International Journal of Diabetes and Clinical Research
Quantitative Models for Evaluating the Correlation between Baseline Hba1c Levels and Sitagliptin as Monotherapy or Dual Therapy Treatment in Type 2 Diabetes: A Meta-Regression Analysis
Jiajing Yin, Yi Lin, Mingyu Gu and Yongde Peng*
Department of Endocrinology and Metabolism, Shanghai General Hospital, Shanghai Jiao Tong University, China
*Corresponding author:
Yongde Peng, M.D Ph.D, Department of Endocrinology and Metabolism, Shanghai General Hospital, Shanghai Jiao Tong University, 100 Haining Road, Shanghai 200080, China, Tel: +86 21-63240090- 3722, +86-13386259649, Fax: +86 21-65830185, E-mail: pengyongde2015@sina.com
Int J Diabetes Clin Res, IJDCR-3-051, (Volume 3, Issue 1), Research Article; ISSN: 2377-3634
Received: November 18, 2015 | Accepted: December 31, 2015 | Published: January 04, 2016
Citation: Yin J, Lin Y, Gu M, Peng Y (2016) Quantitative Models for Evaluating the Correlation between Baseline Hba1c Levels and Sitagliptin as Monotherapy or Dual Therapy Treatment in Type 2 Diabetes: A Meta-Regression Analysis. Int J Diabetes Clin Res 3:051. 10.23937/2377-3634/1410051
Copyright: © 2016 Yin J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aim: Baseline glycated hemoglobin (HbA1c) is critical in assessing anti-diabetic agent efficacy due to its ability to alter the response of HbA1c reduction to dipeptidyl peptidase-IV (DPP-IV) inhibitors. With meta-regression, we aim to investigate the quantitative relationship between baseline HbA1c levels and HbA1c reduction in response to sitagliptin as a monotherapy or metformin + sitagliptin as dual therapy. Additionally, we intend to study the slope difference between monotherapy and dual therapy.
Methods: We conducted an electronic search for randomized controlled trials (RCTs) involving monotherapy (sitagliptin) and dual therapy (sitagliptin and metformin) in type 2 diabetes patients. We analyzed quantitative relationships between baseline HbA1c and the change of HbA1c using meta-regression models.
Results: The mean baseline HbA1c levels ranged from 7.54 (SE = 0.85) to 9.00 (SE = 1.4) for monotherapy and 7.48 (SE = 0.72) to 9.90 (SE = 1.8) for dual therapy respectively. According to meta-regression models, the slopes of meta-regression lines were -0.198 (95% CI [-0.324, -0.072], p = 0.002) for monotherapy and -0.656 (95% CI [-0.925, -0.390], p < 0.001) for dual therapy, respectively.
Conclusions: The association between baseline HbA1c and the HbA1c change differed between monotherapy and dual therapy. Therefore, the effect of baseline HbA1c level on HbA1c changes during monotherapy or dual therapy should be considered when assessing the clinical efficacy of diabetes therapies.
Keywords
Baseline, HbA1c, Sitagliptin, Metformin, Type 2 diabetes
Introduction
Type 2 diabetes mellitus (T2DM) is a disease that causes significant medical and social burdens in developed countries and prevalences of T2DM are gradually increasing in both developed and developing countries [1]. According to International Diabetes Federation, 366 million people were diagnosed as diabetes in 2011. By 2030, the number of diabetes patients is expected to be 552 million, which render previous estimates conservative [2]. Prevention and treatment of T2DM remain a major global public health objective. It was shown that reduction of HbA1c was associated with reductions in risk of diabetes, myocardial infarction, microvascular complications and deaths related to diabetes [3], suggesting that focusing on achieving and sustaining glycemic control during diabetes treatment may be one of the important public health strategies in real-world practice. Current strategies to improve glycemic control include treatments with metformin, sulphonylureas, thiazolidinediones, meglitinides, a-glucosidase inhibitors, insulin, and several new classes of therapies, including amylinomimetics, incretin mimetics and dipeptidyl peptidase-4 (DPP-4) inhibitors (Sitagliptin, Vildagliptin, Saxagliptin, Alogliptin and Linagliptin) [4]. Although metformin is widely accepted as a first-line anti-diabetic drug in the treatment of most type 2 diabetes patients, the choice of ideal agents among various oral anti-diabetic drugs in combination with metformin after metformin's failure is still controversial [5]. A meta-analysis review showed that some randomized controlled trials (RCTs) provided evidence of improved glycemic control with the dipeptidyl peptidase-4 (DPP-4) inhibitors, which has a modest risk of hypoglycemia and no weight gain [4]. Turner et al. reported that in the U.S. the use of DPP-4 inhibitors had increased steadily since 2005, accounting for 21% of ambulatory diabetes treatment visits in 2012 [6].
According to popular therapeutic algorithms, assessment of diabetes therapy should be based on the baseline HbA1c, and a change in therapy is recommended when HbA1c is above the target- intensification of therapy is usually recommended when HbA1c is above 7% [7,8]. Recent articles based on several RCTs around the world supported the concordance in predictors of HbA1c response to glucose-lowering drugs (excluding insulin) between RCTs data and data from routine clinical care. The HbA1c reduction following therapy is greater in the patients with higher baseline HbA1c level [9]. However, little is known about relationships between effects of monotherapy/dual therapy and baseline HbA1c levels. We hypothesize that patients with different baseline HbA1c levels may respond differently to mono and dual therapies. In this study, we provided evidence to support our hypothesis by performing meta-regression analysis based on data from sitagliptin and sitagliptin + metformin randomized control trials. The relationship between mono or dual therapy and baseline HbA1c may provide insight for diabetes health care and maximize the efficacy of available therapies.
Materials and Methods
Search strategy
We performed a literature search using databases including PubMed, Embase, Medline, and Cochrane central register of controlled trials. The search was performed on Apr 2014 and was restricted to publications in English by two independent researchers. Our search strategy included keywords: "Hemoglobin A1c", "HbA1c", "A1C", "Incretins", "DPP-4 inhibitors", "Sitagliptin", "Metformin", "Monotherapy", "Dual therapy", and "Randomized control trial". We searched all fields in PubMed, all text in Medline, but restricted to the fields of abstracts, titles, and keywords in Embase. In addition, a manual search of journals was conducted in personal reference lists of recovered articles to track relevant RCTs that were not indexed by normal keywords.
Inclusion and exclusion criteria
The identified studies were selected according to the following inclusion and exclusion criteria.
(1) Study design: Only RCTs were included. Observational, cohort, case-control, case series, and laboratory studies were excluded. Studies published ahead of print in April 2014 were included.
(2) Durations: For observing changes in HbA1c levels, only the RCTs with follow-up durations longer than 12 weeks were included.
(3) Participants: Only the RCTs on adult, non-pregnant participants aged 18 and older with type 2 diabetes were included, with at least 30 participants in each trial.
(4) Interventions: The interventions have to contain sitagliptin as a monotherapy or sitagliptin as an add-on therapy to metformin. Triple therapy was not included.
(5) Comparators: This included the RCTs employing placebo or any anti-diabetic treatment (mono- or combination therapy). We excluded RCTs employing initiation of triple therapy at the same time.
Outcome
This study included the RCTs measuring HbA1c as the outcome, containing baseline HbA1c levels, and changing of glycosylated hemoglobin (HbA1c, mean and standard error (SE) of change of HbA1c should be reported). Trials were excluded if: (A) trials were extensions of previous RCTs, as extended trials are more likely to be biased, and those patients who had loss of glycemic control were not enrolled in the extension part of the randomized trial; (B) Sitagliptin was added to another anti-diabetic treatment (not included in this analysis); (C) the doses of DPP-4 inhibitors were different from those recommended and approved in the clinical practice.
Study selection and data extraction
The following information was sought from each trial: (A) author identification, (B) year of publication, (C) number of patients in the trial and study design, (D) sitagliptin monotherapy treatment, (E) sitagliptin in dual therapy treatment (sitagliptin + metformin), (F) follow-up time, (G) dose of anti-diabetic agents, (H) baseline HbA1c, (I) change of HbA1c from baseline (mean and standard error (SE) can be retrieved from confidence intervals or exact p-values if necessary), and (J) type of statistical analysis for the outcome. We attempted to contact study authors for additional information when necessary. The relevance of studies was assessed with a hierarchical approach on the basis of title, abstract, and the full manuscript. After the initial screening of titles and abstracts, the studies included by both researchers were compared; disagreement was resolved by consensus.
Quality assessment
We assessed the design, execution, and reporting of the included RCTs to Consolidated Standards of Reporting Trials. We assessed the risk of bias in random sequence generation and allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), and selective reporting (reporting bias). The risk of bias was classified as high, low, or unclear. The quality of each RCT was assessed by one researcher and verified by the other researcher.
The disagreement was resolved by discussion. The evidential level of the outcome was determined in accordance with the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system21 and conducted with GRADE profiler V.3.2 (http://tech.cochrane.org/revman/gradepro).
Data analysis
From each study, the mean decrease of HbA1c and its SE was extracted. Heterogeneity was assessed by using Q statistic and I2, the proportion of total variance due to the heterogeneity across trials rather than sampling error. I2 < 25% was considered as low in heterogeneity, and I2 > 75% was high heterogeneity. Random-effects models were used for the meta-analysis of summarizing the change of HbA1c across different studies. To estimate the slope of HbA1c change by baseline HbA1c level, linear mixed-effects models were used with the mean baseline HbA1c level of each study as a covariate. Both models employed weighted approaches, where the weight of each study is the inverse of the variance associated with the estimated HbA1c change. Change of HbA1c was predicted at different baseline HbA1c levels based on the linear models fitted to either the monotherapy studies or the studies with Sitagliptin added to metformin (dual therapy). All statistical tests were two-tailed and p-values < 0.05 were regarded as significant. All analysis were conducted with R-studio (Boston, MA).
Results
Search results
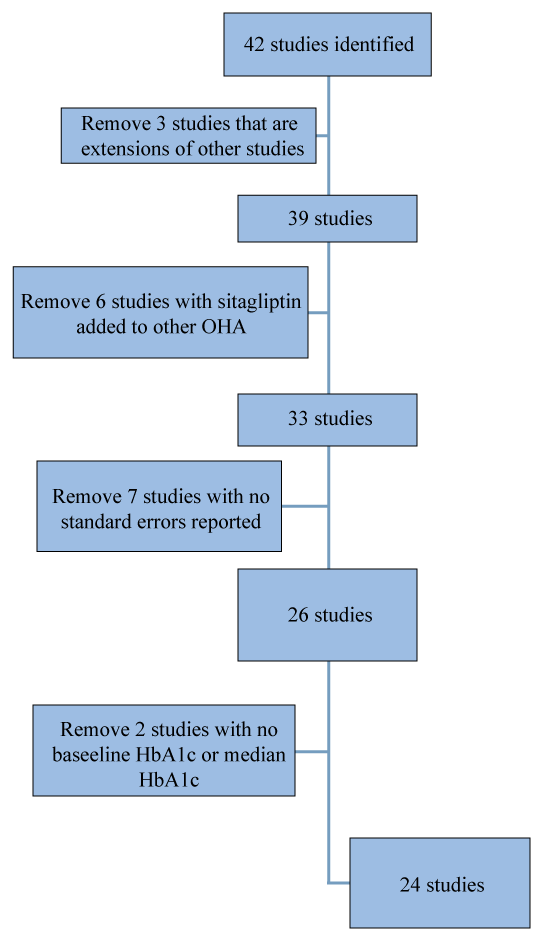
A total of 42 RCTs were identified via bibliographical databases for literature search including PubMed, Embase, Medline, and Cochrane central register of controlled trials (Figure 1). Finally, a total of 24 RCTs, which can be divided into 2 groups (12 articles for each group) according to whether sitagliptin was used as a mono or dual therapy, were included for quantitative synthesis and meta-analysis.
As shown in table 1 and table 2, the participants in all RCTs were patients with type 2 diabetes (≥ 18 years old). The outcome measuring the effects of DPP-4 inhibitors was the change in HbA1c level (in percentage). The data extracted from the included RCTs for meta-analysis were sample sizes and HbA1c change from baseline. Most trials were multinational and sponsored by industry. The trials were published between 2006 and 2012. All trials were of parallel groups: 21 were of a double-blind design, and the remaining 3 were of open-label design. RCTs with follow-up durations longer than 12 weeks were included in this study to observe changes in HbA1c levels. As HbA1c levels decreased in response to metformin and sitagliptin, we only used the most recent RCTs or those with follow-up time closest to 24 weeks. The trials evaluated 5655 patients for the primary endpoint. The patients were either drug naïve or discontinued any previous drug before randomization. The characteristics of trial participants were: mean age range 49.4-71.6 years old, trial duration range 12-104 weeks, and mean baseline HbA1c range 7.2-9.9%.
![]()
Table 1: Twelve studies selected as sitagliptin as mono therapy group (group 1).
View Table 1
![]()
Table 2: Twelve studies selected as sitagliptin as dual therapy group (group 2).
View Table 2
The average quality of search results
According to the Cochrane risk of bias tool, the two common biases (unclear risk of bias) were selection bias (lack of specification of allocation concealment) and detection bias (Data not shown). The average quality of the RCTs was acceptable and the GRADE evaluation indicated that the outcome of HbA1c had a moderate-to-high quality of the evidence.
Primary results
In the group where sitagliptin was used as monotherapy, the mean baseline HbA1c levels ranged from 7.54% (SE = 0.85) to 9.00% (SE = 1.4). The mean changes of HbA1c from baseline after sitagliptin monotherapy treatment was -0.60% (weighted by sample size, 95% CI [-0.68%, -0.51%]) and ranged from -0.43% (95% CI [-0.56%, -0.30%]) to -1.0% (95% CI [-1.15%, -0.85%]) (Figure 2A). In the group where sitagliptin was used with metformin as dual therapy, the mean baseline HbA1c levels ranged from 7.48% (SE = 0.72 in two studies respectively) to 9.90% (SE = 1.8). The mean changes of HbA1c from baseline after sitagliptin dual therapy treatment were -0.98% (weighted by sample size, 95% CI [-1.32%, -0.65%]) and ranged from -0.40% (95% CI [-0.65%, -0.15%]) to -2.40% (95% CI [-2.55%, -2.25%]) (Figure 2B).
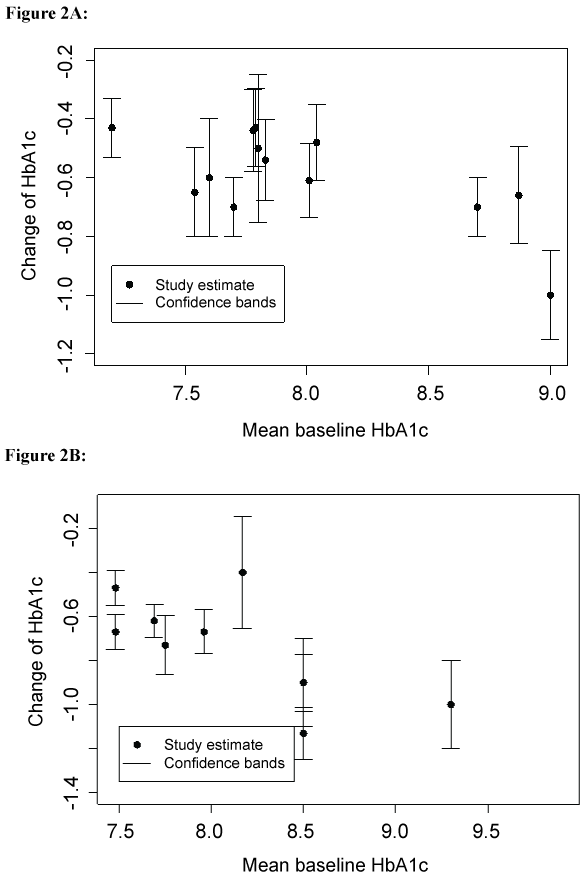
.
Figure 2: Plots of HbA1c change and mean baseline HbA1c in the trials using sitagliptin as mono (A) and dual therapy (B).
View Figure 2
In the meta-regression model where sitagliptin was used as monotherapy (Figure 3A), there was a 0.198% further reduction in HbA1c change from baseline HbA1c when baseline HbA1c level increased by 1% (Slope = -0.198, 95% CI [-0.324, -0.072], p < 0.002) with a modest heterogeneity (I2 = 66.84%, p < 0.0005) and R-square value of 49.97%. A similar relationship between baseline HbA1c and HbA1c reduction was found in the model where sitagliptin was used as dual therapy (Figure 3B), although the absolute value of the decreasing rate of HbA1c reduction is higher than that of monotherapy. When baseline HbA1c increased by 1%, HbA1c change further declined by 0.656% (Slope = -0.656, 95% CI [-0.925, -0.390], p < 0.001) with high heterogeneity (I2 = 96.91%, p < 0.001), and R-square value of 68.29%.
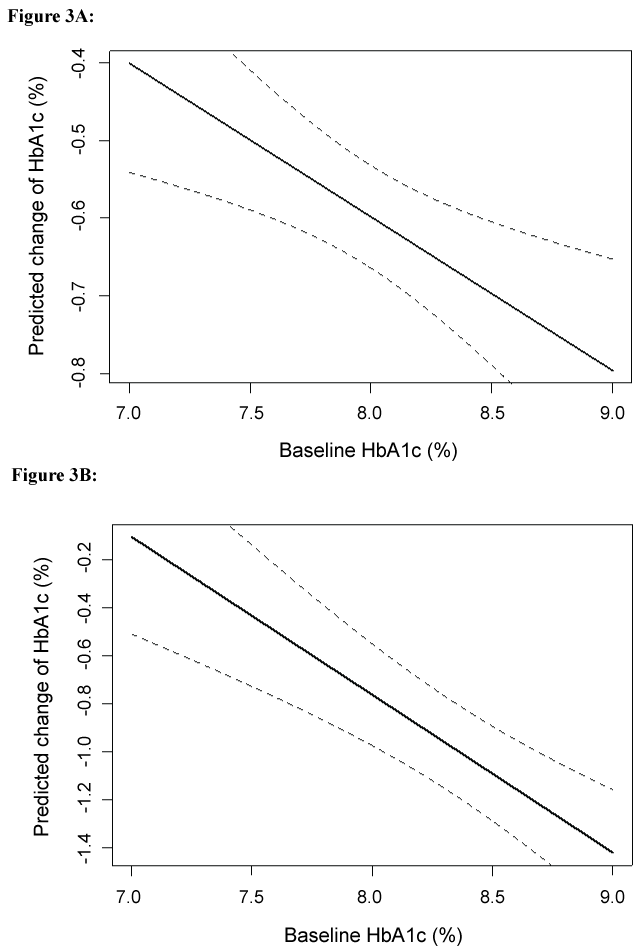
.
Figure 3: Meta-regression analysis of HbA1c change and mean baseline HbA1c in the trials using sitagliptin as mono (A) and dual therapy (B).
View Figure 3
We also conducted meta-regression analysis using relative changes of HbA1c (the change of mean HbA1c level divided by mean HbA1c level before therapy) as the dependent variable and mean baseline HbA1c as the predictor. The mean relative change of HbA1c from baseline after sitagliptin monotherapy treatment was -7.4% and ranged from -5.5% to -11.1% (Figure S1A). The mean relative change of HbA1c from baseline after sitagliptin dual therapy treatment were -11.4% and ranged from -4.9% to -24.2% (Figure S1B).
In the meta-regression model where sitagliptin was used as monotherapy (Figure S2A), there was a 1.480% further reduction in HbA1c relative change from baseline HbA1c when baseline HbA1c level increased by 1% (Slope = -1.480) with a modest heterogeneity (I2 = 66.06%, p = 0.001) and R-square value of 25.1%. In the model where sitagliptin as dual therapy, when baseline HbA1c increased by 1%, the percent HbA1c change further declined by 6.030% (Slope = -6.030) with high heterogeneity (I2 = 95.97%, p < 0.001) and R-square value of 60.50% (Figure S2B).
Discussion
Most recently, diabetes management algorithms recommended a HbA1c target of < 6.5-7.0% for the majority of people with diabetes if the target can be achieved safely [7,8]. Therefore, exploring the relationship between HbA1c reduction and any therapy is the first task for clinicians and patients to meet HbA1c goals. Esposito et al. have made a great contribution in this field [9-11]. Three factors, including baseline HbA1c, baseline fasting glucose, and the type of DPP-4 inhibitors have been identified by meta-regression analysis, which explains 61% of variance between RCTs with different baseline characteristics (such as baseline HbA1c, baseline fasting glucose, mean age, BMI, duration of treatment with a DPP-4 inhibitor, type of DPP-4 inhibitors, previous diabetes treatment in which a DPP-4 inhibitor was added to and statistical evaluation of the trial (per protocol or intention-to-treat)). Intuitively, the HbA1c reduction following a therapy with insulin or noninsulin drugs was greater when baseline HbA1c levels of the patients were higher [9]. A separate analysis for DPP-4 inhibitors indicated the same trend: for the baseline HbA1c higher than 7%, each 1% increase of baseline HbA1c resulted in a greater reduction of HbA1c (0.26% more) in response to DPP-4 inhibitors [10]. However, few studies have been conducted on a combined analysis between monotherapy and dual therapy so far. The present analysis explored the response of baseline HbA1c reduction and baseline HbA1c levels to trial-level covariates of sitagliptin as monotherapy and sitagliptin combined with metformin as a dual therapy treatment.
In our study, the separated analysis of sitagliptin as a monotherapy treatment indicated a significant reduction of HbA1c (0.2%) in response to DPP-4 inhibitors for each 1% increase of baseline HbA1c > 7%. This is lower than what is reported by Esposito et al. (0.26%), where all selected DPP-4 inhibitors were considered as one class [10]. The analysis of sitagliptin combined with metformin as a dual therapy treatment showed a greater reduction in HbA1c (0.656%) in response to DPP-4 inhibitors for each 1% increase of baseline HbA1c > 7%, indicating a significant difference between mono- and dual therapies. Such difference may be caused by different data sources and analysis methods. For example, more trials with sitagliptin as monotherapy were included in this study (12 versus 8). For the dual therapy trials, we focused on sitagliptin + metformin while Esposito et al. considered sitagliptin in combination with any oral medications. Therefore, we may have less heterogeneity in the current study. Another weakness of the design of previous studies is that the same patient cohort may be included more than once. For example, the article published by Seck et al. [12] is an extension of study by Nauck et al. [13], and these two studies were both included in the analysis. Furthermore, we analyzed meta-regression results for sitagliptin and sitagliptin + metformin to support our hypothesis that patients may respond differently to mono- and dual therapies. In previous studies, Esposito et al. performed a pooled analysis and ignored such a difference between mono and dual therapy treatment [9-11]. In addition, we presented quantitative models for HbA1c change (both mono and dual therapy). Esposito et al. computed the slopes but did not present the quantitative models. Based on the quantitative models, we compared HbA1c changes of sitagliptin with other medications at the same baseline level. The quantitative models allow us to compare at any baseline level. Of course, Esposito et al. results are more comprehensive since they conducted a complete meta-analysis with all relevant medications. We used literature data for sitagliptin and sitagliptin combined with other medications, but Esposito et al. can assess other medications. In addition to baseline HbA1c, Esposito et al. also assessed the relationship between the change of HbA1c and other factors such as fasting glucose and age.
This study has several limitations. First, the baseline and changes of HbA1c analyzed were not collected from the same patients, which created potential heterogeneity between studies used in this meta-analysis. Second, due to the limited information available in the qualified articles selected, the other covariates such as age and ethnic group were not included in the meta-regression analysis. Third, by the nature of meta-regression analysis, our study is only considered as an exploratory study, and our findings cannot infer causality.
In summary, our analysis showed the change of HbA1c level decreased in both sitagliptin monotherapy and dual therapy when baseline HbA1c level increased. Our study confirmed the notion that baseline HbA1c should be considered as an important factor when assessing the agent efficacy in diabetic therapy. Moreover, our study demonstrates that different effects of baseline HbA1c on the HbA1c change in response to sitagliptin monotherapy and dual therapy, which fills the gap in the literature.
Conflict of Interest
The authors state that they have no conflict of interest.
References
-
Ginter E, Simko V (2012) Type 2 diabetes mellitus, pandemic in 21st century. Adv Exp Med Biol 771: 42-50.
-
Aguiree F, Brown A, Cho NH, Dahlquist G, Dodd S, et al. (2013) IDF Diabetes Atlas: sixth edition. International Diabetes Federation.
-
Stratton IM, Adler AI, Neil HAW, Matthews DR, Manley SE, et al. (2000) Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 321: 405-412.
-
Karagiannis T, Paschos P, Paletas K, Matthews DR, Tsapas A (2012) Dipeptidyl peptidase-4 inhibitors for treatment of type 2 diabetes mellitus in the clinical setting: systematic review and meta-analysis. BMJ 344: e1369.
-
Esposito K, Ceriello A, Giugliano D (2013) Does personalized diabetology overcome clinical uncertainty and therapeutic inertia in type 2 diabetes? Endocrine 44: 343-345.
-
Turner LW, Nartey D, Stafford RS, Singh S, Alexander GC (2014) Ambulatory treatment of type 2 diabetes in the U.S., 1997-2012. Diabetes Care 37: 985-992.
-
Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, et al. (2014) Management of Hyperglycemia in Type 2 Diabetes, 2015: A Patient-Centered Approach: Update to a Position Statement of the American Diabetes Association and the European Association for the Study of Diabetes. Dia Care 38: 140-149.
-
Tamez-Perez HE, Proskauer-Pena SL, Hernrndez-Coria MI, Garber MJ (2013) AACE Comprehensive Diabetes Management Algorithm 2013 Endocrine Practice. Endocrine Practice 19: 736-737.
-
Esposito K, Chiodini P, Bellastella G, Maiorino MI, Giugliano D (2012) Proportion of patients at HbA1c target < 7% with eight classes of antidiabetic drugs in type 2 diabetes: systematic review of 218 randomized controlled trials with 78 945 patients. Diabetes Obes Metab 14: 228-233.
-
Esposito K, Chiodini P, Capuano A, Maiorino MI, Bellastella G, et al. (2014) Baseline glycemic parameters predict the hemoglobin A1c response to DPP-4 inhibitors: meta-regression analysis of 78 randomized controlled trials with 20,053 patients. Endocrine 46: 43-51.
-
Esposito K, Chiodini P, Maiorino MI, Capuano A, Cozzolino D, et al. (2015) A nomogram to estimate the HbA1c response to different DPP-4 inhibitors in type 2 diabetes: a systematic review and meta-analysis of 98 trials with 24 163 patients. BMJ Open 5: e005892.
-
Seck T, Nauck M, Sheng D, Sunga S, Davies MJ, et al. (2010) Safety and efficacy of treatment with sitagliptin or glipizide in patients with type 2 diabetes inadequately controlled on metformin: a 2-year study. Int J Clin Pract 64: 562-576.
-
Nauck MA, Ellis GC, Fleck PR, Wilson CA, Mekki Q, et al. (2009) Efficacy and safety of adding the dipeptidyl peptidase-4 inhibitor alogliptin to metformin therapy in patients with type 2 diabetes inadequately controlled with metformin monotherapy: a multicentre, randomised, double-blind, placebo-controlled study. Int J Clin Pract 63: 46-55.
-
Aschner P, Chan J, Owens DR, Picard S, Wang E, et al. (2012) Insulin glargine versus sitagliptin in insulin-naive patients with type 2 diabetes mellitus uncontrolled on metformin (EASIE): a multicentre, randomised open-label trial. Lancet 379: 2262-2269.
-
Goldstein BJ, Feinglos MN, Lunceford JK, Johnson J, Williams-Herman DE, et al. Effect of initial combination therapy with sitagliptin, a dipeptidyl peptidase-4 inhibitor, and metformin on glycemic control in patients with type 2 diabetes. Dia Care 30: 1979-1987.
-
Hanefeld M, Herman GA, Wu M, Mickel C, Sanchez M, et al. (2007) Once-daily sitagliptin, a dipeptidyl peptidase-4 inhibitor, for the treatment of patients with type 2 diabetes. Curr Med Res Opin 23: 1329-1339.
-
Mohan V, Yang W, Son HY, Xu L, Noble L, et al. (2009) Efficacy and safety of sitagliptin in the treatment of patients with type 2 diabetes in China, India, and Korea. Diabetes Res Clin Pract 83: 106-116.
-
Nonaka K, Kakikawa T, Sato A, Okuyama K, Fujimoto G, et al. (2008) Efficacy and safety of sitagliptin monotherapy in Japanese patients with type 2 diabetes. Diabetes Res Clin Pract 79: 291-298.
-
Raz I, Hanefeld M, Xu L, Caria C, Williams-Herman D, et al. (2006) Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 49: 2564-2571.
-
Scott R, Wu M, Sanchez M, Stein P (2007) Efficacy and tolerability of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy over 12 weeks in patients with type 2 diabetes. International Journal of Clinical Practice 61: 171-180.
-
Barzilai N, Guo H, Mahoney EM, Caporossi S, Golm GT, et al. (2011) Efficacy and tolerability of sitagliptin monotherapy in elderly patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. Curr Med Res Opin 27: 1049-1058.
-
Iwamoto Y, Tajima N, Kadowaki T, Nonaka K, Taniguchi T, et al. (2010) Efficacy and safety of sitagliptin monotherapy compared with voglibose in Japanese patients with type 2 diabetes: a randomized, double-blind trial. Diabetes Obes Metab 12: 613-622.
-
Aschner P, Katzeff HL, Guo H, Sunga S, Williams-Herman D, et al. (2010) Efficacy and safety of monotherapy of sitagliptin compared with metformin in patients with type 2 diabetes. Diabetes Obes Metab 12: 252-261.
-
Perez-Monteverde, Seck T, Xu L, Lee MA, Sisk CM, et al. (2011) Efficacy and safety of sitagliptin and the fixed-dose combination of sitagliptin and metformin vs. pioglitazone in drug-naïve patients with type 2 diabetes. International Journal of Clinical Practice 65: 930-938.
-
Chan JC, Scott R, Arjona Ferreira JC, Sheng D, Gonzalez E, et al. (2008) Safety and efficacy of sitagliptin in patients with type 2 diabetes and chronic renal insufficiency. Diabetes Obes Metab 10: 545-555.
-
Charbonnel B, Karasik A, Liu J, Wu M, Meininger G, et al. (2006) Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone. Dia Care 29: 2638-2643.
-
Scott R, Loeys T, Davies MJ, Engel SS; Sitagliptin Study 801 Group (2008) Efficacy and safety of sitagliptin when added to ongoing metformin therapy in patients with type 2 diabetes. Diabetes Obes Metab 10: 959-969.
-
Nauck MA, Meininger G, Sheng D, Terranella L, Stein PP, et al. (2007) Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sulfonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: a randomized, double-blind, non-inferiority trial. Diabetes Obes Metab 9: 194-205.
-
Arechavaleta R, Seck T, Chen Y, Krobot KJ, O'Neill EA, et al. (2011) Efficacy and safety of treatment with sitagliptin or glimepiride in patients with type 2 diabetes inadequately controlled on metformin monotherapy: a randomized, double-blind, non-inferiority trial, Diabetes Obes Metab. 13: 160-168.
-
Pratley RE, Nauck M, Bailey T, Montanya E, Cuddihy R, et al. (2010) Liraglutide versus sitagliptin for patients with type 2 diabetes who did not have adequate glycaemic control with metformin: a 26-week, randomised, parallel-group, open-label trial. Lancet 375: 1447-1456.
-
Raz I, Chen Y, Wu M, Hussain S, Kaufman KD, et al. (2008) Efficacy and safety of sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes. Curr Med Res Opin 24: 537-550.
-
Reasner C, Olansky L, Seck TL, Williams-Herman DE, Chen M, et al. (2011) The effect of initial therapy with the fixed-dose combination of sitagliptin and metformin compared with metformin monotherapy in patients with type 2 diabetes mellitus. Diabetes Obes Metab 13: 644-652.
-
Scheen AJ, Charpentier G, Ostgren CJ, Hellqvist A, Gause-Nilsson I (2010) Efficacy and safety of saxagliptin in combination with metformin compared with sitagliptin in combination with metformin in adult patients with type 2 diabetes mellitus. Diabetes Metab Res Rev 26: 540-549.
-
Wainstein J, Katz L, Engel SS, Xu L, Golm GT, et al. (2012) Initial therapy with the fixed-dose combination of sitagliptin and metformin results in greater improvement in glycaemic control compared with pioglitazone monotherapy in patients with type 2 diabetes. Diabetes Obes Metab 14: 409-418.
-
Rigby SP, Handelsman Y, Lai YL, Abby SL, Tao B, et al. (2010) Effects of colesevelam, rosiglitazone, or sitagliptin on glycemic control and lipid profile in patients with type 2 diabetes mellitus inadequately controlled by metformin monotherapy. Endocr Pract 16: 53-63.
-
Bergenstal RM, Wysham C, Macconell L, Malloy J, Walsh B, et al. (2010) Efficacy and safety of exenatide once weekly versus sitagliptin or pioglitazone as an adjunct to metformin for treatment of type 2 diabetes (DURATION-2): a randomised trial. Lancet 376: 431-439.