Journal of Infectious Diseases and Epidemiology
Features and Prognostic Factors of Tuberculous Meningitis in a Tertiary Hospital in Malaysia
Chia Peck Kee1*, Petrick Periyasamy2, Zhe Kang Law2, Norlinah Mohamed Ibrahim2, Wan Nur Nafisah WanYahya2, Hazlina Mahadzir2, Shahrul Azmin Md Rani2, Ramesh Sahathevan2, Rabani Remli2, Raymond Azman Ali2, Hui Jan Tan2, Syazarina Sharis Osman3, Shahizon Azura Mohamed Mukari3 and Shamsul Azhar Shah4
1Department of Medicne, Kuala Lumpur General Hospital, Malaysia
2Department of Medicine, Universiti Kebangsaan Malaysia Medical Centre, Malaysia
3Department of Radiology, Universiti Kebangsaan Malaysia Medical Centre, Malaysia
4Department of Community Health, Universiti Kebangsaan Malaysia Medical Centre, Malaysia
*Corresponding author:
Chia Peck Kee, Mmed, Kuala Lumpur General Hospital, Jalan Pahang, 50586 Wilayah Persekutuan Kuala Lumpur, Malaysia, Tel: +60126619048, E-mail: peckkeec@gmail.com
J Infect Dis Epidemiol, JIDE-3-028, (Volume 3, Issue 1), Research Article; ISSN: 2474-3658
Received: October 22, 2016 | Accepted: March 14, 2017 | Published: March 17, 2017
Citation: Kee CP, Periyasamy P, Law ZK, Ibrahim NM, WanYahya WNN, et al. (2017) Features and Prognostic Factors of Tuberculous Meningitis in a Tertiary Hospital in Malaysia. J Infect Dis Epidemiol 3:028. 10.23937/2474-3658/1510028
Copyright: © 2017 Kee CP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Tuberculous meningitis (TBM) has been one of the major extrapulmonary manifestation with high morbidity and mortality. This paper aims to study the clinical features, prognostic factors and clinical outcome of TBM in a local tertiary hospital. This was an observational study on TBM in patients who were diagnosed in a local tertiary hospital. The patients’ demographic data, medical history, clinical presentation at admission, radiological and microbiological data was reviewed along with the clinical course, treatment and outcome. Sixty one patients were recruited with 37 (60.7%) males and 24 (39.3%) females, with the mean age of 47.4 (SD 18.165). The main presenting features were fever (55.7%), headache (78.7%) and altered conscious level (78.7%). The most common imaging finding was hydrocephalus (43.3% in CT brain and 26.2% in MRI brain), followed by leptomeningeal enhancement (20% in CT brain and 42.9% in MRI brain) and infarct (21.7% in CT brain, 35.7% in MRI brain). The median total antitubercular therapy which patients received was 336 (IQR 332) days. There were 86.9% patients who survived the hospital admission, while 70.5% survived the 6 months follow-up period. Factors associated with poor prognosis were intubation on presentation, surgical intervention (either ventricular-peritoneal shunting or external ventricular drain), elevated CSF protein and presence of tuberculoma in cerebral imaging. This study highlights the importance of recognizing the clinical features and prognostic factors in TBM. A high index of clinical suspicion should always be maintained and supported by cerebrospinal fluid analysis and radiological features.
Keywords
Tuberculous meningitis, Tuberculosis, Meningitis, Malaysia
Introduction
In 2013, it was reported that almost 9 million people had Tuberculosis (TB) globally [1]. The largest number of new TB cases was seen in South East Asia and Western Pacific Regions, accounting 56% of new cases globally. According to WHO report, there were estimated 1,072,931 deaths due to TB (excluding HIV) and 361,774 estimated deaths due to TB/HIV co-infection [1] globally from year 1999 to 2013. More than 95% of TB deaths occurred in low and middle income countries, with South East Asia and Africa Regions topped the chart.
Amongst the reasons for the increment are the burden of HIV, influx of immigrants with the disease and the occurrence of non-communicable diseases such as diabetes mellitus, chronic lung disease and malignancy. Tuberculous meningitis remains one of the major extrapulmonary involvements that carry a high morbidity and mortality. Delay in the diagnosis and the initiation of treatment are the main contributing factors to the morbidity and mortality.
Patients typically present with fever, headache, irritability, neck stiffness, and vomiting; though often has non-specific presentations especially in the early stages [2]. The diagnosis TBM is also supported by cerebrospinal fluid analysis and radiological brain imaging. However, the yield from bacteriological and radiological studies can be low in some cases, thus making the diagnosis of TBM more challenging. The prognosis of TBM largely depends on comorbidities, neurological status at the time of presentation, and treatment initiation.
Many studies have looked at the clinical presentation [2-5] and factors associated with the outcome [5-9] of TBM. But limited literature of TBM in South East Asia particularly addressing the burden, features and clinical predictors of the disease. This paper aims to study the clinical features, prognostic factors and clinical outcome of TBM in a local tertiary hospital.
Methods
This was an observational study on Tuberculous Meningitis (TBM) in patients who were diagnosed in a local tertiary hospital, University Kebangsaan Malaysia Medical Center (UKMMC) from 1st January 2003 to 28th February 2015. The study was approved by the UKMMC Ethics & Research Committee (Ethic Code FF-2014-336). Patients more than 18-years-old with the diagnosis of TBM were included in the study and we excluded those patients with neurodegenerative disorders and diagnosis of other nervous system infections.
The patients demographic data, medical history, clinical presentation at admission, radiological and microbiological data was reviewed along with the clinical course, treatment and outcome. Case definition for tuberculous meningitis included definite, probable and possible, which was based on the Lancet consensus scoring system [2]. The diagnosis of probable or possible tuberculous meningitis was determined by a diagnostic scoring system which included the symptoms and signs of meningitis, cerebrospinal fluid (CSF) analysis, cerebral imaging criteria and evidence of tuberculosis other than CNS, which was tabulated in table 1.
![]()
Table 1: The lancet concensus scoring system [2].
View Table 1
The diagnosis of definite tuberculous meningitis includes the clinical criteria and a positive acid-fast bacilli or Mycobacterium tuberculosis in CSF or positive Mycobacterium nucleic acid amplification test or histological examination. Probable tuberculous meningitis was made from the clinical entry criteria plus a total diagnostic score of twelve points (if cerebral imaging was available), and ten points (if cerebral imaging was not available). At least two points should either come from CSF or cerebral imaging criteria. Possible tuberculous meningitis included clinical criteria, plus a total diagnostic score of six to nine points (when cerebral imaging is not available) or six to eleven points (when cerebral imaging is available). Possible tuberculosis cannot be diagnosed or excluded without doing a lumbar puncture or cerebral imaging.
The neurological status of patients were also classified according to the British Medical Research Council (BMRC) staging system, which grades TBM as follows: grade I, patient with non-specific symptoms and signs, no clouding of consciousness, and no neurologic deficits; grade II, patient with lethargy or behavioral changes, meningeal irritation, or minor neurologic deficits such as cranial nerve palsies; and grade III, patient with stupor or coma, abnormal movements, seizures or severe neurologic deficits such as paresis.
The magnetic resonance imaging (MRI) of the brain was performed with 3.0 - Tesla Signa HDX MR system and computed tomogram (CT) of the brain was performed using a 64-slice Somatom sensation Siemens.
The outcome of patients was taken at the time of discharge from hospital. Patients who were alive on discharge were followed up until a definite outcome was recorded. Phone calls were made for patients who defaulted follow up. The neurological status of patients was assessed using modified Rankin scale (MRS). The grading was as follow; 0: no symptoms, 1: no significant disability, despite symptoms; able to perform all usual duties and activities, 2: Slight disability; unable to perform all previous activities but able to look after own affairs without assistance, 3: moderate disability; requires some help, but able to walk without assistance, 4: moderately severe disability, unable to walk without assistance and unable to attend to own bodily needs without assistance, 5: Severe disability; bedridden, incontinent, and requires constant nursing care and attention.
Statistical Analysis
Non normally distributed parameters were analyzed with non parametric tests (Mann-Whitney test) for quantitative variables. Kruskal-Wallis H test were used to determine if there were statistically significant differences between three groups of an independent variable on a continuous or ordinal dependent variable. Qualitative data were evaluated using chi-square test, and p value < 0.05 was taken as statistically significant. The outcome of patients was investigated by Kaplan-Meier estimates, and univariate cox regression analysis was used to identify the prognostic factors of death. The results were analysed using Statistical Product and Service Solutions (SPSS) version 22.0 [10].
Results
Seventy-eight patients were diagnosed and treated for tuberculous meningitis from year 2003 to February 2015. Cerebrospinal fluids analysis were not obtained in 8 patients, four patients did not fulfil the Lancet Concensus Scoring System, and 9 patients had final diagnoses consisting of cerebral toxoplasmosis, recurrent brain tumor and lymphoma, resulting in 61 patients in this study.
Table 2 showed that 37 (60.7%) males and 24 (39.3%) females had tuberculous meningitis, with the mean age of 47.4 (18.165). A total of 54 patients were natives, whereas seven were foreigners. Majority were Malays (59%), followed by Chinese (26.2%), Burmese (6.6%), Indonesian (4.9%) and Indian (3.3%). Fifty-four (88.5%) patients had no evidence of HIV infection. According to the Lancet Consensus Scoring system for TBM, 40 patients (65.6%) were classified as possible TBM, 11 patients with probable TBM while 10 patients (16.4%) had a definite TBM diagnosis.
![]()
Table 2: Demographic characteristics of study population.
View Table 2
The mean duration of symptoms prior to presentation were 7 (11) days. Thirty-four (55.7%) patients presented with fever, with the median duration of 3 (7) days. Mean body temperature on presentation was 37.7 (0.92) degree celcius. A total of 48 (78.7%) of patients presented with headache and altered conscious level. The median of GCS score was 13 (IQR 3) and 36 (59%) patients had a mild GCS (GCS > 12). Forty-four (72.1%) patients presented with focal neurological signs based on the British Medical Research Council for tuberculous meningitis. Neck stiffness is commonly found in 43 (70.5%) patients in this study.
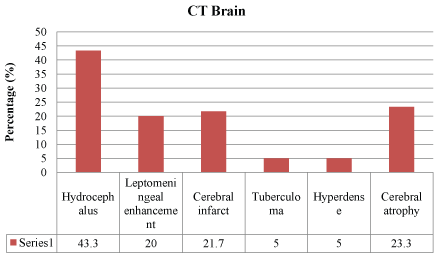
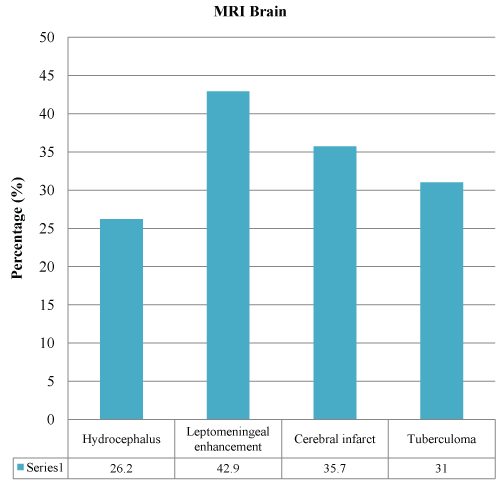
Forty-seven (77%) of the CSF color was clear as compared to 14 (23%) which were turbid. In the CSF analysis, CSF protein was more than 1000 mg/L in 62.3% of the patients, and CSF/serum glucose ratio was 0.37 (0.207) in mean (SD). Forty-five patients (73.7%) had low CSF/serum glucose ratio of less than 0.5. Polymorphonuclear leucocytes (72.1%) were not present in most of the CSF samples, and lymphocytes were detected predominantly in 26.2% of cases. Only one (1.6%) CSF sample yielded acid fast bacilli, and two (3.3%) patients had Mycobacterium tuberculosis in the CSF culture. Six (9.8%) patients had positive MTB PCR from the cerebrospinal fluid samples. The most identified finding in cranial imaging was hydrocephalus (43.3% in CT brain and 26.2% in MRI brain), followed by leptomeningeal enhancement and infarct. There was 26 (43.3%) hydrocephalus, 13 (20%) cerebral infarct and 12 (20%) leptomeningeal enhancement identified from the 60 patients who did CT brain (Figure 1). Out of the 42 patients who had MRI brain, 18 (42.9%) leptomeningeal enhancements, 15 (35.7%) cerebral infarct and 11 (26.2%) hydrocephalus were identified (Figure 2).
All patients had chest radiograph, 27 (44.3%) chest radiographs had abnormal changes: miliary (4.9%), cavitatory (6.6%), effusion (6.6%), or consolidation (37.7%).
Forty-five (73%) of the patients were given the classical regimen without Streptomycin consisting Isoniazid (5 mg/kg), Rifampicin (10 mg/kg), Pyrazinamide (20 mg/kg) and Ethambutol (15 mg/kg), while 12 (19.2%) patients had Streptomycin (15 mg/kg) as they could not tolerate the classical regimen.
Out of 61 patients, 18 (29.5%) had antitubercular therapy for 9-12 months, 3 (4.9%) patients received antitubercular for 12 months, and another 3 (4.9%) for 12-15 months, 10 (16.4%) had antitubercular therapy for more than 15 months (15-24 months). The median total antitubercular therapy which patients received was 336 (IQR 332) days. Seven (11.5%) patients with TBM were diagnosed with HIV. One out of 7 was an existing HIV patients who was already on ART (antiretroviral therapy) prior diagnosis of TBM.
Fifty (82%) patients were discharged alive, and 11 (18%) died. During the 6th and 12th month follow-up period, the number of patients who died were 5 (10%) and 2 (5.3%) respectively. There were 11 (18%) deaths in the hospital, and 33 (54.1%) patients were discharged home with good outcome, and 17 (27.9%) with poor outcome. At 6-months follow-up, 31 (62%) patients had good outcome while 7 (14%) had poor outcome, 2 (10%) did not survive, 3 (6%) were too lost to follow-up and 4 (8%) were transferred to other hospital for further care. At 12-months follow-up, there were 29 (47.5%) patients with good outcome, 5 (8.2%) had MRS 4-5, 2 (5.3%) did not survive, 4 (6.6%) were lost to follow-up, and 5 (8.2%) were transferred to other hospitals.
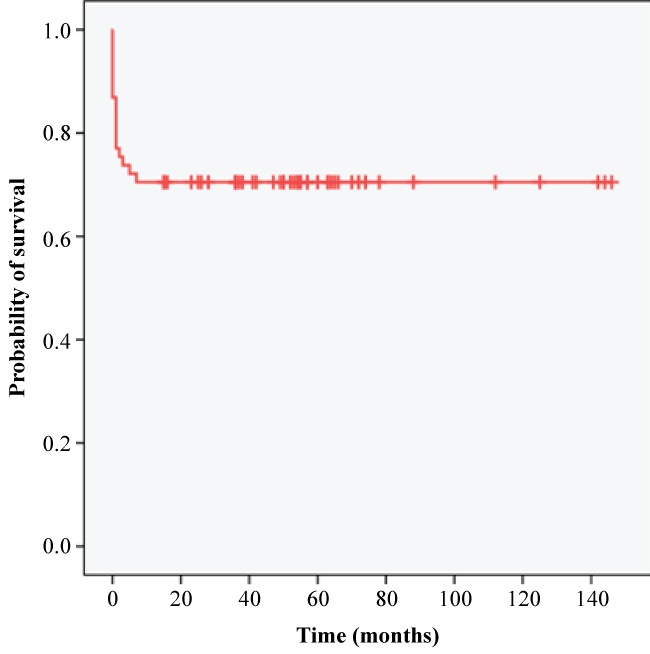
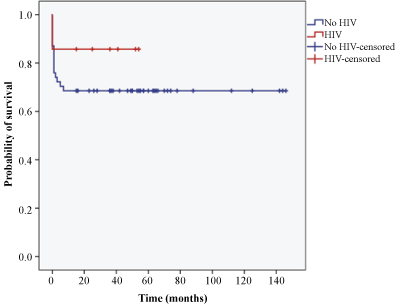
Figure 3 showed the overall survival curve of all TBM patients in this study. The estimated mean survival months for all patients were 103. There were 86.9% patients who survived the hospital admission, while 70.5% survived the 6 months follow-up period. Figure 4 showed the Kaplan-Meier survival curve among TBM patients based on HIV status. The survival rates of patients without HIV were 87% and 68.5% in hospital and 6 months follow-up period respectively. Patients with HIV had a survival rate of 85.7% in hospital and at 6 months follow-up. The estimated mean survival months were 100 for patients without HIV and 46 for patients with HIV.
.
Figure 3: Kaplan-Meier survival curve for all patients with Tuberculous Meningitis.
View Figure 3
.
Figure 4: Kaplan-Meier survival curve among tuberculous meningitis patients based on HIV status.
View Figure 4
Factors associated with 12-months mortality were intubation on presentation, surgical intervention (either ventricular-peritoneal shunting or external ventricular drain), CSF protein of 400-1000 mg/L and presence of tuberculoma in cerebral imaging (Table 3).
![]()
Table 3: Poor prognostic factors among patients with tuberculous meningitis.
View Table 3
Table 4 summarised the subgroup analysis (definite, probable, possible TBM) of the clinical characteristics, CSF analysis and radiological features. There was a significant association between TBM and duration of symptoms prior to presentation (p = 0.02), and duration of fever (p = 0.011). Out of the 60 TBM patients with CT brain, the finding of meningeal enhancement in CT scan was 40% in definite TBM, 36.4% in probable TBM and 10.3% in possible TBM patients. This did show a significant association between meningeal enhancement from CT brain with TBM (p = 0.041). Similarly, finding of meningeal enhancement in MRI brain had a significant association with TBM (p = 0.041).
![]()
Table 4: Clinical characterisitics, CSF analysis and radiological features of patients with definite, probable and possible TBM.
View Table 4
Study Limitation
Firstly, not all relevant information was available in the case notes. These included the absence of a standardized history and neurological examination that could have resulted in misclassification of patients with regard to neurological features, the lack of a complete neurological evaluation at discharge and inadequate registry of follow-up data. Besides, we excluded those patients without cerebrospinal fluid analysis though they were strongly suggestive of TBM based on Thwaites criteria [3]. Furthermore, most patients do not have CT thorax or abdomen to actively screen for TB lymph nodes and miliary in view of our study design. Apart from this, it was not possible to determine the cause of death in all patients, particularly those who died after being discharged from our hospital. For inpatient mortality, we took the cause of death as documented in patient’s medical record. Postmortem examinations are rarely done in Malaysia.
The number of cases may be underestimated from the year 2003 to 2009 due to the coding system. Therefore, only small number of patients were recruited which resulted in the low numbers. Being a hospital-based study in a referral hospital in a city area, it may not capture the real burden of TBM in Malaysia.
However, this study did portray the challenges and dilemma faced by clinicians in diagnosis and managing tuberculous meningitis with its complication. Moreover, getting patients to adhere to long course of antitubercular regimen and not defaulting was a great challenge itself. Finally, the number of TBM patients with HIV infections was relatively low and hence comparison between non-HIV infected patients was not possible.
Discussion
Tuberculosis is prevalent and it is a challenging problem in a developing country, such as Malaysia. In Malaysia, the commonest organ involved in tuberculosis infection is pulmonary [11], while the extrapulmonary involvement constituted about 10% of all the tuberculosis cases with the most common site being lymph node and pleural [6]. However, the most debilitating and fatal form of tuberculosis is tuberculous meningitis. From the year 2003 to year 2015, the incidence rate for tuberculous meningitis at our center ranged from 0.01% to 0.24%. The lower incidence rate in the beginning of our study period (from year 2003 to 2009) was due to the coding system in which only approximately 45% to 50% of the cases were not coded. The reported incidence in a few countries in Asia and Africa ranged 9.1% to 83% [12-15].
In this study, 55.7% of patients had fever, with the mean duration of 3 days. In contrast, other studies observed a higher fever rate among patients from 69% to as high as 91% [5,8,13,16-18]. The most frequent observed symptoms in our patients were headache (78.7%) and altered conscious level (78.7%), and the most frequent clinical signs were neck stiffness (70.5%) and focal neurological signs (72.1%). This was similar to a study conducted in Turkey where the most frequent symptoms observed at the time of admission were headache (86%), altered sensorium (59%) and stiff neck (88%) [19].
Typically, the patients with TBM had pleocytosis with lymphocytic predominance, raised protein concentration and reduced glucose ratio [3,17,20,21], which support our findings on high protein concentration and reduce glucose level. We had 11.5% of patients with normal protein concentration (< 400 mg/L), in fact Berenguer, et al. reported normal CSF protein concentration (< 460 mg/L) in 43% HIV infected patients and 28% non-HIV patients [22]. A study conducted in Kenya which found that 20.7% of TBM patients had acellular CSF and 24.1% had normal CSF cell count (< 5 cells per microliter) [23]. A South African study found a similar finding, in which 21.4% of patients had acellular CSF [15]. The gold standard for the diagnosis of tuberculous meningitis is the growth of Mycobacterium tuberculosis from cerebrospinal fluid, but that would take at least 4 to 8 weeks time for it to become positive. In our study, 90.2% of patients with TBM have negative cerebrospinal fluid culture for Mycobacterium tuberculosis. Out of the 45 (73.7%) cerebrospinal fluid samples that were sent for Polymerase Chain Reaction (PCR), 39 (86.7%) were negative. A few studies have shown that though PCR had a high specificity of 87%-98%, its sensitivity was estimated to be between 56%-75% [24-26].
The common findings of cranial CT imaging in our patients were leptomeningeal enhancement (20%) and hydrocephalus (43.3%). This was similar to a study in Turkey where 45.3% of their patients had hydrocephalus and 23.4% had leptomeningeal enhancement [27]. An observational study in Vietnam found that leptomeningeal enhancement (84%) and hydrocephalus (77%) were the most common findings [28]. Cranial MRI is more superior than cranial CT imaging in identifying meningeal and parenchymal abnormalities [29,30].
The recommended first line agents for the treatment of TBM include isoniazid, rifampicin, pyrazinamide and ethambutol for the initial 2 months, followed by isoniazid and rifampicin for additional 7 to 10 months. A Cochrane review in year 2010 showed that the optimal duration of antitubercular therapy (from 6 months to 24 months) for TBM had not been determined by a randomized controlled trial [31]. Similarly, the duration of antitubercular regimen varied among the patients in this study, from 12 months to 24 months. A systemic review concluded that 6-month treatment is sufficient for tuberculous meningitis with fully susceptible mycobacteria, however the follow-up of patients were limited to 1 year [32].
Univariate analysis in our study showed that TBM patients who required to be intubated on presentation were 2.6 times more likely to die compared to those who need not require invasive ventilator support [Hazard Ratio (HR) 2.619; 95% Confidence Interval (CI) 1.013-6.766; p = 0.047]. Similarly, a French study conducted in an intensive care unit revealed that mechanical ventilation in tuberculosis was a prognosticator of fatality [33]. Our study observed that 13 (31%) of TBM patients who had tuberculoma had a poorer prognosis [Hazard Ratio (HR) 4.153; 95% Confidence Interval (CI) 1.192-14.469; p = 0.025]. A study from Turkey of 160 TBM patients found that tuberculoma [Odds Ratio (OR) 1.9; 95% Confidence Interval (CI) 1.0-4.2; p = 0.048] and cranial nerve palsies [Odds Ratio (OR) 3.9; 95% Confidence Interval (CI) 1.8-8.8; p = 0.001] on admission to be significant predictors of neurological sequelae [19]. In our study, patients who required surgical intervention for hydrocephalus are more likely to die [Hazard Ratio (HR) 2.662; 95% Confidence Interval (CI) 1.047-6.770; p = 0.04]. This was in agreement with a study conducted by Chan, et al. where TBM patients with hydrocephalus had a poorer prognosis even after surgical decompression [34].
The overall survival rate of our patients in this study was high at 86.9% at hospital, with the mean survival months were 103. Twenty-nine (47.5%) of our patients had good outcome (MRS less or equal to 3) and 34 (55.7%) survived at the end of 12 months follow-up. Hosoglu, et al. reported that the mortality of patients with tuberculous meningitis was 44% and only 30.7% completely recovered [8]. In our study, HIV infection did not show a significant association in the mortality of patients with TBM either inpatient or during follow-up, probably because of the small number of HIV patients.
TBM continues to cause high mortality and morbidity. This study highlights the burden of TBM in a tertiary hospital and recognition of its poor prognostic factors such as intubation on presentation, surgical intervention (either ventricular-peritoneal shunting or external ventricular drain), elevated CSF protein and presence of tuberculoma. Despite the rising number of cases, there is no standard test in making the diagnosis of TBM. Therefore a high index of clinical suspicion should always be maintained and supported by cerebrospinal fluid analysis and radiological features.
References
-
(2003) The Global Burden of Tuberculosis, 1990-2013. Health Intelligence.
-
Marais S, Thwaites G, Schoeman JF, Török ME, Misra UK, et al. (2010) Tuberculous meningitis: a uniform case definition for use in clinical research. Lancet Infect Dis 10: 803-812.
-
Thwaites GE, Chau TT, Stepniewska K, Phu NH, Chuong LV, et al. (2002) Diagnosis of adult tuberculous meningitis by use of clinical and laboratory features. Lancet 360: 1287-1292.
-
Yasar KK, Pehlivanoglu F, Sengoz G (2010) Predictors of mortality in tuberculous meningitis: a multivariate analysis of 160 cases. Int J Tuberc Lung Dis 14: 1330-1335.
-
Christensen AS, Andersen AB, Thomsen VO, Andersen PH, Johansen IS (2011) Tuberculous meningitis in Denmark: a review of 50 cases. BMC Infect Dis 11: 47.
-
Nissapatorn V, Kuppusamy I, Rohela M, Anuar AK, Fong MY (2004) Extrapulmonary tuberculosis in Peninsular Malaysia: retrospective study of 195 cases. Southeast Asian J Trop Med Public Health 35: 39-45.
-
Rock RB, Hu S, Gekker G, Sheng WS, May B, et al. (2005) Mycobacterium tuberculosis-induced cytokine and chemokine expression by human microglia and astrocytes: effects of dexamethasone. J Infect Dis 192: 2054-2058.
-
Hosoglu S, Geyik MF, Balik I, Aygen B, Erol S, et al. (2002) Predictors of outcome in patients with tuberculous meningitis. Int J Tuberc Lung Dis 6: 64-70.
-
Bidstrup C, Andersen PH, Skinhøj P, Andersen AB (2002) Tuberculous meningitis in a country with a low incidence of tuberculosis: still a serious disease and a diagnostic challenge. Scand J Infect Dis 34: 811-814.
-
(2013) IBM SPSS Statistics 22.0 - United States.
-
Mohammad Z, Naing NN (2004) Characteristics of HIV-Infected Tuberculosis Patients In Kota Bharu Hospital, Kelantan from 1998 To 2001. Southeast Asian J Trop Med Public Health 35: 140-3.
-
Ganiem AR, Parwati I, Wisaksana R, van der Zanden A, van de Beek D, et al. (2009) The effect of HIV infection on adult meningitis in Indonesia: a prospective cohort study. AIDS 23: 2309-2316.
-
Helbok R, Pongpakdee S, Yenjun S, Dent W, Beer R, et al. (2006) Chronic meningitis in Thailand. Clinical characteristics, laboratory data and outcome in patients with specific reference to tuberculosis and cryptococcosis. Neuroepidemiology 26: 37-44.
-
Hui ACF, Ng KC, Tong PY, Mok V, Chow KM, et al. (2005) Bacterial meningitis in Hong Kong: 10-years' experience. Clin Neurol Neurosurg 107: 366-370.
-
Karstaedt AS, Valtchanova S, Barriere R, Crewe-Brown HH (1998) Tuberculous meningitis in South African urban adults. QJM 91: 743-747.
-
Anderson NE, Somaratne J, Mason DF, Holland D, Thomas MG (2010) A review of tuberculous meningitis at Auckland City Hospital, New Zealand. J Clin Neurosci 17: 1018-1022.
-
Gunawardhana SA, Somaratne SC, Fernando MA, Gunaratne PS (2013) Tuberculous meningitis in adults: a prospective study at a tertiary referral centre in Sri Lanka. Ceylon Med J 58: 21-25.
-
Yechoor VK, Shandera WX, Rodriguez P, Cate TR (1996) Tuberculous meningitis among adults with and without HIV infection. Experience in an urban public hospital. Arch Intern Med 156: 1710-1716.
-
Pehlivanoglu F, Yasar KK, Sengoz G (2010) Prognostic factors of neurological sequel in adult patients with tuberculous meningitis. Neurosciences (Riyadh) 15: 262-267.
-
Luma HN, Tchaleu BCN, Ngahane BHM, Temfack E, Doualla MS, et al. (2013) Tuberculous meningitis: presentation, diagnosis and outcome in hiv-infected patients at the douala general hospital, cameroon: a cross sectional study. AIDS Res Ther 10: 16.
-
Paul Matthew Pasco (2012) Diagnostic features of tuberculous meningitis: a cross-sectional study. BMC Res Notes 5: 49.
-
Berenguer J, Moreno S, Laguna F, Vicente T, Adrados M, et al. (1992) Tuberculous meningitis in patients infected with the human immunodeficiency virus. N Engl J Med 326: 668-672.
-
Hooker JA, Muhindi DW, Amayo EO, Mc'ligeyo SO, Bhatt KM, et al. (2003) Diagnostic utility of cerebrospinal fluid studies in patients with clinically suspected tuberculous meningitis. Int J Tuberc Lung Dis 7: 787-96.
-
Bhigjee AI, Padayachee R, Paruk H, Hallwirth-Pillay KD, Marais S, et al. (2007) Diagnosis of tuberculous meningitis: clinical and laboratory parameters. Int J Infect Dis 11: 348-354.
-
P Shankar, N Manjunath, Shriniwas, KK Mohan, K Prasad, et al. (1991) Rapid diagnosis of tuberculous meningitis by polymerase chain reaction. Lancet 337: 5-7.
-
Caws M, Wilson SM, Clough C, Drobniewski F (2000) Role of IS6110-targeted PCR, culture, biochemical, clinical, and immunological criteria for diagnosis of tuberculous meningitis. J Clin Microbiol 38: 3150-3155.
-
Hosoğlu S, Ayaz C, Geyik MF, Kökoğlu OF, Ceviz A (1998) Tuberculous meningitis in adults: an eleven-year review. Int J Tuberc Lung Dis 2: 553-557.
-
Thwaites GE, Macmullen-Price J, Tran TH, Pham PM, Nguyen TD, et al. (2007) Serial MRI to determine the effect of dexamethasone on the cerebral pathology of tuberculous meningitis: an observational study. Lancet Neurol 6: 230-236.
-
Galimi R (2011) Extrapulmonary tuberculosis: tuberculous meningitis new developments. Eur Rev Med Pharmacol Sci 15: 365-386.
-
Pienaar M, Andronikou S, van Toorn R (2009) MRI to demonstrate diagnostic features and complications of TBM not seen with CT. Childs Nerv Syst 25: 941-947.
-
Prasad K, Sahu JK (2010) Duration of anti-tubercular treatment in tuberculous meningitis: challenges and opportunity. Neurol India 58: 723-726.
-
van Loenhout-Rooyackers JH, Keyser A, Laheij RJ, Verbeek AL, van der Meer JW (2001) Tuberculous meningitis: is a 6-month treatment regimen sufficient?. Int J Tuberc Lung Dis. 5: 1028-1035.
-
Valade S, Raskine L, Aout M, Malissin I, Brun P, et al. (2012) Tuberculosis in the intensive care unit: A retrospective descriptive cohort study with determination of a predictive fatality score. Can J Infect Dis Med Microbiol 23: 173-178.
-
Chan KH, Cheung RTF, Fong CY, Tsang KL, Mak W, et al. (2003) Clinical relevance of hydrocephalus as a presenting feature of tuberculous meningitis. QJM 96: 643-648.