International Journal of Respiratory and Pulmonary Medicine
A Single Center Review of Pleural Fluid Samples
C Walker1, J Mackenzie2, J Aust1, J Port1, D Cooper3, S Parker3, S Bourke3, J Miller3, M Weatherhead3 and A Aujayeb3*
1Foundation Trainees, North Tyneside General Hospital, England
2Specialist Registrar, North Tyneside General Hospital, England
3Respiratory Consultants, North Tyneside General Hospital, England
*Corresponding author:
Dr. Avinash Aujayeb, Respiratory Consultant, North Tyneside General Hospital, Northumbria Healthcare NHS Foundation Trust, Rake Lane, North Shields, Newcastle, England, E-mail: aujayeb@doctors.net.uk
Int J Respir Pulm Med, IJRPM-4-067, (Volume 4, Issue 1), Research Article; ISSN: 2378-3516
Received: December 10, 2016 | Accepted: February 16, 2017 | Published: February 20, 2017
Citation: Walker C, Mackenzie J, Aust J, Port J, Cooper D, et al. (2017) A Single Center Review of Pleural Fluid Samples. Int J Respir Pulm Med 4:067. 10.23937/2378-3516/1410067
Copyright: © 2017 Walker C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction
Large scale epidemiological data of pleural fluid characteristics are lacking. We are an established pleural center with high rates of pleural malignancy in the North East of England. We aim to follow the British Thoracic Guidelines and sought to add to the body of evidence.
MethodologyAll pleural aspirations done between January 1st 2014 and December 31st 2015 were analyzed for fluid biochemistry, microbiology and cytology and final diagnoses. Light's criteria were applied wherever possible.
Results
º Population: 264 patients
º Average age: 74 years
º Gender: 61% Male
º Lung Cancer (n = 44)
º 26/44 (59%) had positive first cytology
º 15 had more than one sample taken, and in 6 patients, this provided the diagnosis (14%)
º 36/44 (82%) were exudates
º 4/44 (9%) were transudates
º 3/44 had a pH < 7.21
º Heart Failure (n = 36)
º 9/36 were exudates
º Empyema (n = 16)
º Only 2/16 (13%) had positive microbiology
º 12/16 (75%) had a pH < 7.21 (2 did not have a pH sent)
º 16/16 were exudates
º Mesothelioma (n = 24)
º 12/24 (50%) had positive first cytology
º 19 went on to have further sampling (done at thoracoscopy). Only 2 further sample provided diagnosis
º 2/24 had a protein < 25
º 14/24 went on to have a biopsy
º 13/14 had a thoracoscopy and 1/14 had a CT guided biopsy
º All Malignancies (Lung Primary/Lung Secondary/Meso) (n = 96)
º 80/95 (84%) were exudates
º 8/95 (8%) had a protein < 25 AND a LDH < 200
º Audit:
º 217/264 didn't meet the BTS guidance stating that every pleural tap should be sent for, microbiology, cytology, protein, LDH
º pH < 7.21 (n = 19)
º 12 were empyemas
º But also:
• 3 adenocarcinomas of the lung
• 2 mesothelioma
• 1 metastatic ovarian carcinoma
• 1 haemothorax
º Microbiology:
• Only positive in 5/241 samples sent in universal bottles
• 3 were empyemas
• 1 was a biliary fistula
• 1 was an infected indwelling pleural catheter
Our findings are consistent with current literature.
We found 60% positivity for cytology in lung cancer, dropping to 50% in mesothelioma, with further sampling helping in the former only. Tissue biopsy should be de rigeur if mesothelioma is suspected, rather than repeat cytology.
8% of all malignant effusions are transudates, and clinical as well as radiological correlation is vital in those cases.
We fail in adherence to BTS guidelines- local guidelines have just been updated and all respiratory doctors have been reminded of them. We will also aim to increase microbiological yield by sending fluid in blood culture bottles.
Keywords
Pleural, Fluid, Exudate, Transudate
Introduction
Anecdotally, the incidence of patients with pleural effusions is increasing, most of the data coming from cancer databases [1].
Pleural units are being set up in almost every UK hospital, based on the financial incentives from the best practice tariff for seeing pleural patients in an outpatient setting and the need to minimise the time these patients spend in hospital [2]. This stems from the fact that if a malignant pleural effusion is diagnosed, median survival is approximately 4 months [3].
We are an established pleural centre that performs medical thoracoscopies and inserts indwelling pleural catheters. We have a very high diagnostic sensitivity for the former, and below national average complication rates for both [4]. As such, we also have a large cohort of patients with pleural effusions. The North East of England has one of the highest rates of mesothelioma in the country and we serve a population of over half a million individuals [4].
We sought to add to the body of evidence about pleural fluid characteristics by analysing that cohort.
Light's criteria revolutionised how to define and diagnose pleural effusions [5]. Pleural fluid is exudate if one of the following is present:
• Effusion protein/serum protein ratio greater than 0.5
• Effusion lactate dehydrogenase (LDH)/serum LDH ratio greater than 0.6
• Effusion LDH level greater than two-thirds the upper limit of the laboratory's reference range of serum LDH
Since then, there have been many studies assimilated into guidelines [6-9]that have been produced, but large scale epidemiological studies, national or local databases about pleural fluid characteristics are missing.
A brief selective summary of the accepted evidence is as follows [6-9]:
• Protein counts, LDH, microscopy and culture (MC&S)and cytology should be requested for every pleural fluid taken off
• Fluid pH and glucose should only be sent if infection or rheumatoid effusions are suspected
• Cytology is only positive in 40-60% of cases, repeated samples can help increase diagnostic sensitivity by 30%- image guided biopsy via computer tomogram or thoracoscopy can help to obtain tissue
• Exudative effusions are often associated with infection or malignancy, transudative ones with cardiac, renal or liver failure
• 80% of patients with bilateral effusions have cardiac failure and 75% of those resolve within 48 hours of starting diuresis. 20% of those become exudative due to protein re-absorption and resolution of venous hypertension
• 20-30% of transudates have positive cytology
• There are many reports of respiratory units not adhering to the BTS guidance
Methods
All pleural aspirations done between January 1st 2014 and December 31st 2015 were analyzed for fluid biochemistry, microbiology, cytology and final diagnoses (n = 264). Light's criteria were applied wherever possible. The patient's diagnosis was taken from clinic letters electronically uploaded to another system called SingleView. Local governance approval was sought and obtained.
Results
264 pleural aspirates were analyzed. The average patient age was 74 years. 61% of the patients were male.
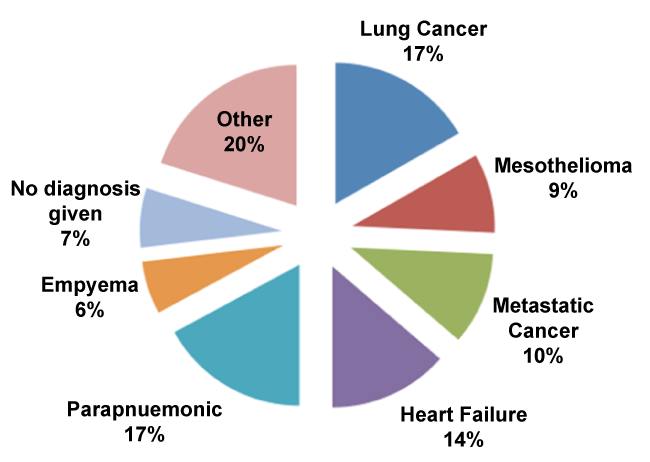
The final diagnoses were as in figure 1.
No diagnosis was given in 18 out of 264 cases as the effusions either resolved spontaneously or patients died before further investigations)
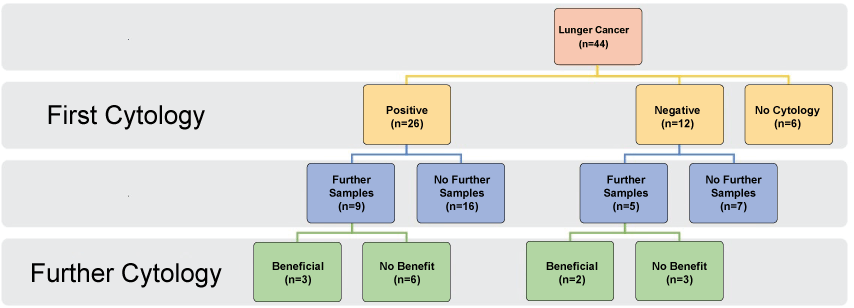
In the patients who had lung cancer (n = 44), figure 2,
.
Figure 2: Analysis of all lung cancer patients, their pleural fluid cytology at first sampling and if further sampling was beneficial or not.
View Figure 2
• 26/44 (59%) had positive first cytology
• 15 had more than one sample taken, and in 6 patients, this provided the diagnosis (14%)
• 36/44 (82%) were exudates
• 4/44 (9%) were transudates
• 3/44 had a pH < 7.21
In the heart failure patients, (n = 36), 9 (25%) were exudates. No other pathology was responsible for the exudative nature.
For those with empyema (n = 16),
• Only 2/16 (13%) had positive microbiology
• 12/16 (75%) had a pH < 7.21 (2 did not have a pH sent)
• 16/16 were exudative
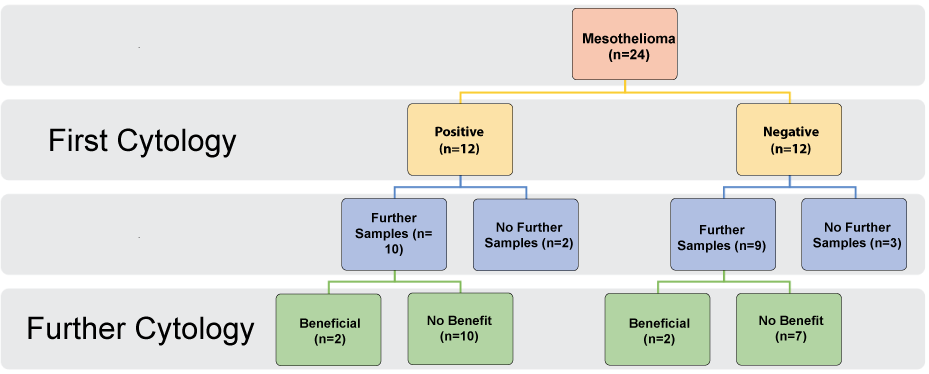
Mesothelioma patients numbered 24 in total (Figure 3)
.
Figure 3: Analysis of all mesothelioma patients, their pleural fluid cytology at first sampling and if further sampling was beneficial or not.
View Figure 3
• 12/24 (50%) had positive first cytology
• 19 went on to have further fluid sampling done at thoracoscopy. Only 2 of those further samples provided a diagnosis. Thoracoscopic biopsies were all diagnostic
• 2/24 had a protein < 25
For all malignancies, n = 96, 80/95 (84%) were exudates, 8/95 (8%) had a protein < 25 and a LDH < 200, and the rest (6) did not have enough data to be classified exactly.
When the pH < 7.21 (n = 19), 12 were empyemas and the rest were 3 adenocarcinomas of the lung, 2 mesotheliomas, 1 metastatic ovarian carcinoma and 1 hemothorax.
With regards to microbiological data, we only had 5 positive results in 241 samples sent. All our samples were sent in universal bottles. 3 were from primary empyemas, 1 from a pleuro-biliary fistula, 1 from an infected indwelling pleural catheter.
With regards to auditing against the BTS guidelines, 217 out of 264 didn't meet the BTS guidance stating that every pleural tap should be sent for microbiology, cytology, protein, and LDH.
Case Vignette
A 62-year-old breathless male is admitted to the cardiology ward with bilateral pleural effusions, pedal edema and a raised jugular venous pressure. He has a history of hypertension, previous heavy smoking and previous deep vein thrombosis. He is diuretic naïve. An echocardiogram shows a left ventricular function at 40% of predicted and his Brain Natriuetic Peptide level is raised at more than 50 times of the upper normal limit. Intravenous diuretics, fluid restriction and blood pressure control help in alleviating some breathlessness but his effusions, though smaller, persist. A respiratory opinion is thus sought, 72 hours post admission.
Some asbestos exposure (previous shipyard worker) is noted about 40 years ago. Bedside thoracic ultrasound reveals a left sided effusion, bigger than the right. A pleural tap shows a pH of 7.32, LDH 300 units/L, protein 33 g/L with respective plasma values of 550 units/L and 89 g/L. Cytology is negative. A chest computer tomogram shows bilateral effusions with mild enhancement of the left pleural surface.
What are the next steps in the management of this patient?
Discussion
The above is a simple retrospective analysis of pleural fluid samples. How can it be used in clinical practice?
Light's criteria have stood the test of time for classifying exudates and transudates, with an overall diagnostic accuracy of 95%. However, care needs to be taken when the protein count approaches 0.5, as some of the sensitivity is lost [10]. Hence, it is suggested to always have a high index of suspicion for alternative pathology. Transudates are not often benign as we found, at a percentage akin to the literature [11]. In our cohort, 8% of all malignant effusions are transudates, and clinical as well as radiological correlation is vital in those cases.
One of the problems we faced was that there was no corresponding serum LDH or protein measurement, but we managed to categorize all the effusions after detailed analysis and looking at serum values a few days before or after the sample was taken. Interestingly, it seems that not having a blood sample does not significantly affect the final clinical interpretation [12].
Cytological yield for malignancy is 40% on average but depends on the tumour type. it can be as high as 83% for ovariancancer, 57% for lung cancer but lowest for mesothelioma and lymphoma [13]. In a region with known high cases of mesothelioma and lung cancer, is it worth doing a pleural aspirate for diagnosis? We found 60% positivity for cytology in lung cancer, dropping to 50% in mesothelioma, with further sampling helping in the former only. Tissue biopsy should be de rigeur if mesothelioma is suspected, rather than repeat cytology. With the advent of immunogenetics and profiling a patient's treatment exactly, there is an ever-increasing need for tissue to be obtained, either from a bronchoscopy, thoracoscopy or image guided biopsy. This is our experience locally and needs to be backed up from national or international surveys. Cytological examination of fluid with immunohistochemistry also costs between 130 to 200 pounds [14]. So, in our opinion, if we suspect that someone has a thoracic malignancy with an effusion and does not need fluid taken off for symptoms, it is better to save the trust money, the patient the pain and risk of complications from a tap or aspirate, and to offer the patient the opportunity to undergo a single procedure (thoracoscopy, if patient is fit to undergo it) for diagnostic and therapeutic purposes.
Our microbiological yield was dire, but reflects other studies. Barnes, et al. [15] found only 15 true-positive cultures (3.2%) among 476 patients with pleural effusions of any cause. This is because of how empyemas or infected parapneumonic effusions occur [16]. The pleural cavity is a closed space and relatively anaerobic. It is widely believed that infections in the pleural space, in the absence of direct instrumentation, occurs from the lung parenchyma (for example a pneumonia) with translocation of pathogens into the cavity. The pathogens then cause an intense cellular reaction with fibrin and collagen deposition and then die off but the inflammation persists and can become systemic. There is some evidence to suggest that yield will increase with direct inoculation into blood culture bottles and we have changed our local guidelines but we only send for microscopy now if we think the fluid might be infected. That requires close correlation with the patient's clinical presentation.
We fail in adherence to British Thoracic Society pleural guidelines [8] local guidelines have just been updated and all respiratory doctors have been reminded of them. We felt that there was no need to disseminate to all the doctors in the trust as since the advent of thoracic ultrasonography, only respiratory registrars or consultants perform pleural procedures. However, is there a need to always adhere to the guidelines? As consultants seeing mostly pleural malignancies, we do not always send a glucose off or a pH sample, as malignancy can cause acidaemia as well as low glucose levels. There is some evidence to suggest that pH values have prognostic value [7] but this is not used extensively.
Our study has some limitations. This is a single center retrospective review of pleural fluid samples and we could have increased the number of patients by going back further in time, but the results above are in par with the available literature. We did not have corresponding serum LDH or protein measurements for every sample but we analysed each sample individually and with the above measurements taken a day before or after we felt we could confidently apply Light's criteria.
The patient above has heart failure and a transudative effusion the high BNP, low ejection fraction and bilateral effusions pointing to this. However, the effusions are not resolving fast and the mild pleural enhancement might be significant. We suggested another week of diuresis, and his weight dramatically decreased but the left sided effusion persisted. He thus underwent medical thoracoscopy 2 weeks after his first presentation. A grossly abnormal parietal pleural surface was biopsied. He was diagnosed with epithelioid mesothelioma, T1N0M0, and was referred for chemotherapy, which he declined. He was referred to palliative care services and is under regular follow up.
Conclusions
We hope that the above will help clinicians in how to investigate pleural effusions. The main learning points are
1. Malignant effusions can be transudative
2. Not having a contemporary blood sample does not affect the eventual interpretation
3. Repeat cytological assessment has limited value
4. To consider a single theurapeutic and diagnostic procedure if a malignant effusion is present
5. To only send for microbiology in a blood culture bottle if an infection is suspected.
References
-
Cancer Research UK Statistical Information Team.
-
2014/15 National Tariff Payment System (2013) Annex 4 A: Additional information on currencies with national prices.
-
Clive AO, Kahan BC, Hooper CE, Bhatnagar R, Morley AJ, et al. (2014) Predicting survival in malignant pleural effusion: development and validation of the LENT prognostic score. Thorax 69: 1098-1104.
-
Aujayeb A, Parker S, Bourke S, Miller J, Cooper D (2016) A review of a pleural service. J R Coll Physicians Edinb 46: 26-31.
-
Light RW, Macgregor MI, Luchsinger PC, Ball WC Jr (1972) Pleural effusions: the diagnostic separation of transudates and exudates. Ann Intern Med 77: 507-513.
-
American Thoracic Society (2000) Management of malignant pleural effusions. Am J RespirCrit Care Med 162: 1987-2001.
-
Roberts ME, Neville E, Berrisford RG, Antunes G, Ali NJ, et al. (2010) Management of a malignant pleural effusion: British Thoracic Society Pleural Disease Guideline 2010. Thorax 65: ii32-ii40.
-
Hooper C, Lee YC, Maskell N, BTS Pleural Guideline Group (2010) Investigation of a unilateral pleural effusion in adults: British Thoracic Society pleural disease guideline 2010. Thorax 65: ii4-ii17.
-
Davies HE, Davies RJ, Davies CW, BTS Pleural Disease Guideline Group (2010) Management of pleural infection in adults: British Thoracic Society pleural disease guideline 2010. Thorax 65: ii41-ii53.
-
Heffner JE, Brown LK, Barbieri CA (1997) Diagnostic value of tests that discriminate between exudative and transudative pleural effusions. Primary Study Investigators. Chest 111: 970-980.
-
Porcel JM, Alvarez M, Salud A,Vives M (1999) Should a cytologic study be ordered in transudative pleural effusions? Chest 116: 1836-1837.
-
Porcel JM, Peña JM, Vicente de Vera C, Esquerda A (2006) Reappraisal of the standard method (Light's criteria) for identifying pleural exudates. Med Clin (Barc) 126: 211-213.
-
Rodríguez-Panadero F (2008) Medical thoracoscopy. Respiration 76: 363-372.
-
Germain A, Doyle R, Nolan K, Gambles M, Roberts A, et al. (2015) PA3 Using the lived experience of volunteers to enhance the care of dying patients and their families. BMJ Support Palliat Care 5: A20.
-
Barnes TW, Olson EJ, Morgenthaler TI, Edson RS, Decker PA, et al. (2005) Low yield of microbiologic studies on pleural fluid specimens. Chest 127: 916-921.
-
Light R, Lee GCY (2016) Textbook of Pleural diseases. (3rd edn), CRC press.