Journal of Infectious Diseases and Epidemiology
Tick-Borne Encephalitis Virus Isolates Features from Natural Foci of Pribaikalie (Eastern Siberia, Russia)
Adelshin RV*, Melnikova OV, Trushina Yu N and Andaev EI
Irkutsk Anti-Plague Research Institute of Siberia and the Far East, Russia
*Corresponding author:
Renat V. Adelshin, Senior Researcher, Irkutsk Anti-Plague Research Institute of Siberia and the Far East, Irkutsk, Russian Federation, 664047, Tel: 73-952-220-135, E-mail: adelshin@gmail.com
J Infect Dis Epidemiol, JIDE-1-002, (Volume 1, Issue 1), Short Communication; ISSN: 2474-3658
Received: July 30, 2015 | Accepted: August 26, 2015 | Published: August 28, 2015
Citation: Adelshin RV, Melnikova OV, Trushina Yu N, Andaev EI (2015) Tick-Borne Encephalitis Virus Isolates Features from Natural Foci of Pribaikalie (Eastern Siberia, Russia). J Infect Dis Epidemiol 1:002. 10.23937/2474-3658/1510002
Copyright: © 2015 Adelshin RV. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Pribaikalie, Tick-Borne Encephalitis Virus (TBEV), Strains, Virulence, E gene sequences
Introduction
Pribaikalie is a mountainous area in the South of Eastern Siberia that borders Lake Baikal between the Irkutsk Region and the Republic of Buryatia. Tick-borne encephalitis (TBE) is endemic to the area. Human cases occur usually within regional centers (Irkutsk and Ulan-Ude) and large industrial centers (Bratsk, Angarsk, Cheremkhovo), where the majority of the population is concentrated.
TBE in Siberia is a seasonal disease, which is closely connected with the activity of its main vector - the hard tick Ixodes persulcatus. Most human cases occur in May and June, which coincides with the peaks of tourism and recreation. Therefore, the territories' high density of tourists and attractive recreational areas for tourists, especially nearby Baikal, become dangerous.
Three subtypes of tick-borne encephalitis virus (TBEV) are accepted today: the European subtype, the Siberian subtype, and the Far-Eastern subtype [1]. It is typical to consider that each of them has its own geographical distribution and causes different symptoms, including mortality [2-7]. The European subtype, which is distributed mainly in Europe, causes mild forms of encephalitis with mortality rate up to 2%. The Far-Eastern subtype is distributed mainly in the Far-East of Russia, Japan, and Northern-East of China and causes severe forms of disease, which can lead to 20-30% of mortality. The Siberian subtype is intermediate and usually causes a milder form of encephalitis compared to the Far-Eastern subtype. Severe forms are registered in 13-15% of cases, mortality rates vary from 3 to 6% depending on the region.
Pribaikalie is a unique territory where all TBEV subtypes circulate, but the Siberian subtype is most prevalent [8]. Most of the strains from the Baikal region that have been studied and described in the literature were isolated up to 2006 and genotyped by the molecular hybridization of nucleic acids (MHNA) reaction [8]. A novel large group of isolates (2006-2014) is characterized in this work by sequencing of the E gene fragment (1193 bp) and their pathogenicity in laboratory mice (LM).
Materials and Methods
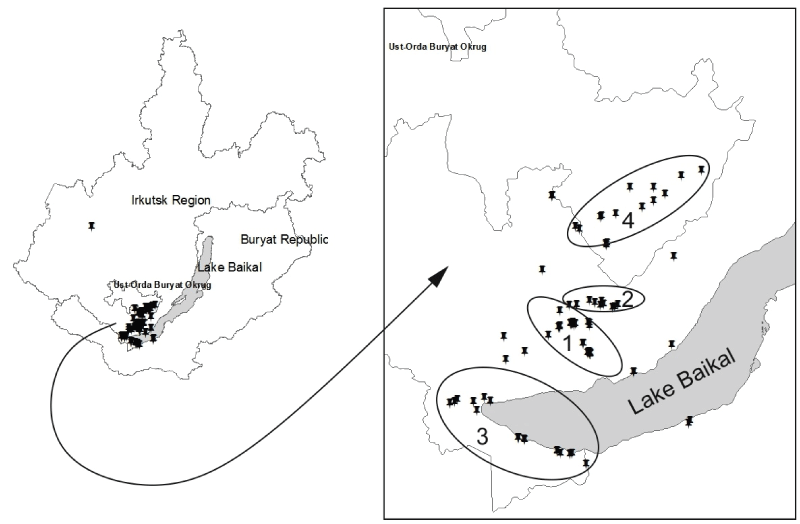
The questing adult I. persulcatus ticks were collected by flagging over vegetation from the end of April up through the beginning of July in 2006-2014 in several areas of Pribaikalie already known to be endemic for TBE (Figure 1). Small mammals were also trapped in the same areas in July-September. The names and geographical coordinates of main sample site localities and the results of their investigation are given in table 1.
.
Figure 1: Map of collection places. 1 - Baikalian Road, 2 - Goloustninsky Road, 3 - Sludyansky district, 4 - UOBO.
View Figure 1
![]()
Table 1: Main sample sites with geographical coordinates and the number of isolates
View Table 1
Each tick and small mammal's brain were homogenized in saline phosphate buffer (10% suspension) and then centrifuged. The suspensions were stored at -60℃ until testing. The TBEV antigen (TBEV AG) was identified by ELISA using the "Microgen" kit (Tomsk-Moscow, Russia). The measuring of optical density at 450 nm was performed no later than 10 min after termination of the reaction. The two-times higher optical density of the studied sample in comparison with background interference was considered a positive result. In total 11,522 ticks and 81 brain specimens were tested individually.
TBEV was isolated by intracerebral (IC) inoculation of newborn LM [9] with suspensions positive and/or doubtful in ELISA. The animals were observed for 21 days. Part of ELISA-negative suspensions, especially from the areas with low tick density, were tested in PS cell culture (96-well plates) for cytopathic effect [9] and then verified by ELISA.
The TBE isolates were titrated (10-3-10-9 LD50/ml dilutions of viral suspension) by IC (0.03 ml) and subcutaneous (SC) (0.25 ml) inoculation of young LM (6-8 g) to estimate the neurovirulence and peripheral activity. The animals were observed for 14 days. The viral titer was estimated according to Reed and Muench [9]. The neuroinvasiveness index (NI) was estimated to be the difference of the intracerebral and subcutaneous titers. All animal experiments were performed in accordance with Guide for Laboratory practice in Russian Federation (GLP) (confirmed by order of the Ministry of Health of Russian Federation N 267 19-6-2003).
Total RNA was extracted from the brain tissue or tick (10% suspension in saline) using Riboprep kit (ILS Ltd., Russia). Amplification of the complete envelope (E) gene of TBE virus strains or isolates was performed via the reverse-transcription polymerase chain reaction (RT-PCR) using Amplisens kit (ILS Ltd., Russia) with primers described in [10]. PCR products were visualized in 1% agarose and then purified according to standard method [11].
The RT-PCR products have been sequenced with the ABI Prism Big Dye Terminator v.1.1 Cycle Sequencing Kit (Applied Biosystems, USA) and the Genetic Analyzer 3500 xL (Applied Biosystems, Japan). The sequences of TBE virus E gene fragment (1193 bp) were aligned with the BioEdit program, version 7.0.5.3 [12]. Phylogenetic analysis was performed in MEGA 5 package using the Maximum Likelihood method [13].
Map with places of material collection was held using ArcGIS soft (ESRI, USA), version 9.3. Statistical significance was assessed at the 0.05 level and hypothesis tests were two-tailed. Descriptive statistics were carried out using Pearson Chi-Square test in Microsoft Office Excel (version 2007).
Results
Sixty five strains of TBEV have been isolated during 2006-2014: 62 from I. persulcatus ticks, 2 from small mammal's brain (Myodes rufocanus and Sorex sp.), 1 from the blood of an infected young woman. There were also four RNA-isolates from tick's suspensions (Table 2). Most of the specimens positive for the TBEV AG were found not to be pathogenic for LM. We managed to isolate the virus from 25% of ELISA-positive ticks' suspensions and only 10.5% of small mammals brains. Elevated TBEV AG in ticks increased the probability of strain isolation (P > 0.01). Usually the mice became ill after the first inoculation (86.8%). The incubation period varied from 4 to 18 days, 6.5 days on average, and decreased to 3-4 days in the following passages.
![]()
Table 2: The results of ELISA test and methods of TBEV isolation from different material
View Table 2
Inoculation of PS cell culture with 299 ELISA-negative or doubtful suspensions led to the isolation of 12 strains. Nine of them were from UOBO, tree - from the Baikalian Road. It should be mentioned that 3 TBE cell culture isolates from ELISA-negative and poor-positive tick suspensions proved to be the European subtype of TBEV (KP331441 IrkutskBR_99-08, KP331442 IrkutskBR_1434-09 and KP331443 IrkutskBR_1456-09).
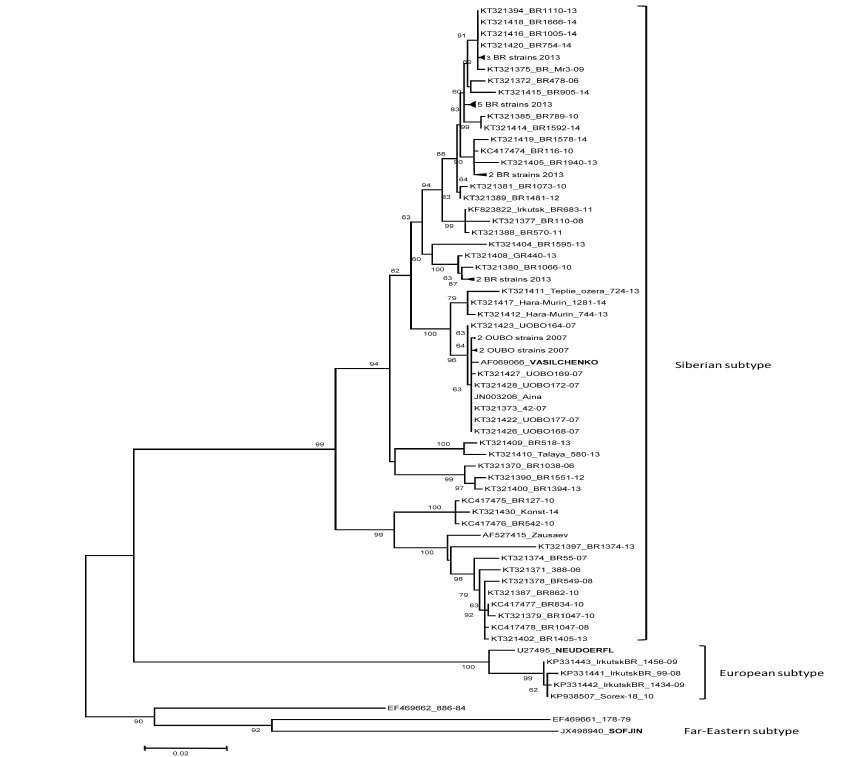
The TBE strains were taken for sequencing no later than the first passage. We managed to sequence the part of E gene directly from the four tick suspensions and in two brain suspensions of LM after the first inoculation (the isolates did not consolidate in next passages). The GenBank accession numbers of the strains used in this study are shown in figure 2.
.
Figure 2: ML tree of envelope gene sequences of TBEV from natural foci of Pribaikalie in 2006-2014. Bootstrap values (> 50 %) are presented. The prototype strains are marked by uppercase. The compressed sequences have next accession No and names: KT321391 BR1052-13, KT321393 BR1103-13, KT321403 BR1485-13, KT321406 BR2023-13, KT321407 BR2024-13, KT321413 BR1121-13, KT321392 BR1099-13, KT321398 BR1389-13, KT321399 BR1392-13, KT321401 BR1401-13, KT321395 BR1112-13, KT321396 BR1169-13, KT321421 UOBO173-07, KT321429 UOBO180-07, KT321424 UOBO167-07, KT321425 UOBO166-07.
View Figure 2
Phylogenetic analysis of 64 strains and four RNA isolates of TBE virus have showed that 94.1% of them belong to the Siberian subtype and 5.9% - to the European one (Figure 2). The Far-Eastern subtype was not found though it always presented in previous studies of Siberian TBEV collections [8,14].
The Siberian subtype isolates divided into two groups on the dendrogram (Figure 2). Most of the Siberian subtype sequences (79.4%) belong to the "Vasilchenko" group, the rest of them - to the "Zausaev" group. Interesting, strains related to the "Zausaev" group were only found in the area of the Baikalian Road (BR), a territory at high risk of TBE according to literature data and our own investigations [15]. Moreover, six of seven "Zausaev" isolates were found near to Irkutsk, in areas of dense concentration of summer houses ("dachas") and cottages. Some researchers associate the prevalence of this TBEV variant in some regions to increased severity of the disease [16].
The clades of strains on the dendrogram are connected more with geographical distribution than with the year of isolation. It should be noted that all modern strains from Ust-Orda Buryat Okrug (UOBO) have the same E gene sequences as the "Aina" strain (JN003206) isolated on this territory 52 years ago from patient's cerebrospinal fluid [14].
Virulence is a complex biological manifestation of viral pathogenic properties. SC inoculation is one of them [17]. It can be used as a marker, indicating the ability of a strain to get over the hematoencephalic barrier.
Most of our strains (88.7%) were of high virulence for LM by SC inoculation (6.0 and more lg LD50/ml), 6 - of moderate virulence (4.1-5.9 lg LD50/ml) and only 2 - of weak virulence (less than 4.0 lg LD50/ml); only one of the weak strains was sequenced. Weak (KT321400 BR1394-13) and moderate-virulent strains (KT321371 388-06, KC417476 BR542-10, KT321387 BR862-10 -"Zausaev" group, KT321389 BR 1481-12, KT321390 BR 1551-12 and KT321419 BR1578-14 - "Vasilchenko" group) in our dendrogram grouped together with the high-virulent ones, unlike the groups of Far-Eastern strains of different pathogenicity [18,19].
All our European subtype strains (n = 4) proved to be highly virulent for LM. IC titers of the European TBEV strains varied from 9.0 to 10.8 lg LD50/ml, SC titers ranged from 7.1 to 8.9 lg LD50/ml. The NI was 0.9-2.4. This observation is in agreement with the changing conception about weak pathogenicity of the European subtype [20,21]. IC and SC titers of the Siberian TBEV strains varied from 6.2 to 11.5 and 3.3-9.9 correspondingly, NI - 0.7-3.7.
The strains from small mammals (KT321375 BR Mr3-09 and KP938507 Sorex-18_10), belonging to the Siberian and the European subtype, as well as strain from human blood (KT321430 Konst-14), were highly virulent for LM by both routes of inoculation and did not differ from most of the "tick" strains by their biological features. The sequences of strains isolated from the small mammals brain (KP938507 Sorex-18_10, KT321375 BR_Mr3-09) and young woman's blood (KT321430 Konst-14) fall into the same clade of the strains isolated from ticks collected on the same place in 2006-2014. This confirms the opinion of other researchers about the correlation between geographic and phylogenetic clustering of the TBEV regardless of host [22].
Additional studies are necessary to understand the differential pathogenicity of the Siberian subtype strains, including complete genomic sequence of large group of strains with different virulence.
Acknowledgements
Authors are very much obliged to their collaborators at the Laboratory for natural-foci infections and Section of zoology and parasitology of Irkutsk Anti-Plague Research Institute of Siberia and the Far East for their participation in collecting of the material and Ivan Botvinkin for the help with figure 1.
References
-
Ecker M, Allison SL, Meixner T, Heinz FX (1999) Sequence analysis and genetic classification of tick-borne encephalitis viruses from Europe and Asia. J Gen Virol 80 : 179-185.
-
Gritsun TS, Lashkevich VA, Gould EA (2003) Tick-borne encephalitis. Antiviral Res 57: 129-146.
-
Ruzek D, Stastna H, Kopecky J, Golovljov I, Grubhoffer L(2007) Rapid subtyping of tick-borne encephalitis virus isolates using multiplex RT-PCR. J Virol Methods 144:133-137.
-
Asghar N, Lindblom P, Melik W, Lindqvist R, Haglund M, et al. (2014) Tick-borne encephalitis virus sequenced directly from questing and blood-feeding ticks reveals quasispecies variance. PLoS One 9.
-
Formanova, P, Cerny J, Bolfíková BC, Valdés JJ, Kozlova I, et al. (2015) Full genome sequences and molecular characterization of tick-borne encephalitis virus strains isolated from human patients. Ticks Tick Borne Dis 6: 38-46.
-
Frey S, Essbauer S, Zöller G, Klemp B, Dobler G, et al. (2014) Full genome sequences and preliminary molecular characterization of three tick-borne encephalitis virus strains isolated from ticks and a bank vole in Slovak Republic. Virus Genes 48: 184-188.
-
Jemersic L, Dezdek D, Brnic D, Prpic J, Janicki Z, et al. (2014) Detection and genetic characterization of tick-borne encephalitis virus (TBEV) derived from ticks removed from red foxes (Vulpes vulpes) and isolated from spleen samples of red deer (Cervus elaphus) in Croatia. Ticks Tick Borne Dis 5:7-13.
-
Demina TV, Dzhioev YP, Verkhozina MM, Kozlova IV, Tkachev SE, et al. (2010) Genotyping and characterization of the geographical distribution of tick-borne encephalitis virus variants with a set of molecular probes. J Med Virol 82: 965-976.
-
(1985) Virology: A practical approach: Edited by B.W.J. Mahy. IRL Press, Oxford, 264.
-
Adelshin RV, Melnikova OV, Karan LS, Andaev EI, Balakhonov SV (2015) Complete Genome Sequences of Four European Subtype Strains of Tick-Borne Encephalitis Virus from Eastern Siberia, Russia. Genome Announc 3.
-
Sambrook J, Russell D (2001) Molecular Cloning: A Laboratory Manual. (3rd editon), Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press, USA.
-
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series 41: 95-98.
-
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, et al. (2011) MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28: 2731-2739.
-
Kulakova NV, Andaev EI, Belikov SI (2012) Tick-borne encephalitis virus in Eastern Siberia: complete genome characteristics. Arch Virol 157: 2253-2255.
-
Melnikova OV, Vershinin EA, Korzun VM, Sidorova EA, Andaev EI (2012) The features of territorial distribution of tick-borne encephalitis morbidity between Irkutsk city population. Bulletin of SB RAMS 2: 104-109.
-
Konkova-Reidman AB, Ter-Bagdasaryan LV, Zlobin VI (2013) Study of genetic structure of natural viral populations and epidemiology of clinical forms of tick-borne encephalitis in Southern Urals. Infectional Diseases 11: 204-205.
-
Leonova GN, Muratkina SM, Kruglyak SP (1990) Study of the virulence of tick-borne encephalitis virus strains isolated in southern part of Soviet Far East. Vopr Virusol 6: 735-741.
-
Leonova GN, Belikov SI, Kondratov IG, Takashima I (2013) Comprehensive assessment of the genetics and virulence of tick-borne encephalitis virus strains isolated from patients with inapparent and clinical forms of the infection in the Russian Far East. Virology 443: 89-98.
-
Belikov SI, Kondratov IG, Potapova UV, Leonova GN (2014) The relationship between the structure of the tick-borne encephalitis virus strains and their pathogenic properties. PLoS One 9: e94946.
-
Gaumann R, Ruzek D, Muhlemann K, Strasser M, Beuret CM (2011) Phylogenetic and virulence analysis of tick-borne encephalitis virus field isolates from Switzerland. J Med Virol 83: 853-863.
-
Tonteri E, Kipar A, Voutilainen L, Vene S, Vaheri A, et al. (2013) The three subtypes of tick-borne encephalitis virus induce encephalitis in a natural host, the bank vole (Myodes glareolus). PLoS One 8: e81214.
-
Fajs L, Durmis E, Knap N, Strle F, Avsic-zupanc T (2012) Phylogeographic characterization of tick-borne encephalitis virus from patients, rodents and ticks in Slovenia. PLoS One 7: e48420.





