Journal of Musculoskeletal Disorders and Treatment
Bone Mineral Density Measured by Quantitative Computed Tomography in Pre-pubertal/Early Pubertal Children with Hypovitaminosis D
Ferenc Péter1*, Ágota Muzsnai1, László Blatniczky1 and Péter Lakatos2
1Pediatric Endocrinology Unit, St. John's Hospital, United Hospitals of North-Buda, Hungary
2Department of Internal Medicine, Semmelweis University Medical School, Hungary
*Corresponding author: F Péter, Pediatric Endocrinology Unit, St. John's Hospital and United Hospitals of North-Buda: Buda Children's Hospital 1026. Budapest, Hargita-u 6, Hungary, Tel: 36-1-3560817, E-mail: ferenc.dr.peter@gmail.com
J Musculoskelet Disord Treat, JMDT-2-008, (Volume 2, Issue 1), Original Article
Received: December 03, 2015: Accepted: March 25, 2016: Published: March 28, 2016
Citation: Péter F, Muzsnai A, Blatniczky L, Lakatos P (2016) Bone Mineral Density Measured by Quantitative Computed Tomography in Pre-pubertal/Early Pubertal Children with Hypovitaminosis D. J Musculoskelet Disord Treat 2:008.
Copyright: © 2016 Péter F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:Clinically significant vitamin D deficiency is associated with decreased bone mineral density (BMD) and increased fracture risk in adults. A similar association is suggested to be present in children. In this study we analyzed serum vitamin D3 level and corresponding BMD in pre-pubertal/early pubertal children.
Methods:272 healthy pre-pubertal/early pubertal schoolchildren (9.74 ± 0.6 years of age) were involved in the study. Serum 25(OH)D3 was measured by HPLC and were correlated to volumetric BMD (vBMD) which was assessed by peripheral quantitative computer tomography (pQCT).
Results:25(OH)D3 levels (samples taken in the autumn) were below 20 ng/ml in 37.1% of the subjects and in the range of 20-30 ng/ml in 26.2%. Therefore, the vitamin D status proved to be insufficient in a significant percentage of children. The vBMD was found to be moderately decreased: Z-score less than -1.0 was found in 10.3% of the children while values “low for age” (Z-score less than -2.0) were extremely rare (0.74 %). Total and trabecular densities, considered together, indicated a trend of decreasing vBMD (Z-score less than -1.0) in only 6.6% of the subjects.
Conclusion:Comparison of the two parameters (vitamin D3 and vBMD) allows to conclude that, in a cross-sectional study of the pre-pubertal/early pubertal age group, 25(OH)D3 levels at present considered as low are usually not associated with significantly low vBMD.
Keywords
25(OH)D3, Hypovitaminosis D, vBMD, pQCT, Pre-puberty, Early puberty
Introduction
Severe childhood vitamin D deficiency is a key factor in the development of rachitis. The disease has become quite rare among Caucasians living in developed countries [1], although there are a number of studies showing that the vitamin D status of children is significantly less than the optimal level all over the world [1]. On the other hand, rachitis is not necessarily manifest even at definitely low vitamin D levels [2]. Pediatric reports on the relationships between vitamin D status and bone health tend to be quite contradictory. Clinically significant vitamin D deficiency is associated with decreased bone mineral density (BMD) and increased fracture risk in adults. It is hypothesized that similar relationship exist in children.
The majority of recent publications in this field are reviews, and many of the authors consider that further studies are necessary [1,3-6]. As part of an extensive screening program of pre-pubertal/early pubertal schoolchildren, we measured the serum level of calcium, phosphorus, alkaline phosphatase, osteocalcin, β-crosslaps and 25(OH)D3 as well as determined volumetric bone mineral density (vBMD) by peripheral quantitative computed tomography (pQCT). The aim of this publication is to present the unexpected results of the comparative evaluation of 25(OH)D3 and vBMD.
Subjects and Methods
These examinations were performed in pre-pubertal and early pubertal white European children living in one of the inner Buda districts of Budapest. This region of the city is populated by slightly more well-to-do families than the average. The selection was made in two schools. All parents of studied children provided written informed consent, enrollment included more than 90% of children in the chosen classes. To avoid the effects of pubertal changes as far as possible, the upper age-limit was 11 years (88.8% were pre-pubertal). The screening examinations (including blood sampling) were conducted in November. The study was approved by the Ethics Committee of the Children's Hospital.
Those having a result for 25(OH)D3 (294 children) were aged 9.741 ± 0.598 years with a gender distribution of 153 boys and 141 girls. 272 of this group were also subjected to vBMD examination (age: 9.737 ± 0.608 years; 140 boys, 132 girls).
Serum concentration of 25(OH)D3 was measured in an HPLC (Bio-Rad) apparatus equipped with a UV detector, using '25-OH Vitamin D3 by HPLC' kits (Bio-Rad, Hercules, CA, USA, Cat. No.: 195-6530). The intra-assay CV of the method is 2.07-5.69% (depending on the concentration range), and the inter-assay CV is 3.59-5.15%. The validated normal range of the performing laboratory is 20-60 ng/ml.
vBMD measurements were performed by mobile pQCT set up at the schools. We chose the Stratec XCT 2000 (XCT 2000 Bone Scanner, Stratec Medizintechnik GmbH, Pforzheim, Germany), the pQCT apparatus most commonly used in pediatric applications [7].
pQCT was employed to determine total and trabecular vBMD on the non-dominant forearm at the 4% position of the distal radial, following routine procedures, especially in screening studies [8]. The lower arm is considered an ideal site for the assessment of fracture risk in children, as this site is the most common location of pre-pubertal and adolescent bone fractures [1,9].
The designation “quantitative” means that measurement is made in three dimensions in a given volume (vBMD), expressed in units of g/cm3. The radiation exposure associated with the test is < 2 μSievert. Its accuracy is characterized by a margin of error of 0.3-2.0 %.
A nation-wide survey to establish vBMD reference values, as assessed by mobile pQCT, was carried out previously in a well-balanced population of Hungarian children using measurements [10]. In this previous study, randomization of the studied children (2237 healthy schoolchildren/1068 boys/age 9.5-15.5 years) was performed according to the localization of the schools (Budapest, East-Hungary, West-Hungary), type of settlements (capital, size of town or village), sex and age (altogether six categories) to gain statistically correct and representative sampling. Total and trabecular vBMD values were grouped according to the sex and age groups [10]. In the present study measured vBMD values were compared to this reference data, which have similar g/cm3 values compared to German and British data [11,12] obtained by the same measurement technique.
Physical examination for height, body weight and pubertal development as well as the routine laboratory parameters were studied; the possible modifying influence of Tanner stages of puberty and BMI on the bone health was also analyzed in this cohort.
Results
There was a slight but significant difference in the ages between the tested boys and girls (boys: 10.18 ± 0.31, girls: 9.27 ± 0.46 years; p < 0.001), although all the children were attending the same year at school. The mean ± SD 25(OH)D3 level of the 294 serum samples was 26.25 ± 13.09 ng/ml (range: 3.3 and 85.5). There was no gender difference the vitamin D levels: boys, 27.17 ± 13.50; girls, 25.25 ± 12.59 ng/ml (p = 0.208).
The distribution of 25(OH)D3 concentrations is summarized in (Table 1). When a lower limit of normal of 20 ng/ml was used, 37.1% of the samples fall into the range of hypovitaminosis D; when the cut-off is moved to 30 ng/ml, nearly two-thirds of the values (63.3%) will rate as low.
![]()
Table 1: Distribution of serum 25(OH)D concentration values.
View Table 1
BMD Z-score values below -2.0 were found in four cases (two total- and two trabecular vBMD). No child had both Z-scores (total and trabecular) below -2.0. Total Z-scores were in 26 (9.6%). trabecular vBMD Z-scores were between -1.0 and -2.0 in 40 (14.7 %) children, while for both parameters were between -1.0 and -2.0 in 18 (6.6 %) cases. All the other Z-score values were above -1.0.
The serum calcium, phosphorus, alkaline phosphatase, osteocalcin and β-crosslaps levels were in the normal range.
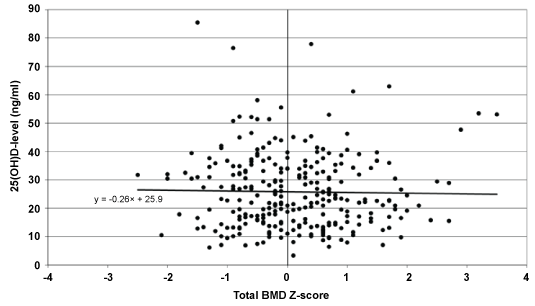
Graphic representation of the individual scores does not reveal any specific trend or any correlation between the two parameters (25(OH)D3 and total vBMD Z-score); the majority of the values below either 20 or 30 ng/ml fall into the normal range of vBMD (Figure 1); Analyzing patients with vBMD Z-scores below -1.0 also does not show a particular trend in Vitamin D levels (Table 2).
![]()
Table 2: Comparison of 25(OH)D3 values and total vBMD Z-scores < -1.0.
View Table 2
In another approach, the vitamin D levels obtained were listed in decreasing order and compared to the associated density Z-scores of those falling into the top and bottom quarter, fifth and eighth. With this statistical technique there is some tendentious parallelism between the two parameters (Table 3). In these extreme cohorts the higher or lower vitamin D mean values are associated with higher or lower mean vBMD Z-scores, but in spite of this trend, the differences of the Z-score values are statistically non-significant. In addition, the bone density in the cohort with low mean vitamin D levels (9.4-11.6 ng/ml) was found to be in the normal range (mean Z-scores: 0.035-0.084).
![]()
Table 3: Vitamin D3 and vBMD values in the highest (top)and lowest (bottom) cohorts (N = number of subjects).
View Table 3
33 children (11.2%), among them 21 girls had already entered puberty at the time of the examination (Tanner II, except one case of one girl, Tanner III). The magnitude of their 25(OH)D3 values showed a distribution identical with that of the pre-pubertal children. Four of them (12.1%) had vBMD Z-scores below -1.0.
The BMI of 27 children was above the 95th percentile. Their 25(OH)D3 level was 25.17 ± 18.16 ng/ml, similar to the whole cohort (26.25 ± 13.09). The mean total vBMD Z-score was not significantly higher (+0.42 ± 1.03 in obese children versus +0.03 ± 1.13).
In summary, in this cross-sectional study the volumetrically determined total bone density of pre-pubertal and early pubertal children with higher or lower vitamin D3 levels was within the normal range (Z-score > -2) with two exceptions; lower vitamin D levels were not associated with decreased bone densities.
Discussion
25(OH)D3 level
The serum 25(OH)D3 concentration is generally accepted to be the best biomarker of the body's vitamin D supply [13]. Recently, however, a lively debate has developed on vitamin D supply and its significance. According to the official statement of a large international group of experts [14], 25(OH)D3 levels above 20 ng/ml are sufficient for maintaining adequate bone health. The population in North-America in general falls therefore into the sufficient category. They considered the extra-skeletal effects of vitamin D, at this point, unproven and suggested that excess intake may actually have deleterious effects [14].
However, according to another group of experts [15], vitamin D deficiency is very common world-wide and there is no doubt about the existence of the numerous significant extra-skeletal effects of vitamin D. They suggest that the levels in the range of 20-30 ng/ml should be considered insufficient, while levels above 30 ng/ml are optimal. In order to achieve this threshold, daily doses exceeding 4000 IU are deemed permissible [15].
Similar difference of opinion exists also in the pediatric literature. Vitamin D3 level was considered sufficient above 20 ng/ml by the Lawson Wilkins Pediatric Endocrine Society [16], the British Paediatric and Adolescent Bone Group [17] and experts of European pediatric gastroenterologist from eleven countries [18]. In a new “expert position statement” and other pediatric publications the 25(OH)D levels < 20 ng/ml are defined as deficiency, between 20 and 30 ng/ml as insufficiency, and only > 30 ng/ml as sufficiency [19-21].
The experts, however, also call attention to the significant inter-assay differences between 25(OH)D3 determination methods, which have been corroborated by numerous investigators. In their opinion, HPLC and its versions supplemented with tandem mass spectrometry (MS/MS) may be the most reliable assays [22-24].
A mean 25(OH)D3 level of 26.25 ± 13.09 ng/ml for nearly three hundred pre-pubertal/early pubertal children determined in the autumn can be considered as low, and the ratio of children with 25(OH)D3 levels below the lower limit of 20 ng/ml (37.1%) is high. If we follow the school of thought that infers vitamin D3 insufficiency from 25(OH)D3 levels below 30 ng/ml, then only 36.7% of the healthy children aged ~10 years, living under relative good conditions in Budapest were optimally supplied with vitamin D at the time of the study. More severe deficiency was measured in 6.1% ( < 10 ng/ml) and 0.37% ( < 5 ng/ml) of the subjects. Similar data was found in another study on adolescents living in Pécs (Hungary), where the average 25(OH)D3 level was 23.12 ng/ml [25].
BMD
The ideal test method for bone health of clinical relevance is the determination of fracture risk on the basis of longitudinal data [5]. The risk of fracture depends - among others - on skeletal fragility, which is affected by bone mass, size, geometry, micro architecture as well as bending strength [26].
The DXA apparatus developed for the measurement of bone mass is considered adequate for practical purposes. The majority of BMD determinations occur by DXA. However, this two-dimensional method measures areal BMD (aBMD in g/cm2), and it is not or hardly suitable for studying bone size, geometry or quality. Elimination of this deficiency has been attempted in multiple ways, especially in children, who are still growing, using various geometric algorithms for correction. Alternatively, data measured are compared to the aBMD values of children matched for gender, Tanner stage, bone age, body height or Z-score rather than age [27]. Advantages of the DXA method in pediatrics are rather the much higher prevalence of devices of this type, and an enormous amount of reference data accumulated over the years [3,5,28].
pQCT, the method used in our work yields volumetric results in units of g/cm3. It can provide information on bone size, geometry and quality, and in addition to total vBMD, it also gives the trabecular fraction separately. The measurement is faster than the one using DXA, it is associated with lower radiation exposure (DXA: 1.5-10.0 μSv; pQCT: < 2.0 μSv), and its accuracy is also acceptable [8]. In follow-up studies, results approaching the data given in the manual were obtained: within-subject CV < 2.7 % [29].
According to our results, only the total density of two subjects and the trabecular density in another two children were found under Z-score of -2.0.
25(OH)D level and vBMD parallel measured with pQCT
Publications comparing the results of simultaneously determined vBMD performed with pQCT on the lower arm of pre-pubertal children and the 25(OH)D levels of both genders are very sparse in the literature, as also demonstrated by the recently published reviews [1,3,5,6].
The investigators usually measured BMD by DXA in the postmenarcheal age group. This material is quite wide-range, but is not fully relevant for comparison. Recent reviews summarizing these results conclude that there is little evidence for a correlation between the 25(OH)D3 level and areal or volumetric bone density in healthy children, and the data are decidedly inconsistent, which is also true for the relationship between low vitamin D status and fracture risk. The observations published also disagree in identifying the circumstances under which vitamin D supplementation improves bone density, although very low 25(OH)D3 levels are probably one of those [6,30].
In a Finnish study, the cortical vBMD values of girls aged 10-12 years (pre-puberty, early puberty) radially measured by pQCT were significantly lower when 25(OH)D values were below 10 ng/ml than in the case of higher vitamin D level [31]. In a study of low subject number (21 subjects), total and trabecular radial vBMD values of African-American girls aged 12-14 years were measured by pQCT. No correlation was found between the very low vitamin D levels (5.0-16.3 ng/ml) and the good vBMD values. The authors explain the observation by the low number of subjects [32]. Another group of investigators found the total and trabecular vBMD Z-scores of postmenarcheal girls measured by the same technique to be in the normal range in spite of the very low 25(OH)D3 levels (mean level 7.2 ng/ml) both before and after vitamin D supplementation [30].
Recently 25(OH)D3 level was found positively related to subsequent cortical bone mass and predicted strength in children (7.6-11.8 years) measured by pQCT at the 50% mid tibia [33]. However, in a study from the US using pQCT also on tibia for the determination of the vBMD Z-scores of children aged 9-13 years, Z-scores were found to be higher in black children with lower vitamin D supply than in their white contemporaries who had higher vitamin D levels [34].
In summary, the total and trabecular radial vBMD Z-scores of nearly 300 children aged 8.5-11 years, measured by pQCT regularly fell into the normal range independently of vitamin D levels considered normal or low; the serum 25(OH)D3 values between 3.3-85.5 ng/ml did not show any correlation with vBMD.
Possible explanations
The continuously boosted effect of PTH enhances bone resorption over bone formation. The concept based on this physiological process suggests that, from the bone health point of view of, the biologically relevant lower limit of 25(OH)D3 is the one which does not yet induce the PTH level to rise [35]. Based on a very careful analysis of the relationship between of 25(OH)D3 and PTH levels, a recently published article [2] puts this PTH-inducing 25(OH)D3 level somewhere between 10.8 and 16.4 ng/ml, suggesting to be the lower limit value for the diagnosis of vitamin D deficiency (as a "biochemical definition"). In adults the corresponding range is higher: 15-30 ng/ml, as bone health needs less vitamin D in pre-puberty before the rapid pubertal growth.
The possibility of yet another explanation is opened by a concept developed a short time ago. According to Australian authors, "the key element of ... rickets in children is a delay in mineralization. It can be resolved by normalization of plasma calcium and phosphate homeostasis independently of vitamin D activity" [36,37]. This opinion is mainly based on the results of in vitro experiments and animal models, which indicate that all the three main bone cell types (osteoblast, osteoclast, osteocyte) are capable of the expression of both the vitamin D receptor and the enzyme 25(OH)D 1α-hydroxylase (CYP27B1), making possible the synthesis of the active hormone (1,25(OH)2D) within the bone tissue. The kidneys can also enhance CYP27B1 enzyme activity e.g. under the effect of PTH, thus, according to observations made on humans with decreased 25(OH)D3 levels, synthesis of the active hormone [38] and preservation of bone quality are possible above a 25(OH)D3 level of ~8 ng/ml, if calcium supply is optimal.
In a recent case-control study from India, low dietary calcium intake was prevalent in nutritional rickets cases compared to the healthy controls, while low vitamin levels were not characteristic [39].
The main limitations of our study are the cross-sectional design rather than longitudinal, the analysis of total and trabecular density out of the possible vBMD parameters, and the lack of the serum PTH values. The schools where our studies were conducted were not "elite" schools, but a slight difference in living standard in this well-to-do area exists compared to the Hungarian average, therefore may represent a further limitation.
Conclusion
Vitamin D screening in healthy pre-pubertal/early pubertal schoolchildren living under favorable conditions in Budapest detected significant hypovitaminosis D. Simultaneous determination of vBMD by pQCT at the distal radius did not indicate decrease in bone density. Based on the analysis of these two parameters, low vitamin D supply is not associated with decreased vBMD in pre-pubertal children.
References
-
Moon RJ, Harvey NC, Davies JH, Cooper C (2014) Vitamin D and skeletal health in infancy and childhood. Osteoporos Int 25: 2673-2684.
-
Atapattu N, Shaw N, Högler W (2013) Relationship between serum 25-hydroxyvitamin D and parathyroid hormone in the search for a biochemical definition of vitamin D deficiency in children. Pediatr Res 74: 552-556.
-
Sopher AB, Fennoy I, Oberfield SE (2015) An update on childhood bone health: mineral accrual, assessment and treatment. Curr Opin Endocrinol Diabetes Obes 22: 35-40.
-
Abrams SA, Tiosano D (2014) Update on vitamin D during childhood. Curr Opin Endocrinol Diabetes Obes 21: 51-55.
-
Golden NH, Abrams SA; Committee on Nutrition (2014) Optimizing bone health in children and adolescents. Pediatrics 134: e1229-1243.
-
Winzenberg T, Jones G (2013) Vitamin D and bone health in childhood and adolescence. Calcif Tissue Int 92: 140-150.
-
Adams JE, Engelke K, Zemel BS, Ward KA, International Society of Clinical Densitometry (2014) Quantitative computer tomography in children and adolescents: the 2013 ISCD Pediatric Official Positions. J Clin Densitom 17: 258-274.
-
Fonseca A, Gordon CL, Barr RD (2013) Peripheral quantitative computed tomography (pQCT) to assess bone health in children, adolescents, and young adults: a review of normative data. J Pediatr Hematol Oncol 35: 581-589.
-
Bishop N, Arundel P, Clark E, Dimitri P, Farr J, et al. (2014) Fracture prediction and the definition of osteoporosis in children and adolescents: the ISCD 2013 Pediatric Official Positions. J Clin Densitom 17: 275-280.
-
Péter F, Muzsnai A, Gyimesi J. Nation-wide survey for normal values of BMD in children measured by mobil pQCT. Horm Res 2002; 58 (Suppl 2): 80.
-
Neu CM, Manz F, Rauch F, Merkel A, Schoenau E (2001) Bone densities and bone size at the distal radius in healthy children and adolescents: a study using peripheral quantitative computed tomography. Bone 28: 227-232.
-
Ashby RL, Ward KA, Roberts SA, Edwards L, Mughal MZ, et al. (2009) A reference database for the Stratec XCT-2000 peripheral quantitative computed tomography (pQCT) scanner in healthy children and young adults aged 6-19 years. Osteoporosis Int 20: 1337-46.
-
Holick MF (2007) Vitamin D deficiency. N Engl J Med 357: 266-281.
-
Institute of Medicine (2011) Dietary reference intakes for calcium and vitamin D,Washington DC, The National Academies Press.
-
Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, et al. (2011) Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 96: 1911-30.
-
Misra M, Pacaud D, Petryk A, Collett-Solberg PF, Kappy M, et al. (2008) Vitamin D deficiency in children and its management: review of current knowledge and recommendations. Pediatrics 122: 398-417.
-
Arundel P, Ahmed SF, Allgrove J, Bishop NJ, Burren CP, et al. (2012) British Paediatric and Adolescent Bone Group's position statement on vitamin D deficiency. BMJ 345: e8182.
-
Braegger C, Campoy C, Colomb V, Decsi T, Domellof M, et al. (2013) Vitamin D in the healthy European paediatric population. J Pediatr Gastroenterol Nutr 56: 692-701.
-
Stagi S, Pelosi P, Strano M, Poggi M, Manoni C, et al. (2014) Determinants of vitamin D levels in Italian children and adolescents: a longitudinal evaluation of cholecalciferol supplementation versus the improvement of factors influencing 25(OH)D status. Intern J Endocrinol: 583039.
-
Mellati AA, Sharifi F, Faghihzade S, Mousaviviri SA, Chiti H, et al. (2015) Vitamin D status and its associations with components of metabolic syndrome in healthy children. J Pediatr Endocrinol Metab 28: 641-648.
-
Saggese G, Vierucci F, Boot AM, Czech-Kowalska J, Weber G, et al. (2015) Vitamin D in childhood and adolescence: an expert position statement. Eur J Pediatr 174: 565-576.
-
Snellman G, Melhus H, Gedeborg R, Byberg L, Berglund L, et al. (2010) Determining vitamin D status: a comparison between commercially available assays. PLoS One 5: e11555.
-
de la Hunty A, Wallace AM, Gibson S, Viljakainen H, Lamberg-Allardt C, et al. (2010) UK Food Standards Agency Workshop Consensus Report: the choice of method for measuring 25- hydroxyvitamin D to estimate vitamin D status for the UK National Diet and Nutrition Survey. Br J Nutr 104: 612-619.
-
Barake M, Daher RT, Salti I, Cortas NK, Al-Shaar L, et al. (2012) 25-hydroxyvitamin D assay variations and impact on clinical decision making. J Clin Endocrinol Metab 97: 835-843.
-
González-Gross M, Valtueña J, Breidenassel C, Moreno LA, Ferrari M, et al. (2012) Vitamin D status among adolescents in Europe: the Healthy Lifestyle in Europe by Nutrition in Adolescence study. Br J Nutr 107: 755-764.
-
Schoenau E, Saggese G, Peter F, Baroncelli GI, Shaw NJ, et al. (2004) From bone biology to bone analysis. Horm Res 61: 257-269.
-
Crabtree N, Ward K (2009) Bone densitometry: current status and future perspectives. Endocr Dev 16: 58-72.
-
Gordon CM, Leonard MB, Zemel BS, International Society for Clinical Densitometry (2014) Pediatric Position Development Conference: executive summary and reflections. J Clin Densitom 17: 219-224.
-
Zemel BS, Paulhamus D, Dilzer C (2004) Precision of peripheral quantitative computed tomography measures of the tibia in children. J Bone Miner Res 19: S232.
-
Ward KA, Das G, Roberts SA, Berry JL, Adams JE, et al. (2010) A randomized, controlled trial of vitamin D supplementation upon musculoskeletal health in postmenarchal females. J Clin Endocrinol Metab 95: 4643-4651.
-
Cheng S, Tylavsky F, Kröger H, Kärkkäinen M, Lyytikäinen A, et al. (2003) Association of low 25-hydroxyvitamin D concentrations with elevated parathyroid hormone concentrations and low cortical bone density in early pubertal and prepubertal Finnish girls. Am J Clin Nutr 78: 485-492.
-
Talwar SA, Swedler J, Yeh J, Pollack S, Aloia JF (2007) Vitamin-D nutrition and bone mass in adolescent black girls. J Natl Med Assoc 99: 650-657.
-
Sayers A, Fraser WD, Lawlor DA, Tobias JH (2012) 25- hydroxyvitamin-D 3 levels are positively related to subsequent cortical bone development in childhood: findings from a large prospective cohort study. Osteoporosis Int 23: 2117-2128.
-
Warden SJ, Hill KM, Ferira AJ, Laing EM, Martin BR, et al. (2013) Racial differences in cortical bone and their relationship to biochemical variables in Black and White children in the early stages of puberty. Osteoporos Int 24: 1869-1879.
-
Srivastava T, Garg U, Ruiz M, Dai H, Alon US (2013) Serum 25(OH)-vitamin D level in children: is there a need to change the reference range based on 2011 Institute of Medicine report? Clin Pediatr (Phila) 52: 178-182.
-
Anderson PH, Turner AG, Morris HA (2012) Vitamin D actions to regulate calcium and skeletal homeostasis. Clin Biochem 45: 880-886.
-
Ryan JW, Anderson PH, Turner AG, Morris HA (2013) Vitamin D activities and metabolic bone disease. Clin Chim Acta 425: 148-152.
-
Need AG, O'Loughlin PD, Morris HA, Coates PS, Horowitz M, et al. (2008) Vitamin D metabolites and calcium absorption in severe vitamin D deficiency. J Bone Miner Res 23: 1859-1863.
-
Aggarwal V, Seth A, Aneja S, Sharma B, Sonkar P, et al. (2012) Role of calcium deficiency in development of nutritional rickets in Indian children: a case control study. J Clin Endocrinol Metab 97: 3461-3466.