International Journal of Respiratory and Pulmonary Medicine
Infective Endarteritis in a Patent Ductus Arteriosus
John Fani Srour*
Department of Internal Medicine, Division of Cardiology, Rhode Island Hospital, Warren Alpert Medical School of Brown University, USA
*Corresponding author: John Fani Srour, MD, Department of Internal Medicine, Cardiovascular Division, Rhode Island Hospital, Warren Alpert Medical School of Brown University, Providence, RI, USA, Tel: 617-777-9286, E-mail: johnfanisrour@yahoo.com
Int J Respir Pulm Med, IJRPM-1-006, (Volume 1, Issue 1), Case Report; ISSN: 2378-3516
Received: December 01, 2014 | Accepted: December 12, 2014 | Published: December 15, 2014
Citation: Srour JF (2014) Infective Endarteritis in a Patent Ductus Arteriosus. Int J Respir Pulm Med 1:006. 10.23937/2378-3516/1410006
Copyright: © 2014 Srour JF. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The ductus arteriosus, an essential fetal structure, normally closes spontaneously soon after birth. It�s persistence into late adulthood is considered rare; infective endarteritis complicating a patent ductus arteriosus (PDA) is an even rarer event. The clinical picture of an infected PDA could be subtle, and the diagnosis is frequently delayed. Symptoms may well be attributed to community acquired pneumonia, pleurisy, or pulmonary infarction from pulmonary embolism. Occasionally, especially in the case of silent PDA, the correct diagnosis can be reached after an extensive work up of fever of unknown origin.
The current case illustrates this uncommon presentation and highlights the role of echocardiography along with other imaging modalities in identifying an infected PDA.
Keywords
Infective endarteritis; Patent ductus arteriosus
Case Report
A 48-year-old man without past medical history presented with 8 weeks of fever, night sweats, fatigue, and weight loss, and one week of pleuritic chest pain and exertional dyspnea. Outpatient workup revealed anemia of chronic disease and negative serologies for tick-borne illnesses. Upon admission, physical examination was notable for a temperature of 38.7�C and a continuous murmur at the left upper sternal border. Computed tomography pulmonary angiogram (CTPA) revealed a wedge-shaped infarct in the lingula (Figure 1) and an intraluminal filling defect on the pulmonary artery end of a patent ductus arteriosus (Figure 2,3). Transthoracic echocardiogram confirmed the presence of a PDA and showed a dilated main pulmonary artery containing mobile echodensities (Figure 4) consistent with vegetations. Multiple blood cultures grew Streptococcus anginosus. The patient was treated with four weeks of intravenous ceftriaxone followed by percutaneous closure of the PDA.
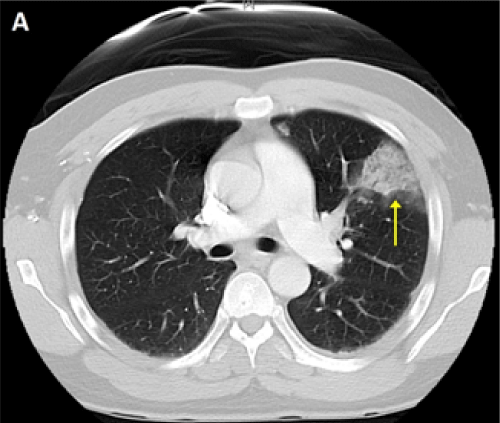
Figure 1: CTPA showing a wedge-shaped infarct in the lingula of the lung.
View Figure 1
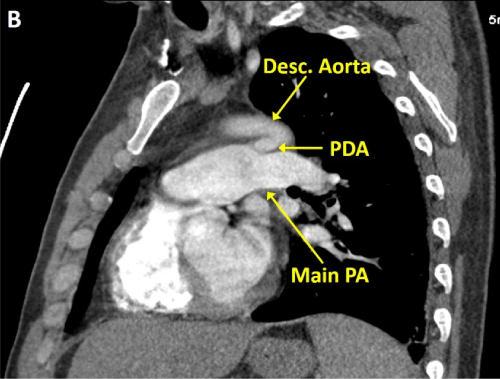
Figure 2: CTPA demonstrating a PDA connecting the proximal descending aorta to the roof of the pulmonary artery.
View Figure 2
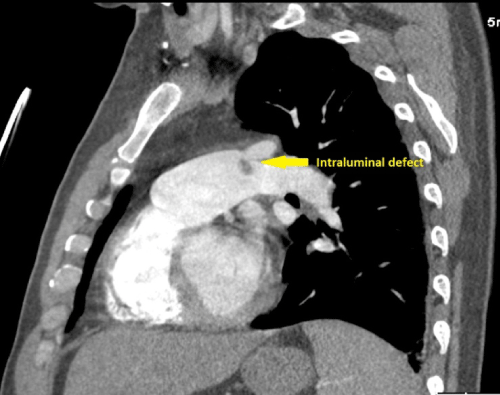
Figure 3: An intraluminal filling defect on the pulmonary artery end of a PDA.
View Figure 3
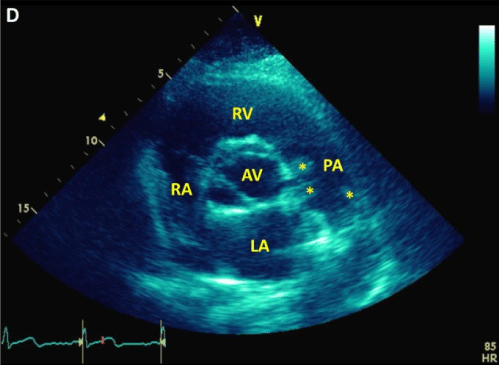
Figure 4: A parasternal short axis view during transthoracic echocardiogram displaying a dilated main pulmonary artery containing mobile echodensities consistent with vegetations.
LA: Left Atrium; RA: Right Atrium; RV: Right Ventricle; PA: Pulmonary Artery; AV: Aortic Valve; stars: vegetations.
View Figure 4
Discussion
Ductus arteriosus is a vascular structure that connects the proximal descending aorta to the roof of the pulmonary artery near the ostium of the left branch. This embryonic structure usually closes after birth. However, the persistence of a PDA after the first few weeks of life is abnormal. PDA accounts for 5% to 10% of all congenital heart disease [1]. The clinical significance of a PDA varies drastically from completely asymptomatic presence "silent PDA" to severe congestive heart failure or Eisenmenger's syndrome. A large duct results in a significant left to right shunt, pulmonary over-circulation, and left heart volume overload. On physical examination, the associated murmur is often noted at the upper left sternal border and is described as continuous sound with "machinery" quality. The murmur often radiates down the left side of the sternum and into the back. A thrill may be present. Diastolic rumble is audible at the cardiac apex in patients with moderate or large shunts.
The incidence of infective arteritis associated with PDA has dramatically decreased because of universal surgical closure, widespread use of antibiotics, and improved dental care. Early natural history studies (pre-antibiotics era) reported an incidence of 1% per year [2]. Silent PDA rarely leads to pulmonary endarteritis. Vegetations usually occur on the pulmonary artery end of the ductus; embolic events are usually of the lung rather than the systemic circulation. Other complications of PDA include aneurysms of the duct itself which may develop after infective endarteritis and surgical or percutaneous closure [3], hoarseness due to left vocal chord paralysis, dissection and/or spontaneous rupture of a dilated pulmonary artery [4], and acute aortic dissection [5].
The first step in the echocardiographic diagnosis lies in the direct visualization of the duct itself, which could be challenging in adults. Doppler imaging improves the diagnostic sensitivity by documenting the left-to-right shunt. The mosaic jet is usually seen entering the distal pulmonary artery retrogradely from the posterolateral direction. The backward flow should be distinguished from pulmonary regurgitation. Additionally, echocardiography plays an important role in estimating the magnitude of the shunt and the degree of left ventricular volume overload and pulmonary hypertension. Cardiac CT and MRI also have significant role in establishing the diagnosis and calculating the shunt volume and its effects on cardiac structures and function. Vegetations typically appear on these imaging modalities as intraluminal filling defects or mobile echodensities as described above.
Conclusion
In the era of antibiotics and routine early surgical closure, infective endarteritis is now a rare complication of PDA especially in late adulthood. However, this entity should remain on the differential diagnosis of adults presenting with fever and pulmonary infiltrates after more common causes are excluded.
References
-
Schneider DJ, Moore JW (2006) Patent ductus arteriosus. below Circulation 114: 1873-1882.
-
Sadiq M, Latif F, Ur-Rehman A (2004) Analysis of infective endarteritis in patent ductus arteriosus. below Am J Cardiol 93: 513-515.
-
Marasini M, Rimini A, Zannini L, Pongiglione G (2000) Giant aneurysm following coil occlusion of patent ductus arteriosus. below Catheter CardiovascInterv 50: 186-189.
-
Green NJ, Rollason TP (1992) Pulmonary artery rupture in pregnancy complicating patent ductus arteriosus. below Br Heart J 68: 616-618.
-
Cusick DA, Frederiksen JW, Mehlman DJ (1996) Acute aortic dissection: association with patent ductus arteriosus. below Am J Card Imaging 10: 200-203.





