Journal of Dermatology Research and Therapy
Cutaneous Manifestations in Patients with Systemic Lupus Erythematosus: Data from a Multiethnic Latin American Cohort (GLADEL)
Maria J Haye Salinas1*, Veronica Saurit1, Alejandro Alvarellos1, Francisco Caeiro1, Daniel Wojdyla2, Cristina Drenkard3, Guillermo J Pons-Estel4, Luis J Catoggio5, Judith Sarano6, Eduardo Ferreira Borba7, Emilia Sato8, Sergio Jacobelli9, Luis A Ramírez10, Marlene Guibert -Toledano11, Virginia Pascual-Ramos12, Mario H Cardiel13, Maria I Segami14, Isaac Abadi15, Graciela S Alarcon16, Bernardo A Pons-Estel17 and Latin American Study Group on Lupus (GLADEL)
1Servicio de Reumatología, Hospital Privado Centro Médico de Córdoba, Argentina
2Associated to GLADEL, Rosario, Argentina
3Department of Medicine, Emory School of Medicine, USA
4Department of Autoimmune Diseases, Institut Clínic de Medicina Dermatologia, Hospital Clínic, Spain
5Hospital Italiano de Buenos Aires Buenos Aires, Argentina
6Instituto de Investigaciones Médicas "Alfredo Lanari", Buenos Aires, Argentina
7Hospital das Clínicas da Facultad de Medicina da Universidade de Brazil
8Disciplina de Reumatología, Universidade Federal da São Paulo (UNIFESP), Brazil
9Departamento de Inmunología Clínica y Reumatología, Pontificia Universidad Católica de Chile, Chile
10Sección de Reumatología, Universidad de Antioquia, Colombia
11Centro de Investigaciones Médico Quirúrgicas, Habana, Cuba
12Instituto Nacional de Ciencias Médicas y Nutrición "Salvador Zubirán", Distrito Federal, Mexico
13Centro de Investigación Clínica de Morelia, Morelia, México
14Servicio de Reumatología, Hospital Nacional Edgardo Rebagliatti Martins, Perú
15Hospital Universitario de Caracas, Caracas, Venezuela
16Department of Medicine, Division of Clinical Immunology and Rheumatology, The University of Alabama at Birmingham, USA
17Hospital Provincial de Rosario, Rosario, Argentina
*Corresponding author:
Maria J Haye Salinas, MD, Hospital Privado Centro Médico de Córdoba SA Naciones Unidas 346- Bo Parque Vélez Sarsfield, Córdoba (X5016KEH), Argentina, Tel: +54-351-4688816, Fax: +54-351-4688865, E-mail:
hayesalinas@gmail.com
J Dermatol Res Ther, JDRT-2-034, (Volume 2, Issue 5), Original Research; ISSN: 2469-5750
Received: August 25, 2016 | Accepted: October 10, 2016 | Published: October 12, 2016
Citation: Salinas MJH, Saurit V, Alvarellos A, Caeiro F, Wojdyla D, et al. (2016) Cutaneous Manifestations in Patients with Systemic Lupus Erythematosus: Data from a Multiethnic Latin American Cohort (GLADEL). J Dermatol Res Ther 2:034. 10.23937/2469-5750/1510034
Copyright: © 2016 Salinas MJH, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: The aim of this study was to assess the prevalence and associated features of cutaneous manifestations in patients with systemic lupus erythematosus (SLE) as well as to evaluate whether cutaneous manifestations are predictors of the occurrence of other clinical manifestations.
Material and Methods: SLE patients from 34 centers in nine Latin American countries with a recent diagnosis (≤ 2 years) were studied. Socioeconomic-demographic characteristics and clinical features according to the presence of cutaneous manifestations were examined by univariable and multivariable logistic regression analyses. Their predictive value for the occurrence of other clinical manifestations was also examined.
Results: Of the 1480 patients included, 93.7% had cutaneous manifestations, 91.0% of them occurred before the diagnosis of SLE. Cutaneous manifestations occurred more frequently in women (90.5% vs. 80.6%, p = 0.002), and in those with systemic (83.1% vs 69.9%, p = 0.002) and musculoskeletal manifestations (93.5% vs. 83.9%, p = 0.002) and anti-Ro antibody positivity (52.5% vs. 31.7%, p = 0.015) but less frequently in those with pleuropulmonary involvement (27.5% vs. 43.0%, p = 0.002). Cutaneous manifestations were protective of the subsequent occurrence of pleuropulmonary (OR 0.519, 95% CI 0.372-0.724) and hematological (OR 0.621, 95% CI 0.440-0.876) manifestations.
Conclusions: Cutaneous manifestations occur frequently and early in SLE. They were associated with female gender, the presence of systemic and musculoskeletal manifestations and anti-Ro antibody positivity. They were protective of the development pleuropulmonary and hematologic manifestations.
Keywords
Systemic lupus erythematosus, Clinical manifestations, Epidemiology
Introduction
Systemic lupus erythematosus (SLE) is a chronic, autoimmune, inflammatory disease which is quite heterogeneous in its clinical manifestations. Cutaneous manifestations occur in 50% to 85% [1-8] of SLE patients making the skin the most commonly affected organ and the most frequent target of initial clinical manifestations following joint involvement [9,10].
Cutaneous manifestations are indeed important features of SLE and have always been included in the classification criteria of this disease. For example, in the revised and updated American College of Rheumatology (ACR) classification criteria lesions such as malar rash, discoid rash, photosensitivity, and oral ulcers had a high specificity for SLE [11,12]. In the recently published SLICC (Systemic Lupus Erythematosus International Collaborating Clinics) criteria, mucocutaneous manifestations otherwise not considered in these previous ACR criteria, such as subacute cutaneous lupus, bullous lupus, lupus panniculitis, scarring alopecia and others were included [13].
In spite of their overall benign appearance, skin lesions may significantly affect the patients' self-esteem, quality of life and job performance [14]. Regardless of gender, skin lesions do affect the patients' self-esteem and may be accompanied by variable degrees of emotional distress [15].
The purpose of the present study was to assess the prevalence of cutaneous manifestations occurring over the course of SLE and their associated features, as well as to evaluate whether cutaneous manifestations are predictive of the occurrence of other clinical manifestations in a Latin American Lupus cohort.
Material and Methods
GLADEL (Grupo Latino Americano De Estudio del Lupus) was established in 1997 as an observational inception lupus cohort constituted by 34 centers distributed among nine Latin American countries. Patients were included with a recent SLE diagnosis (less than two years); fulfillment of four ACR 1982 SLE criteria was not mandatory at enrollment [11], however, 96.0% of patients fulfilled these criteria during their follow-up. In order to have a balanced representation of centers in the initial cohort, each center was asked to incorporate a minimum of 20 and a maximum of 30 randomly selected patients. Randomization was done locally at each center. The first patients were entered in October 1997, and to insure their recent onset they could only be included if the diagnosis of SLE had been made after January 1st 1996 by a rheumatologist or a qualified internist with experience in SLE. After incorporating the initial 30 patients, each group continued to include one new randomly selected patient per month diagnosed within the previous two years. Patients were invited to participate by their treating physician and an informed consent was signed and saved at each participating center. Each patient was interviewed and her or his medical record's information was validated. All researchers followed local regulations according to their institutional review boards.
During each visit, patient data were collected in a common database (ARTHROS) by a clinician trained in the program. At the coordinating center, strict control and supervision of the data received was undertaken, with permanent communication with the submitting center for any queries arising and/or missing data.
History, physical examination and laboratory tests were performed at entry and at all subsequent visits, which took place every six months after the initial visit. Medications taken were also noted; however, the precise data on their average and cumulative dose were not obtained. The average follow-up time was 4.3 years. Ethnicity was defined according to the parents' and all four grandparents' self-reported ethnicity. The following ethnic groups were considered: Caucasian (individuals with all white European ancestors), Mestizo (individuals born in Latin America who had both Amerindian and white ancestors), African-Latin American (ALA) (individuals born in Latin America with at least one African ancestor whether other ancestors were white or not) and other. In short, we used self-reported ethnic definitions rather than ancestry-informative markers (AIMs) to define the different ethnic groups. Socioeconomic status was evaluated using the Graffar index [16]. Disease activity was ascertained with the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) [17] at all visits. Damage was assessed at yearly intervals with the Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index (SDI) [18].
The local ethics committee of each participating center approved the protocol.
Statistical analysis
The socioeconomic, demographic, clinical and laboratory features of patients with and without cutaneous manifestations at any time during the disease course were compared with Pearson Chi squared or Fisher's exact tests for proportions, and the Students t-test for continuous data. The odds ratio (OR) and the 95% confidence interval (CI) were calculated using univariable logistic regression. A p value equal or less than 0.05 was considered statistically significant; variables significant in these analyses were included in a multivariable logistic regression adjusting for gender, age, the average SLEDAI score and the last SDI score excluding cutaneous manifestations. Univariable and multivariable analyses to assess whether cutaneous manifestations are predictors of subsequent occurrence of other clinical manifestations were also performed. For these analyses, only the cutaneous manifestations present before fulfillment of diagnostic criteria were included but not the ones that appeared after the fulfillment of these criteria; recurrences were also excluded.
The different immunological laboratory tests had not been obtained in all patients. Antinuclear antibodies (ANA), anti-DNA antibodies and complement had not been performed in about 15% of the patients; however, the proportion of patients with missing data was comparable in those with and without cutaneous manifestations, so that, the original data were used. The anti-RNP, anti-Sm, anti-La, anti-Ro and anti-cardiolipin antibodies had not performed in about 30% of the patients; however, no significant difference was observed in the proportion of patients with missing data between patients with and without all cutaneous manifestations so again the original data were used. However, the lupus anticoagulant and anti-β2 glycoprotein 1 test had been performed in less than 50% of the patients and thus both tests were excluded from the analyses.
All analyses were carried out using SPSS, version 19 (Chicago Illinois).
Results
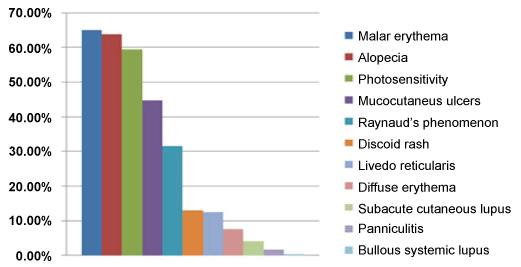
Of the 1480 patients included in this study, 89.9% (n = 1330) were women and 10.1% (n = 150) were men; patients had a mean (SD) age at SLE onset of 27.7 (11.7) years; 93.7% (n = 1387) have had at least one cutaneous manifestation during their follow up and most patients (91.0%, n = 1264) showed cutaneous manifestations before fulfilling the four SLE classification criteria. Most patients had more than one type of skin lesion; the most common manifestations were malar rash 65.0%, alopecia 63.0%, photosensitivity 59.5% and oral ulcers 44.7%. The frequency of each type of cutaneous manifestation is shown in figure 1.
The frequency of the different cutaneous manifestations as a function of ethinicity is shown in table 1. Alopecia was less frequently observed in Caucasians than in the other ethnic groups (59.7% vs 66.8% Mestizos, 64.0% African / Latin American and 72.1% other; p = 0.043) while discoid rash was more frequent in African / Latin American than in the other ethnic groups (19.9% vs 13.2% Caucasians, 11.3% Mestizos, and 4.7% other; p = 0.007).
![]()
Table 1: Cutaneous manifestation frequency by ethnicity.
View Table 1
Table 2 shows the relationship between cutaneous and other clinical manifestations. Patients with cutaneous manifestations had a significantly higher frequency of systemic (fever, prolonged febrile syndrome, asthenia, fatigue, anorexia, weight loss, adenopathy) (83.1% vs. 69.9%; p = 0.002) and musculoskeletal manifestations (93.5% vs. 83.9%; p = 0.002) as well as a higher proportion of patients with a SLEDAI ≥ 4 (58.8% vs. 46.2%; p = 0.022); they were also more frequent users of antimalarials (83.2% vs. 64.5%; p < 0.001) at some point during the course of their disease. They also exhibited anti-RNP (55.3% vs. 33.3%; p = 0.006), anti-Ro (52.5% vs. 31.7%; p = 0.015) and anti-Sm (47.0% vs. 32.6%; p = 0.015) antibodies positivity more frequently than those without cutaneous manifestations. On the other hand, patients with cutaneous manifestations exhibited a lower frequency of pleuropulmonary manifestations (27.5% vs. 43.0%; p = 0.002) and cardiac involvement (20.1% vs. 34.4%; p = 0.002) than those without them.
![]()
Table 2: Sociodemographic, clinical and serological features according to whether cutaneous manifestations were present or not. univariable and multivariable analyses.
View Table 2
Multivariable analysis
The results of the multivariable analysis are also shown in table 2. Variables retained in this analysis were female gender (OR 3.052, 95% CI 1.132 to 8.233), the presence of systemic (OR 2.865, 95% CI 1.290 to 6.364) and musculoskeletal manifestations (OR 5.542, 95% CI 2.071 to 14.836) and anti-Ro antibody positivity (OR 2.485, 95% CI 1.131 to 5.461); on the other hand, cutaneous manifestations were negatively associated with the occurrence of pleuropulmonary manifestations (OR 0.443, 95% CI 0.207 to 0.950).
Cutaneous manifestations: predictor of clinical manifestations
As shown in table 3, the presence of cutaneous manifestations was protective of the subsequent occurrence of pleuropulmonary (OR 0.519, 95% CI 0.372 to 0.724) and hematological (OR 0.621, 95% CI 0.440 to 0.876) manifestations.
![]()
Table 3: Presence of cutaneous manifestations as predictive factor other clinical manifestations.
View Table 3
Discussion
This is the first report on a large number of Latin American SLE patients that describes the cutaneous manifestations of the disease and reflects the reality of daily clinical practice among them. A very high frequency of cutaneous manifestations (93.7%) was observed in this GLADEL cohort; this is even higher than the frequencies described by Dubois, et al. and Harvey, et al. in US patients [3,4], and those found in studies of European (59%), Iranian and Pakistani (82%) populations [2,6,7]. However, the frequency found in our cohort is similar to that found in Brazilians where cutaneous manifestations occurred in over 90% of the patients being the most common malar rash and photosensitivity [19]. A sustained exposure to ultraviolet light among our Latin American patients could be the explanation for these findings. The most frequent manifestations we observed were malar rash, alopecia, photosensitivity and Raynaud phenomenon, similar to what has been described in many other previous studies [6,8,20-24].
Cutaneous manifestations were associated with a higher frequency of systemic manifestations and musculoskeletal manifestations, the latter being the other most common manifestation of the disease; this is, somewhat similar to a study conducted in Spain, in which patients with subacute cutaneous lesions presented arthralgia and systemic manifestations more frequently than patients with chronic cutaneous manifestations [25].
An important finding of our study was the occurrence of a lower frequency of cardiac and pleuropulmonary manifestations in patients with cutaneous involvement; these data, reinforce a previous report [26]. Furthermore, cutaneous manifestations were protective of the subsequent occurrence of hematologic and pleuropulmonary manifestations, fact which has not previously been reported. A protective effect for the occurrence of renal manifestations was not found in our study; however we have previously found discoid lupus (occurring at disease onset) to be protective of the subsequent development of lupus nephritis [27] and that patients with photosensitivity experience a longer time to the occurrence of renal disease [28]. This apparent discrepancy probably relates to the fact that we have examined all cutaneous manifestations together and not individually.
Although we found a higher frequency of patients with a SLEDAI ≥ 4 in those with cutaneous manifestations, this association is of questionable value since these manifestations were not excluded from the SLEDAI; nevertheless, we have included the SLEDAI score in the multivariable analyses as an adjustment variable.
It is widely accepted that anti-Ro antibodies, produced by the exposure of self-antigens from the cell surfaces to ultraviolet B radiation [29], play a role in the pathogenesis of skin lesions association which we have confirmed. Other authors have found these antibodies to be associated with photosensitive rashes, alopecia and subacute cutaneous lesions [8,25,30,31]. It should be noted however, that no relationship between the different skin rashes and the presence of certain antibodies, including anti-Ro has been found in patients of African descend [32].
Anti-RNP antibodies positivity has been associated with cutaneous manifestations; Grönhagen, et al. found these antibodies to be associated with acute cutaneous lesions in a study of 260 SLE patients [7]. Anticardiolipin antibodies positivity has been associated with the occurrence of Raynaud phenomenon and livedo reticularis, among others; these manifestations are well-known components of the antiphospholipid syndrome and have also been associated with anti-Beta 2 glycoprotein 1 antibodies positivity [7]. Hypocomplementaemia was not associated with the presence of cutaneous manifestations; however, other authors have reported hypocomplementemia to be associated primarily with cutaneous vasculitis [33]. However, we have not examined these association because of the paucity of data about complement levels and the fact that we have examined all cutaneous manifestations together.
The association of cutaneous manifestations with the use of antimalarials, more than likely reflects the fact that their presence is a clear indication for therapy with these compounds as has been widely reported and recognized [1,34-36].
An important limitation of our study is that the association of skin lesions and some auto-antibodies could not be examined since they were not available in all patients, had not been obtained at a central laboratory or at the time cutaneous manifestations occurred. This prevents us from making a definitive interpretation of some of the associations we are reporting. Another limitation is that the diagnosis of cutaneous manifestations has not been carried out systematically by dermatologists; however, all patients have been evaluated by rheumatologists trained and experienced in the recognition of cutaneous manifestations of SLE. Finally, we could not examine the relationship between these manifestations and the average and cumulative doses of the drugs used since this detailed information had not been obtained. Nevertheless, we think that the data being reported is quite valuable.
In conclusion, cutaneous manifestations occur quite frequently in Latin American SLE patients and they appear to be an early manifestation of the disease. The most frequent manifestations were malar erythema, alopecia and photosensitivity. Cutaneous manifestations were associated with the presence of systemic and musculoskeletal manifestations and positive anti-Ro antibodies; they were, however, inversely associated with the presence of pleuropulmonary manifestations. Finally, they were protective of the occurrence of pleuropulmonary and hematologic manifestations.
Acknowledgments
We are grateful to Daniel Villalba and Leonardo Grasso for providing expert assistance with the ARTHROS (version 6.0) software.
Author Contributions
All authors were involved in drafting or revising this article critically for important intellectual content, and all authors approved the final version to be published. Dr. María J. Haye Salinas and Bernardo A. Pons-Estel have full access to the dataset used for the study and take responsibility for data integrity and accuracy of the analyses performed.
Financial Support
This project was supported in part by grants from The Pan American League of Associations for Rheumatology (PANLAR). Supported by grants from the Federico Wilhelm Agricola Foundation Research (to B.A.P-E).
On behalf of GLADEL
The following participants are members of GLADEL, have incorporated at least 20 patients into the database with adequate follow-up and in particular provided data related to elderly onset SLE.
ARGENTINA: Enrique R. Soriano, María Flavia Ceballos Recalde and Edson Velozo, Medical Clinic Service, Hospital Italiano and Fundación Dr. Pedro M. Catoggio para el Progreso de la Reumatología, Buenos Aires; Jorge A. Manni and Sebastián Grimaudo, Departamento de Inmunología, Instituto de Investigaciones Médicas "Alfredo Lanari", Buenos Aires; Emilce Schneeberger, María S. Arriola and Graciela Gómez, Servicio de Reumatología Instituto de Rehabilitación Psicofísica, Buenos Aires; Mercedes A. García, Ana Inés Marcos and Juan Carlos Marcos, Servicio de Reumatología, Hospital Interzonal General de Agudos General San Martín, La Plata; Hugo R. Scherbarth, Pilar C. Marino and Estela L. Motta, Servicio de Reumatología, Hospital Interzonal General de Agudos "Dr. Oscar Alende" Mar del Plata; Susana Gamron, Sandra Buliubasich and Laura Onetti, Servicio de Reumatología, UHMI1, Hospital Nacional de Clínicas, Córdoba; Silvana Gentiletti, Norberto Quagliatto, Alberto A. Gentiletti and Daniel Machado, Servicio de Reumatología, Hospital Provincial de Rosario, Rosario; Guillermo Berbotto and Carlos A. Battagliotti, Servicio de Reumatología Hospital Escuela Eva Perón, Granadero Baigorria; Marcelo Abdala and Simón Palatnik, Servicio de Reumatología, Hospital Provincial del Centenario, Rosario.
BRASIL: Elaine M.C. Sella, and Alexandre W. S. Souza, Disciplina de Reumatología, Universidade Federal da São Paulo (UNIFESP), São Paulo; Lilian T. Lavras Costallat, Manoel Barros Bertolo and Ibsen Bellini Coimbra, Divisao de Reumatología, Faculdade de Ciencias Medicas, Universidade Estadual da Campinas, Campinas; Eloisa Bonfa, Divisao de Reumatología, Faculdade da Medicina, Universidade da São Paulo, São Paulo; Joao C. Tavares Brenol, Odirlei Monticielo and Ricardo Xavier, Serviço de Reumatología, Hospital da Clinicas da Porto Alegre, Universidade Federal do Rio Grande do Sul; Fernando de Souza Cavalcanti, Ângela Luzia Branco Duarte e Cláudia Diniz Lopes Marques, Disciplina de Reumatología, Centro de Ciencias da Saúde, Universidade Federal da Pernambuco, Pernambuco; Nilzio Antonio Da Silva, Ana Carolina de O. e Silva and Tatiana Ferracine Pacheco, Serviço da Reumatología, Faculdade de Medicina, Universidade Federal de Goias, Goiania.
COLOMBIA: José Fernando Molina Restrepo and Javier Molina Lopez, Servicio de Reumatología, Hospital Pablo Tobon Uribe, Medellín; Antonio Iglesias Gamarra and Antonio Iglesias Rodríguez, Servicio de Reumatología, Hospital San Juan de Dios, Facultad de Medicina, Universidad Nacional, Bogotá; Eduardo Egea Bermejo, Departamento de Inmunología, Universidad del Norte, Barranquilla; Oscar Uribe-Uribe and Oscar Felipe, Sección de Reumatología, Universidad de Antioquia, Hospital Universitario San Vicente de Paul, Medellín; Renato A. Guzmán Moreno, José F. Restrepo Suarez, Departamento de Medicina Interna e Inmuno-Reumatología, Clínica Saludcoop 104 y Hospital San Juan de Dios, Universidad Nacional de Colombia, Bogotá.
CUBA: Gil A. Reyes Llerena and Alfredo Hernández Martínez, Servicio de Reumatología, Centro de Investigaciones Médico Quirúrgicas (CIMEQ), La Habana.
CHILE: Loreto Massardo and Néstor Gareca, Departamento de Inmunología Clínica y Reumatología, Escuela de Medicina, Pontificia Universidad Católica de Chile, Santiago; Oscar J. Neira, Leonardo R. Guzmán and María A. Alvarado, Sección Reumatología, Hospital del Salvador, Facultad de Medicina, Universidad de Chile, Santiago.
GUATEMALA: Abraham García Kutzbach, Ivette Castro Ampie and Maria Antonieta Tuna, Servicio de Reumatología, Hospital Universitario Esperanza, Ciudad de Guatemala.
MEXICO: Donato Alarcón-Segovia and Antonio R. Villa, Departamento de Inmunología y Reumatología, Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán, México D.F; Leonor A. Barile-Fabris and Juan Manuel Miranda Limón, Unidad de Investigación Médica en Epidemiología Clínica, Hospital de Especialidades, Centro Médico Nacional Siglo XXI, Instituto Mexicano de Seguro Social, México D.F.; Mary-Carmen Amigo Castañeda and Luis H. Silveira Torre, Departamento de Reumatología, Instituto Nacional de Cardiología Ignacio Chávez, México D.F.; Ignacio García De La Torre, Gerardo Orozco Barocio and Magali L. Estrada Contreras, Departamento de Inmunología y Reumatología, Hospital General de Occidente de la Secretaría de Salud, Guadalajara Jal. México; María Josefina Sauza del Pozo, Laura E. Aranda Baca and Adelfia Urenda Quezada, Servicio de Reumatología, Instituto Mexicano de Seguro Social, Hospital de Especialidades No 25, Monterrey, N.L., México; Guillermo F. Huerta Yáñez, Servicio de Reumatología, Hospital de Especialidades Miguel Hidalgo, Aguas Calientes.
PERÚ: Eduardo M. Acevedo Vásquez, José Luis Alfaro Lozano and Jorge M. Cucho Venegas, Servicio de Reumatología, Hospital Nacional Guillermo Almenara Irigoyen, ESSALUD, Lima; Cesar A. Ugarte and Felipe E. Becerra, Servicio de Reumatología, Hospital Nacional Edgardo Rebagliatti Martins, ESSALUD, Lima.
VENEZUELA: Rosa Chacón Díaz, Servicio de Reumatología, Centro Nacional de Enfermedades Reumáticas, Hospital Universitario de Caracas, Caracas; Maria H. Esteva Spinetti, Jorge Vivas and Adriana Bettiol, Unidad de Reumatología, Hospital Central de San Cristobal, San Cristobal; With the corresponding attributes and responsibilities.
References
-
Rothfield N, Sontheimer RD, Bernstein M (2006) Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol 24: 348-62.
-
Font J, Cervera R, Ramos-Casals M, García-Carrasco M, Sents J, et al. (2004) Clusters of clinical and immunologic features in systemic lupus erythematosus: analysis of 600 patients from a single center. Semin Arthritis Rheum 33: 217-230.
-
Dubois EL, Tuffanelli DL (1964) Clinical manifestations of systemic lupus erythematosus. computer analysis of 520 cases. JAMA 190: 104-111.
-
Harvey AM, Shulman LE, Tumulty PA, Conley CL, Schoenrich EH (1954) Systemic lupus erythematosus: review of the literature and clinical analysis of 138 cases. Medicine (Baltimore) 33: 291-437.
-
Mc Cauliffe DP (2001) Cutaneous lupus erythematosus. Semin Cutan Med Surg 20: 14-26.
-
Kapadia N, Haroon TS (1996) Cutaneous manifestations of systemic lupus erythematosus: study from Lahore, Pakistan. Int J Dermatol 35: 408-409.
-
Grönhagen SM, Gunnarsson I, Svenungsson E, Nyberg F (2010) Cutaneous manifestations and serological findings in 260 patients with systemic lupus erythematosus. Lupus 19: 1187-1194.
-
Saurit V, Bell R, Campana R, Ruiz Lascano A, Ducasse C, et al. (2003) Manifestaciones mucocutáneas en pacientes con lupus eritematoso sistémico. Medicina (Buenos Aires) 63: 283-287.
-
Cervera R, Khamashta MA, Font J, Sebastiani GD, Gil A, et al. (1993) Systemic lupus erythematosus: clinical and immunologic pattern of disease expression in a cohort of 1000 patients. The European Working Party on Systemic Lupus Erythematosus. Medicine 72: 113-124.
-
Werth VP (2005) Clinical manifestations of cutaneous lupus erythematosus. Autoimmun Rev 4: 296-302.
-
Tan EM, Cohen AS, Fries JF, Masi AT, McShane DJ, et al. (1982) The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 25: 1271-1277.
-
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40: 1725.
-
Petri M, Orbai AM, Alarcón GS, Gordon C, Merrill JT, et al. (2012) Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 64: 2677-2686.
-
O'Quinn SE, Cole J, Many H (1972) Problems of disability in patients with chronic skin disease. Arch Dermatol 105: 35-41.
-
Klein R, Moghadam-Kia S, Taylor L, Coley C, Okawa J, et al. (2011) Quality of life in cutaneous lupus erythematosus. J Am Acad Dermatol 64: 849-858.
-
Graffar M, Corbisier J, Van Den Berghe F, Asiel M (1961) Comparative study of the health and social status of children in an urban region and in a rural region. Arch Belg Med Soc 19: 77-124.
-
Bombardier C, Gladman DD, Urowitz MB, Caron D, Chang CH (1992) Derivation of the SLEDAI. A disease activity index for lupus patients. The committee on prognosis studies in SLE. Arthritis Rheum 35: 630-640.
-
Gladman D, Ginzler E, Goldsmith C, Fortin P, Liang M, et al. (1996) The development and initial validation of the Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index for Systemic Lupus Erythematosus. Arthritis Rheum 39: 363-369.
-
Borba EF, Araujo DB, Bonfá E, Shinjo SK (2013) Clinical and immunological features of 888 Brazilian systemic lupus patients from a monocentric cohort: comparison with other populations. Lupus 22: 744-749.
-
Biazar C, Sigges J, Patsinakidis N, Ruland V, Amler S, et al. (2013) Cutaneous lupus erythematosus: first multicenter database analysis of 1002 patients from the European Society of Cutaneous Lupus Erythematosus (EUSCLE). Autoimmun Rev 12: 444-454.
-
Rabbani MA, Shah SM, Ahmed A (2003) Cutaneous manifestations of systemic lupus erythematosus in Pakistani patients. J Pak Med Assoc 53: 539-541.
-
Kole A, Ghosh A (2009) Cutaneous manifestations of systemic lupus erythematosus in a tertiary referral center. Indian J Dermatol 54: 132-136.
-
Cardinali C, Caproni M, Bernacchi E (2000) The spectrum of cutaneous manifestations in lupus erythematosus-the Italian experience. Lupus 9: 417-423.
-
Salphale P, Danda D, Chandrashekar L, Peter D, Jayaseeli N, et al. (2011) The study of cutaneous lupus erythematosus disease area and severity index in Indian patients with systemic lupus erythematosus. Lupus 20: 1510-1577.
-
Vera-Recabarren MA, Garcia-Carrasco M, Ramos-Casals M, Herrero C (2010) Comparative analysis of subacute cutaneous lupus erythematosus and chronic cutaneous lupus erythematosus: clinical and immunological study of 270 patients. Br J Dermatol 162: 91-101.
-
Merola JF, Prystowsky SD, Iversen C, Gomez-Puerta JA, Norton T, et al. (2013) Association of discoid lupus erythematosus with other clinical manifestations among patients with systemic lupus erythematosus. J Am Acad Dermatol 69: 19-24.
-
Pons- Estel GJ, Aspey LD, Bao G, Pons-Estel BA, Wojdyla D, et al. (2016) Early discoid lupus erythematosus protects against renal disease in patients with systemic lupus erythematosus: longitudinal data from a large Latin American cohort. Lupus.
-
Pons- Estel GJ, Alarcón GS, Burgos PI, Hachuel L, Boggio G, et al. (2013) Mestizos with systemic lupus erythematosus develop renal disease early while antimalarial retard its appearance: data from a Latin American cohort. Lupus 22: 899-907.
-
Bielsa Marsol I, Rodríguez Caruncho C (2010) Manifestaciones cutáneas del lupus eritematoso. Inmunología 29: 100-110.
-
Harley JB, Scofield RH, Reichlin M (1992) Anti-Ro in Sjögren's syndrome and systemic lupus erythematosus. Rheum Dis Clin North Am 18: 337-358.
-
Mond CB, Peterson MG, Rothfield NF (1989) Correlation of anti-Ro antibody with photosensitivity rash in systemic lupus erythematosus patients. Arthritis Rheum 32: 202-204.
-
Sutej PG, Gear AJ, Morrison RC, Tikly M, de Beer M, et al. (1989) Photosensitivity and anti-Ro (SS-A) antibodies in black patients with systemic lupus erythematosus (SLE). Br J Rheumatol 28: 321-324.
-
Ramos-Casals M, Campoamor MT, Chamorro A, Salvador G, Segura S, et al. (2004) Hypocomplementemia in systemic lupus erythematosus and primary antiphospholipid syndrome: prevalence and clinical significance in 667 patients. Lupus 13: 777-783.
-
Wysenbeek AJ, Guedj D, Amit M, Weinberger A (1992) Rash in systemic lupus erythematosus: prevalence and relation to cutaneous and non-cutaneous disease manifestations. Ann Rheum Dis 51: 717-719.
-
Yokogawa N, Tanikawa A, Amagai M, Kato Y, Momose Y, et al. (2013) Response to hydroxychloroquine in Japanese patients with lupus-related skin disease using the cutaneous lupus erythematosus disease area and severity index (CLASI). Mod Rheumatol 23: 318-322.
-
Jordan N, D'Cruz D (2016) Current and emerging treatment options in the management of lupus. Immunotargets Ther 5: 9-20.