Journal of Nutritional Medicine and Diet Care
Niacin and Oxidative Stress: A Mini-Review
Faranak Ilkhani1, Banafshe Hosseini2 and Ahmad Saedisomeolia2,3,4*
1Department of Animal Science, School of Animal Nutrition, Urmia Agricultural University, Urmia, Iran
2Department of Cellular and Molecular Nutrition, School of Nutritional Sciences and Dietetics, Tehran University of Medical Sciences, Tehran, Iran
3Department of Pharmacology, School of Medicine, Western Sydney University, NSW, Australia
4School of Molecular Bioscience, The University of Sydney, NSW, Australia
*Corresponding author:
Ahmad Saedisomeolia, Department of Pharmacy, School of Medicine, Western Sydney University, Campbelltown Campus, NSW 2560, Australia, Tel: +61246203212, E-mail: A.Saedisomeolia@westernsydney.edu.au
J Nutri Med Diet Care, JNMDC-2-014, (Volume 2, Issue 1), Review Article
Received: February 26, 2016: Accepted: May 14, 2016: Published: May 18, 2016
Citation: Ilkhani F, Hosseini B, Saedisomeolia A (2016) Niacin and Oxidative Stress: A Mini-Review. J Nutri Med Diet Care 2:014.
Copyright: © 2016 Ilkhani F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Oxidative stress has been implicated in the pathogenesis of a variety of chronic diseases. One of the main factors involved in oxidative stress reduction is increased antioxidant potential. Several nutrients such as vitamin C, vitamin E and carotenoids are known to act as antioxidants; however, niacin is one of the neglected antioxidant nutrients that may have an antioxidant action both independently, and also as a component of the glutathione redox cycle. Thus, this study aimed to review the studies that have examined the antioxidant properties of niacin and its effect on oxidative stress reduction. The results of the reviewed studies confirm the antioxidant nature of niacin and indicate that this vitamin can protect the body against oxidative stress, specifically lipid peroxidation and reperfusion oxidative injury. The mechanisms by which niacin protects the body against oxidative stress can be attributed to the glutathione redox cycle and also to other possible roles such as decreasing NADH+H+/NADP+ ratio as well as increasing the NAD+ content.
Keywords
Niacin, Lipid peroxidation, Reperfusion oxidative injury, Glutathione peroxidase
Introduction
Oxidative stress is a phenomenon that reflects an imbalance between the production of reactive oxygen species (ROS) and other oxidants, and their elimination through protective mechanisms. Antioxidative systems can detoxify the reactive intermediates, or repair the consequential damages causing toxic effects through the production of peroxides and free radicals that can destroy all cell components. Oxidative stress is thought to be involved in the development of atherosclerosis, neurodegenerative diseases such as Alzheimer's and Parkinson's disease, cancers, diabetes mellitus, inflammatory diseases, as well as psychological diseases and aging processes.
One of the main factors involved in oxidative stress reduction is increased antioxidant potential, that may be achieved by endogenous antioxidants, such as the enzymes Superoxide Dismutase (SOD), Catalase (CAT) and Glutathione Reductase (GR), and also, exogenous antioxidants like nutritional antioxidants including tocopherols, ascorbic acid, carotenoids, niacin, and trace elements such as chromium and selenium. There are also specific biological defense mechanisms that protect tissues against cellular damage. Niacin acts as a coenzyme of redox enzymes in Nicotinamide Adenine Dinucleotide (NAD) and Nicotinamide Mononucleotide (NMN) forms [1]. Many studies have examined the effects of niacin on various diseases such as anemia [2,3], hypertension [4], cardiovascular diseases (CVD) [5,6], liver diseases [7,8] and some cancers (esophageal, skin, breast, and lung) [9-11]. However, one of the roles through which niacin can have a potential effect on human health is that as an antioxidant, which has not been clearly investigated.
Compelling evidence from both in vitro and in vivo studies supported that Nicotinamide (NA) possesses potent antioxidant properties [12,13]. NA deficiency was associated with increased oxidative stress.
In this review, two aspects of the antioxidant properties of niacin are considered: (1) the role of niacin in the prevention of lipid peroxidation (LPO) and (2) the effect of niacin on the attenuation of reperfusion oxidative injury. Human and animal studies are summarized in table 1.
![]()
Table 1: Application of Niacin to inhibit oxidative stress and lipid peroxidation.
View Table 1
Niacin at a Glance
Niacin and its coenzymes NAD and Nicotinamide Adenine Dinucleotide Phosphate (NADP) have fundamental roles as a part of reduction/oxidation coenzymes involved in energy metabolism, amino acid metabolism, detoxification reactions for drugs and other substances as well as antioxidant protection (Figure 1) [14,15]. Niacin can be synthesized by the essential amino acid tryptophan. Even though this process is not efficient, dietary tryptophan intake seems crucial for the overall niacin status of the body. The Food and Nutrition Board (FNB) has recommended a daily intake of 2-4 mg niacin/day for infants, 6-8 mg niacin/day for children, 12-16 mg niacin/day for adolescents, and 14-18 mg niacin daily by mouth for adults. Considerable amounts of niacin have been found in a wide range of foods including lean meats, poultry, fish, peanuts, and yeasts.
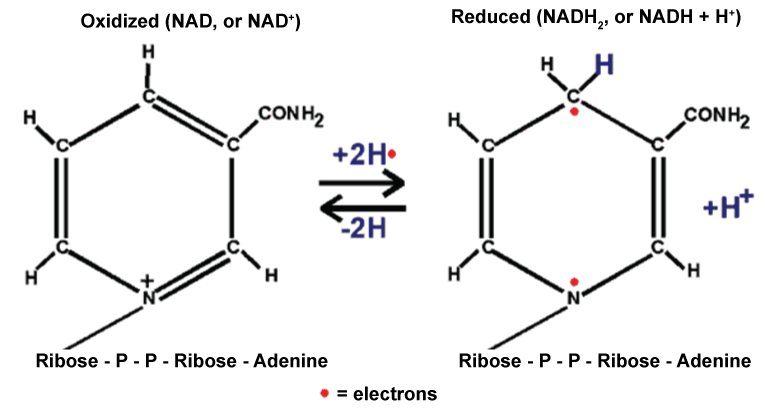
.
Figure 1: Conversion of NADH+H+ to the NAD+-a possible mechanism for its antioxidant nature [15].
View Figure 1
Niacin deficiency can be caused by problems that affect absorption of niacin or tryptophan. Digestive disorders and prolonged treatment with the tuberculosis drug Isoniazid (Laniazid, Nydrazid) can be the risk factors of niacin deficiency. Additionally, alcoholism is regarded as the most common cause of this problem. Cases of niacin deficiency have been also found in patients suffering from Crohn's disease.
Generally, niacin has a low toxicity. High doses of niacin (1-2 g of NA three times per day) can be used in decreasing serum cholesterol. The upper limit (UL) of niacin suggested by the US Food and Nutrition Board is 35 mg/day. It can be suggested that a high dose of niacin could cause an imbalance in the antioxidant state of human body. However, there is not any strong evidence in this area, which recommends further investigations to clarify the possible adverse effects of niacin intake in high amounts.
Role of Niacin in the Prevention of Lipid Peroxidation
Niacin as the glutathione reductase coenzyme
NADPH is an important coenzyme of the GR, which converts oxidized glutathione (GSSG) to its reduced form (GSH) (Figure 2). GSH acts as a co-substrate for glutathione peroxidase (GPx) [16,17], an endogenous antioxidant in various cell types which deactivates ROS. Through its action, by converting this peptide to the oxidized form, it becomes deactivated. Thus, to recovering the antioxidant properties of glutathione, GSSG should be reduced again to GSH by GR. Intracellular NADPH, in turn, is generated through the reduction of NADP+ by glucose-6-phosphate dehydrogenase, the first and rate-limiting enzyme of the pentose phosphate pathway, during the conversion of glucose-6-phosphate to 6-phosphogluconolactone. By generating NADPH, glucose-6-phosphate dehydrogenase is a critical determinant of cytosolic GSH buffering capacity (GSH/GSSG), therefore, it can be considered as an essential, regulatory antioxidant enzyme. It has been suggested that oxidative stress can be increased during niacin deficiency by the ability to regenerate GSH through the use of NADPH in the GR reaction [18]. NADPH adequately maintained by the pentose phosphate pathway, providing sufficient substrate for GSH regeneration in the GSH reductase reaction. This is supported by the lack of any GSH reduction during niacin deficiency [19].
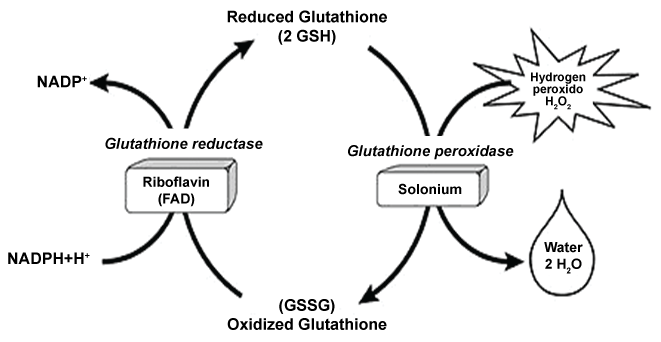
.
Figure 2: Conversion of oxidized glutathione (GSSG) to the reduced form (GSH) by Glutathione Reductase (GR) [19].
View Figure 2
One of the most important antioxidant activities of glutathione is the deactivation of peroxides such as hydroperoxide. This activity of glutathione is mediated by the action of GPx. GPx transfers a hydrogen ion from GSH to lipid peroxide and produces GSSG and alcohol [20]. Based on the aforementioned mechanisms, it is expected that niacin deficiency could increase LPO [21].
Effect of niacin status on glutathione content in tissues
The effect of niacin status on GSH content in tissue has been investigated in a limited number of studies. Benavente et al. [9] found that GSH was increased in restricted cells where GSSG and GSH/GSSG levels were decreased. NA supplementation either alone [4,7,22], or in a combination with other factors [14,23,24] has been shown to moderate the up-regulation of oxidative and inflammatory systems and increase the GSH levels. However, to our knowledge, there are a limited number of human studies that examined the effect of niacin on GSH status. Some human studies reported significant increase in GSH content with pharmacological doses of niacin [2,9,12].
However, some animal studies did not report any changes in GSH content. In an experimental study conducted by Tang et al. [18] no diet-induced change was observed in GSH concentration in total or nucleated bone marrow population in rats, which would indicate that shifts in thiol-dependent transcriptional activity, can play a role in niacin deficiency [18]. The authors reported that measurements of GSSG were elevated, especially in the total cell population. Taken together, these results showed that the reductive roles in oxidant defense were not compromised and increased level of oxidant stress in niacin-deficient bone marrow cells resulted from other events [18].
Effect of niacin status on the activity of antioxidant enzymes
An experimental study indicated that niacin can increase SOD and GPx activities in hepatic tissue in rats [22]. Another study that investigated the effects of dietary niacin on antioxidant defense mechanism of rats, reported a significant increase in levels of SOD, CAT, GPx, glutathione and zinc, while, LPO level was lower in the intervention group [22]. Several studies have reported similar findings on the positive effects of niacin on SOD [14], GPx [7,14,25] and CAT [7,14,23] activities, although, some studies did not observe any association between niacin status and antioxidant enzyme activity [26].
In conclusion, studies indicated that niacin status could affect the activity of several antioxidant enzymes; however, some studies did not attain the similar findings. Furthermore, as studies have been limited to animals, further investigations are warranted in human populations to clarify the role of niacin in the activity of antioxidant enzymes.
Effect of niacin status on lipid peroxidation
Animal studies: Several animal studies indicated that not only niacin deficiency could adversely affect LPO, but also, niacin administration had inhibitory effects on it [4,14,22-24,27]. Studies in which niacin deficiency was induced in animals through a niacin-deficient diet, LPO in different tissues was found to be significantly increased compared to the control groups [4,22,27]. Other studies reported that niacin administration could reduce the production of LPO biomarkers such as malondialdehyde (MDA) in rats [4,22].
Human studies: A limited number of human studies investigated the effects of niacin status on LPO. Arun et al. [2] investigated the effect of nicotinic acid added to the Citrate-Phosphate-Dextrose-Adenine (CPDA) solution on LPO and integrity of red cells. The authors reported that MDA levels decreased, while, concentrations of GSH and vitamin E increased by adding nicotinic acid [2]. In another study, the effects of 27 days of niacin supplementation on intestinal permeability and oxidative stress were investigated in patients suffering from alcoholic pellagra. The study indicated that lipid and protein peroxidation biomarkers had a significant decrease (P < 0.05) after 27 days of niacin supplementation [25]. In the same line with these studies, Hamoud et al. [28] addressed the effects of 3-month niacin supplementation (1 g/day in the first month, followed by 2 g/day for the second and third months) in patients with hypercholesterolemia and low HDL-C levels and in healthy control subjects (received no intervention). The authors reported that niacin treatment in hypercholesterolemic patients led to a significant reduction in oxidative stress, as measured by a significant decrease in the serum concentration of thiobarbituric acid reactive substances, and also, paraoxonase 1(PON1) activity was significantly increased after niacin treatment for 12 weeks, compared with the initial levels. The study also indicated that serum interleukin-6 level significantly decreased following the niacin intervention. However, it should be noted that serum level of C-reactive protein (CRP) was not affected by niacin supplementation. Similarly, findings from a prospective cohort study [29] indicated that plasma oxidized LDL was inversely correlated with dietary niacin intake (r = -0.23, P < 0.05) and was lower in participants with above 22 mg/day vs. below-average niacin intake .Moreover, Ganji et al. [6] investigated the effect of niacin on human hepatocyte fat accumulation, ROS production, and inflammatory mediator interleukin-8 secretion. The authors reported that niacin (at 0.25 and 0.5 mmol/L doses for 24 h) decreased hepatocyte ROS production, palmitic acid-induced interleukin-8 levels and it also inhibited NADPH oxidase activity. However, these findings were not fully supported by all studies. Taylor et al. [30] assessed the changes PON1 activity and concentration after single aerobic exercise sessions conducted before and after 6 weeks of niacin supplementation in men with metabolic syndrome (MetS). Niacin dosage was titrated by 500 mg/week from 500 to 1500 mg daily and maintained at 1500 mg daily for the last 4 weeks. PON1 activity, PON1 concentration, myeloperoxidase (MPO), and ox-LDL had no change following the independent effects of exercise and niacin (P > 0.05 for all). However, PON1 activity increased by 6.1% (P = 0.037) and PON1 levels increased by 11.3% (P = 0.015) with the combination of exercise and niacin. The authors concluded that exercise and niacin works synergistically to enhance PON1 activity and concentration with little or no changes in markers of lipid oxidation.
In summary, although there are discrepant findings regarding the niacin and LPO, the majority of literature indicated that niacin intake/ status is negatively associated with LPO and ROS production.
Effect of niacin on the attenuation of reperfusion oxidative injury
Reperfusion injury is the tissue damage that occurs when blood flows into the tissue after a period of ischemia. It has been shown that free radicals [16,17] and inflammatory cytokines [31] have a key role in the reperfusion injury process. Tai et al. [32] showed that treatment with niacin, as a precursor of NADH and an effective antioxidant, improved kidney I/R-induced cardiac dysfunction and the severity of myocardial LPO through maintaining myocardial peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1a) expression to a level comparable to that of the sham group. The authors also reported that niacin led to a reduction in kidney injury. Niacin is commonly used in treating dyslipidemia because of its ability to decrease lipolysis and the production of very-low-density lipoprotein, and also, it increases the production of high-density lipoprotein [33]. Previously, Brown et al. [34] showed that niacin is effective in reducing myocardial I/R injury in patients with dyslipidemia, potentially through reducing plasma lipid. Furthermore, Lamping et al. [35] showed that the cardiac protective effects of niacin may be independent of the systemic lipids, and associated with a reduction in free fatty acids uptake and utilization of triglyceride. Additionally, Trueblood et al. [33] showed that niacin treatment effectively reduced the cytosolic NADH/NAD+ ratio during myocardial I/R injury, which helps to sustain mitochondrial respiration function and ATP production.
In the study by Thirunavukkarasu et al. [36] the effects of 90 days oral administration of niacin-bound, chromium-based energy formula (40 mg•kg body wt-1•day-1) on the cardiovascular and pathophysiological functions in an isolated rat heart model were investigated in male and female rats. The authors reported that aortic flow, maximum first derivative of developed pressure, left ventricular developed pressure, and infarct size were significantly improved following the niacin formulation comparison with the control group. Energy formula, which includes niacin-bound chromium and D-ribose, might activate adenosine monophosphate-activated protein kinase (AMPK), cause an increase in ATP levels by triggering the ATP-generating pathways and decreasing the energy demand through reducing the ATP consuming process that regulates the cellular energy status, causing cardio protective effects.
Energy formula (EF) supplementation increases the expression of heat shock proteins (HSPs) like HSP-70, -32, and -25. HSP-70 is found to protect cardiac cells against simulated ischemia or thermal stress in vitro [37] as well as in a model of ischemia-reperfusion injury via the suppression of inflammatory cytokines [38]. Additionally, Sammut et al. [39] reported that HSP-70 upregulation may protect the mitochondrial energy metabolism in the injured heart by repairing the ion channels under stress conditions. HSP-25 (also called as HSP-27) is relatively higher in heart tissue [40], and known to play a role in retaining the redox balance, and also, has a regulatory role in muscle contraction [40]. In addition, the results of functional recovery of EF-treated animals after ischemia might be its amino acids contents which are precursors of important molecules like glutathione that may be responsible for ischemic myocardial protection [41].
Future Directions and Conclusion
The role of niacin as an antioxidant micro-nutrient has been established in a variety of research. The mechanisms through which niacin protects the body against oxidative stress can be retaining the glutathione redox cycle, and also, decreasing NADPH/total NADP+ ratio as well as increasing the NAD+ content. However most of the investigations in this area are limited to experimental studies, these mechanistic studies can be useful for further clinical development of niacin and niacin-related compounds for the treatment of oxidative stress and its complications.
Financial Support
This paper was not supported by any organization.
Conflict of Interest
None
References
-
Long AN, Owens K, Schlappal AE, Kristian T, Fishman PS, et al. (2015) Effect of nicotinamide mononucleotide on brain mitochondrial respiratory deficits in an alzheimer's disease-relevant murine model. BMC Neurol 15:19.
-
Arun P, Padmakumaran Nair KG, Manojkumar V, Deepadevi KV, Lakshmi LR, et al. (1999) Decreased hemolysis and lipid peroxidation in blood during storage in the presence of nicotinic acid. Vox Sang 76: 220-225.
-
Boyonoski AC, Gallacher LM, ApSimon MM, Jacobs RM, Shah GM, et al. (2000) Niacin deficiency in rats increases the severity of ethylnitrosourea-induced anemia and leukopenia. J Nutr 130: 1102-1107.
-
Cho KH, Kim HJ, Rodriguez-Iturbe B, Vaziri ND (2009) Niacin ameliorates oxidative stress, inflammation, proteinuria, and hypertension in rats with chronic renal failure. Am J Physiol Renal Physiol 297: F106-13.
-
Brown BG, Zhao XQ, Chait A, Fisher LD, Cheung MC, et al. (2001) Simvastatin and niacin, antioxidant vitamins, or the combination for the prevention of coronary disease. N Engl J Med 345: 1583-1592.
-
Ganji SH, Kashyap ML, Kamanna VS (2014) Niacin inhibits fat accumulation and oxidative stress in human hepatocytes and regresses hepatic steatosis in experimental rat model. J Clin Lipid 8: 349-350.
-
Dou X, Shen C, Wang Z, Li S, Zhang X, et al. (2013) Protection of nicotinic acid against oxidative stress-induced cell death in hepatocytes contributes to its beneficial effect on alcohol-induced liver injury in mice. J Nutr Biochem 24: 1520-1528.
-
Ganji SH, Kashyap ML, Kamanna VS (2015) Niacin inhibits fat accumulation, oxidative stress, and inflammatory cytokine il-8 in cultured hepatocytes: Impact on non-alcoholic fatty liver disease. Metabolism 64: 982-990.
-
Benavente CA, Jacobson EL (2008) Niacin restriction upregulates nadph oxidase and reactive oxygen species (ros) in human keratinocytes. Free Radic Biol Med 44: 527-537.
-
Jacobson EL, Shieh WM, Huang AC (1999) Mapping the role of nad metabolism in prevention and treatment of carcinogenesis. Mol Cell Biochem 193: 69-74.
-
Yuvaraj S, Premkumar VG, Vijayasarathy K, Gangadaran SG, Sachdanandam P (2007) Ameliorating effect of coenzyme q10, riboflavin and niacin in tamoxifen-treated postmenopausal breast cancer patients with special reference to lipids and lipoproteins. Clin Biochem 40: 623-628.
-
Ganji SH, Qin S, Zhang L, Kamanna VS, Kashyap ML (2009) Niacin inhibits vascular oxidative stress, redox-sensitive genes, and monocyte adhesion to human aortic endothelial cells. Atherosclerosis 202: 68-75.
-
Spronck JC, Nickerson JL, Kirkland JB (2007) Niacin deficiency alters p53 expression and impairs etoposide-induced cell cycle arrest and apoptosis in rat bone marrow cells. Nutr Cancer 57: 88-99.
-
Perumal SS, Shanthi P, Sachdanandam P (2005) Augmented efficacy of tamoxifen in rat breast tumorigenesis when gavaged along with riboflavin, niacin, and coq10: Effects on lipid peroxidation and antioxidants in mitochondria. Chem Biol Interact 152: 49-58.
-
(2006) L. C. Glycolysis Lecture 8 biol.
-
Ambrosio G, Flaherty JT, Duilio C, Tritto I, Santoro G, et al. (1991) Oxygen radicals generated at reflow induce peroxidation of membrane lipids in reperfused hearts. J Clin Invest 87: 2056-2066.
-
Bolli R, Zughaib M, Li XY, Tang XL, Sun JZ, et al. (1995) Recurrent ischemia in the canine heart causes recurrent bursts of free radical production that have a cumulative effect on contractile function. A pathophysiological basis for chronic myocardial "stunning". J Clin Invest 96: 1066-1084.
-
Tang K, Sham H, Hui E, Kirkland JB (2008) Niacin deficiency causes oxidative stress in rat bone marrow cells but not through decreased nadph or glutathione status. J Nutr Biochem 19: 746-753.
-
Feng L, Li SQ, Jiang WD, Liu Y, Jiang J, et al. (2015) Deficiency of dietary niacin impaired intestinal mucosal immune function via regulating intestinal nf-kappab, nrf2 and mlck signaling pathways in young grass carp (ctenopharyngodon idella). Fish Shellfish Immunol 49:177-193.
-
Mulherin DM, Thurnham DI, Situnayake RD (1996) Glutathione reductase activity, riboflavin status, and disease activity in rheumatoid arthritis. Ann Rheum Dis 55: 837-840.
-
(2016) Nebraska redox biology center educational portal.
-
Tupe RS, Tupe SG, Agte VV (2011) Dietary nicotinic acid supplementation improves hepatic zinc uptake and offers hepatoprotection against oxidative damage. Br J Nutr 105: 1741-1749.
-
Atac IA, Peksel A, Yanardag R, Sokmen BB, Doger MM, et al. (2006) The effect of combined treatment with niacin and chromium (iii) chloride on the different tissues of hyperlipemic rats. Drug Chem Toxicol 29: 363-377.
-
Ghazi-Khansari M, Nasiri G, Honarjoo M (2005) Decreasing the oxidant stress from paraquat in isolated perfused rat lung using captopril and niacin. Arch Toxicol 79: 341-345.
-
Varella Morandi Junqueira-Franco M, Ernesto Troncon L, Garcia Chiarello P, do Rosario Del Lama Unamuno M, Afonso Jordao A, et al. (2006) Intestinal permeability and oxidative stress in patients with alcoholic pellagra. Clin Nutr 25: 977-983.
-
Bernabucci U, Ronchi B, Lacetera N, Nardone A (2002) Markers of oxidative status in plasma and erythrocytes of transition dairy cows during hot season. J Dairy Sci 85: 2173-2179.
-
Doger MM, Sokmen BB, Yanardag R (2011) Combined effects of niacin and chromium treatment on heart of hyperlipidemic rats. Hum Exp Toxicol 30: 1561-1566.
-
Hamoud S, Hayek T, Hassan A, Meilin E, Kaplan M, et al. (2013) Niacin administration significantly reduces oxidative stress in patients with hypercholesterolemia and low levels of high-density lipoprotein cholesterol. Am J Med Sci 345: 195-199.
-
Kaplon RE, Gano LB, Seals DR (1985) 2014 Vascular endothelial function and oxidative stress are related to dietary niacin intake among healthy middle-aged and older adults. J Appl Physiol 116: 156-163.
-
Taylor JK, Plaisance EP, Mahurin AJ, Mestek ML, Moncada-Jimenez J, et al. (2015) Paraoxonase responses to exercise and niacin therapy in men with metabolic syndrome. Redox Rep 20: 42-48.
-
Kukielka GL, Smith CW, Manning AM, Youker KA, Michael LH, et al. (1995) Induction of interleukin-6 synthesis in the myocardium. Potential role in postreperfusion inflammatory injury. Circulation 92: 1866-1875.
-
Tai ST, Fu YH, Yang YC, Wang JJ (2015) Niacin ameliorates kidney warm ischemia and reperfusion injury-induced ventricular dysfunction and oxidative stress and disturbance in mitochondrial metabolism in rats. Transplant Proc 47: 1079-1082.
-
Trueblood NA, Ramasamy R, Wang LF, Schaefer S (2000) Niacin protects the isolated heart from ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 279: H764-71.
-
Brown G, Albers JJ, Fisher LD, Schaefer SM, Lin JT, et al. (1990) Regression of coronary artery disease as a result of intensive lipid-lowering therapy in men with high levels of apolipoprotein B. N Engl J Med 323:1289-1298.
-
Lamping KA, Menahan LA, Gross GJ (1984) Nicotinic acid, free fatty acids and myocardial function during coronary occlusion and reperfusion in the dog. J Pharmacol Exp Ther 231: 532-538.
-
Thirunavukkarasu M, Penumathsa SV, Juhasz B, Zhan L, Cordis G, et al. (2006) Niacin-bound chromium enhances myocardial protection from ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 291: H820-6.
-
Brar BK, Stephanou A, Wagstaff MJ, Coffin RS, Marber MS, et al. (1999) Heat shock proteins delivered with a virus vector can protect cardiac cells against apoptosis as well as against thermal or hypoxic stress. J Mol Cell Cardiol 31: 135-146.
-
Grunenfelder J, Zund G, Stucki V, Hoerstrup SP, Kadner A, et al. (2001) Heat shock protein upregulation lowers cytokine levels after ischemia and reperfusion. Eur Surg Res 33: 383-387.
-
Sammut IA, Jayakumar J, Latif N, Rothery S, Severs NJ, et al. (2001) Heat stress contributes to the enhancement of cardiac mitochondrial complex activity. Am J Pathol 158: 1821-1831.
-
Huot J, Houle F, Spitz DR, Landry J (1996) Hsp27 phosphorylation-mediated resistance against actin fragmentation and cell death induced by oxidative stress. Cancer Res 56: 273-279.
-
Pasini E, Scarabelli TM, D'Antona G, Dioguardi FS (2004) Effect of amino acid mixture on the isolated ischemic heart. Am J Cardiol 93: 30a-34a.





