Journal of Dermatology Research and Therapy
Quick Detection of Nonmelanoma Skin Cancer by Histopathology: Feasibility and Diagnostic Accuracy of Immediate Cutaneous Diagnosis
Umbert-Millet Pablo1, Sola-Ortigosa Joaquim1, Campos Begoña2 and Bassas-Vila Julio1
1Department of Dermatology, HUSC, Universitat de Barcelona, Spain
2Department of Public Health, HUSC, Universitat de Barcelona, Spain
*Corresponding author:
Joaquim Sola-Ortigosa, Department of Dermatology, Idcsalud Hospital Universitari Sagrat Cor, C/ París 83-85, 08029 Barcelona, Spain, Tel: +34-933-214-998, E-mail: quimsola@yahoo.com
J Dermatol Res Ther, JDRT-1-001, (Volume 1, Issue 1), Original Article; ISSN: 2469-5750
Received: February 14, 2015 | Accepted: March 14, 2015 | Published: March 16, 2015
Citation: Umbert-Millet P, Sola-Ortigosa J, Begona C, Bassas-Vila J (2015) Quick Detection of Nonmelanoma Skin Cancer by Histopathology: Feasibility and Diagnostic Accuracy of Immediate Cutaneous Diagnosis. Int J Dermatol Res Ther 1:001. 10.23937/2469-5750/1510001
Copyright: © 2015 Umbert-Millet P, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are
credited.
Abstract
Background: Dermoscopy, diagnostic cytology, confocal microscopy are used to improve diagnostic accuracy. Frozen section techniques have been used to control margins but not for diagnosis.
Objectives: We used a new frozen section technique called immediate cutaneous diagnosis for nonmelanoma skin cancer, assessing its feasibility and accuracy in obtaining a result within 15 minutes and recording limitations.
Methods and Materials: An observational, prospective clinical study was designed. Biological samples were divided into two: one half was included in paraffin, following the standard procedure, and immediate cutaneous diagnosis was applied in the other. Fresh tissue was examined by frozen sections with perpendicular slides and staining with toluidine blue. Two hundred and sixty patients were enrolled. Results: Immediate cutaneous diagnosis-toluidine blue had a sensitivity of 98.7% (95%CI: 93.0-100%) and a specificity of 92.6% (95%CI: 87.4-96.1%) in detecting nonmelanoma skin cancer. Inter-rater agreement between two dermatopathologists using Cohen's kappa was 0.796 (P< 0.0001) in a subsample of 101 cases. Immediate cutaneous diagnosis-toluidine blue had a sensitivity for squamous epithelial lesions of 78.3% (95%CI: 63.6-89.1%) and a specificity of 98.4% (95%CI: 95.5-99.7%).
Conclusion: Immediate cutaneous diagnosis is a new, ambulatory, minimally invasive, rapid diagnostic technique that is ideal when Mohs surgery infrastructure and staff are available.
Keywords
Frozen sections, Skin neoplasms, Diagnostic techniques, Mohs surgery
Introduction
Nonmelanoma skin cancer is the most common cancer worldwide [1,2] and a wide variety of diagnostic techniques have been developed. Today's society demands rapid reliable diagnosis of skin cancer with minimum morbidity [3]. For the last three decades, we have used Mohs micrographic surgery (MMS) for the removal of high risk skin cancers. MMS is a tissue-sparing technique for skin cancers with a cure rate of 99% for basal cell carcinomas (BCC) and 97% for squamous cell carcinomas (SCC) [4].
Reports suggest the accuracy of frozen section margins of skin cancer in surgical treatment is around 85% versus paraffin sections [5,6]. Specifically, traditional frozen section examinations are accurate in evaluating margins for BCC in approximately 90% to 95% of cases [7,8]. Frozen section techniques have been used to control margins but not for diagnosis. Most histopathologic diagnoses after punch biopsy are reported after the paraffin inclusions, with a minimum delay of two to five days.
In everyday practice, we usually use dermoscopy, diagnostic cytology or other techniques to improve diagnostic accuracy and precision. Although punch biopsy is still the gold standard in the diagnosis of skin cancer, we have developed a new diagnostic technique to improve diagnostic accuracy. This consists of a frozen section technique where the biopsy result and histopathologic report can be obtained in 15 minutes with the same, easily available technical tools and staff requirements as for MMS. We have named this diagnostic technique immediate cutaneous diagnosis.
Objectives
• To verify the feasibility of immediate cutaneous diagnosis of nonmelanoma skin cancer in order to obtain a quick result, and to record the possible limitations of the technique.
• To assess the diagnostic accuracy of immediate cutaneous diagnosis in the clinical setting of MMS and obtain estimates of sensitivity and specificity with respect to the standard paraffin sections and inter-rater reliability among dermopathologists.
Methods
An observational prospective clinical study was designed. The study population consisted of patients of both sexes and all ages with skin lesions suggestive of nonmelanoma skin cancer. Two hundred and sixty patients were recruited by consecutive sampling from outpatients at the Dermatology Department.
Patients were considered for inclusion in the study if the authors suspected lesions with malignancy, poorly defined clinical borders, or residual or relapsing tumors without prior histological diagnosis. Exclusion criteria were clinically evident skin lesions which were easily diagnosed and did not require punch biopsy for diagnosis.
All patients were informed of the study characteristics and provided signed informed consent. The study was approved by the hospital ethics committee.
The anatomic site of the lesion was recorded and classified as: face/neck, scalp, trunk, upper and lower limbs, genitalia or oral mucosa. The suspected clinical diagnosis and age were collected.
Tissue was extracted by 4 millimeter punch biopsy. In exophytic lesions with narrow pedicle, slicing was performed. In extensive or deep lesions, where it was expected that the puncture would provide little material, small spindle removal was done.
Every specimen or biological sample was divided into two halves: one half was included in paraffin to serve as reference [9], and the other half was manipulated to give frozen sections and perform immediate cutaneous diagnosis, which is the index test (Figure 1).
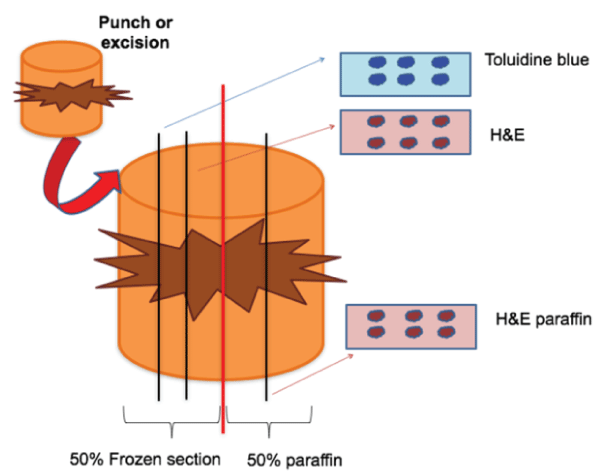
Figure 1: Immediate cutaneous diagnosis procedure. (H&E: Hematoxylin and Eosin stain).
View Figure 1
The half manipulated using paraffin section was processed in the traditional manner: 1-Fix in formaldehyde at different concentrations / 2-Include in paraffin / 3-Cut vertical sections to exhaust the entire piece / 4-Stain with hematoxylin and eosin (H&E).
The other half was examined by frozen sections with perpendicular slides and staining with toluidine blue. The steps of this procedure (6 minutes) were:
95° alcohol (1 min)
Toluidine blue stain (20 sec)
Rinse in tap water and distilled water, twice
95° alcohol, twice
Absolute alcohol, twice
Xylene, twice
Mount with DPX
Immediate cutaneous diagnosis staining with hematoxylin and eosin (10 minutes) was carried out as follows:
95° alcohol (1 min)
Hematoxylin (10 sec)
Rinse with water
Hydrochloric acid wash
Acetic acid wash
Rinse in tap water
Eosin (5 sec)
95° alcohol, twice
Absolute alcohol, twice
Xylene, twice
Mount in with DPX
Tissues were manipulated and processed by two trained histotechnicians with experience in MMS. Immediate cutaneous diagnosis histological preparations dyed with toluidine blue were blindly read in batches by one dermatopathologist who filled in a coded list. In 23 per cent of cases (60/260) immediate cutaneous diagnosis preparations also dyed with hematoxylin and eosin were then read in the same way, and lastly the paraffin sections were read.
For validation, four diagnoses were considered together as squamous epithelial lesions: actinic keratosis, squamous cell carcinoma, Bowen's disease and keratoacanthoma.
A random subset of 101 biological samples was reviewed later by a second dermatopathologist blinded to the previous results, in order to determine the inter-rater reliability of the immediate cutaneous diagnosis test.
The sample size was determined using sample size calculation to estimate a single proportion using the large sample normal approximation. For an expected sensitivity of 0.8, a minimum sample of 62 patients with a diagnosis of cancer, according to the reference standard, was required for a two-sided 95% confidence interval with a precision of 0.1 [10]. The same number of patients without this diagnosis was required to estimate specificity. The total sample size was obtained assuming a 30% detection of cancer as a previous estimate in the biopsies performed.
The sensitivity and specificity of immediate cutaneous diagnosis were evaluated using contingency tables. Two-sided 95% confidence intervals (CI) were obtained using the Agresti-Coull method or the exact binomial method. The statistical significance of diagnostic differences in the frequency distributions of other variables such as the anatomic site was calculated using the Chi-square test and nonparametric Mann-Whitney-Wilcoxon test for continuous variables such as age. A P-value < 0.05 was considered significant. Data were recorded using Microsoft Excel. The statistical analysis was made using the Statistical Package for Social Sciences (SPSS) (version 18.0) for Windows.
Results
Between 1 January 2010 and 1 December 2012, two hundred and sixty patients were enrolled, of whom 177 (68%) were male and 83 (32%) female. The median age was 67 years (IQR: 55.3 to 78). The predominant anatomic sites were the face and neck (60.8%). The baseline characteristics of patients are shown in table 1.
![]()
Table 1: Demographic and clinical characteristics of study patients
View Table 1
Histological findings evaluated using paraffin are summarized in table 2. Twenty-five different diagnoses were found, including no pathology/nonspecific. Ten specimens remained uncertain due to lost or insufficient material, and therefore the total number of patients finally included was two hundred and fifty. The most common pathologic tumor types were BCC, melanocytic nevi, actinic keratoses and SCC, which represented almost 70% of valid cases. Five histopathologic examples are shown in figures 2 to 6.
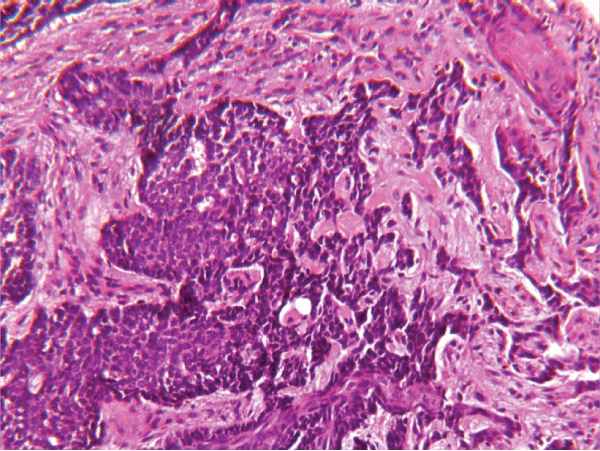
Figure 2: Paraffin with hematoxylin and eosin stain: Infiltrative basal cell carcinoma. The tumor cells are present as narrow strands.
View Figure 2
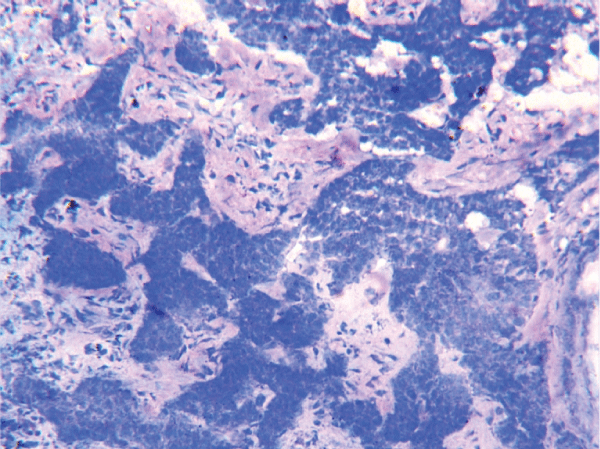
Figure 3: Frozen section with toluidine blue stain: Tissue section of a basal cell carcinoma showing the tumor lobules.
View Figure 3
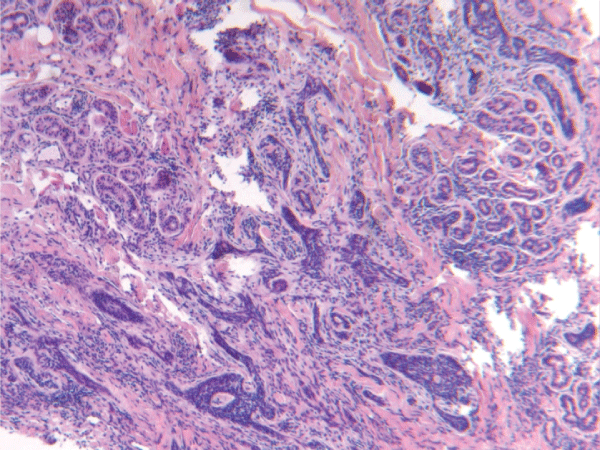
Figure 4: Frozen section with hematoxylin and eosin stain: Infiltrative basal cell carcinoma: close-up view.
View Figure 4
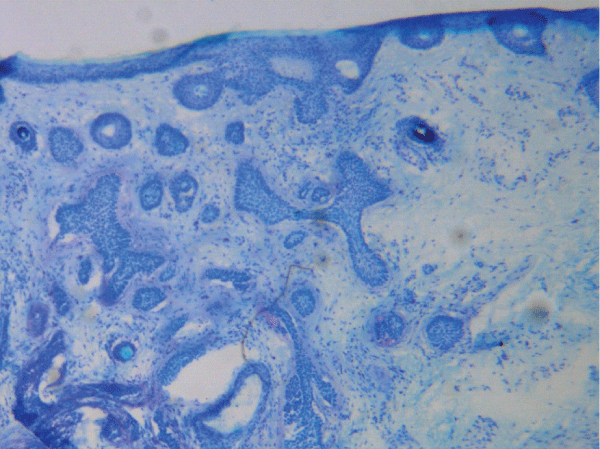
Figure 5: Frozen section with toluidine blue stain: low power view showing the small size and relative uniformity of the tumor nodules.
View Figure 5
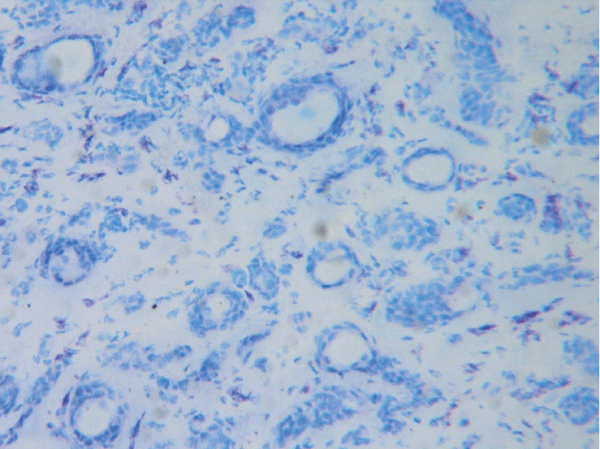
Figure 6: Frozen section with blue toloudine stain shows multiple small ductal structure typical of benigne syringoma.
View Figure 6
![]()
Table 2: Histological diagnoses using all paraffin sections applying H&E stain.
View Table 2
BCC was the most frequent tumor type, in 31.2% (95%CI: 25.8 - 37.2%) of cases. Age comparison between BCC and non BCC groups was statistically significant (P=0.001), with a median age 8.5 years higher in BCC patients (Figure 7). With respect to the anatomic site, the face and neck were significantly more frequent than the scalp, trunk and others (P=0.003). BCC was clinically suspected in 136 out of 250 cases, but only 73 were true positives.
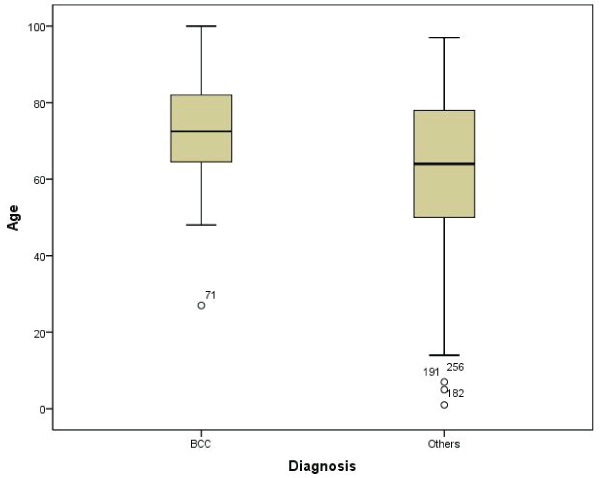
Figure 7: Age comparison with respect to basal cell carcinoma.
View Figure 7
Fifty patients (20%, 95%CI: 15.5% - 25.4%) had squamous epithelial lesions. The median age in patients with squamous epithelial lesion was seven years higher than in patients with non squamous epithelial lesions (P=0.007). Three quarters of squamous epithelial lesions were located in the neck and face, none in the lower limbs and only 4% in the trunk (P=0.021 vs. other sites).
Among all patients only 31 had clinical suspicion of squamous epithelial lesion and 10 of these were false positive. On the other hand, histological test revealed 29 false negative that have would gone unnoticed or misunderstood.
All patients except two underwent immediate cutaneous diagnosis-Toluidine blue testing, but in six patients the sample material was of bad quality or was not sufficient to make a reading, while the outcomes were doubtful in four cases. Cross-classification results according to these data and those obtained by paraffin section method are shown in table 3. Simplifying all diagnoses into three categories (BCC, squamous epithelial lesions and others), the accuracy index or proportion of true results was 90.4% [(76+36+104)/239=0.904].
![]()
Table 3: Results of immediate cutaneous diagnosis-Toluidine blue (ICD-TB)
against gold-standard with H&E. SEL: squamous epithelial lesions.
View Table 3
The results of the use of immediate cutaneous diagnosis in detecting BCC are presented in a flow diagram (Figure 8). Ninety-two test results were positive for BCC, 156 were found to be other pathologies and 10 were inconclusive due to doubts or problems with the material. The gold-standard results confirmed 78 true cases of BCC (Table 4a). Considering only the conclusive results obtained by the two methods (239), the estimated sensitivity of the immediate cutaneous diagnosis in detecting BCC was 98.7% (95%CI: 93.0 - 100%). Of the 162 cases without BCC, only 12 test results were positive giving an estimated specificity of 92.6% (95%CI: 87.4 - 96.1%).
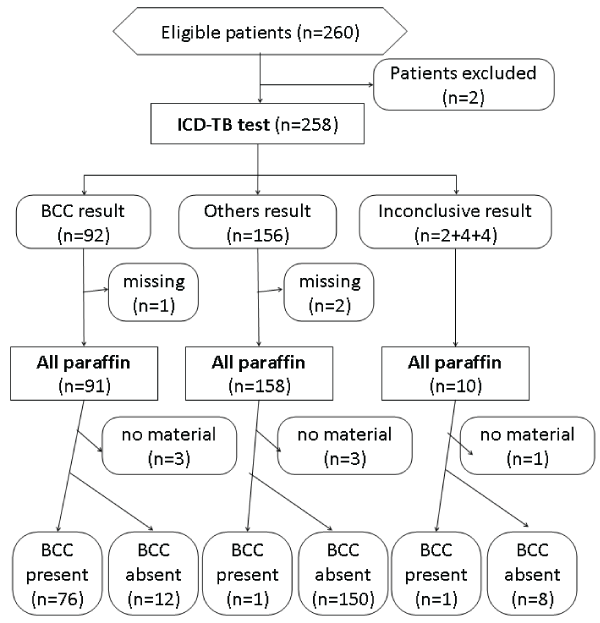
Figure 8: Flow diagram for diagnostic accuracy of immediate cutaneous diagnosis-toluidin blue stain as in STARD initiative.
View Figure 8
![]()
Table 4: Validation of immediate cutaneous diagnosis-Toluidine blue (ICD-TB)
against gold-standard: a) to diagnose BCC, b) to diagnose squamous epithelial
lesions (SEL)
View Table 4
Immediate cutaneous diagnosis showed 39 positive results for squamous epithelial lesions (15 actinic keratosis; 20 squamous cell carcinoma /Bowen's disease and 4 keratoacanthoma) but three of these were false (Table 4b), and therefore the estimated sensitivity was 78.3% (95%CI: 63.6 - 89.1%). There were 200 negative results, but 10 were false, giving an estimated specificity of 98.4 (95%CI: 95.5 - 99.7%).
Immediate cutaneous diagnosis with H&E stain was used in 150 patients (58%). After checking these results against the gold-standard, as done for immediate cutaneous diagnosis- Toluidine blue, the proportion of true results was 95.9% [(46+23+70)/145=0.959]. The estimated sensitivity of immediate cutaneous diagnosis-H&E was 100% (95%CI: 92.3-100%) in diagnosing BCC and 92% (95%CI: 74-99%) in diagnosing squamous epithelial lesions. The estimated specificity was 96% (95%CI: 90.0 - 98.9%) and 100% (95%CI: 97.0 - 100%) for BCC and squamous epithelial lesions respectively (Table 5).
![]()
Table 5: Validation of immediate cutaneous diagnosis with hematoxylin and
eosin (ICD-HE) against gold-standard: a) to diagnose BCC, b) to diagnose
squamous epithelial leisons (SEL)
View Table 5
Inter-rater reliability was assessed in a subsample of 101 patients. Comparison of the diagnoses between two dermatologists using immediate cutaneous diagnosis resulted in 20 disagreements. Subsequently, only two categories were considered: BCC present (+) or BCC absent (-). Cohen's kappa used as a measure of concordance to detect BCC was 0.796 (P< 0.0001). Nine non-coincident cases were evenly distributed in the two cells. When BCC and squamous epithelial lesions were joined as a single category named nonmelanoma epithelial carcinoma, Cohen's kappa was 0.799 (P< 0.0001). Again, the 10 non-coincident cases were equally distributed.
Discussion
Immediate cutaneous diagnosis could represent a new approach to diagnosis in dermatology. The results of this study suggest that evaluation with frozen sections has a high concordance rate with permanent pathologic evaluation, with an average false negative rate for the entire group of 19%, which is in the range of previous reports [5,6,8].
In our study, immediate cutaneous diagnosis test was useful in ruling out BCC vs. doubtful intra-dermal facial nevus and other skin neoplasms. We also found statistically sensitive differences in the diagnosis of two groups: BCC vs. SCC, and actinic keratosis vs. SCC, and for the evaluation of different histopathologic patterns of BCC.
Our results verify the feasibility of the immediate cutaneous diagnosis in the rapid diagnosis of nonmelanoma skin cancer. The estimated sensitivities and specificities provide strong evidence of accuracy of the test.
Although punch biopsy remains the gold standard in the diagnosis of skin cancer, immediate cutaneous diagnostic may improve diagnostic accuracy and save time and money. Our study showed evidence of good internal/external validity and excellent external reliability. Further studies will be necessary to confirm our results and contrast them with other techniques.
The advantages of immediate cutaneous diagnosis are that it allows the choice of the best treatment option in each tumor and surgical margins and the possibility of using topical/external treatment in superficial BCC or actinic keratoses, when required.
Although specificity depends on material quality, the immediate cutaneous diagnosis provides an early diagnosis that helps until a result is obtained by the standard paraffin test. Toluidine blue stain was used in all cases because of its easy availability and its effectiveness in MMS.
Additionally, immediate cutaneous diagnosis reduces diagnostic delay to 15 minutes avoiding patient uncertainty and anxiety.
Evaluation of the costs generated by empiric treatments, histopathologic reports and confirmation biopsies suggests it might be more cost-effective to adapt histological dying techniques and make them accessible to dermatologists, as we suggest with immediate cutaneous diagnosis, as most skin biopsies can be evaluated by an experienced dermopathologist. Initial additional costs would soon be compensated by savings in the number of visits, empiric treatments and administrative processes.
Immediate cutaneous diagnosis is ideal for dermatology departments where MMS infrastructure and staff are available, helping to improve its profitability.
The possible disadvantages of immediate cutaneous diagnosis include the need for specific material and qualified staff experienced in MMS, the availability of MMS infrastructure and an expert dermatopathologist. Likewise, only half of the sample obtained was processed in frozen section, which could lead to some false negatives. In addition, a 4mm punch is only a parcel of possible tumor. Studies have shown that biopsies of a skin cancer are not always indicative of the entire tumor [11,12]. Thus, it is essential to select the ideal biopsy site. We doubt whether immediate cutaneous diagnosis would be suitable for frozen tissue specimens in the diagnosis of melanocytic lesions, such as malignant lentigo melanoma or malignant melanoma. Frozen sections of malignant melanoma and atypical melanocytic skin lesions are controversial [13]. The depth of invasion and degree of cytological atypia cannot be interpreted accurately using frozen section. Additionally, the section may be distorted by the physical process of freezing, and therefore the lesion cannot be accurately measured in the permanent sections. We performed immediate cutaneous diagnosis for evaluation in eight patients with inflammatory disease, but its role in inflammatory patterns will be analyzed in a future report.
The immediate cutaneous diagnosis allows one quickly first step to know with precision the tumor we have dealing in order to perform an immediate treatment. Only in some difficult tumors like Merkel carcinoma, angiosarcoma, adnexial tumors and also to check for perivascular or perineural involvement, we need the histopathology in paraffin and immunochemistry in order to obtain the right diagnosis.
Immediate cutaneous diagnosis could open new diagnostic possibilities in inflammatory and tumoral pathologies. Nowadays, cytology evaluation is useful for herpetic lesions, pustulosis or leishmaniasis, although most histopathologic reports are delayed due to fixation, slide reading or paperwork. Immediate cutaneous diagnosis could represent a standard in outpatient clinics, and could allow dermatologists to read slides immediately, improving clinic-pathological correlation.
The more we simplify diagnostic techniques, such as frozen section samples, the easier it will be for dermatologists or dermatopathologists to make a diagnosis in daily practice in conjunction with clinical information, dermoscopy, confocal microscopy and new rapid and easy techniques such as immediate cutaneous diagnosis.
Although immediate cutaneous diagnosis is only used for tumoral pathology, the procedure could be improved to make possible an immediate diagnosis in outpatient's clinics.
In conclusion, immediate cutaneous diagnosis is a new, ambulatory, minimally invasive, rapid diagnostic technique that is ideal when Mohs surgery infrastructure and staff are available and could aid clinical diagnosis and open new possibilities in clinical practice.
Acknowledgments
The authors gratefully acknowledge the assistance of Nuria Parra-Macías, MD in early phases of the statistical assessment and suggestions, advice and English correction from David Buss. We thank Carmen Castelain for critical reading.
References
-
Mogensen M, Jemec GB (2007) Diagnosis of nonmelanoma skin cancer/keratinocyte carcinoma: a review of diagnostic accuracy of nonmelanoma skin cancer diagnostic tests and technologies. Dermatol Surg 33: 1158-1174.
-
Roenigk RK, Ratz JL, Roenigk HH (2007) Roenigk's dermatology surgery: current techniques in procedural dermatology (3rd edn), New York, NY: inform health care USA: 349.
-
Weinstein MC, Brodell RT, Bordeaux J, Honda K (2012) The art and science of surgical margins for the dermatopathologist. Am J Dermatopathol 34: 737-745.
-
Rowe DE, Carroll RJ, Day CL Jr (1992) Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. Implications for treatment modality selection. J Am Acad Dermatol 26: 976-990.
-
Manstein ME, Manstein CH, Smith R (2003) How accurate is frozen section for skin cancers? Ann Plast Surg 50: 607-609.
-
Dinehart MS, Coldiron BM, Hiatt K, Breau RL (2010) Concordance of frozen and permanent sections for the diagnosis of skin lesions. Dermatol Surg 36: 1111-1115.
-
Ghauri RR, Gunter AA, Weber RA (1999) Frozen section analysis in the management of skin cancers. Ann Plast Surg 43: 156-160.
-
Cataldo PA, Stoddard PB, Reed WP (1990) Use of frozen section analysis in the treatment of basal cell carcinoma. Am J Surg 159: 561-563.
-
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, et al. (2003) Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. BMJ; 326:41-44.
-
Elashoff JD (2002) Query Advisor Version 5.0. User's Guide. Los Angeles, CA.
-
Cohen PR, Schulze KE, Nelson BR (2006) Basal cell carcinoma with mixed histology: a possible pathogenesis for recurrent skin cancer. Dermatol Surg 32: 542-551.
-
Ebede TL, Lee EH, Dusza SW, Busam KJ, Nehal KS (2012) Clinical value of paraffin sections in association with Mohs micrographic surgery for nonmelanoma skin cancers. Dermatol Surg 38: 1631-1638.
-
Smith-Zagone MJ, Schwartz MR (2005) Frozen section of skin specimens. Arch Pathol Lab Med 129: 1536-1543.





