Journal of Clinical Gastroenterology and Treatment
Management of Hemosuccus Pancreaticus after Surgical Drainage of a Pancreatic Pseudocyst - A Rare Cause of Upper Gastrointestinal Bleeding
Petko I Karagyozov1, Bilyana H Teneva1*, Ivan P Tishkov1, Zhenya N Georgieva1 and Marina Vloka2
1Department of Interventional Gastroenterology, Tokuda Hospital Sofia, Bulgaria
2Department of Medical Imaging, Tokuda Hospital Sofia, Bulgaria
*Corresponding author:
Dr. Bilyana H. Teneva, Department of Interventional Gastroenterology, Tokuda Hospital Sofia, 51B N. Vaptzarov Road, Sofia 1407, Bulgaria, Fax: +359-2-403-4010, +359894693513, E-mail: biliana_teneva@yahoo.com
J Clin Gastroenterol Treat, JCGT-2-022, (Volume 2, Issue 2), Case Report; ISSN: 2469-584X
Received: March 06, 2016 | Accepted: April 20, 2016 | Published: April 23, 2016
Citation: Karagyozov PI, Teneva BH, Tishkov IP, Georgieva ZN, Vloka M (2016) Management of Hemosuccus Pancreaticus after Surgical Drainage of a Pancreatic Pseudocyst - A Rare Cause of Upper Gastrointestinal Bleeding. J Clin Gastroenterol Treat 2:022. 10.23937/2469-584X/1510022
Copyright: © 2016 Karagyozov PI, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Haemosuccus pancreaticus (HP) is a rare life threatening and difficult to diagnose cause of upper gastro-intestinal hemorrhage. It presents bleeding from the ampulla of Vater via the pancreatic duct. A 34-year-old woman was discharged from the Surgery Department after operation of pancreatic pseudocysts. Later she was rehospitalized twice due to bleeding from the contact drainage and melena. The upper GI endoscopy found gastric varices without bleeding and no pathologic changes of the papilla of Vater examined with a duodenoscope. Еndoscopic retrograde cholangiopancreatography (ERCP) revealed active bleeding from the papilla. Selective cannulation and contrast of the pancreatic duct found a distal stenosis, a cystic lesion at its level followed by a dilatation of the pancreatic duct in the body with defects in the lumen. Extravasation of the contrast from the duct to the body/tail pancreatic zone with contrasting of the left diaphragm cupola and the end of the contact drainage was observed. A pancreatic stent with side holes was placed to cover the lesion of the pancreatic duct in the tail. A distal stenosis of the choledochal duct was also detected due to possible compression by the pseudocyst in the pancreatic head and a stent was also placed. The control CT proved the correct position of the stents and reduced sizes of the pseudocyst. After ERCP was done a complete stop of the secretion from the retroperitoneal drainage was achieved. We reported a rare case of GI bleeding - HP, where ERCP could play a key role in its diagnosis and treatment.
Introduction
Haemosuccus pancreaticus (HP) is a rare life threatening and difficult to diagnose cause of upper gastro-intestinal(GI) hemorrhage. It presents bleeding fromthe ampulla of Vater via the pancreatic duct. HP is most commonly associated with pancreatic inflammation, erosion of the pancrease by aneurysm or pseudo-aneurysm of the splenic artery. HP is the least frequent cause of upper gastrointestinal bleeding (1/1500), more common in men (male to female ratio 7:1) and mostly occurs as a complication of acute or chronic pancreatitis and pancreatic cancer [1,2]. HP is difficult to diagnose because of its rarity, its anatomical location and the bleeding is often intermittent and cannot be easily diagnosed by esophagogastroduodenoscopy [1-4].
It was first described in 1931 by Lower and Farrell who reported a primary splenicaneurysm rupture into the main pancreatic duct while the name was given by Sandblom in 1970 [2,3]. Approximately 150 cases have been reported in the literature since then.
Case Report
A 34-year-old woman admitted to the Surgery Department of our hospital for elective surgery of pancreatic pseudocysts presented with upper abdominal pain, vomiting, weight consumption and breathing difficulty for the last two months. The patient had no similar conditions before despite she had been previously diagnosed with chronic pancreatitis. On clinical examination she was in bad condition being hypotensive with tachycardia (with a blood pressure of 90/60 mmHg and a heart rate of 110 beats per minute), with decreased breath sounds in the left side of the chest and a painful palpable epigastric mass. Four pancreatic cystic lesions were found on abdominal ultrasound (two in the pancreatic head and two in the body). Chest X-ray showed a large left pleural effusion. Surgeons performed thoracocentesis with drainage of the left pleural cavity, laparotomy with pancreatic necrosectomy and cholecystectomy. The pseudocysts were opened and external and transcystic drainages were placed. The patient was discharged in better condition with a clamped transcystic drainage catheter and a retroperitoneal drainage secretion up to 150 ml/24 h.
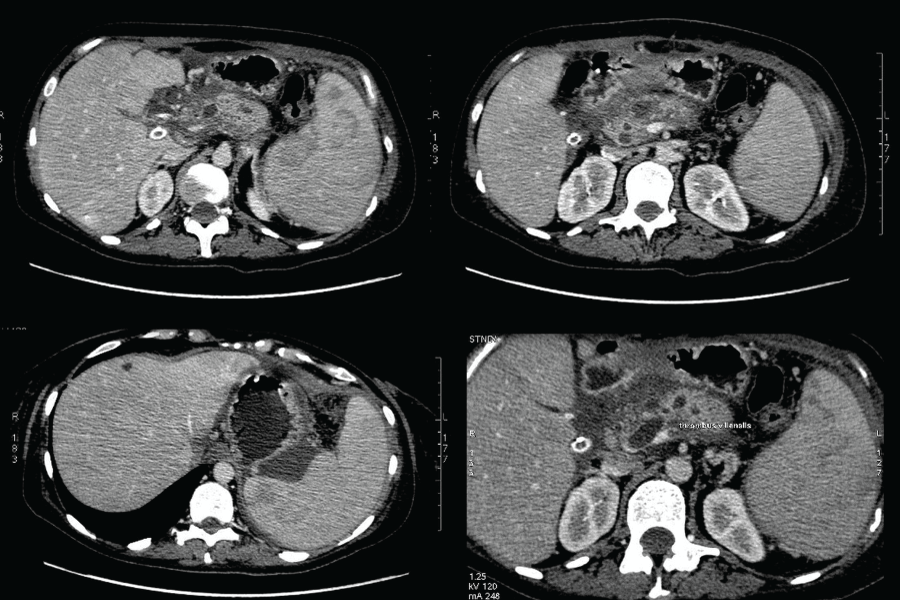
Ten days later she was admitted again with massive fresh bleeding from the contact drainage-500 ml for the last 12 hours. The laboratory tests showed a normocytic normochromic anemia (hemoglobin 9.6 mg/dl, hematocrit 30.0%) and normal coagulation status. Conservative therapy including proton pomp inhibitors (Esomeprasole -80 mg intravenous bolus followed by an 8 mg/h continuous infusion), fresh frozen plasma infusion, Terlipressin (intravenous bolus injection 2 mg/4 h followed by a continuous infusion 2 mg/24 h), vitamin K and Dicynone infusions were administered. However, intermittent bloody secretion from the contact drainage (150-900 ml/24 h) and transcystic biliary secretion (500 ml/24 h) persisted. The abdominal CT showed persistent pancreatic pseudocysts with peripancreatic fluid collections, a dilated pancreatic duct, splenic vein thrombosis and ascites (Figure 1). CT angiography depicteda homogeneously contrasted abdominal aorta without aneurysms or dissection, normal separation and distribution of its visceral branches and homogeneously contrasted lumens without contrast leakage.
A week later the patient presented with several melena stools with persistent intermittent bloody secretion from the drainage. We performed an urgent upper GI endoscopy and found type I isolated gastric varices without bleeding.
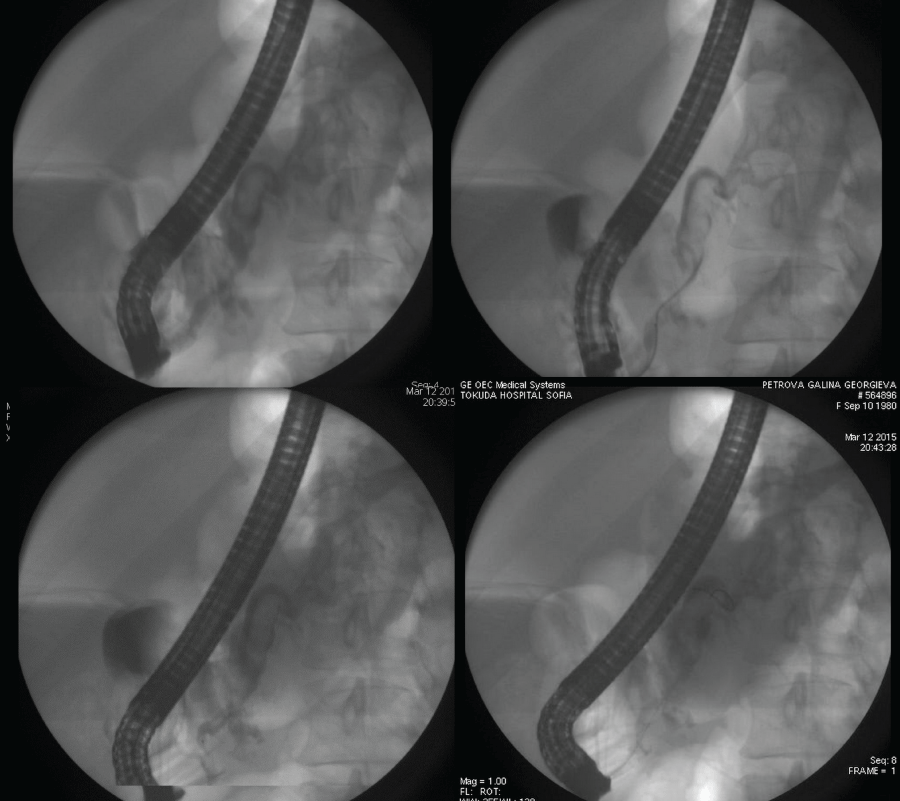
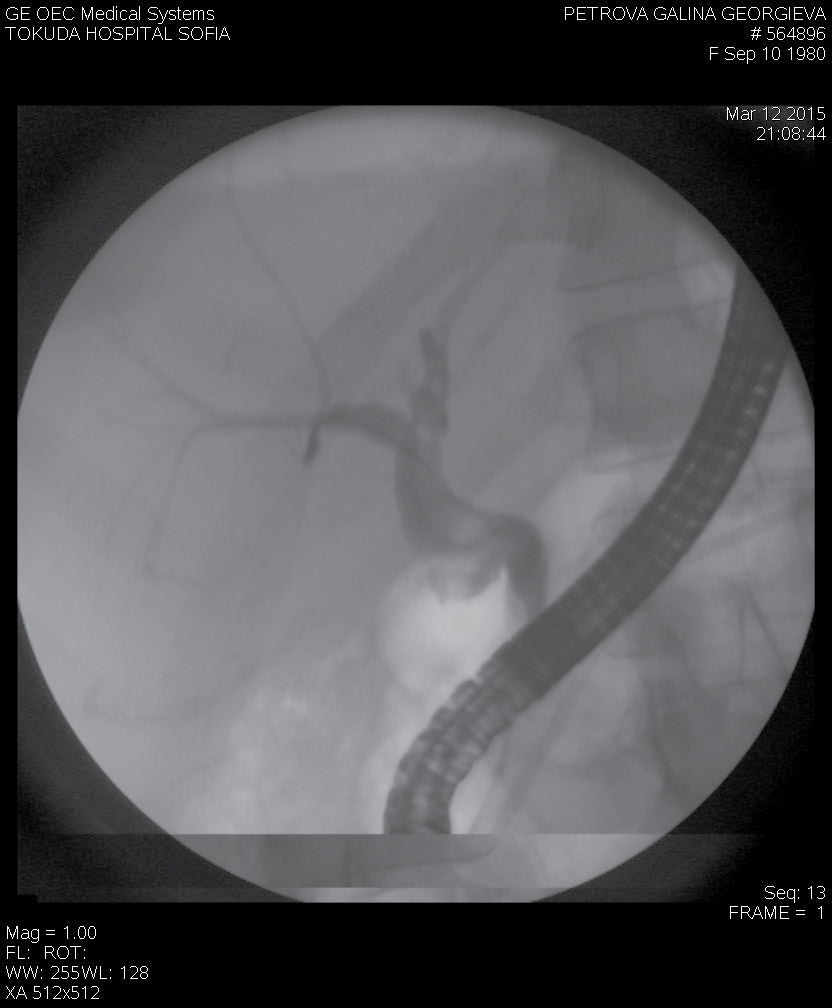
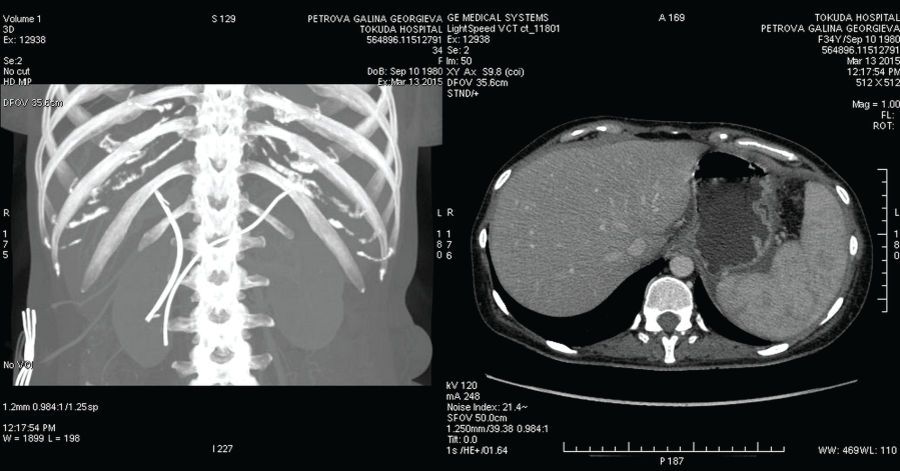
The examination of the papilla of Vater with a duodenoscope found no pathologic changes. Despite conservative therapy that achieved hemodynamic stabilization the patient was again with melena, with blood secretion from the contact drainage (600 ml) and persistent secretion from the transcystic drainage (500- 600 ml/24h). Endoscopic retrograde cholangiopancreatography (ERCP) was performed and revealed fresh bleeding from the ampulla of Vater. Selective cannulation and contrast of the pancreatic duct found a distal stenosis about 20 mm by length. At the level of the stenosis there was a cystic lesion, followed by a dilatation of the pancreatic duct in the body up to 15 mm with defects in the lumen/possible coagulum/, extravasation of the contrast from the duct to the body/tail pancreatic zone with contrasting of the left diaphragm cupola and the end of the contact drainage. A7Fr 13 cm pancreatic stent with side holes was placed to cover the lesion of the pancreatic duct in the tail. A distal stenosis of the choledochal duct was also detected due to a possible compression by the pseudocyst in the pancreatic head. Papilla sphincterotomy (PST) and placing of a 10 Fr 7 сm stent were performed (Figure 2 and Figure 3). The control CT2 days later proved the correct position of the stents, reduced size of the pseudocyst and significantly reduced size of the liquid collection (Figure 4). After the ERCP was done a complete stop of the secretion from the retroperitoneal drainage was achieved. Pancreatic and choledochal stent replacements were performed every three months in our patient and during a 2 years follow-up period no relapse of GI bleeding was observed.
Discussion
We reported a rare case of GI bleeding - HP after surgical drainage of a pancreatic pseudocyst. Patients with HP usually presentwith epigastricpain, intermittent upper GI hemorrhage as evidenced by haematemesis and melena, of which melena is more common, and hyperamylasemia [5]. The hemorrhage is usually intermittent, repetitive and, most often, not severe enough to cause haemodynamic instability.
The various diagnostic modalities in HP include contrast enhanced CT, esophagogastroduodenoscopy, selective angiography and sometimes endoscopic ultrasound and ERCP. A definitive diagnosis can be established only with angiography - selective angiography is the gold standard for its diagnosis with a sensitivity of 96% [6]. Upper GI endoscopy can detectactive bleeding via the papilla only in 30% of patients patients but a negative endoscopy does not exclude the HP [7,8]. Conrast enhanced CT abdomen can be used to visualize splenic artery pseudoaneurysm adjacent to pancreas or pancreatic duct, as well as typical changes of chronic pancreatitis can also be seen with this method [9]. ERCP can detect a lesion under direct visualization with bleeding from the papilla and pancreatographic evidence of poorly outlined filling defects, or identify changes characteristic of a tumor [10,11].
Treatment of HP includes endovascular techniques or surgery.The first ones include inserting coils, particles or gel foam to embolize the bleeding vessel to achieve haemostasis which have a success rate of around 75-85% [9]. The surgical approach depends on the location of the splenic artery aneurysm, the age of patient and operative risk [12].
ERCP can also provide alternative therapeutic management including PST and stenting [10,11]. In our case the distal stenosis of pancreatic duct and its disturbed integrity in the tail leads to a constant contact between the capsule of the drained pseudocyst and the pancreatic secretion that results in erosions of the intracystic arterial vessels and hemorrhage. Blood flows into the pancreatic duct or pseudocyst and as it coagulates it obstructs the drainage passage so that the pressure within these structures increases to temporarily tamponade the hemorrhage. The increased pressure causes the abdominal pain. Hemorrhage recurs days after the intraductal or intracystic clot dissolves. Thus, HP is characterized by intermittent bleeding incl. from the contact drainage in our case. The performed decompression of pancreatic duct by endoscopic pancreatic sphincterotomy and pancreatic stenting stopped the contact of pancreatic liquid with the wall of the psudocyst and the bleeding itself and the patient's condition rapidly improved.
In conclusion, ERCP could play a key role in diagnosis and treatment of HP.
References-
Callinan AM, Samra JS, Smith RC (2004) Hemosuccus pancreaticus. ANZ J Surg 74: 395-397.
-
Sandblom P (1970) Gastrointestinal hemorrhage through the pancreatic duct. Ann Surg 171: 61-66.
-
Lower WE, Farrell JT (1931) Aneurysm of the splenic artery: report of a case and review of literature. Archives of Surgery 23: 182–190.
-
Etienne S, Pessaux P, Tuech JJ, Lada P, Lermite E, et al. (2005) Hemosuccus pancreaticus: a rare cause of gastrointestinal bleeding. Gastroenterol Clin Biol 29: 237-242.
-
Sakorafas GH, Sarr MG, Farley DR, Que FG, Andrews JC, et al. (2000) Haemosuccus Pancreaticus complicating chronic pancreatitis: An obscure cause of upper gastrointestinal bleeding. Langenbacks Arch Surg 385: 124-128.
-
Kim SS, Roberts RR, Nagy KK, Joseph K, Bokhari F, et al. (2000) Hemosuccus pancreaticus after penetrating trauma to the abdomen. J Trauma 49: 948-950.
-
Kapoor S, Rao P, Pal S, Chattopadhyay TK (2004) Hemosuccus pancreaticus: an uncommon cause of gastrointestinal hemorrhage. A case report. JOP 5: 373-376.
-
Peroux JL, Arput JP, Saint-Paul MC, Dumas R, Hastier P, et al. (1994) Wirsungorrhagia complicating chronic pancreatitis associated with a neuroendocrine tumour of the pancrease. Gastroenterol Clin Biol 18: 1142-1145.
-
Gullian R, Garcier JM, Abergel A, Mofid R, Garcia V, et al. (2003) Management of splenic artery aneurysms and false aneurysms with endovascular treatment in 12 patients. Cardiovasc Intervent Radiol 26: 256-260.
-
Levy MJ, Wong Kee Song LM, Farnell MB, Misra S, Sarr MG, et al. (2008) Endoscopic ultrasound (EUS)-guided angiotherapy of refractory gastrointestinal bleeding. Am J Gastroenterol 103: 352-359.
-
Hasaj O, Di Stasi C, Perri V, Tringali A, Costamagna G (2004) Hemosuccus pancreaticus secondary to intraductal rupture of a primary splenic artery aneurysm: diagnosis by ERCP and successful management by interventional radiology. Endoscopy 36: 437-441.
-
De Perrot M, Buhler L, Schneider PA, Mentha G, Morel P (1999) Do Aneurysm and pseudoaneurysm of the splenic artery require different surgical strategy? Hepatogastroenterology 46: 2028-2032.