International Journal of Diabetes and Clinical Research
Effect of Alogliptin, a DPP-4 Inhibitor, on Markers of Lipid Metabolism in Type 2 Diabetic Patients
Yuzuru Kishitani*, Hideyuki Okuda, Daisuke Kinoshita, Kanyun Hata and Toshihiko Kawasaki
Department of Gastroenterology, Endocrinology and Metabolism, Nara Hospital, Kindai University, Japan
*Corresponding author:
Yuzuru Kishitani, M.D., Ph.D., Department of Gastroenterology, Endocrinology and Metabolism, Nara Hospital, Kindai University Faculty of Medicine, 1248-1 Otsuda-cho, Ikoma city, Nara 630-0293 Japan, Tel: +81-743-77-0880; Fax:+81-743-77-0890; E-mail: joe-k@nara.med.kindai.ac.jp
Int J Diabetes Clin Res, IJDCR-1-017, (Volume 1, Issue 3), Research Article; ISSN: 2377-3634
Received: November 24, 2014 | Accepted: December 28, 2014 | Published: December 31, 2014
Citation: Kishitani Y, Okuda H, Kinoshita D, Hata K, Kawasaki T (2014) Effect of Alogliptin, a DPP-4 Inhibitor, on Markers of Lipid Metabolism in Type 2 Diabetic Patients. Int J Diabetes Clin Res 1:017. 10.23937/2377-3634/1410017
Copyright: ©2014 Kishitani Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: DPP-4 inhibitor has been shown to improve not only blood glucose levels but also dyslipidemia, but the effect remains controversial. Further, there are few reports regarding the influence on Apo B, Apo A-1 and Apo B/Apo A-1 in alogliptin.
Objective: Using the alogliptin, we focused on Apo B and Apo A-1, in addition to LDL-C, HDL-C and TG, and investigated the effect of alogliptin on markers predicting cardiovascular events, such as Apo B/Apo A-1 and LDL-C/Apo B.
Methods: This study was carried out with a single arm in an open label manner. 45 consecutive cases that could enroll between September 2011 and March 2012 were selected as the subjects for randomization. Alogliptin 25 mg/day was administered for eight weeks and non-fasting blood samples were collected 2-3 hours after breakfast, once at the beginning of the study and once eight weeks later, and changes in glucose and lipid metabolism markers were compared.
Results: Administration of alogliptin significantly reduced HbA1c levels and non-fasting blood glucose levels, and reduced LDL-C and non-fasting TG. Apo B was significantly reduced by alogliptin treatment but Apo A-1 was unchanged. The Apo B/Apo A-1 ratio was significantly decreased and the LDL-C/Apo B ratio significantly increased by alogliptin treatment. The change in the pre-administration Apo B/Apo A-1 ratio was positively correlated with body weight, TG, HbA1c, blood CPR and pooled urine CPR and negatively correlated with HDL-C
Conclusion: Administration of alogliptin in type 2 diabetic patients significantly improved levels of blood glucose, in addition to those of lipid metabolism markers such as LDL-C and non-fasting TG, as well as sensitive predictive markers for the onset risk of cardiovascular events, such as the Apo B/Apo A-1 ratio and the LDL-C/Apo B ratio. These results suggest that alogliptin could potentially suppress atherosclerosis and prevent cardiovascular events (UMIN 000011470).
Keywords
Alogliptin, Type 2 diabetes, Lipid metabolism, Apo B/Apo A-1, LDL/Apo B
Introduction
It is widely known that patients with diabetes have a higher risk of onset and mortality from cardiovascular events [1,2]. Meta-analysis of UKPDS has shown that the primary risk factor for cardiovascular (CV) events in diabetic patients is neither blood glucose levels nor HbA1c, but rather LDL-C [3]. Accordingly, an improvement not only in blood glucose levels but also in dyslipidemia is considered important for the prevention and avoidance of CV events.
In type 2 diabetes, increased influx of FFA into the liver due to insufficient insulin activity increases VLDL production, while reduced LPL activity increases VLDL, LDL-C, IDL and accelerates atherosclerosis [4]. A number of large-scale clinical studies have proven that statins decrease LDL-C and prevent CV events [5,6]. However, even a reduction in LDL-C leaves a residual risk, and hypertriglyceridemia, in particular, as well as postprandial TG elevation [7] and hypoHDLemia are risk factors.
Given the above data, current guidelines on diabetes treatment recommend appropriate control of hypercholesterolemia, hypertriglyceridemia and hypoHDLemia to prevent CV events [8]. In diabetic patients, abnormalities in both the quantity, and quality of LDL-C, such as increased small dense LDL-C and production of glycosylated LDL-C, are thought to increase the onset risk of CV events [9]. Electrophoresis is the standard method for measuring small dense LDL-C, but a complicated procedure. The LDL-C/Apo B ratio is used as an alternative, and it has been reported that small dense LDL is increased in cases in which the LDL-C/Apo B ratio is 1.2 or lower [10].
In Japan, DPP-4 inhibitors have been used as oral antidiabetic agents since 2011, and have also been shown to improve dyslipidemia. It has been reported that sitagliptin decreased TG and RLP-C without affecting levels of LDL-C and HDL-C [11]. Different from this result, another group showed it could decrease T-C and TG [12]. Thus, effects of sitagliptin toward dyslipidemia are still controversial. A meta-analysis of the effect on TC and TG has demonstrated differences in improvements in lipid metabolism markers and efficacy [13]. The non-fasting Apo B/Apo A-1 ratio has been proven as a useful marker for predicting CV events [14]. According to the INTERHEART study, the non-fasting Apo B/Apo A-1 ratio may be a more sensitive predictive marker for acute myocardial infarction than either non-HDL-C or the T-C/HDL-C ratio. Additionally, the non-fasting Apo B/Apo A-1 ratio was not significantly influenced by age, gender or races. With regard to the influence of DPP-4 inhibitors on apoprotein, previous studies have examined decrease of postprandial chylomicron and Apo B48 levels with a focus on postprandial TG in sitagliptin [10] and in vildagliptin [16]. However, the measurement of both of Apo B, Apo A-1 is reported only in sitagliptin [15], and no report has studied the Apo B/Apo A-1 ratio with any DPP-4 inhibitors.
Thus, in this study, we highlighted Apo B and Apo A-1 and studied the effect of the DPP-4 inhibitors alogliptin [17,18] on Apo B and Apo A-1, in addition to lipid metabolism markers, and investigated the effect of alogliptin on markers predicting CV events, such as Apo B/Apo A-1 and LDL-C/Apo B.
Patients and Methods
Patients
Subjects were type 2 diabetic patients aged 20 years or older at our hospital that were taking oral antidiabetic agents and had HbA1c levels of 6.9% or higher for three months or longer. Exclusion criteria were: patients receiving insulin therapy; severe liver or renal dysfunction; patients with hypersensitivity to alogliptin; patients that had their hypoglycemic agents changed within three months; pregnant women or breast-feeding mothers; and excessive alcohol drinkers.
This study was carried out with a single arm in an open label manner. After informed consent was obtained from patients, 45 consecutive cases that could enroll between September 2011 and March 2012 were selected as the subjects for randomization. In the 45 cases, alogliptin 25 mg/day was administered for eight weeks and non-fasting blood samples were collected 2-3 hours after breakfast, once at the beginning of the study and once eight weeks later, and changes in height, body weight, blood pressure, glucose metabolism markers and lipid metabolism markers were compared. Dietitians analyzed the ingredient of the breakfast by inquiring from 15 patients selected at random. The energy of their breakfast was 450Kcal, of which 32% was from fat. All the subjects take the same menu of the breakfast before study and eight weeks later. During this study, additional drugs that would influence blood pressure and lipid metabolism were not administered. Drug intake compliance was confirmed by interviewing patients at each visit to the hospital. Dietary and exercise therapy were unchanged before, during and after the study, and regular instructions were continued. We registered University hospital Medical Information Network (UMIN), and the registration number is 000011470.
Background information in the 45 cases that received alogliptin is shown in Table 1. Antidiabetic agents were administered as follows: sulfonylurea (SU), 27 cases; a-glucosidase inhibitors (a-GIs), 21; glinides, 14; biguanides (BGs), 18; and TZD, 11. Of these, all a-GIs and glinides were changed to alogliptin. Doses of BGs and TZD were unchanged and co-administered with alogliptin co-administered. With regard to SU, the dose was unchanged in 15 cases, reduced in 10, and increased, or another drug was necessary, in two. During administration, no adverse events occurred and administration was not discontinued in any case.
![]()
Table 1: Patient backgrounds and status of concomitant drug administration
View Table 1
Biochemical analyses
Non-fasting blood samples were collected 2�3 hours after breakfast, once at the beginning of the study and once eight weeks later. Apo A-1 and Apo B were measured by immune nephelometry with BM8060. HbA1c levels were measured by HPLC with HLC-723G8. Measurements were converted to NGSP values with a conversion table recommended by the Japan Diabetes Society [19]. LDL-C and HDL-C were measured by the direct method, while TG was measured by the enzymatic colorimetric method with BM 2250.
Statistical analysis
In this study, all clinical data are expressed as means + standard deviation. The influence of alogliptin on HbA1c, non-fasting blood glucose, LDL-C, HDL-C, non-fasting TG, body weight, BMI, and blood pressure were statistically analyzed by the paired t-test. The effect of alogliptin on Apo B, Apo A-1, Apo B/Apo A-1 and LDL-C/Apo B was statistically analyzed by the non-parametric Wilcoxon matched pair test. SPSS (Version 11) was used for analysis.
Results
Effect of alogliptin on glycemic control
Alogliptin administration significantly reduced HbA1c levels from 7.4 + 1.0% to 7.0 + 0.7 % (p< 0.0001) (Table 2). HbA1c levels were reduced from 20.0% before treatment to lower than 6.9% after administration in 50.6% of cases (p< 0.001). HbA1c levels were improved significantly irrespective of the concomitant drugs, with the exception of the group that switched from glinides (Figure 1a). HbA1c levels were also significantly reduced at -0.5% even in the reduced dose SU group (Figure 1b). There was no significant difference in the degree of reduction by alogliptin between obese patients with BMI = 25 kg/m2 and non-obese patients with BMI< 25 kg/m2 (Figure 1c). Non-fasting blood glucose levels were significantly reduced by alogliptin from 164.6 + 39.2 mg/dL (9.14 + 2.18 mmol/L) to 141.2 + 35.6 mg/dL (7.84 + 1.98 mmol/L) (p< 0.001) (Table 2).
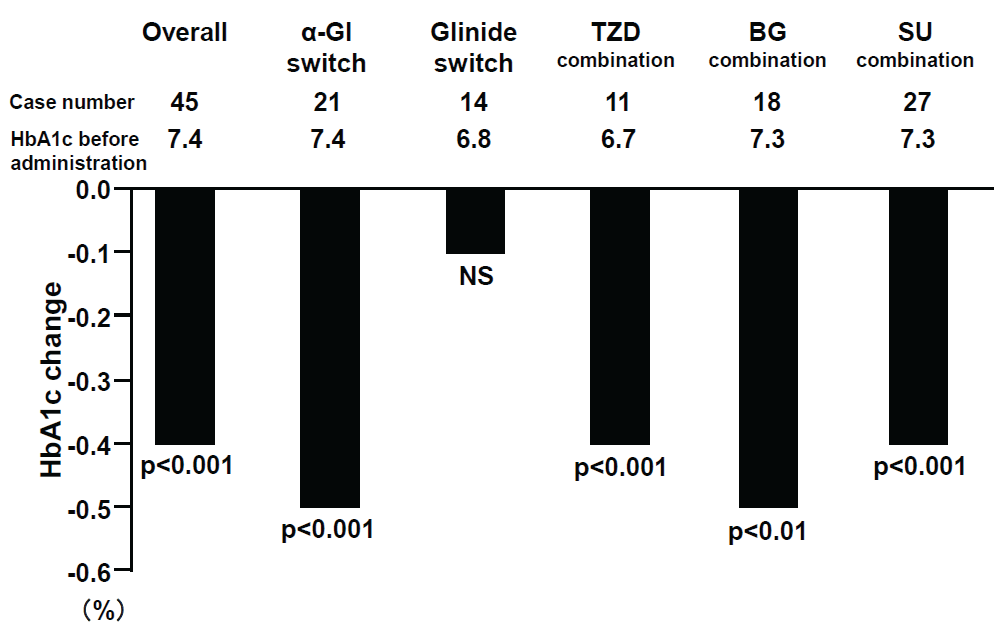
Figure 1a: HbA1c changes and combination drugs with alogliptin
View Figure 1a
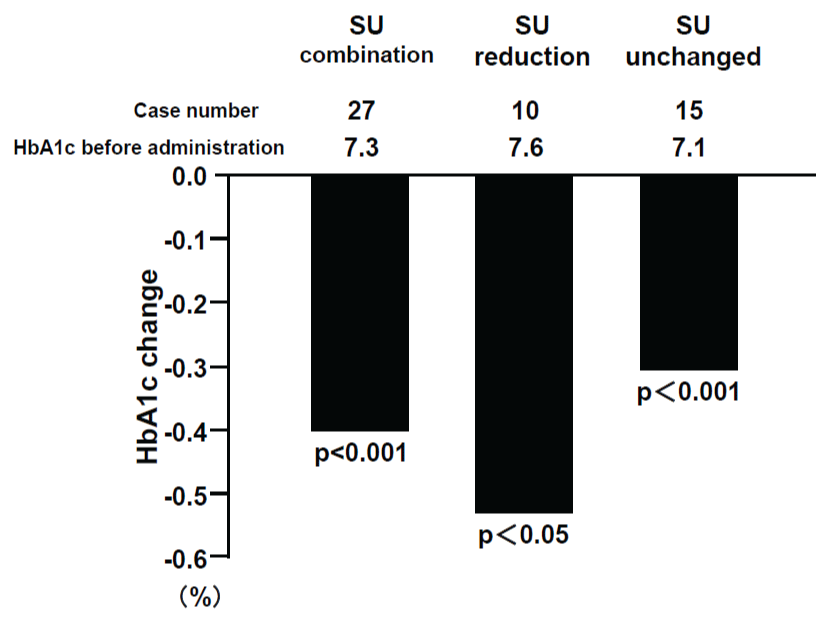
Figure 1b: HbA1c changes by alogliptin and status of SU administration
View Figure 1b
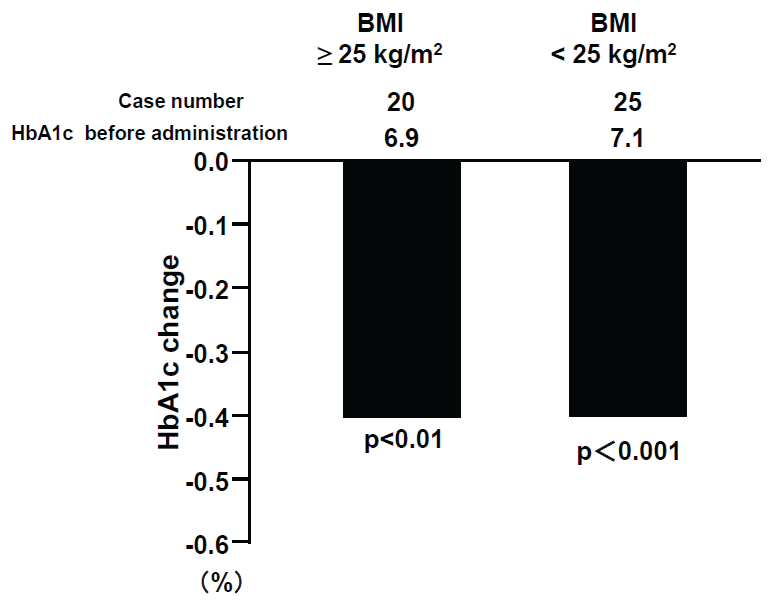
Figure 1c: HbA1c changes by alogliptin and BMI
View Figure 1c
![]()
Table 2: Changes in glucose level and lipid metabolism markers by alogliptin
View Table 2
Effect of alogliptin on lipid metabolism
Alogliptin administration reduced LDL-C from 102.9 + 19.7 mg/dL (2.66 + 0.51 mmol/L) to 97.9 + 22.5mg/dL (2.53 + 0.58 mmol/L) (p< 0.01). Alogliptin elevated HDL-C from 60.7 + 19.3 mg/dL (1.57 + 0.50 mmol/L) to 61.8 + 18.3 mg/dL (1.60 + 0.47 mmol/L), but the change was not significant. In contrast, the LDL-C/HDL-C ratio significantly decreased from 1.83 + 0.60 to 1.68 + 0.53 (p< 0.01). Moreover, non-fasting TG significantly decreased from 137.4 + 93.9 mg/dL (1.55 + 1.06 mmol/L) to 116.0 + 71.1 mg/dL (1.31 + 0.80 mmol/L) (p< 0.05) (Table 2).
The changes (amount and rate) of LDL-C, HDL-C, non-fasting TG were compared between obese (BMI = 25 kg/m2) and non-obese patients (BMI< 25 kg/m2). The changes HDL-C in the obese patients were significantly more than the non-obese patients. There were no significant differences in the changes of LDL-C, HDL-C, and non-fasting TG between anti-diabetic agents (metformin, a-GIs, glinide, pioglitazone and sulfonylureas) combined with alogliptin.
Effect of alogliptin on apoprotein
Apo B was significantly reduced by alogliptin treatment from 91.0 + 16.4 mg/dL (0.91 + 0.16g/L) to 85.0 + 14.0 mg/dL (0.85 + 0.14 g/L) (p< 0.01). Apo A-1 was unchanged in response to alogliptin.
In contrast, the Apo B/Apo A-1 ratio was significantly decreased by alogliptin from 0.61 + 0.15 to 0.57 + 0.12 (p< 0.05), and the LDL-C/Apo B ratio significantly increased from 1.16 + 0.11 to 1.20 + 0.13 (p< 0.01) (Table 3).
![]()
Table 3: Alogliptin-induced changes in apoprotein and predictive markers for the onset risk of cardiovascular events
View Table 3
The changes (amount and rate) of Apo B, Apo A-1, Apo B/Apo A-1, LDL/Apo B were compared between obese (BMI = 25 kg/m2) and non-obese patients (BMI < 25 kg/m2). The changes Apo A-1 in the obese patients were significantly more than the non-obese patients (Table 4). There were no significant differences in the changes of Apo B, Apo A-1, Apo B/Apo A-1, LDL/Apo B between anti-diabetic agents (metformin, a-GIs, glinide, pioglitazone and sulfonylureas) combined with alogliptin.
![]()
Table 4: Changes in lipid metabolism markers by alogliptin between patients with BMI < 25 kg/m2
and BMI > 25 kg/m2
. Data are means � SD. * p < 0.05 between BMI
< 25kg/m2
and BMI > 25kg/m2
View Table 4
The change in the pre-administration Apo B/Apo A-1 ratio was positively correlated with body weight, TG, HbA1c, blood CPR and pooled urine CPR and negatively correlated with HDL-C (Table 5). Changes in LDL and HbA1c before and after administration were positively correlated (Table 6).
![]()
Table 5: Correlations between change in Apo B/Apo A-1 and clinical parameters (values prior to treatment)
View Table 5
![]()
Table 6: Correlations between change in Apo B/Apo A-1 and parameters (changes)
View Table 6
Effect of alogliptin on blood pressure
Alogliptin effected reductions in SBP (125.2 + 12.8 mmHg to 123.6 + 15.8 mmHg) and DBP (67.6 + 9.0 mmHg to 65.9 + 10.7 mmHg) but neither change was significant. However, since blood pressure guidelines recommend a SBP< 130 mmHg, only 18 cases with abnormal SBP (defined as 130 mmHg or higher) were examined, and in these cases, SBP was significantly reduced by alogliptin treatment from 137.9 + 5.5 mmHg to 128.0 + 15.6 mmHg (p< 0.01).
Discussion
We have shown here for the first time that administration of alogliptin over an eight week period at 25 mg/day improved not only blood glucose levels, but also blood levels of LDL-C and non-fasting TG. Furthermore, by improving Apo B levels, alogliptin significantly reduced the Apo B/Apo A-1 ratio, a reliable predictive marker for the onset risk of acute myocardial infarction. It also significantly increased the LDL-C/Apo B ratio, which correlated with small dense LDL, which would be expected to reduce the proportion of small dense LDL.
It has been reported that DPP-4 inhibitors could decrease fasting serum TG. Furthermore, inhibition by alogliptin of TG, Apo B48 and Apo B100 elevation for up to 8 h after meals, has been attributed to improvements in chylomicron TG derived from foods and TG contained in VLDL from the liver [20]. Moreover, it is known that GLP-1 inhibits both TG absorption from the intestine and production of Apo B48 in mice [21]. In human, reduction of Apo B and Apo B48 is reported in sitagliptin [15] and reduction of Apo B is reported in vildagliptin [22]. The reduction in non-fasting TG and Apo B by alogliptin in this study is consistent with the previous reports of TG reduction by GLP-1. In this context, it can be speculated that continuous suppression by alogliptin of the enzymatic activity of DPP-4 results in chronically elevated GLP-1 levels, and suppression of postprandial TG elevation.
In many clinical studies, it has been reported that treatment of LDL-C could prevent CV events. The effect of DPP-4 inhibitors on LDL-C is still controversial. Sitagliptin [12,15] and vildagliptin [23] have been shown to reduce TG, but not LDL-C. In addition, a meta-analysis of the effect of DPP-4 inhibitors on T-C showed that vildagliptin and alogliptin decreased T-C in contrast to sitagliptin [13], suggesting the possibility that distinct DPP-4 inhibitors have different effects on cholesterol metabolism. In this study, alogliptin administration reduced postprandial LDL-C.
According to the INTERHEART study, non-fasting Apo B and Apo B/Apo A-1 are important predictive markers for the onset risk of acute myocardial infarction [14] In particular, the Apo B/Apo A-1 ratio corresponded to the highest odds ratio among predictive markers and it has been considered a marker that would sensitively predict acute myocardial infarction independent of races, gender, age or diet. In this study eight weeks of alogliptin administration significantly reduced Apo B and the Apo B/Apo A-1 ratio. Since the possibility that DPP4 inhibitors may prevent CV events has attracted attention, the results of this study suggest the potential of this drug for suppressing CV events. When we examined factors that influenced the improvement in the Apo B/Apo A-1 ratio by alogliptin, we found significant positive correlations with body weight, TG, HbA1c, and blood and pooled urine CPR prior to administration, and a significant negative correlation with HDL-C.
In metabolic syndrome cases where body weight and TG are increased and intrinsic insulin secretion is maintained or is slightly increased due to insulin resistance, an improvement in Apo B/Apo A-1 is unusual. CETP activity is increased under insulin resistance, and excessive TG levels increase cholesterol transfer from HDL-C to VLDL, and reduce HDL-C. Therefore, alogliptin was more effective in cases where HDL-C levels were reduced. In this study, by comparison of all cases, Apo A-1 and HDL-C did not have the significant difference with about administration of alogliptin. However, when they divided it into the obesity patient and the non-obesity patient, HDL-C and Apo A-1 were significantly more in the obesity patients compared with the non-obesity patient. Administration of alogliptin to the obesity patient may contribute to anti-atherosclerosis by increase HDL-C and Apo A-1.
In the context of DPP-4 inhibitors, the degree of HbA1c reduction has been shown to be larger in cases with higher HbA1c levels before administration [24] and, similarly, we observed an improvement in the Apo B/Apo A-1 ratio. In response to postprandial hyperglycemia, DPP-4 inhibitors, increase GLP-1 and insulin secretion and suppress glucagon secretion. In cases with higher remaining capability of insulin secretion, reactive insulin secretion and suppression of glucagon secretion were more pronounced and both blood glucose levels and lipid metabolism were improved.
We have shown that improvement in Apo B/Apo A-1 has a significant positive correlation with change in LDL-C and HbA1c before and after the administration of alogliptin. A positive correlation with HbA1c suggests that the improvement in glycemic control by alogliptin contributed to an improvement in the Apo B/A-1 ratio. In this study, postprandial blood CPR was not significantly different before and after the administration, which we speculate is related to reduced insulin resistance by suppression of glucagon secretion.
In animal experiments, GLP-1 signal activation has been shown to suppress cholesterol absorption from the intestine [21] and GLP-1 analog administration is known to upregulate LDL receptor expression in the liver [25], suggesting a possible mechanism of not only LDL-C reduction via insulin but also direct reduction of GLP-1. In this study, alogliptin administration increased the LDL-C/Apo B ratio. The LDL/Apo B ratio is considered a marker for LDL-C particle diameter and a reduction in this marker indicates an increase in levels of small dense LDL, a smaller diameter particle [11]. While several reports have shown that alogliptin increases LDL-C particle diameter [26-28], this effect was also observed in surrogate markers.
Finally, alogliptin administration had no significant overall effect on blood pressure although blood pressure decreased in patients with SBP 130mmHg or higher prior to administration. Ogawa et al. have reported that administration of sitagliptin [29] and the GLP-1 agonist exenatide [30] reduce blood pressure, and suggesting that the reduction in blood pressure is attributable to the effect of GLP-1 per se.
Taken together, alogliptin had the effect of reducing lipid and blood pressure in addition to blood glucose levels in type 2 diabetic patients and, based on these data, is expected to suppress the advancement of atherosclerosis. This is in agreement with a meta-analysis of phase II and phase III clinical trials in the U.S. which has shown that alogliptin reduced the major adverse CV events [31]. In EXAMINE trial presented in 2013, alogliptin didn�t demonstrate the superior effects on the prevention of death from CV causes, nonfatal myocardial infarction or nonfatal stroke in patients with type 2 diabetes with a recent acute coronary syndrome [32]. It is possible that the observation period was too short, and the subjects were too little for evaluation of the effect on prevention of CV events. In UKPDS trial, observation more than 10 years was necessary so that significant difference appeared by CV events. Therefore, the EXAMINE study may prove the beneficial effects of alogliptin if it has a longer observation period. In addition, it may influence it that the patients with very high risk for CVD were subjected. In the view of alogliptin effect, improved glucose and lipid metabolism and direct action of GLP-1 for cardiac function may be beneficial for CVD. However the risk reduction in the diabetic patients with CVD is difficult and earlier intervention should be needed.
Conclusions
Alogliptin administration in type 2 diabetic patients significantly improved levels of blood glucose, in addition to those of lipid metabolism markers such as LDL-C and non-fasting TG, as well as sensitive predictive markers for the onset risk of CV events, such as the Apo B/Apo A-1 ratio and the LDL-C/Apo B ratio. These results suggest the possibility that alogliptin could potentially suppress atherosclerosis and prevent CV events.
References
-
Booth GL, Kapral MK, Fung K, Tu JV (2006) Relation between age and cardiovascular disease in men and women with diabetes compared with non-diabetic people: a population-based retrospective cohort study. Lancet 368: 29-36.
-
Rana JS, Dunning A, Achenbach S, Al-Mallah M, Budoff MJ, et al. (2012) Differences in Prevalence, Extent, Severity, and Prognosis of Coronary Artery Disease Among Patients With and Without Diabetes Undergoing Coronary Computed Tomography Angiography: Results from 10,110 individuals from the CONFIRM (COronary CT Angiography EvaluatioN For Clinical Outcomes): an InteRnational Multicenter registry. Diabetes Care 35: 1787-1794.
-
Turner RC, Millns H, Neil HA, Stratton IM, Manley SE, et al. (1998) Risk factors for coronary artery disease in non-insulin dependent diabetes mellitus: United Kingdom Prospective Diabetes Study (UKPDS: 23). BMJ 316: 823-828.
-
Avramoglu RK, Basciano H, Adeli K (2006) Lipid and lipoprotein dysregulation in insulin resistant states. Clin Chim Acta 368: 1-19.
-
Nakamura H, Arakawa K, Itakura H, Kitabatake A, Goto Y, et al. (2006) Primary prevention of cardiovascular disease with pravastatin in Japan (MEGA Study): a prospective randomised controlled trial. Lancet 368: 1155-1163.
-
Matsuzaki M, Kita T, Mabuchi H, Matsuzawa Y, Nakaya N, et al. (2002) J-LIT Study Group. Japan Lipid Intervention Trial.; Large scale cohort study of the relationship between serum cholesterol concentration and coronary events with low-dose simvastatin therapy in Japanese patients with hypercholesterolemia. Circ J 66: 1087-1095.
-
Nordestgaard BG, Benn M, Schnohr P, Tybjaerg-Hansen A (2007) Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 298: 299-308.
-
American Diabetes Association (2012) Standards of medical care in diabetes--2012. Diabetes Care 35 Suppl 1: S11-63.
-
Koba S, Hirano T, Yoshino G, Sakai K, Sakaue T, et al. (2002) Remarkably high prevalence of small dense low-density lipoprotein in Japanese men with coronary artery disease, irrespective of the presence of diabetes. Atherosclerosis 60: 249-256.
-
Hirano T, Ito Y, Yoshino G (2005) Measurement of small dense low-density lipoprotein particles. J Atheroscler Thromb 12: 67-72.
-
Saito M, Hattori Y, Eto M (2011) Effects of 10-month Sitagliptin therapy on HbA1c, triglyceride and remnant cholesterol levels in type2 diabetic patients. [in Japanese] Jpn Pharmacol Ther 39: 217-223.
-
Charbonnel B, Karasik A, Liu J, Wu M, Meininger G, et al. (2006) Efficacy and safety of the Dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone. Diabetes care 29: 2638-2643.
-
Monami M, Lamanna C, Desideri CM, Mannucci E (2012) DPP-4 inhibitors and lipids: systematic review and meta-analysis. Adv Ther 29: 14-25.
-
McQueen MJ, Hawken S, Wang X, Ounpuu S, Sniderman A, et al. (2008) Lipids, lipoproteins, and apolipoproteins as risk markers of myocardial infarction in 52 countries (the INTERHEART study): a case-control study. Lancet 372: 224-233.
-
Tremblay AJ, Lamarche B, Deacon CF, Weisnagel SJ, Couture P (2011) Effect of sitagliptin therapy on postprandial lipoprotein levels in patients with type 2 diabetes. Diabetes Obes Metab 13: 366-373.
-
Matikainen N, M�ntt�ri S, Schweizer A, Ulvestad A, Mills D, et al. (2006) Vildagliptin therapy reduces postprandial intestinal triglyceride-rich lipoprotein particles in patients with type 2 diabetes. Diabetologia 49: 2049-2057.
-
Parsa KV, Pal M (2011) Preclinical development of dipeptidyl peptidase IV inhibitor alogliptin: a brief overview. Expert Opin Drug Discov 6: 855-869.
-
Covington P, Christopher R, Davenport M, Fleck P, Mekki QA, et al. (2008) Pharmacokinetic, pharmacodynamic, and tolerability profiles of the dipeptidyl peptidase-4 inhibitor alogliptin: a randomized, double-blind, placebo-controlled, multiple-dose study in adult patients with type 2 diabetes. Clin Ther 30: 499-512.
-
Kashiwagi A, Kasuga M, Araki E, Oka Y, Hanafusa T, et al. (2012) International clinical harmonization of glycated hemoglobin in Japan: From Japan Diabetes Society to national glycohemoglobin standardization program values. J Diabetes Investig 3: 39-40.
-
Eliasson B, M�ller-Goede D, Eeg-Olofsson K, Wilson C, Cederholm J, et al. (2012) Lowering of postprandial lipids in individuals with type 2 diabetes treated with alogliptin and/or pioglitazone: a randomized double-blind placebo-controlled study. Diabetologia 55: 915-925.
-
Hsieh J, Longuet C, Baker CL, Qin B, Federico LM, et al. (2010) The glucagon-like peptide 1 receptor is essential for postprandial lipoprotein synthesis and secretion in hamsters and mice. Diabetologia 53: 552-561.
-
Tani S, Nagao K, Hirayama A (2013) Association between urinary albumin excretion and low-density lipoprotein heterogeneity following treatment of type 2 diabetes patients with the dipeptidyl peptidase-4 inhibitor, vildagliptin: A pilot study. Am J Cardiovasc Drugs 13: 443-450.
-
Saito M, Baba M, Abe T (2011) Vildagliptin reduces postprandial levels of triglyceride and remnant cholesterol, and blood pressure in type2 diabetic patients. [in Japanese] Jpn Pharmacol Ther 39: 805-811.
-
Deacon CF (2011) Dipeptidyl peptidase-4 inhibitors in the treatment of type 2 diabetes: a comparative review. Diabetes Obes Metab 13: 7-18.
-
Li L1, Miao Z, Liu R, Yang M, Liu H, et al. (2011) Liraglutide prevents hypoadiponectinemia-induced insulin resistance and alterations of gene expression involved in glucose and lipid metabolism. Mol Med 17: 1168-1178.
-
Tsuchiya M (2011) Alogliptin Ameliorates Dyslipidemia and LDL-Size in Patients with IGT or Type 2 Diabetes: Comparing Alogliptin and Voglibose. Diabetes 60: A280.
-
Fleck P, Wilson C, Deerfield IL (2012) Effect of Alogliptin combined with Pioglitazone on lipids and lipoprotein particles in patients with Type 2 Diabetes. Diabetes 61: A300.
-
Tsuchiya M, Nishio Y, Maegawa H (2012) Long-term effect of DPP4 Inhibition on dyslipidemia and the size of LDL particles in patients with IGT or Type 2 Diabetes. Diabetes 61: A577.
-
Ogawa S, Ishiki M, Nako K, Okamura M, Senda M, et al. (2011) Sitagliptin, a dipeptidyl peptidase-4 inhibitor, decreases systolic blood pressure in Japanese hypertensive patients with type 2 diabetes. Tohoku J Exp Med 223: 133-135.
-
Okerson T, Yan P, Stonehouse A, Brodows R (2010) Effects of Exenatide on systolic blood pressure in subjects with type 2 diabetes. Am J Hypertens 23: 334-339.
-
White WB, Pratley R, Fleck P, Munsaka M, Hisada M, et al. (2013) Cardiovascular safety of the dipetidyl peptidase-4 inhibitor alogliptin in type 2 diabetes mellitus. Diabetes Obes Metab 15: 668-673.
-
White WB, Cannon CP, Heller SR, Nissen SE, Bergenstal RM, et al. (2013) Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med 369: 1327-1335.





