Journal of Clinical Gastroenterology and Treatment
Listeriosis Complicating Infliximab Treatment in Crohn's Disease
FW Chen1*, W Matar2, M Hersch2 and J Freiman1
1Department of Gastroenterology and Hepatology, St George Hospital, New South Wales, Australia
2Department of Neurology, St George Hospital, New South Wales, Australia
*Corresponding author:
Fei Chen, Department of Gastroenterology and Hepatology, St George Hospital, New South Wales, Australia, E-mail: feiw.chen@gmail.com
J Clin Gastroenterol Treat, JCGT-2-024, (Volume 2, Issue 2), Case Report; ISSN: 2469-584X
Received: October 11, 2015 | Accepted: May 04, 2016 | Published: May 06, 2016
Citation: Chen FW, Matar W, Hersch M, Freiman J (2016) Listeriosis Complicating Infliximab Treatment in Crohn's Disease. J Clin Gastroenterol Treat 2:024. 10.23937/2469-584X/1510024
Copyright: © 2016 Chen FW, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Listeria monocytogenes, a gram-positive rod, infects the central nervous system in neonates, pregnant woman and those immunosuppressed by naturally occurring illnesses and by therapeutic agents, including agents such as infliximab. We report here the first published case of Listeriosis complicating Infliximab therapy in Crohn's disease in Australia.
Keywords
Listeria, Brain abscess, Infliximab, Crohn's disease
Case Report
A 45-year-old Caucasian woman presented to the emergency department with a two-day history of increasing right lower limb weakness. Her sandals would slip off her right foot due to toe weakness and at times her foot would drag. There were no associated pain, sensory changes or sphincter-related symptoms. Two days earlier she had experienced chills, rigors, myalgia and mild headache, but no respiratory or gastrointestinal symptoms.
She had a 6-year history of small bowel Crohn's disease, which had been poorly controlled despite treatment with Methotrexate (20 mg per week with folic acid supplementation), Prednisone (50 mg daily, then weaned to 10 mg daily) and Mesalazine (2 g twice daily). Azathioprine had not been tolerated. However, she had excellent clinical response to Infliximab (5 mg/kg) infused on two occasions, most recently a month prior to the current presentation.
On examination, she was comfortable and afebrile. The only consistent abnormality was marked weakness of flexion and extension of all the toes on her right foot. Power at the right hip, knee and ankle varied from near normal (with encouragement) to moderately weak in the absence of pain. Sensation was unimpaired. The plantar responses were flexor and the tendon jerks were present and symmetrical in all four limbs. Examination of the right arm, left limbs and cranial nerves was unremarkable. At no stage was there headache, meningism or altered sensorium.
Initial blood work showed normal total white cell count (4.32 × 109/L) but a lymphopenia (0.6 × 109/L). Electrolytes and liver function tests were normal. The admission Computed Tomography (CT) brain (without contrast) was reported to be normal.
She was admitted for investigation, including Magnetic Resonance Imaging (MRI) to exclude Infliximab-induced myelopathy and neurophysiologic studies to exclude focal neuropathy but her condition deteriorated significantly overnight. Power in all right lower limb muscles from hip to toes was reduced to Medical Research Council (MRC) grade 3 (easily overcome). Tendon jerks remained symmetrical initially but there was unsustained (initially) clonus at the right ankle, on which side the plantar response was extensor. Sensation remained normal.
MRI of the entire spine (to exclude infliximab-induced demyelination) was normal. Repeat CT brain with contrast excluded sinus thrombosis but showed subtle low attenuation in the white matter at the vertex of the left frontoparietal junction without definite enhancement; in retrospect, this was discernible on the admission CT. Expedited MRI brain (Figure 1) confirmed a left frontal cortex abnormality consistent with a cerebral abscess, in addition to further regions of early abscess formation in the left frontal and left temporal lobes. Lumbar puncture was unremarkable (cells, protein, glucose, microscopy and culture). Ceftriaxone 2 g twice daily, and Metronidazole 500 mg twice daily were commenced intravenously after blood cultures were taken from the afebrile patient. The regimen was modified to Ampicillin 8 g daily and Gentamicin 1mg/kg three times daily intravenously after Listeria monocytogenes was cultured from the blood. Methotrexate and Infliximab were ceased but Prednisolone 10mg daily was continued. Gentamicin was administered for 3 weeks, followed by Benzylpenicillin by Baxter pump for 2 months followed by Sulfamethoxazole/Trimethoprim for 4 months.
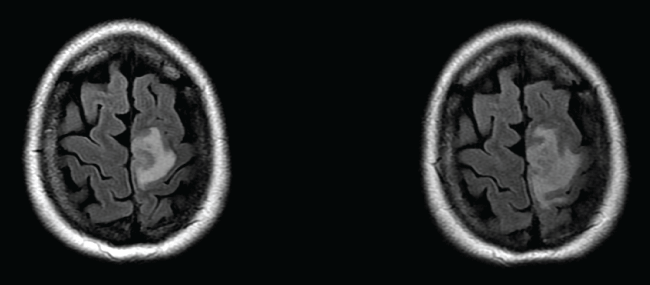
.
Figure 1: MRI brain at similar level on day 2 (left) and 9 (right) post admission. Early abscess/cerebritis of the left medial frontal lobe is apparent.
View Figure 1
Neurologic function and MRI imaging began to improve about three weeks after commencement of antibiotics. Lower limb function had recovered fully by 5 months post presentation, and the Crohn's disease remained in remission on Mesalazine 2 g twice daily and Prednisone 7.5 mg daily. The patient has been advised to avoid unpasteurised dairy products.
Discussion
Listeria monocytogenes, a Gram-positive rod, infects the central nervous system in neonates, pregnant woman and those immunosuppressed by naturally occurring illnesses (e.g. diabetes, alcoholism) and by therapeutic agents, including agents such as Infliximab. However, about a third of patients infected by Listeria are immune-competent [1].
Typically central nervous system Listerosis manifests as meningitis or meningo-encephalitis, which was not the case in our patient. Cerebritis and abscess formation occur in about 1% of patients, predominantly in men and carry a mortality three times higher than other brain abscesses [2].
In our patient, Infliximab, a TNF-α blocker, was very effective in remediating her inflammatory bowel disease. Such agents have revolutionised the management of Crohn's disease, exerting their effect by binding and clearing soluble TNF-α, neutralizing its pro-inflammatory actions, as well as binding to cell-bound TNF-α on macrophages and T-cells, interfering with direct cell-to-cell interactions [3-5].
However, Infliximab and similar agents impair the crucial role of TNF in host defences against intracellular pathogens including Listeria, Histoplasma, Salmonella and Mycobacterium [4]. The US Food and Drug Administration has documented 38 cases of serious infection due to Listeria in patients on Infliximab. Most were being treated for rheumatoid arthritis or Crohn's disease and most were on an additional immunosuppressant [6,7]. In two thirds of cases, Listeria infection was evident within 3 infusions [6], as was the case in our patient.
Other neurological complication of Infliximab include demyelination of the central or peripheral nervous systems, hence the initial MRI of the spinal cord in our patient. Optic neuritis is also described.
It is unclear whether Listeria infection in immuno-compromised hosts originates from contaminated foodstuffs or from chronic faecal carriage [8]. The occurrence of infection shortly after the initiation of therapy with TNF-α blocker favours reactivation of latent infection [9], but vulnerable patients should avoid unpasteurised dairy products and should reheat processed meats [8]. The extent to which other immuno-modulating drugs influence susceptibility to serious infections in patients on Infliximab is unclear; although it is likely that concomitant use of these agents may increase the risk significantly [8,10].
To this date, this is the first reported case of Listeria abscess induced by Infliximab infusion in Australia. This case highlights the need for physicians to consider Listeria infection in patients taking TNF-α inhibitors, alone or in combination with other immunosuppressants, presenting with neurological illness even when the clinical presentation is atypical for this opportunistic pathogen. To this end, the US FDA includes Listeria in a boxed warning for the entire class of TNF-α blockers.
References
-
Mylonakis E, Hohmann EL, Calderwood SB (1998) Central nervous system infection with Listeria monocytogenes. 33 years' experience at a general hospital and review of 776 episodes from the literature. Medicine 77: 313-336.
-
Cone LA, Leung MM, Byrd RG, Annunziata GM, Lam RY, et al. (2003) Multiple cerebral abscesses because of Listeria monocytogenes: three case reports and a literature review of supratentorial listerial brain abscess(es). Surgical neurology 59: 320-328.
-
Camussi G, Albano E, Tetta C, Bussolino F (1991) The molecular action of tumor necrosis factor-alpha. Eur J Biochem 202: 3-14.
-
Bodro M, Paterson DL (2013) Listeriosis in patients receiving biologic therapies. European journal of clinical microbiology & infectious diseases 32: 1225-1230.
-
Murch SH, Braegger CP, Walker-Smith JA, MacDonald TT (1993) Location of tumour necrosis factor alpha by immunohistochemistry in chronic inflammatory bowel disease. Gut 34: 1705-1709.
-
Abreu C, Magro F, Vilas-Boas F, Lopes S, Macedo G, et al. (2013) Listeria infection in patients on anti-TNF treatment: report of two cases and review of the literature. J Crohns Colitis 7: 175-182.
-
Grainne M, Daniel SM, Gerard HB, Sinead H (2009) Systemic listeriosis with adalimumab therapy. Journal of clinical rheumatology 15: 369-370.
-
Williams G, Khan AA, Schweiger F (2005) Listeria meningitis complicating infliximab treatment for Crohn's disease. Can J Infect Dis Med Microbiol 16: 289-292.
-
Wallis RS, Broder MS, Wong JY, Hanson ME, Beenhouwer DO (2004) Granulomatous infectious diseases associated with tumor necrosis factor antagonists. Clin Infect Dis 38: 1261-1265.
-
Colombel JF, Loftus EV Jr, Tremaine WJ, Egan LJ, Harmsen WS, et al. (2004) The safety profile of infliximab in patients with Crohn's disease: the Mayo clinic experience in 500 patients. Gastroenterology 126: 19-31.





