International Journal of Virology and AIDS
Salviae and Cinnamomi Herbal Medicines have Antiviral Activity against a Broad Range of Human Immunodeficiency Virus
Fu-Shi Quan1, Young Jae Lee2, Richard W Compans3 and Sang-Moo Kang4*
1Department of Medical Zoology, Kyung Hee University School of Medicine, Seoul, Korea
2College of Veterinary Medicine, Jeju National University, 66 Jejudaehakno, Jeju 690-756, Republic of Korea
3Department of Microbiology and Immunology and Emory Vaccine Center, Emory University School of Medicine, Atlanta, GA 30322, USA
4Center for Inflammation, Immunity & Infection, Institute of Biomedical Sciences, Georgia State University, Atlanta, GA 30303, USA
*Corresponding author: Sang-Moo Kang, Center for Inflammation, Immunity & Infection, Institute of Biomedical Sciences, Georgia State University, Atlanta, GA 30303, USA, Tel: 404-413-3588, E-mail: skang24@gsu.edu
Int J Virol AIDS, IJVA-2-013, (Volume 2, Issue 2), Research Article; ISSN: 2469-567X
Received: August 31, 2015 | Accepted: October 17, 2015 | Published: October 23, 2015
Citation: Fu-Shi Q, Lee YJ, Compans RW, Sang-Moo K (2015) Salviae and Cinnamomi Herbal Medicines have Antiviral Activity against a Broad Range of Human Immunodeficiency Virus. Int J Virol AIDS 2:013. 10.23937/2469-567X/1510013
Copyright: © 2015 Fu-Shi Q, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Development of broadly effective anti-human immunodeficiency virus (HIV)-1 compounds is a high priority since the number of individuals having HIV infections continues to grow worldwide and vaccines are not available yet. From herbal extracts commonly used in humans, Salviae miltiorrhiza (Salviae), Cinnamomi Ramulus (Cinnamomi), Astragali Radix (Astragali), Asari Radix et Rhizome (Asari), Panax Ginseng (Korean red ginseng), we screened candidate herbal medicines and investigated their antiviral activity against a broad range of HIV-1 strains including clades A and C primary isolates that are dominantly found in Africa and India. Antiviral activity was determined based on the levels of inhibiting viral replication of infectious HIV-1 or infectivity of pseudovirion particles. Ginseng but not Astragali and Asari showed low to moderate levels of antiviral activity against HIV-1 strains. Most significantly, we found that Salviae and Cinnamomi showed a potent antiviral activity against antigenically and geographically different strains of HIV-1. In particular, Salviae extract reduced over 90% of infectivity of all strains tested including clades A, B, and C. The results in the present study provide evidence that Salviae and Cinnamomi extracts may have the potential to be developed for the discovery of novel anti-IV-1 microbicides or drugs which are applicable to a broad range of HIV-1 strains found in the developing and developed countries.
Keywords
HIV-1, Antiviral activity, Herbs, Salviae, Cinnamomi
Introduction
Human immunodeficiency virus type-1 (HIV-1) is the cause of a major pandemic, seriously threatening the public health worldwide particularly in developing countries. Despite enormous efforts, effective vaccines against HIV-1 are not available yet for human use and the number of individuals having HIV infections continues to grow. Therefore, there is an urgent need to develop alternative means to prevent or control HIV infections. Herbal medicines including ginseng are widely used in the United States, with approximately one quarter of adults reporting the use of an herb to treat a medical illness. Ginseng is reported to have a wide range of therapeutic and pharmacological benefits. Ginsenosides, the major pharmacologically active ingredients of ginseng, appear to be responsible for most of the activities of ginseng including vasorelaxation, antioxidation, anti-inflammation and anti-cancer {Lu, 2009 #34}. Natural products have provided a source for the discovery of many novel therapeutic agents. Approximately one third of human drugs were originally derived from plant sources [1]. Astragali Radix is used in treatment of abscess, ulcers and skin eruptions. The root extracts of Salviae miltiorrhiza are commonly used in China, Japan, the United States, and European countries. The root extracts of Salviae miltiorrhiza are known to promote the flow of bloodstream and used for the treatment of cardiovascular and cerebrovascular diseases. In China, Salvia miltiorrhiza is used to treat variety of diseases such as angina pectoris, myocardial infarction, hypertension, hyperlipidemia, and acute ischemic stroke. Cinnamomi Ramulus is used for gas (flatulence), muscle and stomach spasms, preventing nausea and vomiting, diarrhea, infections, the common cold, and loss of appetite. Asari Radix et Rhizome is widely used as an antitussive and analgesic agent in the treatment of cough and dyspnoea, headache, toothache, rheumatoid arthritis and sinusitis. Although traditional Chinese medicines are widely used in humans, our knowledge on their therapeutic effects on human disease is very limited. It is suggested that research efforts should be highly encouraged towards identifying novel herbal products that have efficacy for treating or preventing disease. It was previously demonstrated that HIV-1 infected individuals who had taken Korean red ginseng showed significantly improved clinical parameters including slower decreases in CD4 T cells, lower copy numbers of HIV RNA and a decrease in serum soluble CD8 antigen, resulting in fewer cases requiring anti-retroviral drug treatments and hospital admission [2].
In an effort to understand the potential mechanism by which ginseng exhibits its positive clinical effects in HIV-1 infected individuals, we tested the possibility of potential anti- HIV-1 activities of ginseng and several other common herbal medicines that are routinely prescribed to healthy individuals and patients.
Materials and Methods
Virus and cells
HIV-1 89.6 strain and single-cycle replication-competent HIV-1 pseudovirions containing envelope proteins from diverse HIV-1 primary isolates of clades A, B, and C were used as described in table 1. JC53-BL cells was used which are derivative of HeLa cells expressing β-galactosidase from an HIV-1 LTR promoter were used. Pseudovirions HIV-1 containing env gene YU2 (NIH AIDS reagent program) and consensus HIV-1 env of clade B (ConB) were used.
Herbs and herbal extract preparation
Korean red ginseng root powder (Panax Ginseng, The Korean Ginseng Co. Ltd.) that was the same type of ginseng products prescribed for HIV-1 infected patients was used for preparation of extracts. Other herbal medicines used in this study include the roots of Salviae (Botanical Name Salvia miltiorrhiza as described [3-6], the branches of Cinnamomi (Cinnamomi Ramulus) [7], the roots of Asari (Asari radix et rhizome), and the roots of Astragali (Astragali Radix) (Figure 1A, Figure 1B, Figure 1C, Figure 1D, Figure 1E). These herbal medicines are routinely prescribed by licensed oriental medicine doctors. Extracts of herbal drugs were prepared and kindly provided by Dr. In- Seok Jung (Grace Acupuncture and Herbs Clinic, Duluth, GA, USA). Briefly, dried herb plants were grounded into small pieces. Water extracts of herbs were prepared as follows. Herbs were mixed with distilled water and then heated to 80 - 90°C for 2 h. The aqueous extracts were 0.45 μm membrane-filtered and kept at 4°C until use (Ginseng: 2.5 mg/ml, Salviae: 4.2 mg/ml, Cinamomi: 1.3 mg/ml, Astragali: 0.76 mg/ml, Asari: 1.2 mg/ml based on dried herbal plant weight after removing undissolved solid plant materials). Approximately less than 20% of the weight of dried herbs was extracted into the aqueous solution. Batch to batch preparations were maintained consistent. Since an herbal extract contains a mixture of various components including proteins and organic compounds, the protein concentrations were determined and normalized for cytotoxicity assays.

.
Figure 1: Herbal medicines used in this study. A). Ginseng (Panax Ginseng), B). Cinnamomi (Cinnamomi Ramulus), C). Salviae (Salviae miltiorrhiza), D). Astragali (Astragali Radix), E). Asari (Asari Radix et Rhizome).
View Figure 1
Virus neutralization assay
JC53-BL cells that were used for neutralization activity assays of HIV-1 were incubated with various concentrations of herbal extracts to determine the cytotoxicity of herbal extract. After culture for 3 days, we counted the live cells (unstained) based on resistance to penetration of trypan blue dye, indicating the membrane integrity, and the dead cells (stained). Use of pseudovirions to assess neutralizing activities of antibodies against HIV-1 primary isolates in vitro has been well described [8-11]. In brief, plasmid pHIVSG3 (a kind gift from Dr. Beatrice Hahn, University of Alabama at Birmingham) containing the entire virus genome with a deleted env gene was mixed with a plasmid containing the HIV env gene YU2 (NIH AIDS reagent program) or consensus HIV-1 env of clade B (ConB), and transfected into 293T cells using calcium phosphate precipitation. Then pseudovirion particles were harvested at day 2 post transfection and used for anti-HIV-1 activity assays. The codon optimized ConB sequence was described [12] and obtained from Dr. Beatrice Hahn.
Toxicity of herbal extract in mice
Regarding safety and toxicity concerns of herbal extracts, mice were injected subcutaneously daily for 10 days (250 μg per mouse based on the amount of protein) and did not show any sign of side effects or changes in body weight. From the cytotoxicity results, anti-HIV-1 activities were performed in a range of 20 to 100 fold lower than maximum concentrations with no observed cytotoxicity.
Results and Discussion
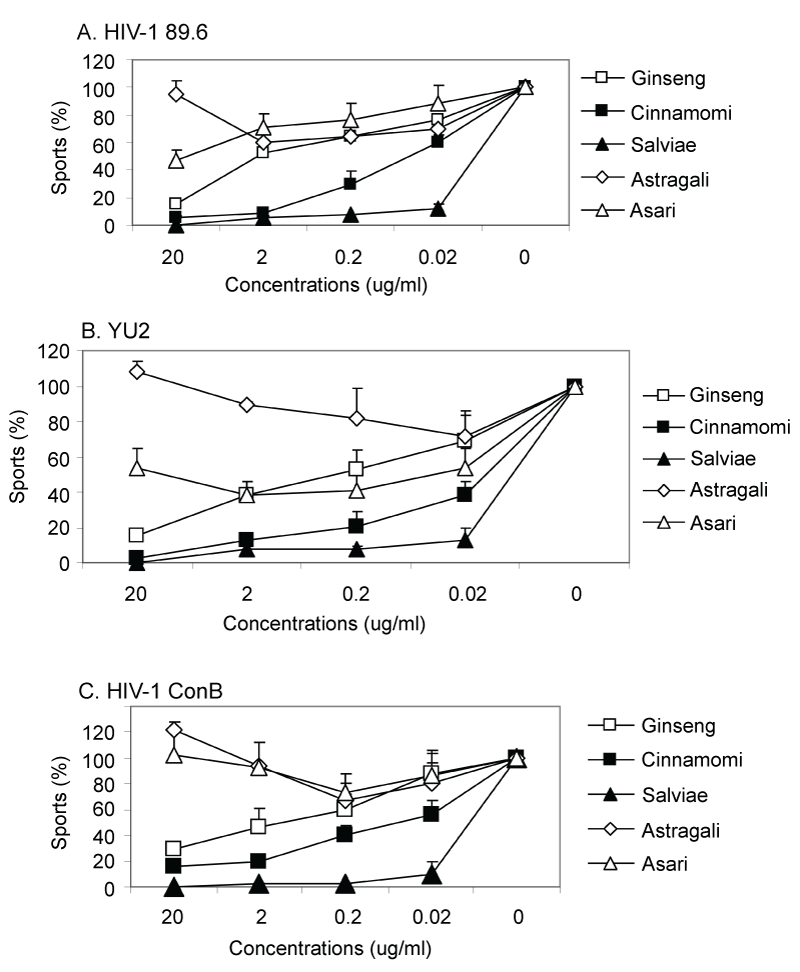
Initially we determined virucidal activities of several herbal extracts including ginseng against an infectious virus, the HIV-1 89.6 strain. The neutralization assay was based on an indicator cell line, JC53-BL, which is a derivative of HeLa cells expressing β-galactosidase from an HIV-1 LTR promoter [8,9]. Infection with HIV-1 induces the expression of β-galactosidase in infected cells and blue spots were counted as a measure of viral infection. The detailed procedure was previously described [13,14]. Ginseng showed a moderate level of anti-HIV-1 89.6 virucidal activity (Figure 2A). Interestingly, Cinnamomi and Salviae herbal extracts showed much more potent anti-HIV-1 activities. In particular, Salviae exhibited antiviral activity against HIV-1 at as low as 0.02 μg/ml concentration whereas Asari and Astragali did not show any significant levels of antiviral activity. Only minimal cytotoxicity of 5-10% cell death was observed up until the 1000 μg/ml concentrations of ginseng extract. Similarly, no visible cytototoxicity of JC53-BL cells was observed after incubation with Cinnamomi, Salviae, Astragali, Asari herbal extracts up to 500 μg/ml concentrations.
From the unexpected promising results of virucidal effects of Cinnamomi and Salvia, we further extended the study of their antiviral effects on different HIV-1 strains. As shown in Figure 2B and 2C, ginseng showed a moderate level of antiviral activity against HIV-1 YU2 and ConB pseudotypes. Low levels of antiviral activity were observed in Asari herbal extracts but not in Astragali. Surprisingly, highest levels of antiviral activity against both YU2 and ConB strains were observed in Salviae herbal extract. Cinnamomi extract also showed high antiviral activity against both YU2 and ConB strains but slightly lower than Salviae. Elicitation of broadly neutralizing antibodies against HIV-1 is a challenging and difficult hurdle. Thus, an antiviral drug that is broadly reactive against different clades of HIV-1 would be desirable. We tested anti-viral activities of herbal extracts against a more extended panel of primary isolates of HIV-1 including clades A, B and C, which are the strains causing serious AIDS epidemic in developing countries. The herbal extracts were tested against one simian human immunodeficiency virus (SHIV) and nine primary HIV-1 isolates (Table 1). The HIV-1 stocks tested were human PBMC-grown SHIV-89.6P and 293T cell-produced pseudotyped viruses with Env of primary HIV-1 isolates (Table 1).
.
Figure 2: Virucidal activity of herbal extracts against infectious live HIV-1 89.6 and pseudovirions containing HIV-1 envelope protein from primary isolates. Approximately 100 infectious particles of HIV-1 89.6, pseudovirions of HIV-1 YU2 or HIV-1 ConB were incubated with various concentrations of herbal extracts and transferred to monolayer cell cultures of JC53 cells. The remaining infectious centers were counted under the microscope and results are represented as percentile compared to the control well without herbal extracts. (A) HIV-1 89.6, (B) Pseudovirion HIV-1 YU2, (C) Pseudovirion HIV-1 ConB.
View Figure 2
![]()
Table 1: Antiviral activities of herbal extracts against diverse HIV-1 primary isolates of clades A, B, and C.
View Table 1
The antiviral titers are represented as dilutions of herbal extracts at which relative luminescence units were reduced to 50% compared to virus control wells without the test inhibitor. Titers of 100 to 400 were considered to be moderate and titers over 400 to be potent antiviral activities. Ginseng showed moderate antiviral activities against HIV-1 primary isolates of clade B, QH0692.42 and PVO.4. Broadly cross-reactive HIV-1 antiviral activities against all HIV-1 primary isolates tested were consistently observed with Salviae and Cinnamomi extracts. Particularly, Salviae showed potent antiviral activities (> 400) against 4 out of 6 clade B primary isolates and 3 out of 4 clades A, or C isolates. Astragali and Asari extracts were inactive against most primary HIV-1 isolates tested. Also, all herbal samples were tested against a murine retrovirus (SVA-MLV) and showed low activity.
To evaluate the antiviral activities of herbal extracts as a potential candidate for a microbicide against HIV-1, 90% reductions of infectivity were similarly determined (Table 1). The herbal extracts of Salviae and Cinnamomi but not ginseng, Asari, or Astragali could decrease the infectivity of various HIV-1 primary isolates up to 90%. The capacity of these Salviae and Cinnamomi to inactivate 90% of infectious particles indicates a highly potent strength of antiviral activity. The active components in Salviae and/or Cinnamomi may have virucidal activity against clades B, C, and A of HIV-1 isolates, which would be a desirable property for being a promising microbicide and/or antiviral drugs.
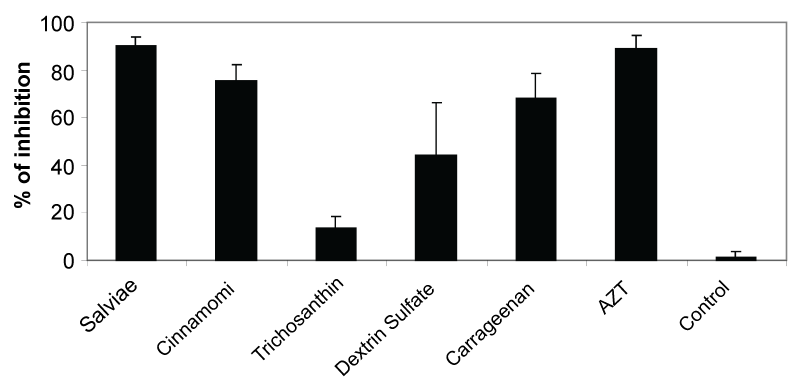
Encouraged by the unusually potent anti-HIV-1 activities of Salviae and Cinnamomi herbal extracts, virucidal activities of Salviae and Cinnamomi herbal extracts were further compared with previously demonstrated antiviral compounds including AZT (3'-azido-3'-deoxythymidine) as a positive control. Trichosanthin, a ribosome-inactivating protein extracted from the root tuber of Chinese medicinal herb has been extensively investigated in vitro and clinically as a potential anti-HIV-1 herb, but has some limitations due to its antigenicity, short half-life, and toxicity [15-22]. Dextran sulfate has also been studied for its virucidal activity despite development of resistant virus and low efficacy against HIV-1 R5 primary isolates, and toxicity in a clinical trial [23-26]. Carrageenan, a sulfated polysaccharide, is considered to be a promising microbicide although its safety and efficacy remains to be determined [27-30]. Compared to previously described virucidal compounds, Salviae and Cinnamomi herbal extracts consistently showed similar or higher potency in activity against HIV-1 primary isolate SF162 pseudovirions. It was also reported that aqueous extracts of Chinese medicinal herbs (Prunella vulgaris, Viola yedoensis) inhibited the in vitro growth of HIV at a subtoxic concentration [31,32] although they did not inactivate HIV extracellularly (i.e., they were not virucidal) and showed activities only at relatively high concentrations. It is also important to note that non-toxic inhibitors of HIV-1 integrase were isolated from the Salviae extract [33]. HIV-1 integrase inhibitors from Salviae did not affect the entry and replication of HIV-1, indicating no virucidal activity [33], which is different from our observation of potent virucidal activity of the whole Salviae extract against a broad range of HIV-1 isolates. Therefore, these promising results validate the present study supporting further analysis of the potential active components (Figure 3).
.
Figure 3: Comparison of virucidal activities for HIV-1 primary isolate SF162 pseudovirions. Levels of inhibition were presented compared to that of a control well with medium alone. Concentrations of test samples are as follows: Salviae (0.2 μg/ml), Cinnamomi (0.2 μg/ml), Trichosanthin (0.2 μg/ml), Dextran Sulfate (1 μg/ml), Carrageenan (κ and λ) (0.5 μg/ml), AZT (3'-azido-3'-deoxythymidine; Zidovudine, 0.032
μmole /ml, an anti-HIV-1 drug).
View Figure 3
In summary, the results in the present study suggest that Salviae and Cinnamomi have potent anti-HIV-1 virucidal activities. Future studies are needed to identify the active components of the Salviae and Cinnamomi herbal extract, and to better understand the possible action mechanisms of purified components.
Acknowledgment
We thank David C. Montefiori (Duke University Medical Center, Durham, North Carolina) for helping in anti-HIV assays of herbal samples. This work was partially supported by NIH/NIAID grants AI093772(S.M.K.), AI105170 (S.M.K), AI119366 (S.M.K.), the Korean Society of Ginseng (S.M.K), a research grant of the Jeju National University (Y.J.L.), the National Research Foundation of Korea (NRF) grant NRF-2014R1A2A2A01004899 (F.S.Q.), and Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry and Fisheries (IPET) through Agri-Bio industry Technology Development Program, funded by Ministry of Agriculture, Food and Rural Affairs (MAFRA) (315030-03-1-HD020) (F.S.Q.). The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the funding agencies.
References
-
Winslow LC, Kroll DJ (1998) Herbs as medicines. Arch Intern Med 158: 2192-2199.
-
Sung H, Kang SM, Lee MS, Kim TG, Cho YK (2005) Korean red ginseng slows depletion of CD4 T cells in human immunodeficiency virus type 1-infected patients. Clin Diagn Lab Immunol 12: 497-501.
-
Jeong JC, Hwang WM, Yoon CH, Kim YK (2001) Salviae radix extract prevents cisplatin-induced acute renal failure in rabbits. Nephron 88: 241-246.
-
Kim HM, Lee EH, Lee JH, Jung JA, Kim JJ (1999) Salviae radix root extract inhibits immunoglobulin E-mediated allergic reaction. Gen Pharmacol 32: 603-608.
-
Xie F, Li X, Sun K, Chu Y, Cao H, et al. (2001) An experimental study on drugs for improving blood circulation and removing blood stasis in treating mild chronic hepatic damage. J Tradit Chin Med 21: 225-231.
-
Yao XX, Cui DL, Sun YF, Li XT (1998) Clinical and experimental study of effect of Raondix Salviae Militiorrhiza and other blood-activating and stasis-eliminating Chinese herbs on hemodynamics of portal hypertension. World J Gastroenterol 4: 439-442.
-
Xu CJ, Liang YZ, Song YQ, Li JS (2001) Resolution of the essential constituents of Ramulus cinnamomi by an evolving chemometric approach. Fresenius J Anal Chem 371: 331-336.
-
Derdeyn CA, Decker JM, Sfakianos JN, Wu X, O'Brien WA, et al. (2000) Sensitivity of human immunodeficiency virus type 1 to the fusion inhibitor T-20 is modulated by coreceptor specificity defined by the V3 loop of gp120. J Virol 74: 8358-8367.
-
Wei X, Decker JM, Liu H, Zhang Z, Arani RB, et al. (2002) Emergence of resistant human immunodeficiency virus type 1 in patients receiving fusion inhibitor (T-20) monotherapy. Antimicrob Agents Chemother 46: 1896-1905.
-
Bures R, Morris L, Williamson C, Ramjee G, Deers M, et al. (2002) Regional clustering of shared neutralization determinants on primary isolates of clade C human immunodeficiency virus type 1 from South Africa. J Virol 76: 2233-2244.
-
Li M, Gao F, Mascola JR, Stamatatos L, Polonis VR, et al. (2005) Human immunodeficiency virus type 1 env clones from acute and early subtype B infections for standardized assessments of vaccine-elicited neutralizing antibodies. J Virol 79: 10108-10125.
-
Kothe DL, Nathan M, Pushko P, Weng Z, Moldoveanu Z, et al. (2005) Characterization and Immunogenicity of Virus-like Particles Produced by a Baculovirus-Expression System. AIDS Vaccine. Montreal, Quebec, Canada.
-
Kang SM, Compans RW (2003) Enhancement of mucosal immunization with virus-like particles of simian immunodeficiency virus. J Virol 77: 3615-3623.
-
Kang SM, Yao Q, Guo L, Compans RW (2003) Mucosal immunization with virus-like particles of simian immunodeficiency virus conjugated with cholera toxin subunit B. J Virol 77: 9823-9830.
-
Byers VS, Baldwin PW (1991) Trichosanthin treatment of HIV disease. AIDS 5: 1150-1151.
-
Byers VS, Levin AS, Malvino A, Waites L, Robins RA, et al. (1994) A phase II study of effect of addition of trichosanthin to zidovudine in patients with HIV disease and failing antiretroviral agents. AIDS Res Hum Retroviruses 10: 413-420.
-
Byers VS, Levin AS, Waites LA, Starrett BA, Mayer RA, et al. (1990) A phase I/II study of trichosanthin treatment of HIV disease. AIDS 4: 1189-1196.
-
McGrath MS, Santulli S, Gaston I (1990) Effects of GLQ223 on HIV replication in human monocyte/macrophages chronically infected in vitro with HIV. AIDS Res Hum Retroviruses 6: 1039-1043.
-
Shaw PC, Lee KM, Wong KB (2005) Recent advances in trichosanthin, a ribosome-inactivating protein with multiple pharmacological properties. Toxicon 45: 683-689.
-
Shaw PC, Mulot S, Ma SK, Xu QF, Yao HB, et al. (1997) Structure/function relationship study of Tyr14 and Arg22 in trichosanthin, a ribosome-inactivating protein. Eur J Biochem 245: 423-427.
-
Wang JH, Nie HL, Tam SC, Huang H, Zheng YT (2002) Anti-HIV-1 property of trichosanthin correlates with its ribosome inactivating activity. FEBS Lett 531: 295-298.
-
Zheng YT, Zhang WF, Ben KL, Wang JH (1995) In vitro immunotoxicity and cytotoxicity of trichosanthin against human normal immunocytes and leukemia-lymphoma cells. Immunopharmacol Immunotoxicol 17: 69-79.
-
Hantson A, Fikkert V, Van Remoortel B, Pannecouque C, Cherepanov P, et al. (2005) Mutations in both env and gag genes are required for HIV-1 resistance to the polysulfonic dendrimer SPL2923, as corroborated by chimeric virus technology. Antivir Chem Chemother 16: 253-266.
-
Moulard M, Lortat-Jacob H, Mondor I, Roca G, Wyatt R, et al. (2000) Selective interactions of polyanions with basic surfaces on human immunodeficiency virus type 1 gp120. J Virol 74: 1948-1960.
-
Flexner C, Barditch-Crovo PA, Kornhauser DM, Farzadegan H, Nerhood LJ, et al. (1991) Pharmacokinetics, toxicity, and activity of intravenous dextran sulfate in human immunodeficiency virus infection. Antimicrob Agents Chemother 35: 2544-2550.
-
Kensinger RD, Catalone BJ, Krebs FC, Wigdahl B, Schengrund CL (2004) Novel polysulfated galactose-derivatized dendrimers as binding antagonists of human immunodeficiency virus type 1 infection. Antimicrob Agents Chemother 48: 1614-1623.
-
Spieler R (2002) Seaweed compound's anti-HIV efficacy will be tested in southern Africa. Lancet 359: 1675.
-
Perotti ME, Pirovano A, Phillips DM (2003) Carrageenan formulation prevents macrophage trafficking from vagina: implications for microbicide development. Biol Reprod 69: 933-939.
-
Schaeffer DJ, Krylov VS (2000) Anti-HIV activity of extracts and compounds from algae and cyanobacteria. Ecotoxicol Environ Saf 45: 208-227.
-
D'Cruz OJ, Uckun FM (2004) Clinical development of microbicides for the prevention of HIV infection. Curr Pharm Des 10: 315-336.
-
Tabba HD, Chang RS, Smith KM (1989) Isolation, purification, and partial characterization of prunellin, an anti-HIV component from aqueous extracts of Prunella vulgaris. Antiviral Res 11: 263-273.
-
Chang RS, Yeung HW (1988) Inhibition of growth of human immunodeficiency virus in vitro by crude extracts of Chinese medicinal herbs. Antiviral Res 9: 163-175.
-
Abd-Elazem IS, Chen HS, Bates RB, Huang RC (2002) Isolation of two highly potent and non-toxic inhibitors of human immunodeficiency virus type 1 (HIV-1) integrase from Salvia miltiorrhiza. Antiviral Res 55: 91-106.





