.
Breast neoplasia was confirmed on mammography. The patient underwent breast quadrantectomy and resection of the distal small and proximal large bowel for suspected primary breast cancer and a recurrence of midgut carcinoid, respectively. Histologic sections of the bowel were negative for neoplasic cells but showed chronic flogosis and hyperplasia. Immunohistochemical staining of breast tissue was diagnostic for neuroendocrine metastasis.
Cardiac magnetic resonance (CMR) showed interventricular septum thickening due to an inhomogeneous infiltrating mass (35 mm × 19 mm) protruding in the right ventricle. Areas of hypersignal intensity were noted on T2-weighted images indicating flogosis and inhomogeneous enhancement on late gadolinium enhancement images.
Transthoracic echocardiography confirmed the presence of a voluminous inhomogeneous mass within the interventricular septum jutting in the right ventricle (Figure 1). Contrast perfusion imaging was used for characterizing mass vascularization. Following a slow intravenous injection of a second generation contrast agent (Sonovue, Bracco, Milano, Italy) hyperintense inhomogeneous enhancement was attained (Figure 2).
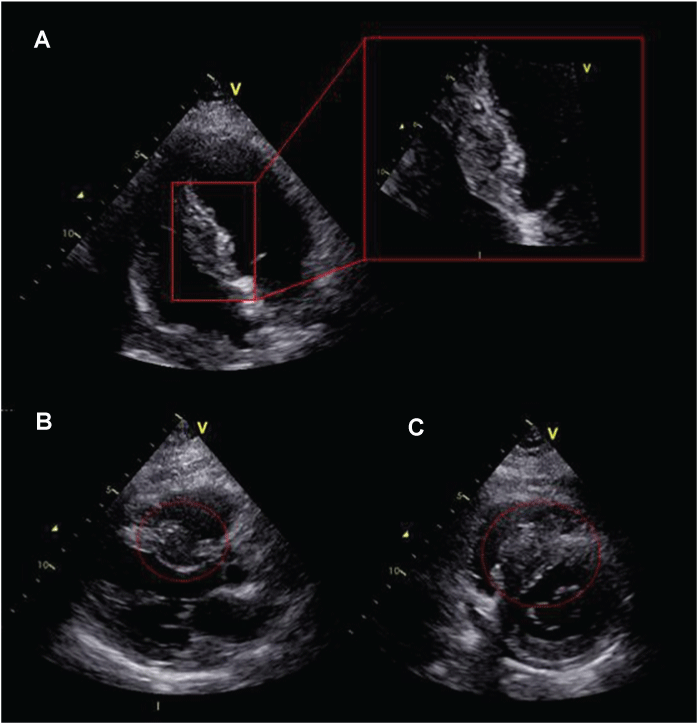
.
Figure 1: (A) Two-dimensional (2D) echocardiography apical 4-chamber view; (B) Apical long axis view; (C) Short axis view. The interventricular septum appears thickened for the presence of a inhomogeneous infiltrating mass jutting in the right ventricle.
View Figure 1
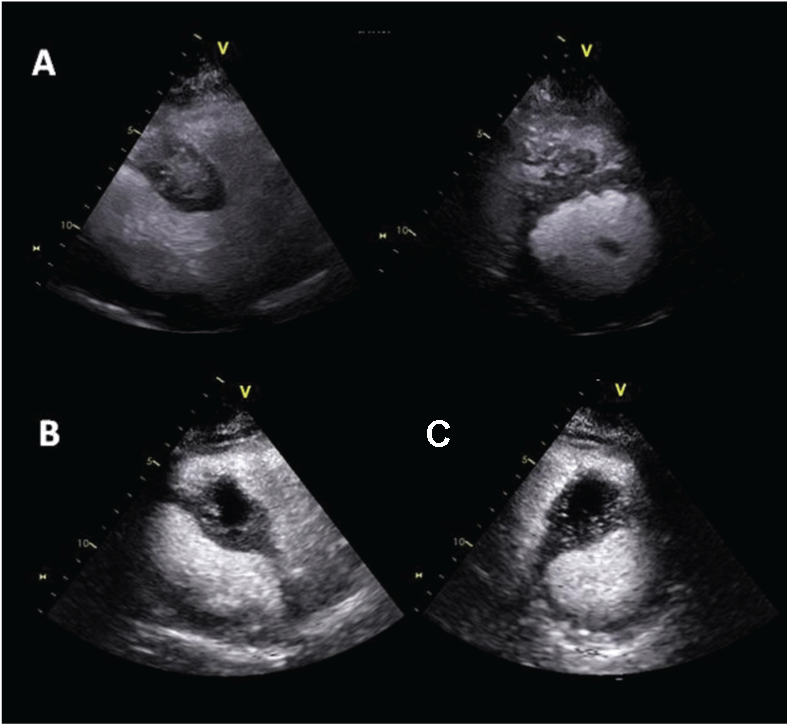
.
Figure 2: (A) Contrast echocardiography off axis and short axis views showing inhomogeneous contrast enhancement of cardiac carcinoid metastasis within the interventricular septum; (B) Contrast echocardiography showing the appearance of an area of hypoperfusion suggestive of partial necrosis of the mass.
View Figure 2
Echocardiography evidenced cardiac chambers having normal dimension; preserved biventricular function and no significant valve disease; of note, typical characteristics of carcinoid valvular disease were absent.
A myocardial biopsy was not performed for clinical history indicating cardiac carcinoid metastasis. Metastasis localization excluded cardiac surgery and the patient was treated with somatostatin analogues and referred for adjuvant therapy.
Follow-up of the myocardial neoplastic lesion included serial standard and contrast echocardiography. Standard echocardiography showed a slow progressive growth of the mass mainly toward the right ventricle (26 mm × 35 mm). Contrast echocardiography evaluation demonstrated inhomogeneous perfusion of the mass with new appearance of an area of hypoperfusion suggestive of partial necrosis of the mass (Figure 2). A CMR confirmed these findings.
Tricuspid valve insufficiency worsened by interference of the metastatic mass with the subvalvular apparatus (Figure 3). This tricuspid valve regurgitation was not secondary to the thickening of the valvular leaflet as in typical carcinoid heart disease occurrence (Figure 3).
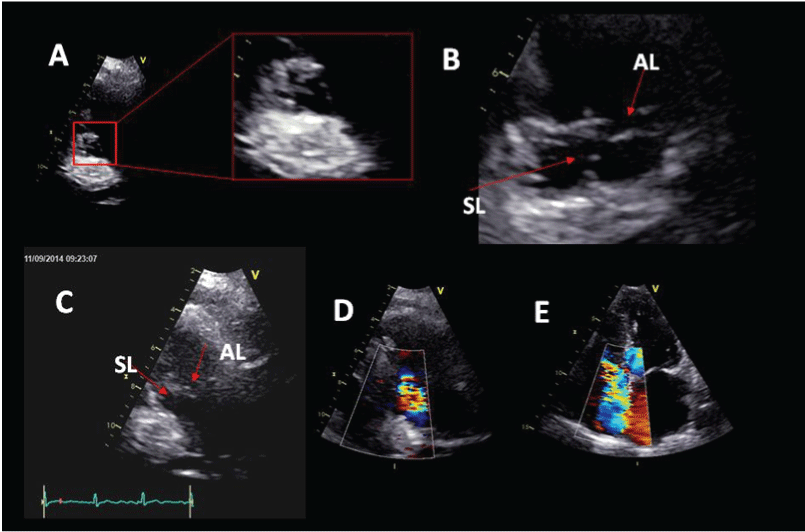
.
Figure 3: (A) Parasternal long-axis view for right ventricle inflow. In diastole tricuspid valve opening is preserved. The septal leaflet of tricuspid valve is thin but chordae tentineae are entrapped by metastasis; (B) Both septal and anterior leaflet are thin, but during systole the septal leaflet; (C) Restriction of the septal leaflet in end systole with protrusion of the anterior leaflet; (D) Moderate eccentric tricuspid valve insufficiency; (E) Apical 4-chamber view showing moderate tricuspid valve regurgitation directed toward interatrial septum. AL anterior leaflet. SL septal leaflet.
View Figure 3
Echocardiography also revealed new onset of mild pericardial effusion. MRI documented hypersignal intensity on T2-weighted images suggestive of pericardial flogosis. Post three months a thoracic computed tomography showed appearance of multiple pericardial metastasis. During the three years follow-up the patient had also developed multiple bone and lung metastasis and she died for tumour progression. No liver metastasis were observed.
Discussion
As reported in literature about 50% of patients with liver metastasis of midgut neuroendocrine tumor develop carcinoid syndrome and approximately 50% of them manifest carcinoid heart disease (CHD). CHD is generally characterized by dysfunction of right sided valves due to fibrotic plaque deposits, which usually leads to heart failure and poor prognosis [1]. This patient had carcinoid syndrome without liver metastases or typical carcinoid valve disease.
In carcinoid valve disease the tricuspid valve leaflets are classically thickened, shortened and retracted, fixed in a semi-open position, the chordae tendineae are thickened and retracted, the valve annulus is narrowed. Functionally this results in a combination of valvular regurgitation and stenosis [1]. Usually also pulmonary valve dysfunction is present.
In this patient tricuspid valve regurgitation was due to interference of the mass with the subvalvular apparatus, not thickening and shortening of the tricuspid leaflets as in typical carcinoid heart disease occurrence. Chordae tendineae of the septal leaflet were entrapped by metastasis causing hypomobility of the septal leaflet with systolic protrusion of the anterior leaflet resulting in moderate eccentric insufficiency (Figure 3). Tricuspid leaflets were relatively thin and valve opening was preserved. Pulmonary valve had normal morphology and function.
In this patient cardiac manifestation of carcinoid heart disease was represented by pericardial and myocardial neoplastic involvement, rarely reported in literature. Cardiac metastasis remained asymptomatic and the patient manifested only symptoms related to carcinoid syndrome.
Echocardiographic evaluation evidenced the cardiac mass appearing inhomogeneous and infiltrating, not as previously described. Contrast echocardiography allowed for defining the metastasis profile, size measurement and growth monitoring during follow-up. This also showed its vascularization demonstrating hyperenhancement indicating neovascularization and a hypoenhanced area of partial necrosis (Figure 1 and Figure 2). We believe this is a novel case describing echo contrast features of a cardiac neuroendocrine tumor metastasis.
We conclude that a complete echocardiographic evaluation in carcinoid syndrome is of primary importance for the assessment of new valve disease, intramyocardial metastasis, or suspicion of pericardial involvement, this includes standard and contrast echocardiography for monitoring and follow-up in cardiac metastasis.
References
-
Bhattacharyya S, Davar J, Dreyfus G, Caplin ME (2007) Carcinoid heart disease. Circulation 116: 2860-2865.
-
Collins N, Bellamy G, Hayes P (2004) Intrapericardial carcinoid metastasis. J Am Soc Echocardiogr 17: 675-676.
-
Rich LL, Lisa CP, Nasser WK (1973) Carcinoid pericarditis. Am J Med 54: 522-527.
-
Pellikka PA, Tajik AJ, Khandheria BK, Seward JB, Callahan JA, et al. (1993) Carcinoid heart disease. Clinical and echocardiographic spectrum in 74 patients. Circulation 87: 1188-1196.
-
Fine SN, Gaynor ML, Isom OW, Dannenberg AJ (1990) Carcinoid tumor metastatic to the heart. Am J Med 89: 690-692.
-
Kasi VS, Ahsanuddin AN, Gilbert C, Orr L, Moran J, et al. (2002) Isolated metastatic myocardial carcinoid tumor in a 48-year-old man. Mayo Clin Proc 77: 591-594.
-
Patel CN, Anthoney A, Treanor D, Scarsbrook AF (2009) Solitary myocardial metastasis from small-bowel neuroendocrine carcinoma. J Clin Oncol 27: 1724-1726.
-
Pandya UH, Pellikka PA, Enriquez-Sarano M, Edwards WD, Schaff HV, et al. (2002) Metastatic carcinoid tumor to the heart: echocardiographic-pathologic study of 11 patients. J Am Coll Cardiol 40: 1328-1332.
-
Haverkamp MC, Scholte AJ, Holman ER, Jongbloed MR, Schippers EF, et al. (2005) Contrast echocardiography as a useful additional diagnostic tool in evaluating a primary cardiac tumor. Eur J Echocardiogr 6: 388-391.
-
Kirkpatrick JN, Wong T, Bednarz JE, Spencer KT, Sugeng L, et al. (2004) Differential diagnosis of cardiac masses using contrast echocardiographic perfusion imaging. J Am Coll Cardiol 43: 1412-1419.





