Journal of Clinical Gastroenterology and Treatment
Impairment of Salivary Protection in Esophageal Defense: Its Implications in Reflux Esophagitis
Harathi Yandrapu1, Marek Marcinkiewicz2, Tomasz Zbroch3, Cezary Poplawski4, Irene Sarosiek1, Zbigniew Namiot3 and Jerzy Sarosiek1*
1Department of Internal Medicine, Molecular Medicine Research Laboratory, Paul L. Foster School of Medicine, Texas Tech University Health Sciences Center, El Paso, TX, USA
2Department of Medicine, Colombia County Health System, Dayton, WA, USA
3Medical University of Bialystok, Bialystok, Poland
4Nicolaus Copernicus University of Torun, Torun, Poland
*Corresponding author:
Jerzy Sarosiek, MD, PhD, AGAF, FACG, Professor of Medicine, Vice Chair for Research, Department of Internal Medicine, Internal Medicine Research Committee Chairman, Texas Tech University Health Sciences Center, Director of Molecular Medicine Research Laboratory, 4800 Alberta Avenue, El Paso, TX, 79905, USA, Tel: (915) 215-5255, E-mail: jerzy.sarosiek@ttuhsc.edu
J Clin Gastroenterol Treat, JCGT-1-006, (Volume 1, Issue 1), Original Article; ISSN: 2469-584X
Received: August 26, 2015 | Accepted: September 25, 2015 | Published: September 28, 2015
Citation: Yandrapu H, Marcinkiewicz M, Zbroch T, Poplawski C, Sarosiek I, et al. (2015) Impairment of Salivary Protection in Esophageal Defense: Its Implications in Reflux Esophagitis. J Clin Gastroenterol Treat 1:006. 10.23937/2469-584X/1510006
Copyright: © 2015 Yandrapu H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: It has been previously demonstrated that patients with reflux esophagitis (RE) exhibit a significant impairment of salivary epidermal growth factor (EGF) secretion. However, the secretion of other salivary protective components contributing to mucosal defense in RE patients remains to be established.
Aim: To investigate the secretion of salivary volume, pH, bicarbonate, non-bicarbonate, glycoconjugate, protein, transforming growth factor α (TGFα) and prostaglandin E2 (PGE2) during basal conditions, mastication, mechanical and chemical stimulations evoked by esophago-salivary reflex in patients with RE and to compare with the corresponding values in controls (CTRL).
Methods: Salivary secretion was collected during basal conditions, mastication, intraesophageal mechanical (tubing, balloon) and chemical (initial saline, acid, acid/pepsin and final saline) stimulations respectively mimicking the natural gastroesophageal reflux in RE and control groups.
Results: Salivary pH in RE patients was significantly lower than controls during mechanical stimulation with tubing (p < 0.05) and chemical stimulation with initial saline (p < 0.05), acid (p < 0.05), acid/pepsin (p < 0.01) and final saline (p < 0.01). Salivary bicarbonate secretion in RE group was significantly lower than in CTRL group during mechanical stimulation with tubing (p < 0.05). Salivary glycoconjugate secretion in RE group was significantly lower than in the control group (p < 0.05), not only at basal level but also during mastication, intraesophageal mechanical and chemical stimulations. Salivary protein output was significantly lower in RE patients during chemical stimulation with initial saline (p < 0.05) and acid (p < 0.01). There were no significant differences in the secretions of salivary volume, non-bicarbonate buffers, TGFα and PGE2 between the two groups.
Conclusions: Patients with RE demonstrated a compromised salivary pH along with significant impairment of salivary secretions of bicarbonate, glycoconjugate and protein. This impairment could predispose to the development of accelerated mucosal injury.
Keywords
Reflux esophagitis, Salivary protection, Salivary pH, Salivary bicarbonate, Glycoconjugate, Salivary protein
Abbreviations
RE: Reflux esophagitis, CTRL: Control group, GERD: Gastroesophageal reflux disease, NERD: Non-erosive reflux disease, Basal: Basal conditions, Mastication: Mastication, Tubing: Mechanical stimulation with tubing, Balloon: Mechanical stimulation with balloon, NaCl: Normal saline, Initial NaCl: Initial saline perfusion, HCl: Hydrochloric acid, HCl/pepsin: Hydrochloric acid and pepsin, Final NaCl: Final saline perfusion, EGF - Epidermal growth factor, TGFα: Transforming growth factor α, PGE2: Prostaglandin E2
Introduction
Gastroesophageal Reflux Disease (GERD) is a highly prevalent disease in the western world and affects approximately up to 20% of adults and nearly 25 million experience heartburn on a daily basis [1,2]. Reflux esophagitis (RE) is caused by the excessive exposure of esophageal mucosa to aggressive factors like acid and pepsin leading to inflammation [3-5]. Gastroesophageal reflux is a common phenomenon which would not normally cause esophageal mucosal damage because of presence of defense mechanisms [5]. Impairment of salivary protective mechanisms predisposes the esophageal epithelium to accelerated mucosal injury when exposed to acid and pepsin.
Organic and inorganic protective components of salivary secretion within the esophageal pre-epithelial barrier play a pivotal role in the maintenance of integrity of the esophageal epithelium [1,6-10]. Salivary secretion of water and inorganic components (electrolytes, bicarbonate and non-bicarbonate buffers) is mediated predominantly by parasympathetic pathways whereas secretion of organic components (glycoconjugate, proteins, growth factors and prostaglandins) by sympathetic pathways [1,6,7,10].
It has been previously demonstrated that patients with RE exhibit a significant impairment of salivary epidermal growth factor (EGF) secretion during intraesophageal mechanical and chemical stimulations when compared to controls [11-13]. However, the basal rate of salivary EGF secretion is similar in RE and control groups, which strongly implicates that esophago-salivary reflex pathway is impaired in response to the presence of acid and pepsin within the esophageal lumen [13]. However, the secretion of other salivary protective components of mucosal defense in patients with RE remain to be established.
We therefore have studied the secretion of salivary volume, pH, bicarbonate, non-bicarbonate, glycoconjugate, protein, transforming growth factor α (TGFα) and prostaglandin E2 (PGE2) in basal conditions, during mastication, intraesophageal mechanical and chemical stimulations evoked by esophago-salivary reflex in patients with RE and compared with the corresponding values in controls (CTRL).
Materials and Methods
Subjects
The study was approved by the Human Subject Committee and conducted on 33 Caucasian asymptomatic volunteers (15 females and 18 males, mean age of 39, range 26-56) and 20 Caucasian patients with RE (7 females and 13 males, mean age of 40, range 34-59) with a history of GERD (heartburn and acid regurgitation as predominant symptoms weekly or more for at least 5 years). Our assumption that the esophageal mucosa in our asymptomatic volunteers is truly healthy was based on the fact that they never complained of heartburn and acid regurgitation and therefore, potential 24-hour pH monitoring was unlikely to reveal any significant changes. Endoscopically RE patients had erosive reflux esophagitis of grade B and C as per Los Angeles classification [14-16]. Informed consent was obtained from all subjects. All subjects were not afflicted with any acute illness, did not receive any antisecretory medication at least 7 days before the salivary sample collection and never had any dysfunction of mastication.
Salivary sample collection
Subjects expectorated all saliva collected in their mouth every 10 seconds and were instructed not to swallow during the procedure. The salivary samples were sequentially collected on ice during the same time of the day for each subject as follows: (1) basal saliva during the first 10 minutes, (2) saliva produced during stimulation by parafilm chewing (mastication) during the following 5 minutes, (3) saliva produced by tubing following the placement of the intraesophageal catheter during two consecutive 1.5-minute intervals, (4) saliva produced following inflation of both intraesophageal balloons during two consecutive 1.5-minute intervals and (5) saliva produced during the esophageal perfusion with initial saline (initial NaCl), hydrochloric acid (HCl), HCl/pepsin (acid/pepsin) and final saline (final NaCl) consecutively, four samples of each totaling 16 consecutive 1.5-minute intervals.
Esophageal perfusion catheter
Esophageal perfusion was performed with a specially designed six-channel catheter manufactured by Wilson-Cook Company, Chapel Hill, NC, as described in detail by Sarosiek et al. [17]. The four larger diameter channels were used for infusion and aspiration of the perfusate, gastric juice and incidentally swallowed saliva, which is retained above the upper balloon. The two smaller diameter channels were used for inflation of the upper and lower balloons to compartmentalize the segment of the lower esophagus [6-9,12,17].
Perfusing solutions
Esophageal perfusion in all subjects was performed using fresh 10 ml solutions for each 1.5-minute interval: (1) NaCl (0.15 M) that corresponds to 0.9% saline. (2) HCl (0.01M; pH 2.1). This concentration and pH of HCl was chosen to closely resemble the content of gastroesophageal refluxate [21]. (3) HCl (0.01M; pH 2.1) with pepsin. Pepsin (0.5 mg/ml; Sigma Chemical Co.) was dissolved in the concentration, which corresponds to the average proteolytic activity of human gastric juice [1,11,18,19].
Esophageal perfusion procedure
Subjects were placed in the semi-recumbent position. The nasopharynx was anaesthetized with xylocaine gel, the esophageal catheter was inserted into the esophagus through the nares and the balloons of the catheter were gently insufflated to seal the esophageal lumen. This procedure allows the compartmentalization of 3.75 cm segment of the esophagus between the balloons [11,17,20-22]. During each perfusion period of 1.5-minute interval, the entire 10 ml solution of perfusate was circulated within the isolated segment of esophagus for a total duration of 24 minutes for each subject (16 consecutive 1.5-minute intervals).
Analysis of salivary secretion components
Salivary volume was assessed using a sialo meter (Proflow Incorporated, Amityville, NY) [1,8,12,13]. Salivary pH was monitored using the Expandable Ion Analyzer EA 940 (Orion Res., Boston, MA).
The salivary bicarbonate and non-bicarbonate buffers were analyzed by titration and back-titration methodology using TitraLab 90 (Radiometer America Inc., Chicago, IL) [23]. Secretions form a thin film on the mucosa and allows the evolution of carbon dioxide (CO2) formed from acid-base interactions. Therefore, the esophageal bicarbonate buffer value would be equilibrated with CO2 tension of the lumen [4,18,23,24]. This was the rationale for choosing titration to pH of 4.0 for assessment of esophageal bicarbonate in an open system (without covering with a layer of liquid paraffin oil) with continuous CO2-free bubbling. The bicarbonate concentration was calculated using the difference in the amount of acid initially required to titrate the sample from it's starting pH to pH 4.0 and the amount of base required to back-titrate the sample to its original pH after development of the CO2. The difference between the back-titration from pH 4.0 to its original starting value and the similar run of the buffer-free blank solution was used to calculate non-bicarbonate buffers [11,23,24]. In addition, this methodology was always validated by the titration of known concentrations of bicarbonate and non-bicarbonate in the standard solutions.
Salivary glycoconjugate (predominantly mucin) was measured using the periodic acid Schiff (PAS) methodology [11,21-23]. Salivary EGF was assessed by RIA using a commercially available kit (Amersham, Arlington Heights, IL) [11-13,17,23].
Salivary TGFα was recorded using a commercially available RIA kit based on highly specific sheep anti-human TGFα antibodies (Biomedical Technologies Inc., Stoughton, MA) [23,25]. The separation between bound and unbound TGFα was performed using donkey anti-sheep IgG and polyethylene glycol (PEG). Human recombinant TGFα was used for a standard curve. All samples were centrifuged at 4℃ and 3000 rpm for 20 min, which are the conditions required to spin down cellular debris, plasma membrane sheets and nuclei [1,25].
Salivary PGE2 was measured using a RIA kit (Amersham, Arlington Heights, MA) [25]. This RIA method is based on highly specific antibodies directed to oximated form of PGE2. Salivary protein was monitored by the Lowry methodology [11,21].
Data processing and statistical analysis
Data were measured as mean values of salivary collections at basal level, during parafilm chewing, following placement of tubing, following inflation of balloons and during the perfusion intervals. All results were expressed as means ± SEM. Statistical analysis by non-paired t-test was performed using Σ-Stat software (Jandel Scientific, San Rafael, CA).
Results
Salivary inorganic protective components (Table 1)
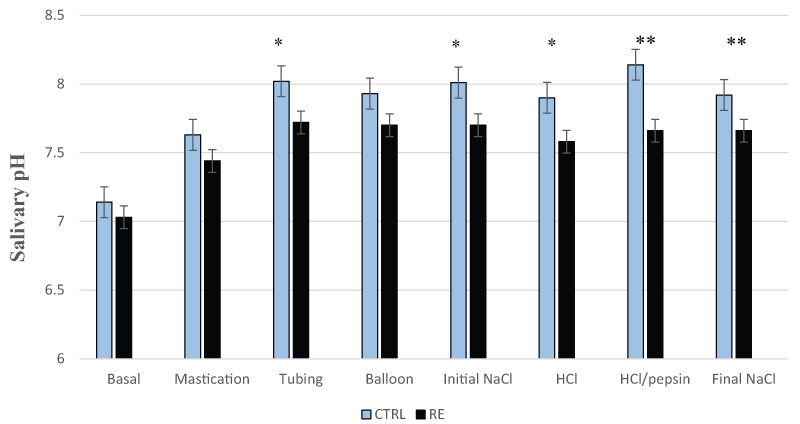
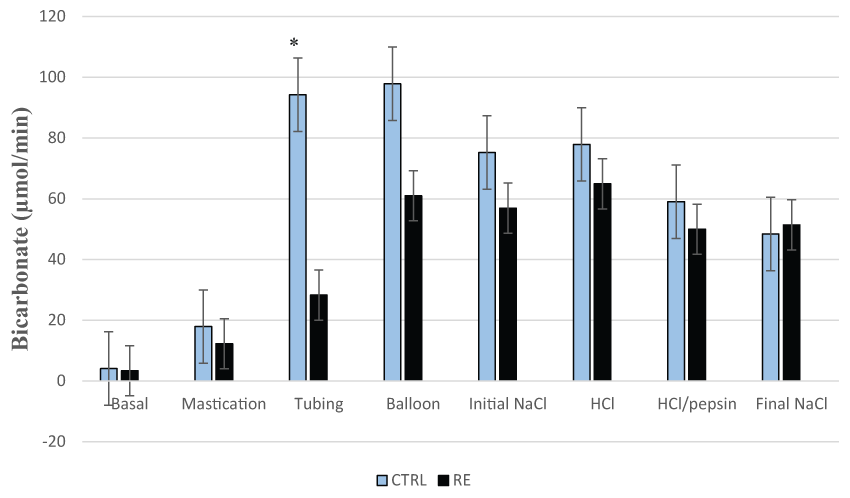
Salivary pH in patients with RE was significantly lower than controls during mechanical stimulation with tubing (7.72 ± 0.09 versus 8.02 ± 0.06, p < 0.05) and chemical stimulation with initial saline ±7.70 ± 0.08 versus 8.01 ± 0.08, p < 0.05), HCl (7.58 ± 0.11 versus 7.90 ± 0.09, p < 0.05), HCl/pepsin (7.66 ± 0.10 versus 8.14 ± 0.08, p < 0.01) and final saline (7.66 ± 0.09 versus 7.92 ± 0.18, p < 0.01), as shown in figure 1. Salivary bicarbonate output in RE group was also significantly lower than CTRL group during mechanical stimulation with tubing (28.3 ± 6.95 versus 94.2 ± 35.2 μmol/min, p < 0.05) as shown in figure 2. No significant differences were found in the salivary volume and non-bicarbonate buffers between the two groups.
.
Figure 1: Salivary pH in the control group (CTRL) and patients with reflux esophagitis (RE)
Salivary pH in patients with RE is significantly lower than CTRL during mechanical stimulation with tubing and chemical stimulation with initial saline (initial NaCl), HCl, HCl/pepsin and final saline (final NaCl).
* = p < 0.05; **= p < 0.01 are significant
View Figure 1
.
Figure 2: Salivary bicarbonate secretion in the control group (CTRL) and patients with reflux esophagitis (RE)
Salivary bicarbonate secretion in RE group is significantly lower than CTRL group during mechanical stimulation with tubing.
* = p < 0.05 is significant
View Figure 2
Salivary organic protective components (Table 2)
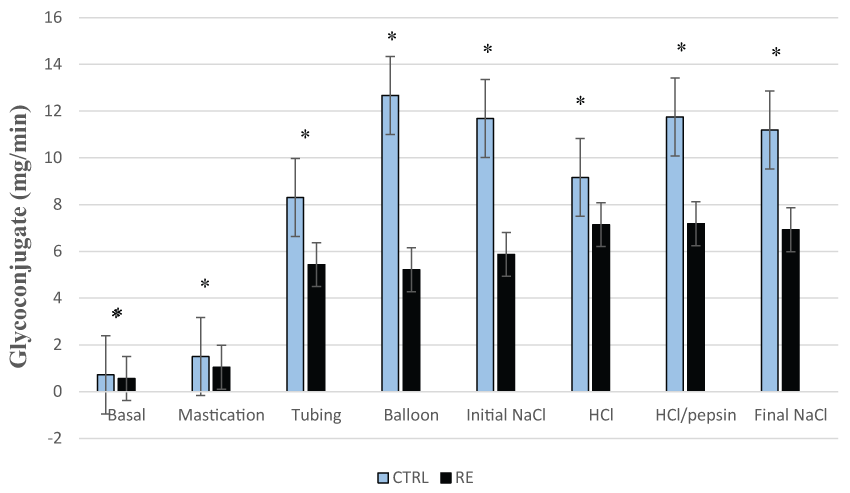
Salivary glycoconjugate secretion in patients with RE was significantly lower than controls in basal conditions (0.56 ± 0.09 versus 0.75 ± 0.08 mg/min, p < 0.05), during mastication (1.04 ± 0.16 versus 1.50 ± 0.20 mg/min, p < 0.05), mechanical stimulation with tubing (5.43 ± 1.27 versus 8.30 ± 0.99 mg/min, p < 0.05), balloons (5.21 ± 0.91 versus 12.67 ± 1.59 mg/min, p < 0.05), chemical stimulation with initial saline (5.87 ± 0.97 versus 11.68 ± 1.68 mg/min, p < 0.05), HCl (7.14 ± 1.03 versus 9.16 ± 1.32 mg/min, p < 0.05), HCl/pepsin (7.18 ± 1.19 versus 11.75 ± 1.70 mg/min, p < 0.05) and final saline (6.92 ± 0.97 versus 11.19 ± 1.65 mg/min, p < 0.05) as shown in figure 3. Thus, salivary glycoconjugate is the only protective component that is significantly lower in patients with RE not only during basal conditions and mastication, but also throughout the mechanical and chemical stimulations.
.
Figure 3: Salivary glycoconjugate secretion in the control group (CTRL) and patients with reflux esophagitis (RE)
Salivary glycoconjugate secretion is significantly lower in patients with RE throughout the basal conditions, mastication, mechanical and chemical stimulations.
* = p < 0.05 is significant
View Figure 3
![]()
Table 1: Salivary inorganic protective components in patients with reflux esophagitis (RE) and control group (CTRL) (Mean± SEM)
View Table 1
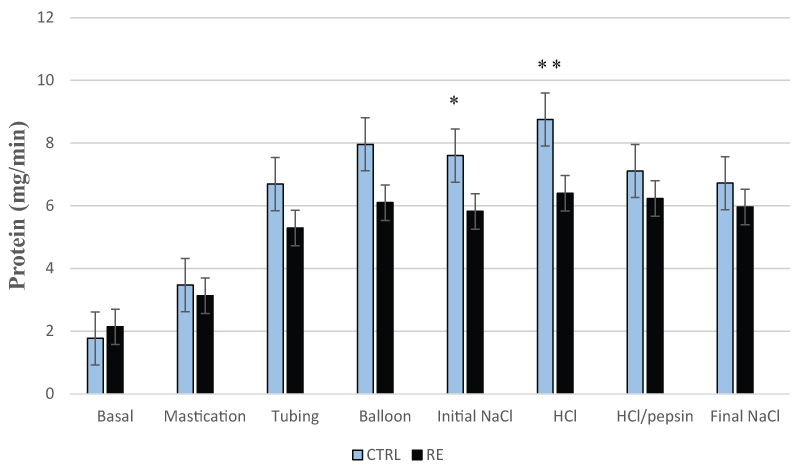
Salivary protein output was significantly lower in RE patients than controls during chemical stimulation with initial saline (5.82 ± 1.29 versus 7.60 ± 0.89 mg/min, p < 0.05) and HCl (6.40 ± 1.19 versus 8.75 ± 1.06 mg/min, p < 0.01) as shown in figure 4. The secretions of salivary TGFα and PGE2 were not significantly different in both groups throughout the entire process of salivary collection.
![]()
Table 2: Salivary organic protective components in patients with reflux esophagitis (RE) and control group (CTRL) (Mean ± SEM)
View Table 2
.
Figure 4: Salivary protein secretion in the control group (CTRL) and patients with reflux esophagitis (RE)
Salivary protein secretion is significantly lower in RE patients than controls during chemical stimulation with initial saline and HCl.
* = p < 0.05; **= p < 0.01 are significant
View Figure 4
Discussion
The integrity of the esophageal mucosa depends upon an equilibrium between aggressive factors (acid, pepsin and bile components) and defense mechanisms [1,4,11]. Chronic exposure to aggressive factors in the setting of failed defense mechanisms leads to inflammation of the esophagus with reflux symptoms such as heartburn and regurgitation, known as GERD potentially setting the stage for development of reflux esophagitis [5].
In some GERD patients, absence of inflammatory erosive abnormalities on endoscopy with the presence of reflux associated symptoms is defined as non-erosive reflux disease (NERD). It has been recently demonstrated by Yandrapu et al that patients with NERD elicit a significantly stronger salivary secretory response when compared to controls. It potentially provides enhanced salivary esophagoprotection mediating resistance to the development of endoscopic mucosal changes [1]. Yandrapu et al. also demonstrated a significant reduction of salivary protective component's secretion leading to mucosal injury in patients with Barrett's esophagus [11]. Therefore, salivary secretory components play a pivotal role in the restoration of the mucosal pH and integrity of the esophagus [1,6-10].
Our current study demonstrated a significant impairment of salivary protective components in patients with RE with respect to bicarbonate, glycoconjugate and protein along with compromised pH, mediated by esophago-salivary reflex. Salivary pH was more acidic in RE patients during intraesophageal mechanical stimulation with tubing and chemical stimulation with initial saline, HCl, HCl/pepsin and final saline when compared to controls. Salivary bicarbonate, the most effective component of salivary buffering capacity [4], was significantly low in RE patients during mechanical stimulation with tubing. Salivary protein output was low during chemical stimulation with initial saline and HCl.
The most interesting factor about the secretion of salivary glycoconjugate in our present study was its persistent and significant impairment in patients with RE not only during basal conditions and mastication but also throughout the mechanical and chemical stimulations. Glycoconjugate has a protective role by retarding hydrogen ion diffusion through hydrophobicity and interaction with phospholipids, and providing an architectural framework for the unstirred layer of the mucus bicarbonate pre-epithelial barrier [26,27]. No significant differences were found in the salivary volume, non-bicarbonate buffers, TGFα and PGE2 between the two groups in our present study. Furthermore, the rate of secretion of salivary EGF was significantly impaired in similar settings of mechanical and chemical stimulations originated in the squamous epithelium of the esophageal mucosa in patients with RE, as demonstrated by Rourk et al. [13].
Overall our present study indicates a significant impairment of salivary secretory function in patients with RE, mediated by esophago-salivary reflex evoked by intraesophageal mechanical and chemical stimulations mimicking the natural gastroesophageal reflux scenario. This can be a pathogenically important phenomenon facilitating the damage to the mucosa by reflux contents like acid and pepsin thereby contributing to its etiology. The potential impact of gender and age on the secretion of salivary protective factors in patients with RE requires further exploration.
Conclusions
In summary, a significant impairment of the secretion of salivary bicarbonate, glycoconjugate and protein along with compromised low salivary pH in patients with RE can diminish the protective potential leading to accelerated esophageal mucosal injury. Potential restoration of this impairment by the masticatory stimulation of all salivary protective components using sugarless chewing gum justifies further clinical exploration.
Conflict of Interest
All authors have no conflict of interest
Financial Disclosures
None
Human and Animal Rights and Informed Consent
With regard to the author's research cited in this paper, all procedures were followed in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975, as revised in 2000 and 2008.
References
-
Yandrapu H, Marcinkiewicz M, Poplawski C, Han K, Zbroch T, et al. (2015) Role of saliva in esophageal defense: implications in patients with nonerosive reflux disease. Am J Med Sci 349: 385-391.
-
Camilleri M, Dubois D, Coulie B, Jones M, Kahrilas PJ, et al. (2005) Prevalence and socioeconomic impact of upper gastrointestinal disorders in the United States: results of the US Upper Gastrointestinal Study. Clin Gastroenterol Hepatol 3: 543-552.
-
Mattox HE 3rd, Richter JE (1990) Prolonged ambulatory esophageal pH monitoring in the evaluation of gastroesophageal reflux disease. Am J Med 89: 345-356.
-
Skoczylas T, Yandrapu H, Poplawski C, Asadi M, Wallner G, et al. (2014) Salivary bicarbonate as a major factor in the prevention of upper esophageal mucosal injury in gastroesophageal reflux disease. Dig Dis Sci 59: 2411-2416.
-
Wang RH (2015) From reflux esophagitis to Barrett's esophagus and esophageal adenocarcinoma. World J Gastroenterol 21: 5210-5219.
-
Namiot Z, Rourk RM, Piascik R, Hetzel DP, Sarosiek J, et al. (1994) Interrelationship between esophageal challenge with mechanical and chemical stimuli and salivary protective mechanisms. Am J Gastroenterol 89: 581-587.
-
Sarosiek J, McCallum RW (1995) What role do salivary inorganic components play in health and disease of the esophageal mucosa? Digestion 56 Suppl 1: 24-31.
-
Sarosiek J, Rourk RM, Piascik R, Namiot Z, Hetzel DP, et al. (1994) The effect of esophageal mechanical and chemical stimuli on salivary mucin secretion in healthy individuals. Am J Med Sci 308: 23-31.
-
Sarosiek J, Scheurich CJ, Marcinkiewicz M, McCallum RW (1996) Enhancement of salivary esophagoprotection: rationale for a physiological approach to gastroesophageal reflux disease. Gastroenterology 110: 675-681.
-
Sarosiek J, McCallum RW (1995) Do salivary organic components play a protective role in health and disease of the esophageal mucosa? Digestion 56: 32-37.
-
Yandrapu H, Marcinkiewicz M, Poplawski C, Namiot Z, Zbroch T, et al. (2014) A distinct salivary secretory response mediated by the esophago-salivary reflex in patients with Barrett's esophagus: its potential pathogenetic implications. Adv Med Sci 59: 281-287.
-
Li L, Yu Z, Piascik R, Hetzel DP, Rourk RM, et al. (1993) Effect of esophageal intraluminal mechanical and chemical stressors on salivary epidermal growth factor in humans. Am J Gastroenterol 88: 1749-1755.
-
Rourk RM, Namiot Z, Sarosiek J, Yu Z, McCallum RW (1994) Impairment of salivary epidermal growth factor secretory response to esophageal mechanical and chemical stimulation in patients with reflux esophagitis. Am J Gastroenterol 89: 237-244.
-
Lundell LR, Dent J, Bennett JR, Blum AL, Armstrong D, et al. (1999) Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut 45: 172-180.
-
Genta RM, Spechler SJ, Kielhorn AF (2011) The Los Angeles and Savary-Miller systems for grading esophagitis: utilization and correlation with histology. Dis Esophagus 24: 10-17.
-
Katz PO, Gerson LB, Vela MF (2013) Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterol 108: 308-328.
-
Sarosiek J, Hetzel DP, Yu Z, Piascik R, Li L, et al. (1993) Evidence on secretion of epidermal growth factor by the esophageal mucosa in humans. Am J Gastroenterol 88: 1081-1087.
-
Hirschowitz BI (1991) A critical analysis, with appropriate controls, of gastric acid and pepsin secretion in clinical esophagitis. Gastroenterology 101: 1149-1158.
-
Walker V, Taylor WH (1980) Pepsin 1 secretion in chronic peptic ulceration. Gut 21: 766-771.
-
Sarosiek J, Yu Z, Namiot Z, Rourk RM, Hetzel DP, et al. (1994) Impact of acid and pepsin on human esophageal prostaglandins. Am J Gastroenterol 89: 588-594.
-
Namiot Z, Sarosiek J, Rourk RM, Hetzel DP, McCallum RW (1994) Human esophageal secretion: mucosal response to luminal acid and pepsin. Gastroenterology 106: 973-981.
-
Namiot Z, Sarosiek J, Marcinkiewicz M, Edmunds MC, McCallum RW (1994) Declined human esophageal mucin secretion in patients with severe reflux esophagitis. Dig Dis Sci 39: 2523-2529.
-
Marcinkiewicz M, Han K, Zbroch T, Poplawski C, Gramley W, et al. (2000) The potential role of the esophageal pre-epithelial barrier components in the maintenance of integrity of the esophageal mucosa in patients with endoscopically negative gastroesophageal reflux disease. Am J Gastroenterol 95:1652-1660.
-
Izutsu KT (1981) Theory and Measurement of the buffer value of bicarbonate in saliva. J Theor Biol 90: 397-403.
-
Marcinkiewicz M, Namiot Z, Edmunds MC, McCallum RW, Sarosiek J (1996) Detrimental impact of acid and pepsin on the rate of luminal release of transforming growth factor alpha. Its potential pathogenetic role in the development of reflux esophagitis. J Clin Gastroenterol 23: 261-268.
-
Slomiany BL, Sarosiek J, Slomiany A (1987) Gastric mucus and the mucosal barrier. Dig Dis 5: 125-145.
-
Sarosiek J, Slomiany A, Slomiany BL (1983) Retardation of hydrogen ion diffusion by gastric mucus constituents: effect of proteolysis. Biochem Biophys Res Commun 115: 1053-1060.





