Journal of Clinical Gastroenterology and Treatment
Validation of Steroid-Free Immunosuppression Regimen after Liver Transplantation
Ken Fukumitsu1, Ahmed Hammad1,2, Toshimi Kaido1*, Kohei Ogawa1, Yasuhiro Fujimoto1, Akira Mori1, Hideaki Okajima1 and Shinji Uemoto1
1Division of Hepato-Biliary-Pancreatic and Transplant Surgery, Department of Surgery, Graduate School of Medicine, Kyoto University, Japan
2Department of Surgery, Mansoura University, Egypt
*Corresponding author:
Toshimi Kaido, Division of Hepato-Biliary-Pancreatic and Transplant Surgery, Department of Surgery, Graduate School of Medicine, Kyoto University, 54 Kawahara-cho, Shogoin, Sakyo-ku, Kyoto 606-8507, Japan, Tel: +81-75-751-4323, Fax: +81-75-751-4348, E-mail: kaido@kuhp.kyoto-u.ac.jp
J Clin Gastroenterol Treat, JCGT-1-008, (Volume 1, Issue 1), Original Article; ISSN: 2469-584X
Received: October 15, 2015 | Accepted: November 25, 2015 | Published: November 27, 2015
Citation: Fukumitsu K, Hammad A, Kaido T, Ogawa K, Fujimoto Y, et al. (2015) Validation of Steroid-Free Immunosuppression Regimen after Liver Transplantation. J Clin Gastroenterol Treat 1:008. 10.23937/2469-584X/1510008
Copyright: © 2015 Fukumitsu K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Various regimens are used for immunosuppression for liver transplantation (LT). Even though steroid is one of the key drugs, it has many adverse effects, especially infection. A steroid-free immunosuppression protocol has been recently adopted at our institute. In this report, we evaluate usability of such protocol.
Methods: The study comprised 141 patients who underwent LT from January 2009 until November 2013 excluding the cases with ABO-incompatible transplantation or with steroid administration before LT. After February 2011, steroid has been replaced with mycophenolate mofetil in the immunosuppression protocol. We analyzed the rate of bacteremia, acute cellular rejection within 3 months after LT and patients' overall survival between the earlier steroid-containing protocol and latter steroid-free protocol.
Result: The incidence of post-transplant bacteremia tended to decrease, but insignificantly (P = 0.086), while rejection tended to occur more frequently, but insignificantly (P = 0.367) after LT under the steroid-free protocol, however, most of rejection cases were controlled by steroid pulse treatment. There was no significant difference in overall survival between both protocols (P = 0.281).
Conclusion: Steroid-free immunosuppression could be of value in LT in decreasing risk of bacteremia without the complication of rejection after LT.
Keywords
Steroid-free immunosuppression, Liver transplantation, Outcomes, Bacteremia, Overall survival
Introduction
Liver transplantation (LT) is performed worldwide with immunosuppression being an indispensable component of the perioperative management. Calcineurin inhibitors and steroids have been the most popular and widely used drugs for a long time. However, steroids hold long-term adverse effects, as increased susceptibility to infection, hyperglycemia, hyperlipidemia, obesity, hypertension and osteopenia [1]. Furthermore, steroids have been implicated in hepatitis C (HCV) recurrence after LT and graft failure [2-4]. The immunosuppression regimen at our center comprised originally tacrolimus and steroids. However, as of February 2011, a steroid-free protocol using tacrolimus and mycophenolate mofetil (MMF) has been employed instead. In the present study, we evaluate this new steroid-free protocol against the old steroid-containing one with respect to the incidence of bacteremia, acute cellular rejection and long-term patient survival.
Patients and Methods
Patients
A total of 141 patients among those who underwent LT at Kyoto University hospital between January 2009 and November 2013 were analyzed in this study. ABO-incompatible recipients and those who received steroid-administration before transplantation were excluded. One hundred twenty eight patients received living donor LT (LDLT) while the other 13 patients underwent deceased donor LT (DDLT). Sixty-three patients underwent LT between January 2009 and January 2011 (former period) and were thus administered the conventional steroid-containing immunosuppression protocol of tacrolimus and steroid. From February 2011 till November 2013 (latter period), 76 patients underwent LT and received the steroid-free immunosuppression protocol of tacrolimus and MMF. There were 2 patients who were given steroid after the change to the steroid-free protocol. The reason was that one patient was a 17 years old female and she received the child's protocol which contains steroid. The other patient developed acute respiratory distress syndrome (ARDS) right after transplantation and she was administered steroid as a part of ARDS treatment. Patient characteristics are described in table 1. There were no significant differences between both groups in age, gender, graft to recipient body weight ratio (GRWR), Child-Pugh classification or model for end-stage liver diseases (MELD) score. The study was approved by the Ethics Committee of Kyoto University and conducted in accordance with the Declaration of Helsinki of 1996.
![]()
Table 1: Patient characteristics.
GRWR: Graft-to-Recipient Weight Ratio, MELD score: Model for End-stage Liver Disease score, Lt: Left lobe graft, Rt: Right lobe graft, Whole: Whole liver graft, Post: Posterior segment graft, PBC: Primary biliary cirrhosis, PSC: Primary Sclerosing Cholangitis, Steroid (+): Steroid-containing protocol, Steroid (-): Steroid-free protocol
View Table 1
Immunosuppression regimens used in this study were described elsewhere in details [5]. Briefly, tacrolimus was given within the first 12 hours after LT, adjusted to a trough level 10 ng/ml which was decreased gradually to 5-8 ng/ml according to each patient's condition. All patients received a bolus injection of 10 mg/kg of steroid immediately after reflow during the surgery. In the steroid-administered group, we then routinely administered 1, 0.5 and 0.3 mg/kg/day of intravenous steroid from post-operative days (POD) 1-3, 4-6 and 7, respectively. Thereafter we administered 0.3 mg/kg/day p.o. of steroid until POD 28 and 0.1 mg/kg/day until POD 90. In the steroid-free group, MMF was given at a starting dose of 10-15 mg/kg/day, and then was gradually increased to 30 mg/kg/day depending on each patient's condition. Tacrolimus, prednisolone and MMF were given either by oral intake or via a naso-gastric tube.
Bacteremia was used in this study as an index of infection. We compared the rate of positive blood culture (other than common skin contaminants) within three months after liver transplantation between patients in the former period (before January 31, 2011) and in the latter period (after February 1, 2011). Blood culture samples were collected within a few days after transplantation from all patients, and then, samples were collected after that whenever high fever or any kind of symptoms suggesting infections occur. Bacteremia caused by common skin contaminants was considered significant only when the organism was isolated from 2 individual blood cultures and this finding was accompanied by clinical signs of infection. Broad-spectrum antimicrobials are administered when we suspect bacteremia due to high fever and increase in white blood cell count. When the clinical symptoms or laboratory data indicating infection such as C-reactive protein or white blood cell counts improved, we change antibiotics into narrow spectrum one according to the antimicrobial susceptibility test or stop antibiotics treatment with reference to the infection control team.
Rejection was diagnosed by liver biopsy. The acute cellular rejection (ACR) grading was assessed as mild, moderate and severer according to the Banff criteria [6]. Two cases were diagnosed as a humoral rejection. The incidence and severity of acute cellular rejection were compared between patients in the former period and in the latter period.
Steroid pulse treatment was given when a patient was diagnosed as ACR by histological evidence or by clinical judgment. In case of moderate to severe rejection, 10 mg/kg/day methyl prednisolone was administered 3 days then was tapered gradually. In case of mild to moderate rejection, 20 mg/kg//day hydrocortisone was administered then was tapered gradually thereafter. Some patients who were diagnosed as having mild rejection by histology received only oral prednisolone.
Overall survival was also compared between patients in the former period and in the latter period.
Statistical analysis
Data were summarized as mean ± standard deviation (SD) for continuous variables. Categorical variables were compared using the χ2 test or Fisher's exact test where appropriate. Survival rate was calculated using Kaplan-Meier methods with differences evaluated using log-rank or Mantel-Cox test. Two tailed P < 0.05 was considered significant. Any variable identified as significant (P < 0.05) in univariate analysis was considered a candidate for multivariate analysis using multiple logistic regression models. All statistical data were generated using JMP 11 (SAS Institute, Cary, NC) and Prism 6.02 (GraphPad Software, Inc., La Jolla, CA, USA).
Results
Bacteremia
The rate of positive blood culture was 47.7% (31 cases out of 65 patients) in the steroid-given group and 32.9% (25 cases out of 76 patients) in the steroid-free group. These proportions were also not significantly different (P = 0.086), although the rate of bacteremia had a tendency to decrease in the steroid-free group. Regarding to species, the percentage of Pseudomonas aeruginosa was significantly lower in the steroid-free group compared with the steroid-given group (P = 0.028). Methicillin-resistant Staphylococcus aureus was less detected in the steroid-free group than the steroid-given group (P = 0.123). Moreover, the incidence of death due to bacteremia decreased in the steroid-free group (11.9%) compared with the steroid-given group (20.3%) (P = 0.181). Moreover, in case where patients diagnosed with ACR who received steroid pulse treatment were categorized as steroid-positive, the rate of bacteremia in the steroid-free group (27.0%) was lower than that in the steroid-given group (44.0%) (P = 0.061).
Rejection
There were 27.7% (18 cases out of 65 patients) under the steroid-containing protocol and 35.5% (27 cases out of 76 patients) under the steroid-free protocol who were diagnosed as rejection, which showed no significant difference (P = 0.367) (Table 2). However, rejection tended to be more frequent in the steroid-free protocol.
![]()
Table 2: Comparisons of the incidence of post-transplant rejection and severity of acute cellular rejection between the steroid-free and steroid-containing protocols.
View Table 2
Furthermore, the histological severity of rejection was additionally analyzed within the patients who were histologically diagnosed as rejection, 14 cases were ACR-indeterminate to mild and 4 cases were ACR-moderate to severe under the steroid containing protocol, and 15 cases were ACR-indeterminate to mild, 10 cases were ACR-moderate to severe and 2 cases of humoral rejection under the steroid-free protocol. The two groups also revealed no significant difference (P = 0.325), however, rejection tended to be severer in steroid-free protocol than under the steroid-containing protocol (Table 2).
![]()
Table 3: Univariate analysis of factors affecting bacteremia.
View Table 3
Five factors including age, gender, steroid administration, GRWR, Child-Pugh score (A, B vs. C), MELD score (< 19 vs. > 20) were analyzed as risk factors for bacteremia or rejection. As for bacteremia, steroid administration and MELD score were of prognostic significance at P < 0.10 (Table 3), then only MELD score > 20 was an independent risk factor for bacteremia (P = 0.0032) (Table 4). None of the analyzed 5 factors was a significant risk factor of rejection (Table 5).
![]()
Table 4: Multivariate analysis of factors affecting bacteremia.
View Table 4
![]()
Table 5: Univariate analysis of factors affecting rejection.
View Table 5
Overall survival
Overall survival in the former period did not differ significantly from that in the latter period (P = 0.281) (Figure 1).
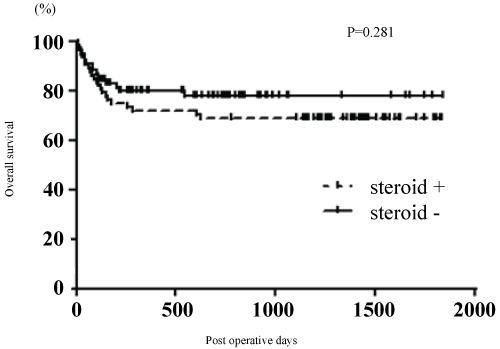
.
Figure 1: Overall survival has no significant difference between steroid (+) and (-), but a better survival positive tended to occur in the steroid-free protocol (P = 0.281). Steroid (+), steroid-containing protocol; steroid (-), steroid-free protocol.
View Figure 1
Discussion
Steroid has been used popularly as a part of immunosuppressive therapy despite of its long-term adverse effects. Recently, several clinical trials to address the feasibility of steroid avoidance have been performed. Some studies reported steroid early-withdrawal or minimization regimens to show no significant difference in graft failure, however, with a better control of adverse effects, as hyperglycemia [7-10]. Other studies used steroid administration via vein injection during surgery followed by steroid-free protocols or completely steroid-free protocols [11-14]. Recently, meta-analysis reported that steroid-free protocol was of lower risk of hyperlipidemia and cytomegalovirus infection than in the steroid-containing protocols [3,15]. Furthermore, Cuervas-Mons et al. found the same steroid-free regimen with tacrolimus and MMF to be associated with a trend toward better preservation of kidney function and reduction of cardiovascular risk score [16]. To the best of our knowledge, this is the first report to show that the incidence of post-transplant bacteremia tends to insignificantly decrease while rejection tended to be more frequent after LT under the steroid-free protocol, however most of rejection cases were controlled by steroid pulse treatment.
In the present study, the rate of bacteremia is high, especially in the steroid-given group. As for possible reason, our study includes severe patients with a little high MELD score and more than 70% of the patients had Child-Pugh class C cirrhosis. Moreover, our measures against postoperative infection including hand hygiene and contact precautions might be insufficient. In fact, this is the reason why we changed our immunosuppression protocol after February 2011 to try to reduce the rate of bacteremia in some way.
Bacteremia is considered the major cause of graft morbidity and in-hospital death [17], and infection control has a pivotal role in improving early outcomes of LT [18]. Even though there was no significant difference observed between both groups, the incidence of bacteremia tended to decrease in the steroid-free group in this study.
All cases of rejection under the steroid-free protocol received steroid pulse treatment, which often leads to remission. Moreover, since the early ACR does not affect the morbidity and graft survival, ACR is recently thought to be no longer a critical issue, and complete prevention of ACR early after LT is no longer needed [14,19].
Only high pre-transplant MELD score was as significant risk factor for post-transplant bacteremia. Previously we reported that MELD score was not associated to occurrence of bacteremia after LT [17]. The discrepancy of these results might be attributed to the fact that ABO-incompatible transplantation and patients who were given steroids before transplantation were excluded from the present study.
Several limitations should be born in mind. First, this was a retrospective single center study. Bacteremia tended to decrease and ACR tended to increase with steroid-free protocol, however, insignificantly. One of the reasons could be that the sample volume was not enough for analysis. As for diagnosis of the rejection, some cases were diagnosed without histology by a subjective personal judge of an attending doctor, and steroid pulse was administered accordingly. A further study with a consensus for rejection diagnosis is therefore necessary.
In conclusion, steroid-free regimen had a trend of decreased risk of post-transplant bacteremia, increased risk of rejection and no difference in overall survival from the steroid-containing regimen. Thus, steroid-free immunosuppression could be of value in LT. Further prospective randomized controlled trials are warranted to precisely evaluate the steroid-free protocol.
Conflict of Interest
The authors who have taken part in this study declare that they do not have anything to disclose regarding funding or conflict of interest with respect to this manuscript.
References
-
Ramirez CB, Doria C, Frank AM, Armenti ST, Marino IR (2013) Completely steroid-free immunosuppression in liver transplantation: a randomized study. Clin Transplant 27: 463-471.
-
Turner AP, Knechtle SJ (2013) Induction immunosuppression in liver transplantation: a review. Transpl Int 26: 673-683.
-
Segev DL, Sozio SM, Shin EJ, Nazarian SM, Nathan H, et al. (2008) Steroid avoidance in liver transplantation: meta-analysis and meta-regression of randomized trials. Liver Transpl 14: 512-525.
-
Teixeira R, Pastacaldi S, Papatheodoridis GV, Burroughs AK (2000) Recurrent hepatitis C after liver transplantation. J Med Virol 61: 443-454.
-
Takada Y, Kaido T, Asonuma K, Sakurai H, Kubo S, et al. (2013) Randomized, multicenter trial comparing tacrolimus plus mycophenolate mofetil to tacrolimus plus steroids in hepatitis C virus-positive recipients of living donor liver transplantation. Liver Transpl 19: 896-906.
-
(1997) Banff schema for grading liver allograft rejection: an international consensus document. Hepatology 25: 658-663.
-
Pirenne J, Aerts R, Koshiba T, Van Gelder F, Roskams T, et al. (2003) Steroid-free immunosuppression during and after liver transplantation- a 3-yr follow-up report. Clin Transplant 17: 177-182.
-
Everson GT, Trouillot T, Wachs M, Bak T, Steinberg T, et al. (1999) Early steroid withdrawal in liver transplantation is safe and beneficial. Liver Transpl Surg 5: S48-57.
-
Greig P, Lilly L, Scudamore C, Erb S, Yoshida E, et al. (2003) Early steroid withdrawal after liver transplantation: the Canadian tacrolimus versus microemulsion cyclosporin A trial: 1-year follow-up. Liver Transpl 9: 587-595.
-
Stegall MD, Wachs ME, Everson G, Steinberg T, Bilir B, et al. (1997) Prednisone withdrawal 14 days after liver transplantation with mycophenolate: a prospective trial of cyclosporine and tacrolimus. Transplantation 64: 1755-1760.
-
Eason JD, Loss GE, Blazek J, Nair S, Mason AL (2001) Steroid-free liver transplantation using rabbit antithymocyte globulin induction: results of a prospective randomized trial. Liver Transpl 7: 693-697.
-
Tisone G, Angelico M, Palmieri G, Pisani F, Anselmo A, et al. (1999) A pilot study on the safety and effectiveness of immunosuppression without prednisone after liver transplantation. Transplantation 67: 1308-1313.
-
Pelletier SJ, Vanderwall K, Debroy MA, Englesbe MJ, Sung RS, et al. (2005) Preliminary analysis of early outcomes of a prospective, randomized trial of complete steroid avoidance in liver transplantation. Transplant Proc 37: 1214-1216.
-
Reggiani P, Arru M, Regazzi M, Gatti S, Molinaro MD, et al. (2005) A "steroid-free" tacrolimus and low-dose mycophenolate mofetil primary immunosuppression does not prevent early acute rejection after liver transplantation. Transplant Proc 37: 1697-1699.
-
Sgourakis G, Radtke A, Fouzas I, Mylona S, Goumas K, et al. (2009) Corticosteroid-free immunosuppression in liver transplantation: a meta-analysis and meta-regression of outcomes. Transpl Int 22: 892-905.
-
Cuervas-Mons V, Herrero JI, Gomez MA, Gonzalez-Pinto I, Serrano T, et al. (2015) Impact of tacrolimus and mycophenolate mofetil regimen vs. a conventional therapy with steroids on cardiovascular risk in liver transplant patients. Clin Transplant 29: 667-667.
-
Kaido T, Mori A, Ogura Y, Ogawa K, Hata K, et al. (2012) Pre- and perioperative factors affecting infection after living donor liver transplantation. Nutrition 28: 1104-1108.
-
Iida T, Kaido T, Yagi S, Yoshizawa A, Hata K, et al. (2010) Posttransplant bacteremia in adult living donor liver transplant recipients. Liver Transpl 16: 1379-1385.
-
Rodriguez-Peralvarez M, Manousou P, Lerut J, De la Mata M, Burroughs AK (2014) How much immunosuppression is needed after liver transplantation? Clin Transplant 28: 6-7.





