Journal of Musculoskeletal Disorders and Treatment
Treatment of Recalcitrant Lateral Epicondylitis with Autologous Conditioning Plasma
Wing Yum Man1*, Paul Y Lee2 and Andrew Rogers1
1Trauma and Orthopaedics Department, Royal Gwent Hospital, Newport, UK
2WelshBone, UK
*Corresponding author:
Wing Yum Man, Trauma and Orthopaedics Department, The Royal Gwent Hospital, Lovely Lane, Newport, NP20 2UB, UK, E-mail: wingyum@gmail.com
J Musculoskelet Disord Treat,
JMDT-1-005, (Volume 1, Issue 1),
Research Article
Received: November 06, 2015: Accepted: December 29, 2015: Published: December 31, 2015
Citation: Man WY, Lee PY, Rogers A (2015) Treatment of Recalcitrant Lateral Epicondylitis with Autologous Conditioning Plasma. J Musculoskelet Disord Treat 1:005
Copyright: © 2015 Man WY, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:Platelet-rich plasma (PRP) injections are part of a developing therapy believed to promote healing using the patient's own blood components. Certain studies have shown PRP treatment to be effective in the treatment of various tendinopathies such as lateral epicondylitis. Currently there is no standard method for PRP preparation with manufacturers each claiming various benefits for their system.
Purpose:Autologous Conditioning serum (ACP) (Arthrex, Naples, Florida) is a recent development that claims to have the same effects of PRP but with reduced cost and procedural time. The aim of this study is to investigate whether the use of ACP injections can have a beneficial effect in the treatment of recalcitrant lateral epicondylitis.
Study Design:Case Series
Methods:Data was collected prospectively from 76 patients between September 2010 and February 2013, who had undertaken ACP injections for lateral epicondylitis. All patients had failed primary care interventions including physiotherapy, corticosteroid injection and elbow clasps. Clinical outcome was recorded at 6 weeks and 3 months post procedure. Medical records were reviewed at 1-year to check for reoccurrence of symptoms.
Results:90 ACP injections were performed in 76 patients. 55 (61%) patients were symptom free at 3 months after a single injection and 8 (9%) patients had partial improvement. 13 patients underwent a 2nd injection and 6 became symptom free.
Conclusion:The results from this study suggest that ACP is an effective treatment option with 61% of patients symptom free after a single injection, 67.8% symptom free after a second injection. These findings demonstrate that ACP can be of benefit in cases that have failed to respond to traditional therapies.
Keywords
Lateral epicondylitis, Platelet-Rich plasma, PRP, Autologous condition serum, ACP
Introduction
Lateral epicondylitis or "tennis elbow" is the most commonly diagnosed condition of the elbow [1]. It affects 1-3% of the population and is thought to be due to the combination of mechanical overloading and abnormal microvascular responses [2,3]. It tends to effect those between 30 and 64 years of age, peaking between 45 and 54. Typically it will effect the dominant upper extremity, and is associated with a repetitive and forceful activity. Pain is felt on the lateral aspect of the elbow, and is often pronounced with wrist extension [4]. Despite the name "tennis elbow" the condition is mostly associated with work-related activities that require repetitive wrist flexion and extension, such as cutting meat, plumbing and working on cars [5].
Strides in our understanding of pathogenesis of the condition have shaped our treatment strategies. Histological studies have demonstrated lack of inflammatory cells, and have shown invasion of immature fibroblasts and disorganised and hypercellular change to the tissue of the ECRB tendon [6]. These findings suggest that the pathophysiology of lateral epicondylitis resembles that of a tendinopathy rather than a tendinitis and hence popularised the use of platelet-rich plasma (PRP) injections [1,7-9].
Preparation of a PRP injection can be a lengthy process; requiring harvesting of an autologous blood sample, and centrifugation. A multitude of PRP preparation systems are now available. These systems differ in their method of isolation: (one-step or two-step centrifugation), type and operation of collecting tubes, speed and duration of centrifugation all of which results in varying concentrations of platelets, growth factors, white blood cells and red blood cells [10-12].
Autologous Conditioning serum (ACP) (Arthrex, Naples, Florida) is a form of PRP developed to have the effects of PRP but at a reduced cost and preparation time due to its one-step centrifugation process [13,14]. Unlike other platelet-rich plasma formulations, ACP is characterised by its low concentration of white blood cells such as neutrophil granulocytes that can be detrimental to the healing process in high concentrations.
There have been numerous studies supporting the use of PRP injections for the treatment of lateral epicondylitis, there have not been any studies investigating the efficacy of ACP, which is claimed to be equally effective but cheaper alternative [15]. This study investigates the use of ACP in the management of lateral epicondylitis that has previously failed to respond to corticosteroid injection therapy.
Methods
A single centred prospective observational study was carried out at a district general NHS hospital during February 2010 to September 2013. The authors have no financial or other interest in the product or distributor of the product. The study was approved by the local research and development department.
Patient selection
Patients were pooled from consecutive primary or secondary referrals with recalcitrant lateral epicondylitis. For the purposes of this study, recalcitrant lateral epicondylitis was defined.
Inclusion criteria
• Diagnosis of lateral epicondylitis for more than 2 years
• Used conservative measures such as the tennis elbow clasp for 6 months or more
• Previously received an injection of corticosteroid
• Undertaken at least 2 courses of physiotherapy
Exclusion criteria
• Received a corticosteroid injection within the last 6 months
• Previously received a form of PRP injection therapy
• Previously undergone surgical intervention for their symptoms
The autologous conditioning plasma (ACP) was prepared using the ACP Double Syringe system in accordance with manufacture's guidance (Arthrex ABS-10014). 15ml of autologous whole blood was withdrawn into the proprietary double syringe. No optional anti-coagulant was added. The sample was then centrifuged at 5000rpm for 6 minutes. The ACP supernatant was extracted using the inner syringe ready for use.
All injections were performed in a similar technique as favoured by the senior author under his supervision without the use of ultrasound. Prior to ACP injection the tender point would be identified with direct palpation. Upon agreement with the patient that this point was the most tender, it would be marked with a marker pen. All patients received 5ml of 1% lidocaine for local anaesthesia. The skin, subcutaneous tissue, the tendon and periosteum would be infiltrated, while awaiting the centrifugation process. Under aseptic conditions 3-4 ml of ACP was injected to the periosteum and tendon using a single needle puncture needle technique with an 18-gauge needle to the marked area.
Clinical outcome was recorded at 6 weeks and 3 months following ACP injection and was determined by a subjective assessment of the patient's response to treatment using an interview style questionnaire with a 3-point scale: no improvement, partial improvement, and complete resolution of symptoms. Medical records were also checked at 1 year to see if included patients re-attended with a reoccurrence of symptoms.
All statistical analyses were performed using the Statistical Package for Social Scientists (SPSS, version 20, USA). Data was analyzed using one-way ANOVA tests with repeated measures. Prior to running all ANOVA tests, data was checked for sphericity using Mauchly's test. Where sphericity violations were noted Huyn-Feldt corrections were used to modify the degrees of freedom employed in the subsequent statistical analyses. Statistical significance was accepted at p ≤ 0.05 and, where statistical significance was noted, post-hoc (Bonferroni) pair-wise comparisons were made to determine specifically where differences existed. Multiple regression analysis was also performed for correlation of age, sex, BMI or duration of symptoms.
Results
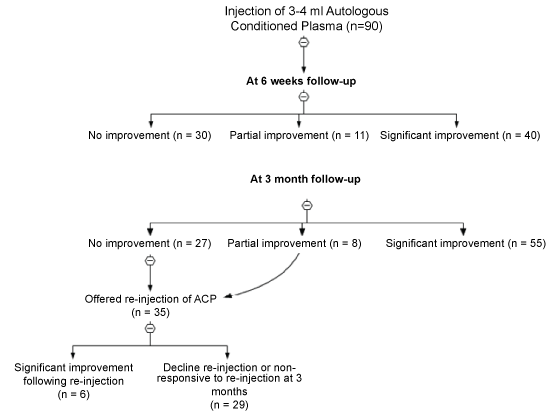
During the study period (3 years 7 months) 90 injections were given to 76 patients with non-responsive lateral epicondylitis. Patient's mean age was 49 years (27-65, SD 10.3) (58% female, 42% male). The mean BMI was 28 (20-38, SD 5.3). Prior to entering the study the patients reported a mean duration of symptoms of 33.6 months (25-50, SD 3.2 months). All patients were followed-up for a minimum of 3 months (Figure 1).
At 6 weeks post ACP injection 60 (66.7%) reported a significant or partial improvement in their symptoms. No improvement was recorded in 30 (33.0%). Of the patients who had noticed improvement 49 (54.4%) patients had significant improvement and 11 (12.2%) reported only partial improvement.
At the 3-month follow-up the 49 (54.4%) patients who reported significant improvement at 6 weeks remained asymptomatic. A further 6 (6.7%) patients now reported significant improvement, meaning that a total of 55 (61.1%) patients at the 3 month mark were now symptom free. 27 (30.0%) patients remained symptomatic with no improvement, and 8 (8.9%) now reported partial improvement.
Those who had partial or no response at 3 months were offered a 2nd injection of ACP. In the group of 8 partial responders all underwent further injections. Six (75%) of these reported significant improvement, with the remaining 2 (25%) staying symptomatic at 3 months post 2nd injection.
2nd injections were also offered to the 27 (30.0%) patients who reported no improvement at 3 months. Only 5 patients of this group had a 2nd injection but none of them reported any improvement at 3 months post reinjection.
At 1-year medical records of the 55 (61.1%) patients who showed significant improvement revealed that none had re-attended clinic with reoccurrence or residual symptoms.
A multi-regression analysis was performed. No correlation for age, sex, BMI or duration of symptoms was found in those who reported significant improvement at any time interval.
Discussion
The patients in our study all suffered with lateral epicondylitis that had not responded to one or more local corticosteroid injection. Following a single injection of ACP 70% (63/90) reported a partial or significant improvement in their symptoms at 3 months. These findings are comparable to previous similar studies on PRP [1,7]. In 2006 Mishra et al compared the use of a single local injection of PRP to a single injection of bupivacaine for the treatment of chronic lateral epicondylitis. At 8 weeks the PRP treated group reported an improvement of 60% in their visual analog pain scores, compared to 16% in the group treated with bupivacaine [7]. However, studies have suggested that bupivacaine may be toxic to tendon cells, thereby affecting the validity of the study [16]. More recently Peerbooms et al performed a randomised controlled trial comparing the efficacy of PRP injection versus a conventional corticosteroid injection for the management of lateral epicondylitis. The group treated with the corticosteroid injection reported better functional scores initially, but at 1-year only 49% had significant improvement, against 79% of the PRP group [1]. The findings of Peerbooms et al seem to suggest that PRP may not relieve symptoms as rapidly as corticosteroid injections but the reoccurrence rates are lower. Our findings at 1-year also show that none of the 55 patients whose symptoms were relieved after a single injection of ACP had any reoccurrence.
There is concern whether the reduced centrifugation time used in the ACP procedure will lead to lower concentration of platelets reducing its efficacy. However, in vitro studies have defined platelet numbers of > 200 × 103/μL as sufficient for a therapeutic effect [17]. In 2012 Mazzuco et al. compared the constituents of plasma preparations produced by ACP and a two-step centrifugation PRP system. This revealed that the one-step single spin of ACP procured a platelet concentration of 378.3 ± 58.64 × 103/μL, compared to a two-step yield of 873.8 ± 207.82 × 103/μL. Despite the significantly higher concentration of platelets in the two-step process, both methods satisfy the aforementioned therapeutic concentration of > 200 × 103/μL [14].
In our study patients who reported a partial or no response at 3 months were offered a 2nd injection of ACP. Of the 8 patients who initially reported a partial response 75% (6/8) reported resolution of symptoms 3 months after the second injection. The patients who did not report any improvement after the first injection, also failed to report any improvement after the 2nd injection. Thus the patients who exhibited a partial response to the first injection benefitted from a repeat injection, whereas the non-responders still did not.
The repeat injection could have helped by increasing the platelet concentration in the target area. Literature has shown a correlation between platelet concentration and the regenerative effects of PRP up to a point [18]. However, with the injections being at least 6-months apart, this is unlikely to be the case. An in vitro study by Kajikawa et al. demonstrated a two-fold increase in cells crucial for healing (macrophages and fibroblasts) at 3 and 7 days post PRP injection for tendon injury. However, at 14 days in both the control and PRP group there was a marked decrease in circulation-derived cells [19]. As such a repeat injection after a given interval may produce a second spike in the proliferation of macrophages and fibroblasts and complete the healing response.
With regards to the non-responders it appears that despite creating an optimal environment for tissue regeneration symptoms are unaffected. As such, in the group of non-responders the authors suspect that the underlying pathophysiology may not be related to tendinosis of the ECRB tendon.
Our findings have shown ACP produces clinical outcomes comparable to PRP systems. Arthrexsuggests its system to be more cost-effective than alternative PRP systems. PRP injections are quoted at £250 per injection as opposed to ACP injections; £70 [13]. Based on these figures (Figure 1 and Figure2) using ACP over PRP systems based on equipment alone would give a hypothetical saving of £16200 for the injections carried out in this study. In addition, the preparation time for ACP (5 min) is significantly quicker compared with some PRP systems which have a preparation time of over 30 minutes [13]. In fact the faster single-step process which ACP uses has been shown to demonstrate significantly higher myocyte and tenocyte proliferation rates compared to the traditional two-step PRP counterparts [18].
.
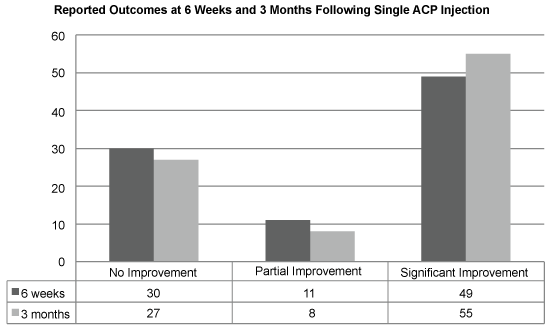
Figures 2: A graph illustrating the outcomes at 6 weeks and 3 months.
Non-responders failed to improve at 6 weeks and at 3 months. After one injection 61% (55/60) demonstrated a significant improvement at 3 months.
View Figures 2
Conclusion
Our findings have shown that the ACP system is a cost-effective alternative with comparable therapeutic effect to other PRP system in treating recalcitrant lateral epicondylitis. Patients who experience partial benefit from a first injection of ACP may benefit from a repeat injection. Other treatment strategies must be considered for patients who show no improvement following their first injection as repeat injections were ineffective.
References
-
Peerbooms JC, Sluimer J, Bruijn DJ, Gosens T (2010) Positive effect of an autologous platelet concentrate in lateral epicondylitis in a double-blind randomized controlled trial: platelet-rich plasma versus corticosteroid injection with a 1-year follow-up. Am J Sports Med 38:255-262.
-
Nirschl RP, Ashman ES (2003) Elbow tendinopathy: tennis elbow. Clin Sports Med 22: 813-836.
-
Smith RW, Papadopolous E, Mani R, Cawley MI (1994) Abnormal microvascular responses in a lateral epicondylitis. Br J Rheumatol 33: 1166-1168.
-
Sims SE, Miller K, Elfar JC, Hammert WC (2014) Non-surgical treatment of lateral epicondylitis: a systematic review of randomized controlled trials. Hand (N Y) 9: 419-446.
-
Long L, Briscoe S, Cooper C, Hyde C, Crathorne L (2015) What is the clinical effectiveness and cost-effectiveness of conservative interventions for tendinopathy? An overview of systematic reviews of clinical effectiveness and systematic review of economic evaluations. Health Technol Assess 19: 1-134.
-
Faro F, Wolf JM (2007) Lateral epicondylitis: review and current concepts. J Hand Surg Am 32: 1271-1279.
-
Mishra A, Pavelko T (2006) Treatment of chronic elbow tendinosis with buffered platelet-rich plasma. Am J Sports Med 34: 1774-1778.
-
Gosens T, Peerbooms JC, van Laar W, den Oudsten BL (2011) Ongoing positive effect of platelet-rich plasma versus corticosteroid injection in lateral epicondylitis: a double-blind randomized controlled trial with 2-year follow-up. Am J Sports Med 39: 1200-1208.
-
Thanasas C, Papadimitriou G, Charalambidis C, Paraskevopoulos I, Papanikolaou A (2011) Platelet-rich plasma versus autologous whole blood for the treatment of chronic lateral elbow epicondylitis: a randomized controlled clinical trial. Am J Sports Med 39: 2130-2134.
-
Foster TE, Puskas BL, Mandelbaum BR, Gerhardt MB, Rodeo SA (2009) Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med 37: 2259-2272.
-
Alsousou J, Thompson M, Hulley P, Noble A, Willett K (2009) The biology of platelet-rich plasma and its application in trauma and orthopaedic surgery: a review of the literature. J Bone Joint Surg Br 91: 987-996.
-
Lopez-Vidriero E, Goulding KA, Simon DA, Sanchez M, Johnson DH (2010) The use of platelet-rich plasma in arthroscopy and sports medicine: optimizing the healing environment. Arthroscopy 26: 269-278.
-
Arthrex. Arthrex ACPTM Double Syringe System Autologous Conditioned Plasma
-
Mazzocca AD, McCarthy MB, Chowaniec DM, Cote MP, Romeo AA, et al. (2012) Platelet-rich plasma differs according to preparation method and human variability. J Bone Joint Surg Am 94: 308-316.
-
(2011) Arthrex.com. Arthrex ACP Double Syringe. Arthrexcom 39.
-
Haasters F, Polzer H, Prall WC, Maximilian Michael Saller, Julia Kohler, et al. (2011) Bupivacaine, ropivacaine, and morphine: comparison of toxicity on human hamstring-derived stem/progenitor cells. Knee Surgery, Sports Traumatology, Arthroscopy 19: 2138-2144.
-
Mazzucco L, Balbo V, Cattana E, Guaschino R, Borzini P (2009) Not every PRP-gel is born equal Evaluation of growth factor availability for tissues through four PRP-gel preparations: Fibrinet®, RegenPRP-Kit®, Plateltex® and one manual procedure. Vox Sang 97: 110-118.
-
Mazzocca AD, McCarthy MB, Chowaniec DM, Dugdale EM, Hansen D, et al. (2012) The positive effects of different platelet-rich plasma methods on human muscle, bone, and tendon cells. Am J Sports Med 40: 1742-1749.
-
Kajikawa Y, Morihara T, Sakamoto H, Matsuda K, Oshima Y, et al. (2008) Platelet-rich plasma enhances the initial mobilization of circulation-derived cells for tendon healing. J Cell Physiol 215: 837-845.