International Journal of Respiratory and Pulmonary Medicine
Management of a Refractory Malignant Pleural Effusion in a Patient with Small Cell Lung Cancer: A Case Report
Maria M Wilczynska1* and Helen E Davies2
1Department of Respiratory Medicine, Forth Valley Royal Hospital, UK
2Department of Respiratory Medicine, University Hospital of Llandough, UK
*Corresponding author: Maria M Wilczynska, Department of Respiratory Medicine, Forth Valley Royal Hospital, Stirling Road, Larbert FK5 4WR, UK, E-mail: mwilczynska@doctors.org.uk
Int J Respir Pulm Med, IJRPM-2-022, (Volume 2, Issue 3), Case Report; ISSN: 2378-3516
Received: April 19, 2015 | Accepted: July 17, 2015 | Published: July 20, 2015
Citation: Wilczynska MM, Davies HE (2015) Management of a Refractory Malignant Pleural Effusion in a Patient with Small Cell Lung Cancer: A Case Report. Int J Respir Pulm Med 2:022. 10.23937/2378-3516/1410022
Copyright: © 2015 Wilczynska MM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Malignant pleural effusion (MPE) is a common presenting feature of underlying malignancy and frequently results in debilitating symptoms. In chemosensitive malignancies, such as lymphoma, small cell lung or ovarian cancers, chemotherapy is often considered the first line treatment, however if unsuccessful, pleural intervention to achieve fluid control is required. This may include therapeutic thoracentesis, chest drain insertion, thoracoscopy, chemical pleurodesis, and use of an ambulatory indwelling pleural catheter (IPC) or a combination of the above procedures.
Our case describes the management of a patient with a refractory MPE secondary to metastatic small cell lung cancer. No consensus exists to guide the management of patients with MPE secondary to chemosensitive disease and a personalised strategy should be adopted. Patients should be informed of all currently available therapeutic options, and further research performed focusing on the effect of these approaches, in combination with palliative chemotherapy, on patient related outcome measures.
Keywords
Indwelling pleural catheter, Lung cancer, Pleural effusion, Thoracentesis
Introduction
Lung cancer is the most common cause of malignant pleural effusion (MPE) followed by carcinoma of the breast. These malignancies account for 50-65% of all malignant effusions. Lymphoma, ovarian and stomach cancer account for a further 25%. Median survival in patients with MPE ranges from 3-12 months, varying with tumour type and stage [1].
Although small cell lung cancer (SCLC), lymphoma, ovarian and breast cancer classically respond to chemotherapy, associated recurrent symptomatic MPE may require additional intervention during the course of patients' treatment. Options for management include observation, therapeutic pleural aspiration, and intercostal tube drainage with instillation of sclerosant, thoracoscopic pleurodesis and placement of an indwelling pleural catheter (IPC).
Our case highlights a clinical scenario and encountered therapeutic dilemmas commonly faced by physicians managing patients with a MPE resultant from chemosensitive disease.
Case Presentation
A 69-year-old female presented with a 3-month history of a cough, three weeks of worsening breathlessness and generalised left arm pain. She was otherwise well with no anorexia or weight loss. She had no significant co-morbidities but was a current smoker of 20 cigarettes daily having accumulated a 30 pack year history; there was no history of asbestos exposure. At presentation, the patient was still working as a school cleaner. Her breathlessness was not limiting functional capabilities.
On initial examination, a swollen, engorged left arm with dilated superficial veins was noted. The latter was also evident on the anterior left chest wall. There was no upper limb neuro-vascular compromise. On chest auscultation, breath sounds were reduced with a reduced percussion note throughout left hemithorax.
Chest radiography and subsequent thoracic ultrasonography confirmed a moderate echogenic left-sided pleural effusion. Computed tomography imaging showed a 7cm left apical mass encasing the subclavian vessels; a central nodal mass occluding the left main bronchus, and a left pleural effusion (Figure 1). Radiologically, the patient had advanced primary bronchogenic carcinoma (stage 4; T4 N2 M1a). A diagnostic pleural aspiration was cytologically positive confirming metastatic small cell lung cancer.
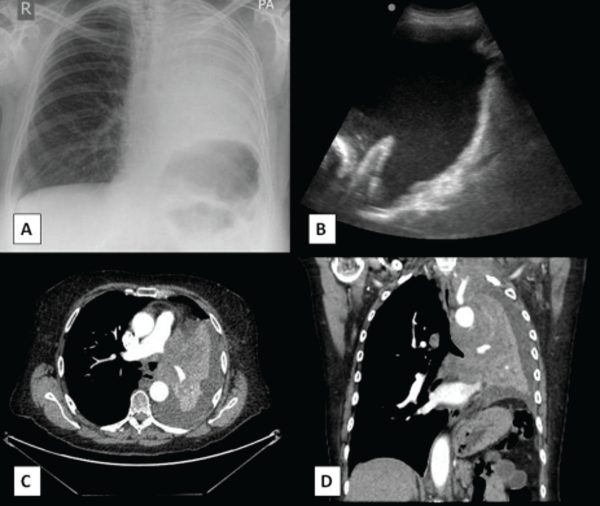
.
Figure 1: (A) Chest radiograph on presentation showing complete whiteout of left hemithorax the central location of the trachea concurs with the presence of underlying lung collapse and pleural effusion. (B) Thoracic ultrasound showing an echogenic pleural effusion, diaphragmatic inversion and nodularity. Transverse (C) and coronal (D) computed tomography images confirming complete left lung collapse and an associated pleural effusion.
View Figure 1
The patient's case was discussed with the lung cancer multidisciplinary team, and referral for chemotherapy made. She received four cycles of palliative carboplatin and etoposide with a good radiological and clinical response to treatment - her tumour burden lessened and the pleural effusion resolved. Consolidation thoracic radiotherapy (5 fractions of 20 Gy) and prophylactic cranial irradiation were therefore administered.
Five months after the initial treatment, the patient developed worsening dyspnoea. A chest x-ray confirmed recurrence of pleural effusion. She was admitted to a local hospital where an intercostal chest drain was inserted. Her symptoms improved and an x-ray the following day confirmed lung re-expansion with pleural apposition. Talc pleurodesis was performed with instillation of 4g of sterile talc slurry. Second-line chemotherapy with cyclophosphamide, doxorubicin, and vincristine was started, however, was discontinued after the first cycle as her symptoms worsened. Repeat imaging revealed recurrence of the left pleural effusion. Therapeutic options were discussed and an IPC was inserted. This resulted in significant improvement in breathlessness and quality of life.
Our patient gained independence through self-management of her IPC. She lived another four months and spent this time with family and community palliative care team support.
Discussion
MPE is a common clinical problem and is a frequent presenting feature of metastatic malignancy. Over 1 million people in Europe develop a MPE from lung cancer alone [2]. The burden of MPE continues to grow as the population's life expectancy rises, oncological therapies advance and cancer rates increase. The predicted human cost for patients with a MPE, with a median survival of 3 months, is significant.
Palliation of common symptoms of MPE, such as breathlessness, cough and chest pain; with the improvement of patient related outcome measures, should be foremost in MPE patient management. Early pharmacological treatment alongside pleural intervention is frequently required.
In patients with the chemosensitive disease, systemic chemotherapy may resolve the pleural effusion, as in the featured case; however after an initial response, pleural fluid may re-accumulate.
When considering treatment options, patients' symptoms, expectations and wishes, their performance status and life expectancy; and the nature of their underlying malignancy should be taken into account. In all cases, alleviation of symptoms to achieve improvement in patients' quality of life is paramount.
Most recent treatment algorithms for MPE advocate that if there is response to initial therapeutic pleural aspiration and there is no evidence of trapped lung on CT thorax several therapeutic intervention could be undertaken as the first line treatment depending on patient's choice and predicted life expectancy. It could be medical pleurodesis, thoracoscopy with a sclerosant instillation, or placement of an IPC [3].
Therapeutic pleural aspiration
At presentation pleural fluid aspiration can be performed for diagnostic and therapeutic purpose. Cytological analysis of pleural fluid provides a diagnostic sensitivity of approximately 60% although this is higher in certain malignancies e.g. adenocarcinoma than others e.g. mesothelioma. Although therapeutic (large volume) thoracentesis can be readily performed; between 98-100% of patients experience fluid re-accumulation with associated symptoms within 30 days and the procedure is not without risk [3].
Pleurodesis
Pleurodesis is the iatrogenic fusion of the pleural surfaces to prevent the accumulation of fluid or air. It may be performed chemically through instillation of a sclerosant into the pleural cavity, or mechanically through thoracoscopic pleural abrasion or parietal pleurectomy. The latter are seldom indicated in patients with MPE. Prior to performing pleurodesis a chest drain is sited and the effusion drained. Pleural apposition is a prerequisite for successful pleurodesis and, if this is not seen or the lung is 'trapped', pleurodesis is contraindicated [1]. Talc is the favoured (and most effective) sclerosant worldwide; an observation supported by a recent Cochrane review [4].
Thoracoscopy
Thoracoscopy is another therapeutic procedure that can be performed under local anaesthesia in patients with MPE. It allows for lysis of adhesions and administration of chemical sclerosants before a chest drain is placed [3]. Two randomised controlled trials have shown no significant difference in the 30 day pleurodesis success rate (radiologically measured) between surgical (thoracoscopic) talc insufflation and thoracostomy with talc slurry for patients with documented MPE [5,6]. However meta-analysis conducted by Xia et al. [7] showed that thoracoscopic talc poudrage was more effective than bedside talc slurry in controlling MPE especially in subjects with life-expectancy longer than one month [7].
Indwelling pleural catheter (IPC)
An alternative approach is the placement of a long term tunneled IPC. Insertion should be considered as a first-line option alongside pleurodesis and offers patients the choice of outpatient management versus the previous 'standard' inpatient drainage and pleurodesis [8]. Their insertion is indicated as the primary treatment for symptomatic MPE patients with an underlying 'trapped lung'. In RCT Davies et al. [8] compared the effectiveness of IPC with chest tube and talc slurry pleurodesis. There was no observed difference in the primary outcome measure, daily visual analogue scale scores of dyspnoea over 42 days, with both groups reporting an improvement in dyspnoea from baseline. Length of hospitalisation over the 12 month study period was significantly shorter in the IPC group than the talc group (-3.5 days, p<.001). There was no significant difference in quality of life or in the mean cost of managing patients with IPCs compared with talc pleurodesis. Furthermore, for patients with a survival of <14 weeks, IPC was significantly less costly than talc pleurodesis [8,9]. Although complications were higher in the IPC group in this study, other authors have reported low rates of IPC related adverse effects. In the meta-analysis by Van Meter et al. [10] the rates of empyema (2.8%), pneumothorax (5.9%), cellulitis (3.4%), catheter obstruction/blockage (3.7%) and unspecified malfunction of the catheter (9.1%) were all low and use of an IPC was without complication in the majority (87.5%) of patients [10]. There is ongoing multicentre RCT which aims to compare the efficacy of an IPC placement versus IPC placement plus talc sclerosant in patients with MPE managed exclusively as outpatients (IPC-PLUS). The primary outcome is successful pleurodesis at five weeks post-randomisation [11]. Initial data based on 24 patients with a symptomatic MPE who underwent talc pleurodesis via IPCs confirmed the safety and efficacy of administering talc slurry through IPCs in an outpatient setting [12].
Conclusions
The decision to undertake definitive therapy (pleurodesis, IPC, or both) for MPEs is decided on a case-by-case basis. The goals should concentrate on the prompt relief of symptoms, minimizing discomfort and disruption of the patient's everyday life. There are several effective therapeutic interventions available for patients with recurrent MPE which might be combined to achieve optimal control of symptoms.
The ultimate decision as to which strategy to adopt should be made following dialogue between the patient and the managing physicians, supported by the multi-disciplinary team.
No consensus exists regarding the ideal timing of definitive treatment of MPE in patients with chemosensitive disease; at present, this remains at the discretion of the individual physician and patient. Further research is required.
References
-
Roberts ME, Neville E, Berrisford RG, Antunes G, Ali NJ; BTS Pleural Disease Guideline Group (2010) Management of a malignant pleural effusion: British Thoracic Society Pleural Disease Guideline 2010. Thorax 65 2: 32-40.
-
Mishra E, Davies H, Lee Y (2009) Malignant pleural disease in primary lung cancer. Thoracic Malignancies Sheffield, UK European Respiratory Society Journals Ltd: 318-335.
-
Hsia D, Musani AI (2012) Management of malignant pleural effusions. Curr Respir Care Rep 1: 73-81.
-
Shaw P, Agarwal R (2004) Pleurodesis for malignant pleural effusions. Cochrane Database Syst Rev: CD002916.
-
Dresler CM, Olak J, Herndon JE 2nd, Richards WG, Scalzetti E, et al. (2005) Phase III intergroup study of talc poudrage vs talc slurry sclerosis for malignant pleural effusion. Chest 127: 909-915.
-
Yim AP, Liu HP (1996) Complications and failures of video-assisted thoracic surgery: experience from two centers in Asia. Ann Thorac Surg 61: 538-541.
-
Xia H, Wang XJ, Zhou Q, Shi HZ, Tong ZH (2014) Efficacy and safety of talc pleurodesis for malignant pleural effusion: a meta-analysis. PLoS One 9: 87060.
-
Davies HE, Mishra EK, Kahan BC, Wrightson JM, Stanton AE, et al. (2012) Effect of an indwelling pleural catheter vs chest tube and talc pleurodesis for relieving dyspnea in patients with malignant pleural effusion: the TIME2 randomized controlled trial. JAMA 307: 2383-2389.
-
Penz ED, Mishra EK, Davies HE, Manns BJ, Miller RF, et al. (2014) Comparing cost of indwelling pleural catheter vs talc pleurodesis for malignant pleural effusion. Chest 146: 991-1000.
-
Van Meter ME, McKee KY, Kohlwes RJ (2011) Efficacy and safety of tunneled pleural catheters in adults with malignant pleural effusions: a systematic review. J Gen Intern Med 26: 70-76.
-
Bhatnagar R, Kahan BC, Morley AJ, Keenan EK, Miller RF, et al. (2015) The efficacy of indwelling pleural catheter placement versus placement plus talc sclerosant in patients with malignant pleural effusions managed exclusively as outpatients (IPC-PLUS): study protocol for a randomised controlled trial. Trials 16: 48.
-
Ahmed L, Ip H, Rao D, Patel N, Noorzad F (2014) Talc pleurodesis through indwelling pleural catheters for malignant pleural effusions: retrospective case series of a novel clinical pathway. Chest 146: 190-194.





