Journal of Infectious Diseases and Epidemiology
Incidence of Hospital Acquired Multidrug Resistant Organisms in a Tertiary Care Facility
Kamran Baig1*, Shaikh Muhammad Saif Din2, Noura A Elkhizzi3 and Daifallah J Al Nakhli1
1Department of Infection Prevention and Control, Prince Sultan Military Medical City, Saudi Arabia
2Department of Infection Prevention and Control, Al Hada Armed Forces Hospital, Saudi Arabia
3Division of Medical Microbiology, Prince Sultan Military Medical City, Saudi Arabia
*Corresponding author:
Kamran Baig, Department of Infection Prevention and Control, Prince Sultan Military Medical City, Riyadh, Kingdom of Saudi Arabia, Tel: 966 11 4777714, extension 42404, E-mail: kbaig82@gmail.com
J Infect Dis Epidemiol, JIDE-1-004, (Volume 1, Issue 1), Original Research; ISSN: 2474-3658
Received: October 14, 2015 | Accepted: November 16, 2015 | Published: November 18, 2015
Citation: Baig K, Din SMS, Elkhizzi NA, Al Nakhli DJ (2015) Incidence of Hospital Acquired Multidrug Resistant Organisms in a Tertiary Care Facility. J Infect Dis Epidemiol 1:004. 10.23937/2474-3658/1510004
Copyright: © 2015 Baig K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Irrational use of antimicrobials and gaps in infection control practices have resulted in alarmingly high prevalence of multidrug resistant organisms (MDRO) globally. The objective of our study was to highlight the incidence of hospital acquired MDROs in our facility.
Method: A retrospective analysis of surveillance data collected from January - December 2013 in a tertiary care hospital of Saudi Arabia. The Centre for Disease Prevention and Control (CDC) surveillance definitions were used, while the MDRO definition was modified. Descriptive analysis was performed and incidence density was calculated. SPSS version 20 (IBM, Chicago, USA) was used for analysis.
Result: In total 1737 MDRO isolates were identified. Of these n = 1,326 (76%) were hospital acquired and the mean incidence rate was 4.8 cases/1,000 patient-days. The most common risk factors were prolonged stay (≥ 5 days) n = 1134 (85.5%), indwelling medical devices 955 (72%) and antimicrobial therapy in past 90 days 949 (71.5%). Klebsiella pneumoniae n = 307(23%), Acinetobacter baumannii n = 270 (20%), Methicillin Resistant Staphylococcus aureus n = 183 (14%), Pseudomonas aeruginosa n = 160 (12%) and Escherichia coli n = 159 (12%) were the most prominent nosocomial pathogens. Resistance among gram negatives for cephalosporins ranged from 94% to 98%. A. baumannii (98%) and P. aeruginosa (89%) were resistant to carbapanems. More than 90% of A. baumannii were extensive drug resistant.
Conclusion: The study indicated an alarmingly high incidence of hospital acquired MDRO in a single center. The increasing resistance warrants the need for a robust and continuous surveillance system for monitoring regional trends in MDRO susceptibility patterns and comprehensive infection control and antimicrobial stewardship programs.
Background
The World Health Organization (WH)) defines "rational use of drug" as "patients receive medications appropriate to their clinical needs, in doses that meet their own individual requirements, for an adequate period of time, and at the lowest cost to them and their community" [1]. The increasing irrational use of antimicrobials in humans and veterinary medicine has consequently increased the emergence of antibiotic resistant organisms or multidrug resistant organisms (MDRO) [2]. Primary reasons for concern about MDRO are the diminishing treatment options increasing cost of care, length of stay, mortality and morbidity [3-6]. Antimicrobial resistance has been a global problem. The published literature from the Gulf Cooperation Council (GCC) region had highlighted an alarming prevalence of antimicrobial resistance and the dawn of post antibiotic era [7-11]. In Saudi Arabia too, the increasing misuse and overuse of antibiotics resulted in the emergence of antimicrobial resistance among organisms of epidemiological importance [7,9,12].
However, very little efforts had been implemented at national and regional level to control the emergence of antimicrobial resistance. It has been documented that the resistance patterns depend on the antibiotic consumption [13,14]. Unfortunately, we do not have a centralized and standardized surveillance system that could better explain the resistance patterns in our region. There is a tremendous need to formulate a robust and continuous surveillance system for monitoring regional trends in antimicrobial consumption and MDRO susceptibility patterns.
The purpose of this study was to highlight the incidence of hospital acquired MDRO in our facility, justifying the need of developing and implementing a comprehensive local and national infection control and antimicrobial stewardship programs. It will also provide a benchmark for similar facilities using the same surveillance definitions and methodology.
Methods
Facility
Prince Sultan Military Medical City (PSMMC) is one of the largest tertiary care military hospitals for armed forces personnel and their dependents, in Riyadh, Saudi Arabia. The PSMMC provides a broad range of services to a wide variety of patient populations. It is a 1,100 bed facility with most of the clinical areas being multi-bedded.
Microbiological testing method
All specimens received at the microbiology laboratory were processed according to the standard operating procedures of the laboratory. Organisms were identified biochemically by catalase, coagulase, oxidase and using API 20 E and API20NE (Biomerieux, France) for gram negatives. The antimicrobial sensitivity testing was performed with an automated system (MicroScan Walkaway 96 plus System, Siemens Healthcare Diagnostics, USA) using gram negative & gram positive minimum inhibitory concentration panels. Antibiotic agents tested were:penicillins (methicillin, amoxicillin, amoxicillin-clavulanate, piperacillin-tazobactam), cephalosporins (cefuroxime, cetriaxone, ceftazidime), flouroquinolones (ciprofloxacin, norfloxacin, levofloxacin), carbapanems (imipenem, meropenem), aminoglycosides (gentamicin, amikacin),trimethoprim-sulphamethoxazole, clindamycin, teicoplanim, vancomycin, linezolid, colistin and rifampicin. Clinical Laboratory Standards (CLSI, M100-S23) criteria were used for the interpretation of susceptibility results.
Definition of MDRO
The United States Center for Disease Control and Prevention (CDC) and European Centre for Disease Prevention and Control (ECDC) came up with a consensual surveillance definition for MDRO, which states, "acquired non-susceptibility to at least one agent in three or more antimicrobial categories" [15]. The same definition is being used by the GCC - Centre for Infection Control. However, the observed practice within our facility indicated irrational use of antimicrobials. The published literature also suggests increasing use and misuse of antimicrobials in different areas of Saudi Arabia [16-19]. Therefore, the Infection Control Department at PSMMC in collaboration with Division of Infectious Diseases and Department of Microbiology devised an MDRO definition for our facility. This definition was a modified version of CDC and GCC MDRO definitions and stated that "a gram-negative organism is considered MDRO if it's resistant to all agents tested in at least three classes of antimicrobials". The antimicrobials classes are: - β-lactams (penicilins and cephalosporins), aminoglycosides, fluoroquinolones and carbapenems.
According to the CDC, a laboratory identified (LabID) MDRO is one which is isolated from a specimen that was collected for therapeutic reasons and will not include the samples taken for active screening. The MDRO which were isolated after 3rd day of admission i.e. on day 4 or after will be considered hospital acquired MDRO otherwise will be labeled as community acquired. Any duplicate isolate from the same patient was not included in the analysis. The MDRO identified by the laboratory were immediately informed to Infection Control Practitioners (ICPs) who then thoroughly reviewed patients clinical and laboratory records to check if it was a new episode of infection or continuation of previous infection. The data was then reviewed by senior ICP and an Infection Control Coordinator before admission in the surveillance system. This process ensured that contaminants, duplicates and screening isolates were not entered in the database. The database was created for our facility using Microsoft Access (Microsoft Inc., Redmond, USA) and was accessible only by the infection prevention and control staff.
Statistical analysis
A retrospective surveillance study based on data collected during 2013. The incidence of hospital acquired MDRO was calculated as incidence density (events per 1,000 patient days) and descriptive analysis with frequency and percentage was performed for risk factors, isolates and susceptibility. SPSS version 20 (IBM, Chicago, USA) was used for analysis.
Ethical approval
The study proposal was approved by research ethics committee of Prince Sultan Military Medical City
Results
During 2013, a total of 1737MDRO isolates were identified among 278,528 patient days. Of these isolates, 1326 (76%) were hospital acquired whereas 365 (21%) were community acquired and 46 (3%) were transferred from other hospitals (if MDRO was isolated within 3 days of transfer from another facility).
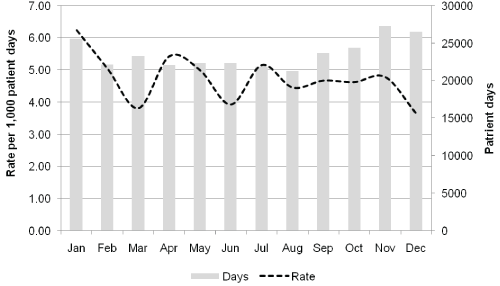
The overall incidence density was 4.8 cases per 1,000 patient days. The rate remained stable throughout the study period and did not show any statistically significant change in trend (R-square = 0.12 and p value = 0.093). No seasonal variation was detected in the rates, as indicated in figure 1. Though the rate decreased in March, June and December, but this was statistically insignificant. Hypothetically, the decrease in March could be attributed to admission of patients with Middle Eastern Respiratory Syndrome (MERS) corona virus, the decrease in June could be due to preceding hand hygiene campaign in May and infection control link nurse program while the lower rate in December could be due to the preparation for mock survey for accreditation. During all these three points, the infection control activities and compliance to practices was increased, hence could have resulted in decreased rates.
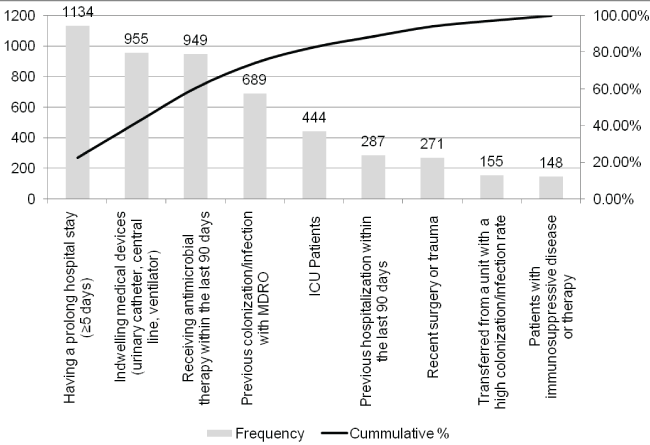
The most common risk factors were prolonged stay (≥ 5 days) n = 1134 (85.5%), indwelling medical devices 955 (72%), antimicrobial therapy in past 90 days 949 (71.5%) and previous colonization/infection of MDRO 689 (52%). The pareto analysis, in figure 2, indicated that controlling these factors might help in reducing the MDRO incidence rate by approximately 80%.
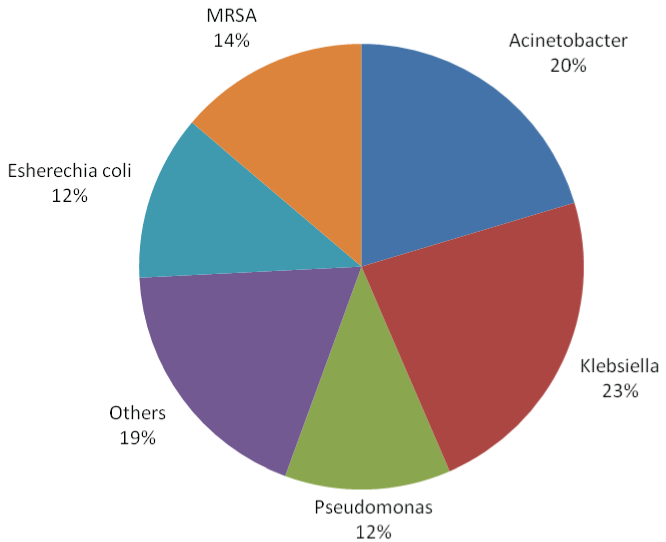
The most common organisms were gram negative (1090, 82.2%). Among all the hospital acquired MDROs K. pneumoniae (307, 23%), A. baumannii (270, 20%), MRSA (183, 14%), P. aeruginosa (160, 12%) and E. coli (159, 12%) were the most prominent pathogens (Figure 3).
The antibiotic susceptibility analysis for these organisms, presented in table 1, indicate that almost all of the A. baumannii isolates were resistant to penicillins, carbapanems, fluroquinolones and cephalosporins and three-fourth of the isolates were resistant to aminoglycosides. Approximately all the K. pneumoniae isolates were resistant to cephalosporins and penicillins and a majority was resistant to fluroquinolones (76%) also. However, approximately 16% were resistant to carbapanems. Similarly P. aeruginosa were resistant to cephalosporins and carbapanems and approximately three fourth were resistant to fluroquinolones and aminoglycosides each. Nearly all the E. coli isolates were resistant to cephalosporins and a big number to fluroquinolones also, but majority were susceptible to carbapanems and aminoglycosides.
![]()
Table 1: Antibiotic Resistance for Most Common Pathogens
View Table 1
Discussion
The multi drug resistance among healthcare acquired organisms is an emerging global threat. The irrational use of antimicrobials has played an eminent role in developing such organisms [13,20]. The prevalence of such organisms have induced direct and indirect financial burden on both the patients and the providers and have limited the treatment options [3,6]. Like other parts of the world, the emergence of MDRO has created an alarming situation in the gulf region [10,21,22]. Though very little has been published from the region and it does not address any substantial initiative by the local authorities.
In our study, we presented the findings from a single center surveillance data. Most of our MDROs were acquired within the hospital setting. This indicated an alarming gap in infection control practices. As our facility is multi-bedded so lapse in proper implementation of isolation precautions cannot be ignored. However, isolation is just one intervention which would be more effective when combined with other prevention strategies [23]. As reported by other researchers, most of the healthcare acquired MDRO from our facility were gram negatives [10,24-26]. Among gram negatives, the greatest concern which has been repeatedly highlighted was the presence of MDR A. baumannii [27,28]. The A. baumannii isolates detected during the study period were almost resistant to all the drugs being tested. If the definition given by Manchada et al., was followed, all the A. baumannii isolates would have been classified as extensive drug resistant (XDR) i.e. resistant to three classes (all penicillins, cephalosporins, fluroquinolones, and aminoglycosides) and carbapanems [29]. The treatment for carbapenem resistant and XDR A. baumannii has been widely studied and indicates limited therapeutic options for the organism [30-32].
Similarly a large number of our P. aeruginosa isolates were resistant to carbapenems. Almost all the isolates were resistant to cephalosporins, therefore excessive use of carbapanems might have resulted in increased resistance. The same phenomenon has been reported by Rahal et al., [33]. The emergence of carbapanems resistant organisms has been in discussion for decades but unfortunately no substantial initiative has been taken to overcome the issue. More than 90% of the 4 gram negative isolates were resistant to cephalosporins, indicating the limited therapeutic use of the drug without manipulation of pharmacokinetics and pharmacodynamics. Similar results were reported from Taiwan, Romania and Kenya [14,34-36].
Our risk factors analysis indicated that prolong hospital stay, indwelling medical devices, antimicrobial therapy and previous colonization or infection were the most common and frequently occurring risk factors. These predictors have been reported several times by other researchers [37-39]. They can be controlled through a comprehensive infection prevention and control program. In addition to continuous surveillance, the program should incorporate strategies to minimize hospital stay, introduce and enhance bundles implementation for reducing device associated infections and implement an aggressive antimicrobial stewardship program [40].
Based on our analysis, we observed that our findings are not different from rest of the world. An integrated and robust surveillance system is deemed necessary to understand the resistance patterns and prevent the new emergence. The local and national authorities should pay heed to the emerging global threat of antimicrobial resistance and should devise a national antimicrobial stewardship program for promoting judicious use of antimicrobial. This might not help us to revert the existing resistance pattern but will prevent further emergence.
References
-
http://who.int/medicines/areas/policy/world_medicines_situation/en/
-
Laxminarayan R, Duse A, Wattal C, Zaidi AK, Wertheim HF, et al. (2013) Antibiotic resistance-the need for global solutions. Lancet Infect Dis 13: 1057-1098.
-
Cartaxo Salgado FX, Carneiro Goncalves J, Monteiro De Souza C, Barbosa Da Silva N, Gavilanes Sanchez TE, et al. (2011) Cost of antimicrobial treatment of patients infected with multidrug-resistant organisms in the Intensive Care Unit. Medicina (B Aires) 71: 531-535.
-
Cosgrove SE, Sakoulas G, Perencevich EN, Schwaber MJ, Karchmer AW, et al. (2003) Comparison of mortality associated with methicillin-resistant and methicillin-susceptible Staphylococcus aureus bacteremia: a meta-analysis. Clin Infect Dis 36: 53-59.
-
Patel R (2003) Clinical impact of vancomycin-resistant enterococci. J Antimicrob Chemother 51 Suppl 3: iii13-21.
-
Tansarli GS, Karageorgopoulos DE, Kapaskelis A, Falagas ME (2013) Impact of antimicrobial multidrug resistance on inpatient care cost: an evaluation of the evidence. Expert Rev Anti Infect Ther 11: 321-331.
-
Al Johani SM, Akhter J, Balkhy H, El-Saed A, Younan M, et al. (2010) Prevalence of antimicrobial resistance among gram-negative isolates in an adult intensive care unit at a tertiary care center in Saudi Arabia. Ann Saudi Med 30: 364-369.
-
Al-Mendalawi MD, Asghar AH (2010) Frequency and antimicrobial susceptibility of Gram-negative bacteria isolated from 2 hospitals in Makkah, Saudi Arabia. Saudi Med J 31: 338.
-
Babay HAH (2007) Antimicrobial resistance among clinical isolates of Pseudomonas aeruginosa from patients in a teaching hospital, Riyadh, Saudi Arabia, 2001-2005. Jpn J Infect Dis 60: 123-125.
-
Balkhair A, Al-Farsi YM2, Al-Muharrmi Z, Al-Rashdi R, Al-Jabri M, et al. (2014) Epidemiology of multi-drug resistant organisms in a teaching hospital in oman: a one-year hospital-based study. ScientificWorldJournal 2014: 157102.
-
Rotimi VO, al-Sweih NA, Feteih J (1998) The prevalence and antibiotic susceptibility pattern of gram-negative bacterial isolates in two ICUs in Saudi Arabia and Kuwait. Diagn Microbiol Infect Dis 30: 53-59.
-
Alghaithy AA, Bilal NE, Gedebou M, Weily AH (2000) Nasal carriage and antibiotic resistance of Staphylococcus aureus isolates from hospital and non-hospital personnel in Abha, Saudi Arabia. Trans R Soc Trop Med Hyg 94: 504-507.
-
Goossens H, Ferech M, Vander Stichele R, Elseviers M, Group EP (2005) Outpatient antibiotic use in Europe and association with resistance: a cross-national database study. Lancet 365: 579-587.
-
Hsueh P-R, Chen W-H, Luh K-T (2005) Relationships between antimicrobial use and antimicrobial resistance in Gram-negative bacteria causing nosocomial infections from 1991-2003 at a university hospital in Taiwan. Int J Antimicrob Agents 26: 463-472.
-
Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, et al. (2012) Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clinical microbiology and infection 18: 268-281.
-
Bawazir SA (1993) Prescribing patterns of ambulatory care physicians in Saudi Arabia. Ann Saudi Med 13: 172-177.
-
Neyaz Y, Khoja T, Qureshi NA, Magzoub MA, Haycox A, et al. (2011) [Medication prescribing pattern in primary care in Riyadh City, Saudi Arabia]. East Mediterr Health J 17: 149-155.
-
Bawazir S (1992) Prescribing pattern at community pharmacies in Saudi Arabia.
-
Al-Ghamdi S, Gedebou M, Bilal NE (2002) Nosocomial infections and misuse of antibiotics in a provincial community hospital, Saudi Arabia. J Hosp Infect 50: 115-121.
-
Neuhauser MM, Weinstein RA, Rydman R, Danziger LH, Karam G, et al. (2003) Antibiotic resistance among gram-negative bacilli in US intensive care units: implications for fluoroquinolone use. JAMA 289: 885-888.
-
Balkhy HH, Bawazeer MS, Kattan RF, Tamim HM, Al Johani SM, et al. (2012) Epidemiology of Acinetobacter spp.-associated healthcare infections and colonization among children at a tertiary-care hospital in Saudi Arabia: a 6-year retrospective cohort study. Eur J Clin Microbiol Infect Dis 31: 2645-2651.
-
Mwanri L, AlSaleh E (2014) Multi-drug Resistant Organisms and Patients' Risk Factors in the Intensive Care Unit of King Fahad Hofuf Hospital, Saudi Arabia. IJHPR 2: 8-25.
-
Landelle C, Pagani L, Harbarth S (2013) Is patient isolation the single most important measure to prevent the spread of multidrug-resistant pathogens? Virulence 4: 163-171.
-
Maechler F, Pena Diaz LA, Schroder C, Geffers C, Behnke M, et al. (2015) Prevalence of carbapenem-resistant organisms and other Gram-negative MDRO in German ICUs: first results from the national nosocomial infection surveillance system (KISS). Infection 43:163-168.
-
Asghar AH, Faidah HS (2009) Frequency and antimicrobial susceptibility of gram-negative bacteria isolated from 2 hospitals in Makkah, Saudi Arabia. Saudi Med J 30: 1017-1023.
-
Zaidi AK, Huskins WC, Thaver D, Bhutta ZA, Abbas Z, et al. (2005) Hospital-acquired neonatal infections in developing countries. Lancet 365: 1175-1188.
-
Smolyakov R, Borer A, Riesenberg K, Schlaeffer F, Alkan M, et al.(2003) Nosocomial multi-drug resistant Acinetobacter baumannii bloodstream infection: risk factors and outcome with ampicillin-sulbactam treatment J Hosp Infect 54: 32-38.
-
Lee K, Kim M-N, Kim J-S, Hong HL, Kang JO, et al. (2011) Further increases in carbapenem-, amikacin-, and fluoroquinolone-resistant isolates of Acinetobacter spp. and P. aeruginosa in Korea: KONSAR study 2009 Yonsei Med J 52:793-802.
-
Manchanda V, Sanchaita S, Singh N (2010) Multidrug resistant acinetobacter. J Glob Infect Dis 2: 291-304.
-
Viehman JA, Nguyen MH, Doi Y (2014) Treatment options for carbapenem-resistant and extensively drug-resistant Acinetobacter baumannii infections. Drugs 74: 1315-1333.
-
Durante-Mangoni E, Signoriello G, Andini R, Mattei A, De Cristoforo M, et al. (2013) Colistin and rifampicin compared with colistin alone for the treatment of serious infections due to extensively drug-resistant Acinetobacter baumannii: a multicenter, randomized clinical trial. Clin Infect Dis 57: 349-358.
-
Karaiskos I, Galani L, Baziaka F, Giamarellou H (2013) Intraventricular and intrathecal colistin as the last therapeutic resort for the treatment of multidrug-resistant and extensively drug-resistant Acinetobacter baumannii ventriculitis and meningitis: a literature review. Int J Antimicrob Agents 41: 499-508.
-
Rahal JJ, Urban C, Horn D, Freeman K, Segal-Maurer S, et al. (1998) Class restriction of cephalosporin use to control total cephalosporin resistance in nosocomial klebsiella. JAMA 280: 1233-1237.
-
Botnarciuc M, Stan I, Ispas S (2015) Cephalosporin resistant bacterial strains isolated from respiratory infections. ARS Medica Tomitana 21: 7-11.
-
Livermore DM (2012) Fourteen years in resistance. Int J Antimicrob Agents 39: 283-294.
-
Huber CA, Sartor AL, McOdimba F, Shah R, Shivachi P, et al. (2014) Outbreaks of multidrug-resistant Acinetobacter baumannii strains in a Kenyan teaching hospital. J Glob Antimicrob Resist 2:190-193.
-
Lautenbach E, Patel JB, Bilker WB, Edelstein PH, Fishman NO (2001) Extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae: risk factors for infection and impact of resistance on outcomes. Clin Infect Dis 32:1162-1171.
-
Safdar N, Maki DG (2002) The commonality of risk factors for nosocomial colonization and infection with antimicrobial-resistant Staphylococcus aureus, enterococcus, gram-negative bacilli, Clostridium difficile, and Candida. Ann Intern Med 136: 834-844.
-
Aloush V, Navon-Venezia S, Seigman-Igra Y, Cabili S, Carmeli Y (2006) Multidrug-resistant Pseudomonas aeruginosa: risk factors and clinical impact. Antimicrob Agents Chemother 50: 43-48.
-
Bartlett JG, Gilbert DN, Spellberg B (2013) Seven ways to preserve the miracle of antibiotics. Clin Infect Dis 56: 1445-1450.