International Journal of Ophthalmology and Clinical Research
Multimodal Imaging of Retinal Angiomatous Proliferation Showing Finger-Like Lesions of the Outer Retina
Shiraaz I Rahman, Mihai Mititelu and Rukhsana G Mirza*
Department of Ophthalmology, Northwestern University, Feinberg School of Medicine, USA
*Corresponding author:
Rukhsana G. Mirza, MD, Department of Ophthalmology, Northwestern University, Feinberg School of Medicine, 645 North Michigan Avenue, Suite 440, Chicago, IL, USA, 60611, Tel: (312) 908-8152, Fax: (312) 503-8152, E-mail: r-mirza@northwestern.edu
Int J Ophthalmol Clin Res, IJOCR-2-024, (Volume 2, Issue 3), Case Report; ISSN: 2378-346X
Received: March 31, 2015 | Accepted: May 22, 2015 | Published: May 25, 2015
Citation: Rahman SI, Mititelu M, Mirza RG (2015) Multimodal Imaging of Retinal Angiomatous Proliferation Showing Finger-Like Lesions of the Outer Retina. Int J Ophthalmol Clin Res 2:024. 10.23937/2378-346X/1410024
Copyright: © 2015 Rahman SI, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Retinal Angiomatous Proliferation (RAP) is a process that occurs in patients with Age-Related Macular Degeneration (AMD) in which retinal capillaries proliferate and invade posteriorly into the choroid. RAP lesions have a characteristic appearance of finger-like projections at the level of the outer retina which can resemble inflammatory neovascularization. This case report describes a patient with RAP who received consecutive intravitreal injections of aflibercept. The patient was followed with multi-modal imaging, including en face spectral domain optical coherence tomography (SD-OCT), which showed finger-like lesions and profound response and improvement in retinal architecture. RAP lesions must be considered in the differential of finger-like lesions in the outer retina and aflibercept is an effective treatment choice.
Keywords
Age-related macular degeneration (AMD), Retinal angiomatous proliferation (RAP), Anti-vascular endothelial growth factor (Anti-VEGF), Aflibercept, Multimodal Imaging, Ocular coherence tomography (OCT)
Introduction
Choroidal neovascularization (CNV) is a well-characterized process that can occur in individuals with age-related macular degeneration (AMD). As the name implies, CNV originates in the choroid and has the potential to erode the retinal pigment epithelium and invade the neurosensory retina. The reverse has also been described, in which the retinal capillaries proliferate and invade posteriorly into the choroid. This process is referred to as Retinal Angiomatous Proliferation (RAP) [1].
The appearance of these lesions can be described as "finger-like" and therefore have a strong similarity to inflammatory CNV as described by Hoang et al. [2] While similar in appearance, the etiology of RAP lesions and inflammatory lesions is distinct as RAP appears to be more related to ischemia and retinal capillary proliferation rather than inflammation [1].
In terms of treatment, anti-vascular endothelial growth factor (anti-VEGF) therapies have been shown to be effective specifically for RAP lesions either as monotherapy or in combination with other treatment modalities [3-7].
We present a case of a patient with AMD-associated RAP with characteristic finger-like lesions in the outer retina who was treated with intravitreal aflibercept and followed with multimodal imaging.
Case Report
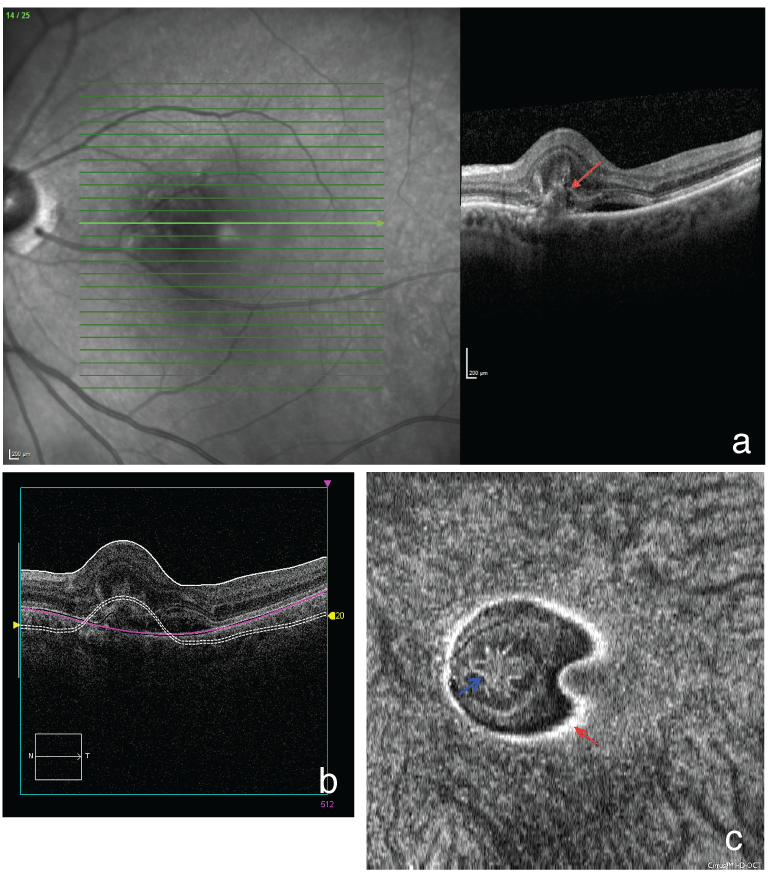
A 69-year-old man presented with a complaint of worsening near vision for 1-2 weeks along with new-onset metamorphopsia in his left eye for the previous 2-3 days. He described a circular structure obscuring his vision. He had no past ocular or relevant medical history. His visual acuity was 20/20 in right eye and 20/70 in the left eye. His anterior segment exam was mostly unremarkable aside from 1+ cortical cataract bilaterally. His fundus examination at presentation revealed elevation of the macula nasally in his left eye with one small pinpoint intra-retinal hemorrhage. The fundus exam in the right eye showed some small drusen. Fluorescein angiogram (FA) along with Heidelberg SD-OCT (Spectralis OCT, Heidelberg Engineering, Heidelberg, GmbH, Dossenheim, Germany) and Cirrus SD-OCT (Carl Zeiss Meditec) were obtained. FA showed a prominent, circular hyper-reflective area of hyperfluorescence nasal to the foveal avascular zone that demonstrated leakage on late images. His OCT studies revealed a RAP lesion in the left eye consisting of solid pigment epithelial detachment (PED) with subretinal fluid (SRF), associated with intraretinal hyper-reflective finger-like lesions shown in en face images, primarily located in the outer nuclear layer (ONL) of the retina but abutting the outer plexiform layer (OPL) (Figure 1a-1c). He received aflibercept in the left eye on this visit, which he tolerated well.
.
Figure 1a: Heidelberg spectral-domain optical coherence tomography (SD-OCT) of the left eye shows a pigment epithelial detachment (PED) with subretinal fluid (SRF) (red arrow). There are hyper-reflective branching lesions with a finger-like appearance, primarily located in the outer nuclear layer (ONL) of the retina but abutting the outer plexiform layer (OPL). The photoreceptor layer is poorly visualized in this complex. There is no intraretinal fluid. There is retinal thickening.
Figure 1b: Cross sectional Cirrus SD-OCT scan showing location of slab (section). A double-stranded interrupted line created by the Cirrus software follows the contour of the retinal inner limiting membrane (ILM).
Figure 1c: En face Cirrus SD-OCT of the left eye shows a finger-like intraretinal hyper-reflective complex (blue arrow). The hyper-reflective edge of the RPE is highlighted by a red arrow.
View Figure 1
He returned to the clinic 4 weeks later and reported that the central "spot" was lighter with the ability to see color through it. His vision was now 20/40 in the affected eye. His OCT at this visit showed smaller PED with less retinal branches visualized and with significant improvement in the amount of subretinal fluid (SRF) nasal to the PED. He then received a second injection of aflibercept.
On his next visit, 4 weeks later, he continued to describe subjective improvement in his vision. His visual acuity had returned to 20/20 in his left eye. Fundus exam revealed resolution of the intraretinal hemorrhage near the fovea. There was improvement on Heidelberg SD-OCT, including resolution of SRF and he received a third injection.
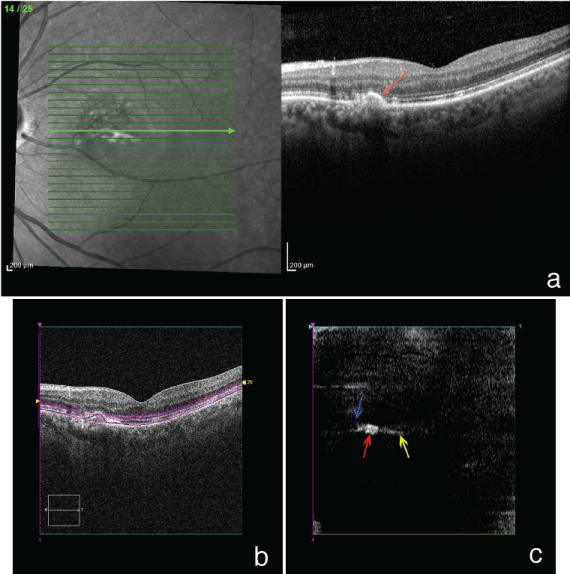
At his next 4-week follow-up visit, his OCT showed continued improvement of the RAP lesion, with no residual intraretinal fluid or subretinal fluid and complete resolution of intraretinal hyper-reflective branching (Figure 2a-2c). In keeping with aflibercept dosing guidelines, an injection was not given at this visit [8]. The patient was instructed to follow-up closely for future treatments as needed. Of note, one year after initial presentation, the patient remained dry with bimonthly treatment and maintained a visual acuity of 20/20 over this time period.
.
Figure 2a: Heidelberg SD-OCT obtained four weeks after the third intravitreal injection shows a small, shrinking PED (red arrow) with mild residual RPE and retinal photoreceptor layer disruption nasal to it. There is complete resolution of intraretinal hyper-reflective branching. There is smoothening of the contour of the ONL and OPL. There is no intraretinal or subretinal fluid. The foveal contour is restored.
Figure 2b: On Cirrus cross-sectional SD-OCT, a double-stranded interrupted purple RPE slab line is placed through the roof of the lesion to highlight the level of axial cut.
Figure 2c: En face Cirrus SD-OCT obtained four weeks after the third intravitreal injection shows significant decrease in the size of the PED nasal to the fovea (red arrow), as well as resolution of the previously seen finger-like hyper-reflective lesion (blue arrow). There is less perifoveal hyper-reflective debris (yellow arrow) compared to prior scan.
View Figure 2
Discussion
AMD is a common disorder with a variety of clinical manifestations. The prevalence of RAP in AMD with occult CNV has been reported at up to 20-28% [9,10]. Furthermore, this phenomenon has been divided into 3 stages, each with distinct imaging characteristics. Stage 1 involves capillary proliferation within the retina. Stage 2 is characterized by extension of intraretinal neovascularization into the subretinal space. Stage 3 marks the presence of detectable CNV [1].
The use of OCT has highly enhanced the ability to detect and follow neovascular lesions associated with AMD, including the relatively recent characterization of RAP. On fluorescein angiography, RAP can mimic occult or minimally classic CNV, and is therefore less revealing than OCT [11]. The advent of SD-OCT has further enhanced the characterization of these lesions. The neovascularization appears to originate from the deep retinal capillaries and progress toward the retinal pigment epithelium (RPE) with development of pigment epithelial detachment (PED) once the sub-RPE space is penetrated [12]. This process is well shown in the Heidelberg and en face Cirrus images in this patient and can be categorized as stage 2 RAP.
After three injections, our patient showed both anatomic and functional improvement, along with subjective improvement of visual distortion. There was rapid shrinking of choroidal neovascular membranes and regression of vascular branching over a short time interval with resolution of intraretinal fluid, return of foveal contour that was well documented in the SD-OCT images. Finally, the patient returned to normal, pre-treatment vision.
As we show in this patient's images, the lesions have a finger-like and branching pattern which is not as typical of classic CNV. This is highly similar to the pattern attributed to inflammatory CNV by Hoang et al. [2]. The overlap in the findings suggests that the "pitchfork sign" might not be as specific to inflammatory etiologies such as Punctate Inner Choroidopathy (PIC) and/or Multifocal Choroiditis (MFC) as previously proposed. Therefore, RAP lesions must be considered in the differential of finger-like lesions in the outer retina and aflibercept is an effective treatment choice.
Acknowledgement
Supported by an unrestricted grant from Research to Prevent Blindness, New York, NY. The supporting source had no involvement in the study design, collection, analysis or interpretation of data.
References
-
Yannuzzi LA, Negr�o S, Iida T, Carvalho C, Rodriguez-Coleman H, et al. (2001) Retinal angiomatous proliferation in age-related macular degeneration. Retina 21: 416-434.
-
Hoang QV, Cunningham ET Jr, Sorenson JA, Freund KB (2013) The "pitchfork sign" a distinctive optical coherence tomography finding in inflammatory choroidal neovascularization. Retina 33: 1049-1055.
-
Hemeida TS, Keane PA, Dustin L, Sadda SR, Fawzi AA (2010) Long-term visual and anatomical outcomes following anti-VEGF monotherapy for retinal angiomatous proliferation. Br J Ophthalmol 94: 701-705.
-
Hufendiek K, Hufendiek K, Panagakis G, Helbig H, Gamulescu MA (2012) Visual and morphological outcomes of bevacizumab (Avastin�) versus ranibizumab (Lucentis�) treatment for retinal angiomatous proliferation. Int Ophthalmol 32: 259-268.
-
Kramann CA, Sch�pfer K, Lorenz K, Zwiener I, Stoffelns BM, et al. (2012) Intravitreal ranibizumab treatment of retinal angiomatous proliferation. Acta Ophthalmol 90: 487-491.
-
Lee MY, Kim KS, Lee WK (2011) Combination therapy of ranibizumab and photodynamic therapy for retinal angiomatous proliferation with serous pigment epithelial detachment in Korean patients: twelve-month results. Retina 31: 65-73.
-
Parodi MB, Iacono P, Menchini F, Sheth S, Polini G, et al. (2013) Intravitreal bevacizumab versus ranibizumab for the treatment of retinal angiomatous proliferation. Acta Ophthalmol 91: 267-273.
-
(2013) Highlights of Prescribing Information. Regeneron,
-
Axer-Siegler R, Bourla D, Priel E, Yassur Y, Weinberger D (2002) Angiographic and flow patterns of retinal choroidal anastamoses in age-related macular degeneration with occult choroidal neovascularization. Ophthalmology 109: 1726-1736
-
Massacesi AL, Sacchi L, Bergamini F, Bottoni F (2008) The prevalence of retinal angiomatous proliferation in age-related macular degeneration with occult choroidal neovascularization. Graefes Arch Clin Exp Ophthalmol 246: 89-92.
-
Rouvas AA, Papakostas TD, Ntouraki A, Douvali M, Vergados I, et al. (2010) Angiographic and OCT features of retinal angiomatous proliferation. Eye (Lond) 24: 1633-1642.
-
Matsumoto H, Sato T, Kishi S (2010) Tomographic features of intraretinal neovascularization in retinal angiomatous proliferation. Retina 30: 425-430.





