Journal of Infectious Diseases and Epidemiology
Bluetongue in Morocco 2004 to 2015: An Overview
Lhor Youssef1*, Kyriaki Nomikou2, Khayli Mounir1, Bouslikhane Mohammed3, Fassi Fihri Ouafaa3 and El Harrak Mehdi4
1National Office of Food Safety (ONSSA), Rabat, Morocco
2Vector-Borne Viral Diseases Programme, The Pirbright Institute, United Kingdom
3Institut Agronomique et vétérinaire Hassan II, Rabat, Morocco
4MCI Santé Animale, Morocco
*Corresponding author:
Lhor Youssef, National Office of Food Safety (ONSSA), Rabat, Morocco, E-mail: y.lhor@yahoo.fr
J Infect Dis Epidemiol, JIDE-3-023, (Volume 3, Issue 1), Overview; ISSN: 2474-3658
Received: June 29, 2016 | Accepted: February 15, 2017 | Published: February 18, 2017
Citation: Youssef L, Nomikou K, Mounir K, Mohammed B, Ouafaa FF, et al. (2017) Bluetongue in Morocco 2004 to 2015: An Overview. J Infect Dis Epidemiol 3:023. 10.23937/2474-3658/1510023
Copyright: © 2017 Youssef L, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Bluetongue (BT) is an infectious, arthropod borne viral disease of domestic and wild ruminants. BT is a notifiable disease of huge socio-economic concern and of major importance in the international trade of animals and animal products. Due to the segmented nature of genome, there is existence of high diversity in BT virus serotypes too as a consequence of genetic reassortments. Until 2004, BT was considered as an exotic disease in Morocco. However, from 2004 to 2015, at least two BTV serotypes (BTV-1 and BTV-4) were reported in the country. BTV-4 was introduced in September 2004 and produced an epizootic in northern Morocco but large vaccination campaigns with live attenuated vaccine contributed to the disease disappearance from the country. However, in 2006 and 2007, the widespread recrudescence and extension of a severe BTV infection in new areas with unfavorable environmental conditions suggest the introduction of a new serotype, BTV-1. Implementation of vaccination campaigns using a live bivalent BTV-1 and 4 vaccine allowed control of the disease, but, in 2009, BTV-4 re-emerged in the country after an epidemiological silence of 5 years, showing severe symptoms similar to BTV-1. Sequencing of the new BTV-4 virus showed a reassortant with at least 4 segments of BTV-1, likely to be a combination of the field virulent BTV-1 and BTV-4. The radial extension of BT infection across borders with the coexistence of many serotypes increases the risk for reassortment between different wild and attenuated serotypes, particularly those that occur in the Mediterranean Basin, where vector activity continues throughout the year. This review focuses on the current understanding, genetic diversity and transmission dynamics of BT, and the possible factors that may play a role in the spread of the virus in Morocco or neighboring countries.
Keywords
Morocco, Bluetongue, Serotype 1, Serotype 4, Culicoides spp
Introduction
With 18 million sheep, 5 million goats and 3.3 million cattle, livestock represents the main livelihood for the majority of the rural population in Morocco and plays a crucial role at both national and household levels. Over the year, Morocco enjoys a Mediterranean soft climate in the green belt part of the country between the Mediterranean Sea in the North, the Atlantic Ocean in the West and Atlas Mountains in the Southeast. Most of the human population and livestock are distributed in this northwestern area.
Bluetongue (BT) is an arthropod-transmitted viral disease of ruminants and certain other animals. BT infection of ruminants and vector Culicoides insects are enzootic throughout tropical and temperate regions of the world [1]. Bluetongue virus (BTV) is the type species of the genus Orbivirus, family Reoviridae [2]. Twenty-seven serotypes have been recognized for this virus [3-5]. The virus particle consists of ten segments of double-stranded RNA surrounded by three protein shells. Unlike other arbo viruses, BTV lacks a lipid envelope. The particle has a diameter of 86 nm. The structure of the 70 nm core was determined in 1998 and was at the time the largest atomic structure to be solved [6,7].
BT is a notifiable disease for the World Organization of Animal Health (WOAH) and is of considerable socio-economic concern and major importance in the international trade of animals and animal products. In recent years there have been drastic alterations in the global distribution of BTV infection, particularly since 1998 in Europe possibly due to global climate changes, travel and trade [8-13].
Since 1999 at least 7 different serotypes have invaded the West Mediterranean basin, BTV-1, -2, -4, -8, -9, -14 and -16 [14-20].
Serotype 2 caused outbreaks for the first time in Tunisia in areas North East of the country at the end of 1999 [21], and further spread in the eastern and central parts of the country as well as in other countries in North Africa during the following years. It also spread across the Mediterranean basin in the Balearic, Corsica, Sardinia, Sicily and southern Italy [14,17,18,22].
Concerning serotype 4, a different strain of BTV-4 was circulating in the eastern Mediterranean basin, was also reported and found to spread from North Africa to Spain, Sicily, Sardinia, Corsica, the Balearic and southern Italy in 2003-2005 [17,18,23]. The North Africa origin of serotype 4 was suspected but no cases reported from this region at this time.
In Morocco, before 2004, BT was considered an exotic disease. A limited incursion of BTV-10 occurred in 1956 in the southern area of Larache and West of Arbaoua from Portugal [17,24-27].
Outbreaks of BT appeared for the first time in 2004, caused by BTV-4 following the emergence and expansion of outbreaks caused by this serotype in several other countries of the West Mediterranean basin [17,18,23].
In 2006, the disease reappeared in the country with the introduction of a new serotype, BTV-1, from the eastern borders with Algeria [17,28,29]. Since then, the resurgence of the disease has taken a seasonal peak settled by favorable climatic factors of temperature and humidity to the abundance of Culicoides spp.
A vaccination program implemented in affected areas from 2004 to 2006, and a mass vaccination carried out throughout the country in 2007 and 2008, covering nearly 90% of the national herd. Despite the vaccination programs implemented, Morocco is now an endemic country with periodic reappearance of the disease, caused by at least 2 serotypes. The reemergence of BTV-4 in 2009 after 5 years silence raises questions regarding the disease pattern and the efficacy of the vaccination program.
In this paper, our aim is to provide a review of the epidemiological situation of BT in Morocco since its introduction in 2004. We provide a historic description of yearly reported outbreaks, the vaccination efforts to contain the virus and observed results with new distribution of circulating serotypes (Table 1). The capacity and competence of the Culicoides spp. vectors and an outline of the mode of entry and amplification mechanisms oh BTV are also discussed.
![]()
Table 1: Bluetongue epizootics in Morocco (2004-2014) with vaccination campaigns.
View Table 1
The BT situation before 2004
In 1956, a limited incursion of BT was reported in the southern area of Larache and West of Arbaoua due to BTV serotype 10. Typical clinical signs were observed only in adult sheep during this outbreak with an incidence of 20% [24]. The rapid implementation of a vaccination program using a polyvalent vaccine combined with unfavorable climatic conditions for the vectors halted the spread of the disease. The wind at this time could have carried offshore infected Culicoides midges from North Africa causing the simultaneous epidemic of BT in Portugal. Bluetongue had not been reported at that time in Morocco, but the presence of the virus in animals without clinical signs might be possible [30].
No clinical cases notified since then in Morocco. In 1999, following BT notification in Tunisia for the first time [31], and the spread of the virus (serotype 2) in the European Islands [22,32] Morocco straightened vigilance and implemented an epidemiological surveillance in the whole territory focusing on humid zones. Light traps have been installed in selected areas with an active surveillance program for the clinical disease.
Around 7040 bovine sera from 32 provinces were analyzed in 1999 using radial immuno diffusion test as described by Abu El Zein, 1984 showed 2% of positive sera over the country [33]. In 2001, a second survey was conducted analyzing 4000 sheep sera. Almost 8.7% of them tested positive. This increase in the percentage of BT positive sera was the first alert of the virus circulation in the country. No determination of virus serotypes was performed at that time.
The BTV-4 outbreak of 2004
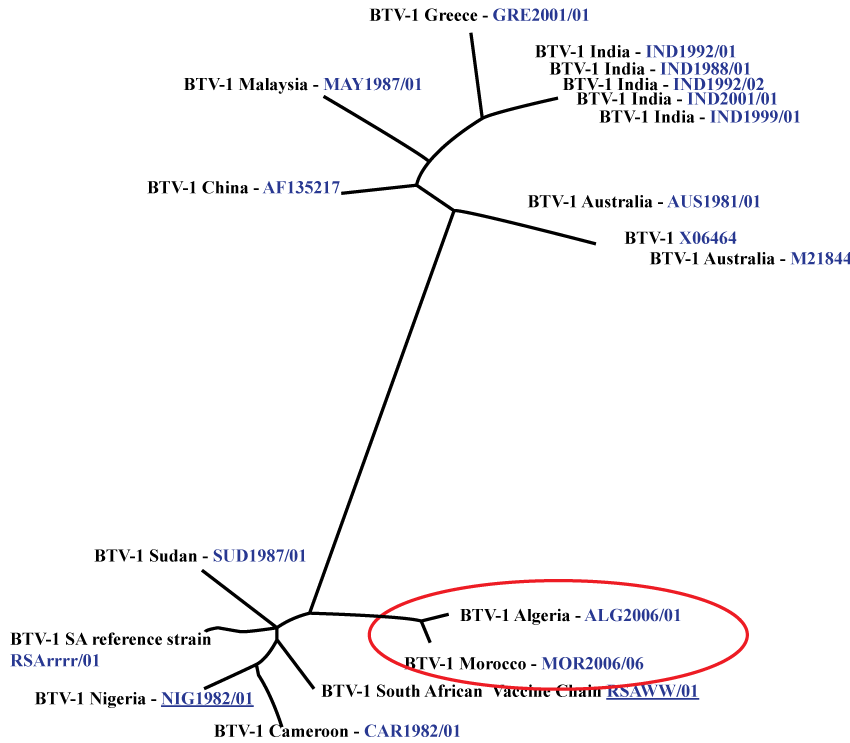
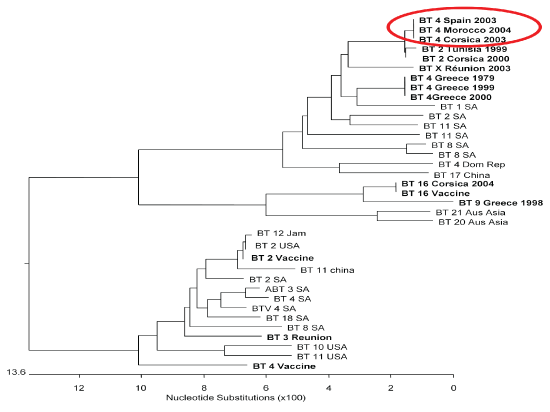
After many decades of epidemiological silence, several outbreaks of BT were reported in 2004 in sheep flocks in Larache province at the Northwest Atlantic littoral of Morocco [34]. The virus spread to central region causing over 230 outbreaks with morbidity and mortality rates of 8.2% and 1.3% respectively (Figure 1). Clinical symptoms were observed in adult sheep with hyperthermia, nasal discharge, salivation, oedema and congestion of the mucous membranes. The epidemic lasted from September 3rd to November 22nd 2004. BTV-4 was laboratory confirmed, and based on its genetic similarity with European isolates and epidemiological modeling, it is reasonable to predict that the virus most likely originated from Spain or Corsica (Figure 2).
.
Figure 2: Phylogenetic relationships: Nucleotide sequences (segment 10, NS3, GenBank).
View Figure 2
No clinical disease was recorded in cattle. In 2005 a serological survey conducted in the infected area using an ELISA test (commercial VRMD kit) showed 60% positivity among the ruminant population in the infected areas and 23% positivity in the non-infected areas. The high percentage of seropositive of BTV in cattle (87% in the affected zone and 44% in the non-affected zone) and goats (79% in the affected zone and 32% in the non-affected zone) suggest that these two ruminant species may play a role as reservoir of the virus in the country. Seropositivity in sheep population was lower, 59% in the affected zone and 15% in the other non-affected areas (Table 2).
![]()
Table 2: Serological survey for bluetongue conducted on livestock population of Morocco in 2005.
View Table 2
A vaccination program was implemented in the affected zone using a monovalent live attenuated vaccine serotype 4 produced locally in Rabat. Around 5.5 million doses were applied in 2005 and 7.8 million doses in 2006 with a respective coverage rate of 76% and 98% of the susceptible sheep population in the affected zone. Cattle and goat populations were not included in this campaign. This vaccination effort allowed eradication of the disease since no BTV-4 circulation was detected in the country until 2009. Despite close and active virological and serological surveillance, testing of the unvaccinated flocks by group-specific real-time RT-PCR [35] or ELISA (commercial VRMD kit) was also carried out for efficient control and management of BTV infection.
BTV-1 outbreak of 2006-07
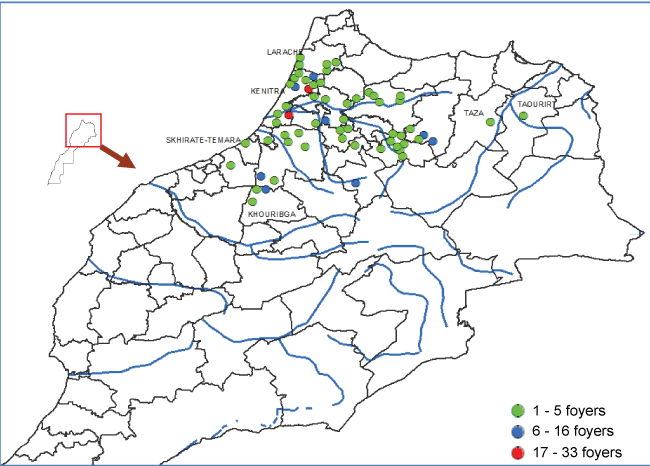
However, in September 2006, BTV-1 was isolated from flocks of sheep presenting typical symptoms of BT in the Eastern part of the country, near the Algerian borders. The isolated virus was genetically identical to the strain identified in Algeria few months before [29] (Figure 3). In the following weeks, the virus spreaded across Morocco, causing, 505 outbreaks in the small ruminant population (Figure 4). The reported disease seemed to be clinically more severe than the previous BT outbreak due to serotype 4. The disease entered Morocco likely from Algeria through smuggling of infected sheep or culicoides insect vector transported by wind.
The newly introduced BTV-1 serotype distributed in the northwestern part of Morocco with 2 peaks registered early October and mid-December 2006. Due to mild winter, the disease reemerged in the country as soon as May 2007. It caused huge economic losses because of severe clinical symptoms in a naive population living even in altitudes of Atlas Mountains, in zones never affected in the past. The epizootic lasted till November 2007 in the southeast with 1076 notified outbreaks, 5222 affected sheep and 2180 reported mortality (Figure 5). Morbidity registered rate in 2007 was 3.9% against 2.6% in 2006 and mortality rate 1.6% in 2007 against 0.7% in 2006.
A compulsory vaccination campaign was initiated among small ruminants population using a bivalent BTV-1 and -4 live attenuated vaccine, locally produced, allowed control of the disease since no BT clinical cases were reported in 2008. However, virological surveillance using group-specific real-time RT-PCR [35] detected circulation of BTV-1 in two sentinel cattle in the eastern region of the country.
Re-emergence of the reassortant BTV-4 in 2009
Because of the occurrence of PPR (Peste des Petits Ruminants) for the first time in Morocco in 2008 [36], all vaccination efforts of the authorities where focused to eradicate the new pathogen. For BT prevention, measures were limited to perifocal vaccination in high-risk areas, associated with disinsectization of farms and prevention of vector contact with susceptible animals.
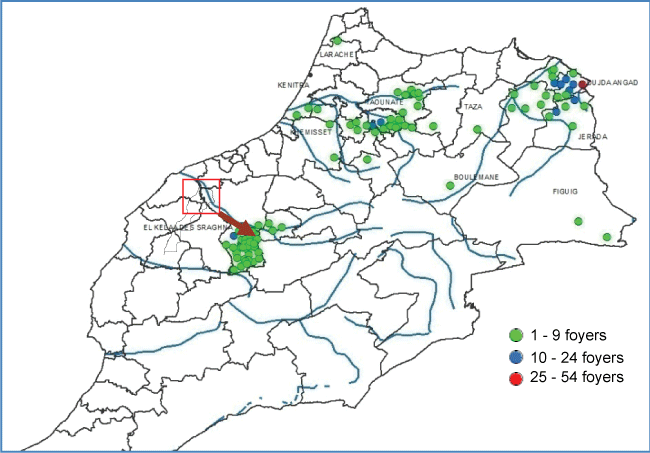
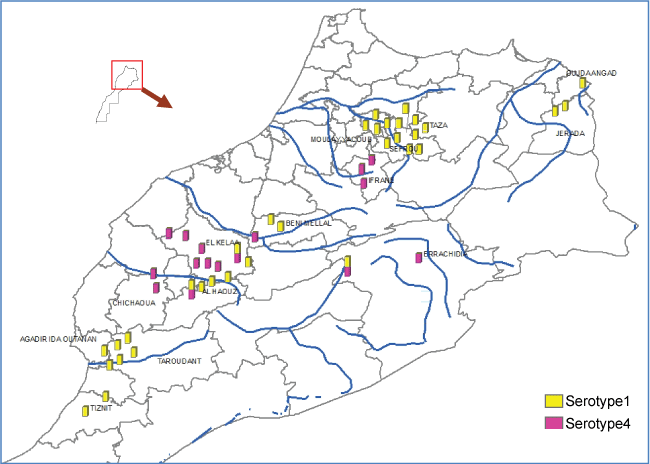
In 2009, clinical disease in sheep was highly reported in several location in the North and few in the South of the country. The northern outbreaks were caused by BTV-1, but in the South, circulation of BTV-4 was laboratory confirmed for the first time since November 2004 (Figure 6). Flocks with mixed infection has also been detected by real time PCR (qPCR) in Marrakech region as well as few clinical cases in cattle, probably due to BTV-8 in Beni Mellal region as reported by [37].
For BTV-1, all of the genome segments that have been sequenced from previous strains from the region, are from a single western lineage represented by MOR2006/06 (Table 3). For BTV-4, the evidence from full sequence analysis suggests that MOR2009/09 is a reassortant strain (BTV-4R), containing sequences from previous BTV-1 and BTV-4 strains that spread in Morocco and neighboring regions. However, since both Seg-2 and Seg-6 (encoding outer capsid proteins VP2 and VP5) are from BTV-4, the BTV-4 serotype remain unchanged (Table 4).
![]()
Table 3: Similarity between BTV-1 (MOR 2009) and other BTV-1 strains.
View Table 3
![]()
Table 4: Similarity between BTV4 (MOR 2009) and previous strains of BTV-1 and 4 from Morocco showing evidence of reassortment.
View Table 4
The new BTV-4 reassortant showed similar clinical severity as BTV-1 in the field, which was much more pathogenic compared to the previous disease due to BTV-4 in 2004. Emergency perifocal vaccination carried out with the live attenuated bivalent vaccine allowed control of the virus spread. However, the disease re-emerged in 2010 dominated by BTV-4 reassortant in the North and few outbreaks due to BTV-1 in the South, exactly the opposite of the situation observed in 2009. In accordance with [38] observations, it seems that the two serotypes affected the vector favorable areas alternatively in the North and in the South of the country. This observation may be explained by the active immunity of the population after BT outbreaks and vaccination.
Full genome sequencing of the BTV-4 isolate of 2010 showed presence of segments from three serotypes 1, 4 and 8 which confirm circulation of a third serotype in the country which is BTV-8. This circulation is not accompanied with clinical cases in cattle as seen in Europe during recent notified outbreaks in this continent.
The current situation
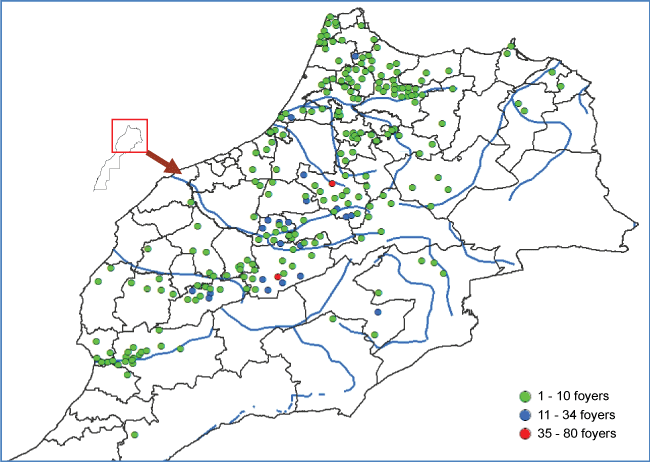
Between 2009 and 2014, the epidemiological situation remained identical, BTV-1 and BTV-4 continued to co-circulate with an average of 166 outbreaks reported annually. The government implemented several control measures with active surveillance, perifocal vaccination (about 3 km around the outbreak) and other sanitary measures.
In 2015, BT clinical cases were confirmed in several regions dominated by BTV-1 serotype. The qRT-PCR results confirmed the disease in 2193 sheep belonging to 526 premises among 30 provinces throughout the country. The average morbidity rate is 2.45% and mortality rate is 0.67%.
Interesting to note that a great difference in sheep's breed susceptibility was observed mainly during the 2007 outbreaks. In the eastern part of the country where Beniguil sheep breed is dominant, registered morbidity rate was 1.8% and mortality 0.3%.
In the Atlas Mountains, the Timahdit sheep breed presented 4.7% morbidity and 1.3% mortality while the Sardi sheep at the Atlantic littoral reported 3.7% morbidity and 2.1% mortality. The Sardi and Timahdit sheep breeds seem to be more sensitive to BTV infection than the other breeds present in the country.
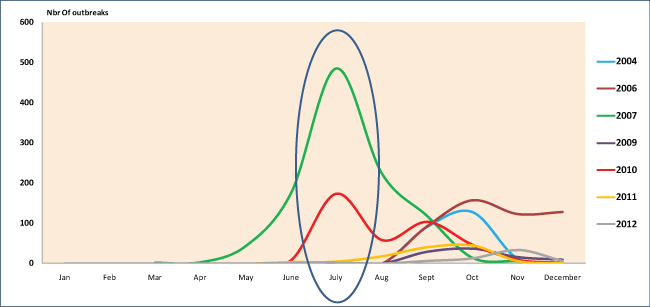
The seasonal effect of BTV in Morocco was highlighted especially around the summer period in 2007 and 2010 and autumn period in 2004, 2006, 2009, 2011 and 2012 (Figure 7). Both periods coincide with peaks of vector activity validated by the entomological surveillance of BT vectors, implemented at the country level. The main vector was reported to be Culicoides imicola. The involvement of a novel vector was subsequently suspected because of the high density of the Culicoides obsoletus in the North of Morocco where BT is regularly reported. This finding was further supported by the fact that the disease penetrated into new areas in altitude where Culicoides obsoletus is dominant [39].
Vector species of Culicoides spp in Morocco - entomological surveillance
Following the epizootic of African Horse Sickness in 1966 and 1989 in Morocco [25,40-45] extensive field studies were conducted that identified 49 species of Culicoides spp. Culicoides imicola has been first identified in 1968 [46]. In 1994-1995 a total of 3887 light trap samples were collected from 22 sites distributed all over the country [45]. Culicoides imicola was found to be widely distributed with the greatest catches in the low-lying northwestern areas between Tangier and Rabat and in Marrakech region. In general, the numbers of Culicoides imicola peaked in late summer and autumn, with a smaller peak in spring [44].
In 2001-2002, a trapping program was carried out to update the distribution of Culicoides spp. More than 44 sites have been sampled across Morocco. The study showed practically the same distribution of the vector [47]. From April 2009 to March 2010, a second round of vector traping design was carried out at 14 sites all over the country. The average collected midges at the central regions were 2.1 times higher than those collected at the southeastern areas and 2.6 times higher than northern regions. Culicoides identified were belonging to 10 different species namely: C. imicola, C. pulicaris, C. newsteadi, C. punctatus, C. nubeculosus, C. circumscriptus, C. newsteadi, C. oxystoma, C. obsoletus and C. helviticus. Culicoides imicola were abundant in the northern center areas of the country. Overall, the result indicated that the two most abundant Culicoides species were Culicoides imicola (94.2%-95.85%) and Culicoides newsteadi (2.21%-2.72%). Interestingly, Culicoides newsteadi is also present in abundance at the same locations as the C. imicola.
The rate of prevalence of these species was bi-phasic and higher from April to June and October to November. Noteworthy that the infectivity rate of BTV was also higher during those periods indicating that high risk of infection seems to be proportional to the vectors abundance [39].
Conclusions
BTV is currently endemic in Morocco, at least 2 serotypes (BTV-1 and BTV-4) have been continuously present since 2006 in different parts of the country with variable incidences. Although the imminent risk, the circumstances which led to endemicity in Morocco are largely unchanged; limited control of movements of the susceptible livestock and lack of an efficient and sustainable preventive vaccination program, mean that recrudescence of the disease is a real possibility. In addition, poor vaccination coverage, the use of live attenuated vaccines with the coexistence of more than one virulent field strains causes the emergence of new reassortants with different pattern.
Live attenuated BTV vaccines cause significant levels of viraemia post vaccination, leading to infection of Culicoides and onward virus-transmission [48-51]. Monitoring the field situation is therefore essential to identify BTV circulating strains.
The emergence of BT into new areas never affected before, is mainly attributed to climatic changes and linked to the northern expansion of the major vector Culicoides imicola. Additionally, novel vector species of Culicoides within the obsoletus and pulicaris complexes may have been involved.
References
-
Roy P (1992) Bluetongue virus proteins. J Gen Virol 73: 3051-3064.
-
Mertens MS, Samuel A, Attoui H (2005) Orbiviruses, Reoviridae. In: Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA, Virus Taxonomy Eighth Report of the International Committee on Taxonomy of Viruses. London, Elsevier/Academic Press, USA, 466.
-
Hofmann MA, Renzullo S, Mader M, Chaignat V, Worwa G, et al. (2008) Genetic characterization of toggenburg orbivirus, a new bluetongue virus, from goats, Switzerland. Emerg Infect Dis 14: 1855-1861.
-
Maan S, Maan NS, Nomikou K, Veronesi E, Bachanek-Bankowska K, et al. (2011) Complete genome characterisation of a novel 26th bluetongue virus serotype from Kuwait. PLoS One 6: e26147.
-
Zientara S, Sailleau C, Viarouge C, Höper D, Beer M, et al. (2014) Novel bluetongue virus in goats, Corsica, France, 2014. Emerg Infect Dis 20: 2123-2125.
-
Verwoerd DW, Louw H, Oellermann RA (1970) Characterization of bluetongue virus ribonucleic acid. J Virol 5: 1-7.
-
Grimes JM, Burroughs JN, Gouet P, Diprose JM, Malby R, et al. (1998) The atomic structure of the bluetongue virus core. Nature 395: 470-478.
-
Maclachlan NJ, Guthrie AJ (2010) Re-emergence of bluetongue, African horse sickness, and other orbivirus diseases. Vet Res 41: 35.
-
Maclachlan NJ, Wilson WC, Crossley BM, Mayo CE, Jasperson DC, et al. (2013) Novel serotype of bluetongue virus, western North America. Emerg Infect Dis 19: 665-666.
-
Purse BV, Mellor PS, Rogers DJ, Samuel AR, Mertens PP, et al. (2005) Climate change and the recent emergence of bluetongue in Europe. Nat Rev Microbiol 3: 171-181.
-
Purse BV, Brown HE, Harrup L, Mertens PP, Rogers DJ (2008) Invasion of bluetongue and other orbivirus infections into Europe: the role of biological and climatic processes. Rev Sci Tech 27: 427-442.
-
C Saegerman, M Hubaux , B Urbain, L Lengelé, D Berkvens (2007) Regulatory aspects concerning temporary authorisation of animal vaccination in case of an emergency situation: example of bluetongue in Europe. Rev. sci. tech. Off. int. Epiz
-
Randolph SE, Rogers DJ (2010) The arrival, establishment and spread of exotic diseases: patterns and predictions. Nat Rev Microbiol 8: 361-371.
-
Mellor PS, Wittmann EJ (2002) Bluetongue virus in the Mediterranean Basin 1998-2001. Vet J 164: 20-37.
-
Mellor PS, Carpenter S, Harrup L, Baylis M, Mertens PP (2008) Bluetongue in Europe and the Mediterranean Basin: history of occurrence prior to 2006. Prev Vet Med 87: 4-20.
-
Saegerman C, Berkvens D, Mellor PS (2008) Bluetongue epidemiology in the European Union. Emerg Infect Dis 14: 539-544.
-
Mellor PS, Carpenter S, Harrup L, Baylis M, Wilson A, et al. (2009) Bluetongue in Europe and the Mediterranean Basin. In: Mellor PS, Baylis M, Mertens PPC editors. Bluetongue monograph. (1st edn), Elsevier/Academic Press, London, USA, 235-264.
-
Wilson AJ, Mellor PS (2009) Bluetongue in Europe: past, present and future. Philos Trans R Soc Lond B Biol Sci 364: 2669-2681.
-
García-Bocanegra I, Arenas-Montes A, Lorca-Oró C, Pujols J, González MA, et al. (2011) Role of wild ruminants in the epidemiology of bluetongue virus serotypes 1, 4 and 8 in Spain. Vet Res 42: 88.
-
Lorca-Oró C, Pujols J, Arenas A, Gómez-Guillamón F, Zorrilla I, et al. (2011) Epidemiological surveillance of bluetongue virus serotypes 1, 4 and 8 in Spanish ibex (Capra pyrenaica hispanica) in southern Spain. Vet Microbiol 149: 230-235.
-
Hammami S (2004) North Africa: a regional overview of bluetongue virus, vectors, surveillance and unique features. Veterinaria Italiana 40: 43-46.
-
Zientara S, de La Rocque S, Gourreau JM, Gregory M, Diallo A (2000) Epidemiol. Sante Aim 38: 133-144.
-
Breard E, Sailleau C, Nomikou K, Hamblin C, Mertens PP, et al. (2007) Molecular epidemiology of bluetongue virus serotype 4 isolated in the Mediterranean Basin between 1979 and 2004. Virus Res 125: 191-197.
-
Placidi L (1957) La bluetongue au Maroc. Vigot Freres, Eds Bull Acad Vet, XXX, 79- 84.
-
Mellor PS, Boorman J (1995) The transmission and geographical spread of African horse sickness and bluetongue viruses. Ann Trop Med Parasitol 89: 1-15.
-
Manso-Ribeiro J, Rosa-Azevedo J, Noronha F, Braco-Forte-Junior M, Vasco-Fernandez M (1957) Fievre catarrhale du mouton (bluetongue). Bulletin de l'Office International des Epizooties 48: 350-367.
-
Gibbs EP, Greiner EC (1994) The epidemiology of bluetongue. Comp Immunol Microbiol Infect Dis 17: 207-220.
-
WOAH (2006) Bluetongue in Morocco. OIE Disease Information, Wahid.
-
Cêtre-Sossah C, Madani H, Sailleau C, Nomikou K, Sadaoui H, et al. (2011) Molecular epidemiology of bluetongue virus serotype 1 isolated in 2006 from Algeria. Res Vet Sci 91: 486-497.
-
Sellers RF, Pedgley DE, Tucker MR (1978) Possible windborne spread of bluetongue to Portugal, June-July 1956. J Hyg (Lond) 81: 189-196.
-
Hammami S, Ben Said M.S & Bahri S (2000). La fièvre catarrhale du mouton : éléments de diagnostic et moyens de lutte. BEIV 17: 3-6.
-
De Diego AC, Sánchez-Cordón PJ, Sánchez-Vizcaíno JM (2014) Bluetongue in Spain: From the First Outbreak to 2012. Transbound Emerg Dis 61: e1-e11.
-
Abu Elzien (1984) Rapid detection of bluetongue virus antigen in the sera and plasma of camel, sheep and cattle in the Sudan using the gel immune diffusion test. Archives of Virology 79: 131-134.
-
WOAH (2004) Bluetongue in Morocco. OIE Disease Information, Handistatus.
-
Shaw AE, Monaghan P, Alpar HO, Anthony S, Darpel KE, et al. (2007) Development and initial evaluation of a real-time RT-PCR assay to detect bluetongue virus genome segment 1. J Virol Methods 145: 115-126.
-
WOAH (2008) Bluetongue in Morocco. OIE Disease Information, Wahid.
-
Drif Kamar, Fassi Fihri Ouafaa, Loutfi Chafiqa, Sebbar Ghizlane, Chaffai Nadia, et al. (2013) Phylogeny of BTV in Morocco: determination of new reassortants.15th International Congress of Immunology (ICI), Milan, Italy, 22-27.
-
Drif Kamar, Elharrak Mehdi, Loutfi Chafiqa, Fassi Fihri Ouafaa (2015) Bluetongue Virus in Morocco from 2004-2012. Journal of Animal Health and Production 3: 51.
-
Youssef Lhor, Mounir Khayli, Mohamed Bouslikhane, Mehdi El Harrak, Ouafaa Fassi Fihri (2015) Spatial and seasonal distribution of Culicoides species in Moroccoin relation to the transmission of bluetongue viruses. British Journal of Virology 2: 88-95.
-
Fassi-Fehri M, Mouslifi M, Nguyen Thanh Cac (1967) [On various aspects of African horse sickness in Morocco]. Maroc Med 47: 112-119.
-
RF Sellers, DE Pedgley, MR Tucker (1977) Possible spread of African horse sickness on the wind. J Hyg (Lond) 79: 279-298.
-
Baylis M, el Hasnaoui H, Bouayoune H, Touti J, Mellor PS (1997) The spatial and seasonal distribution of African horse sickness and its potential Culicoides vectors in Morocco. Med Vet Entomol 11: 203-212.
-
Baylis M, Bouayoune H, Touti J, El Hasnaoui H (1998) Use of climatic data and satellite imagery to model the abundance of Culicoides imicola, the vector of African horse sickness virus, in Morocco. Med Vet Entomol 12: 255-266.
-
Bouayoune H, Touti J, el Hasnaoui H, Baylis M, Mellor PS (1998) The Culicoides vectors of African horse sickness virus in Morocco: distribution and epidemiological implications. Arch Virol Suppl 14: 113-125.
-
Baylis M, Rawlings P (1998) Modelling the distribution and abundance of Culicoides imicola in Morocco and Iberia using climatic data and satellite imagery. Arch Virol Suppl 14: 137-153.
-
Bailly-Choumara, HM Kremer (1970) "Deuxième contribution à l'étude des Culicoides du Maroc (Diptera, Ceratopogonidae)." Cahiers de l'ORSTOM, série entomologie médicale et parasitologie 8: 383-391.
-
Lhor Y (2014) The economic impact of bluetongue in northern Africa. Bluetongue and related orbiviruses, IV international conference, Rome, 5-7 November, 2014.
-
Batten CA, Maan S, Shaw AE, Maan NS, Mertens PP (2008) A European field strain of bluetongue virus derived from two parental vaccine strains by genome segment reassortment. Virus Res 137: 56-63.
-
Veronesi E, Darpel KE, Hamblin C, Carpenter S, Takamatsu HH, et al. (2010) Viraemia and clinical disease in Dorset Poll sheep following vaccination with live attenuated bluetongue virus vaccines serotypes 16 and 4. Vaccine 28: 1397-1403.
-
Ferrari G, De Liberato C, Scavia G, Lorenzetti R, Zini M, et al. (2005) Active circulation of bluetongue vaccine virus serotype-2 among unvaccinated cattle in central Italy. Prev Vet Med 68: 103-113.
-
Kyriaki Nomikou, Joseph Hughes, Rachael Wash, Paul Kellam, Emmanuel Breard, et al. (2015) Widespread Reassortment Shapes the Evolution and Epidemiology of Bluetongue Virus following European Invasion. PLoS Pathog 11: e1005056.