Journal of Rheumatic Diseases and Treatment
A MRI Assessment of the Response of Chronic, Occult, Synovial-Based Inflammation of Gout to Serum Urate Lowering Therapy
John D. Carter*, Michelle Patelli, Scott R. Anderson, Neelesh Prakash, Ernesto J. Rodriquez and Louis R. Ricca
University of South Florida, Tampa, FL, USA
*Corresponding author: John D Carter, M.D. University of South Florida; Tampa, FL, USA, 12901 Bruce B. Downs Blvd, MDC 81, Tampa, FL 33612, USA, Tel: (813) 974-2681, Fax: (813) 974-5229, E-mail: jocarter@health.usf.edu
J Rheum Dis Treat, JRDT-1-019, (Volume 1, Issue 3), Research Article; ISSN: 2469-5726
Received: August 13, 2015 | Accepted: September 07, 2015 | Published: September 09, 2015
Citation: Carter JD, Patelli M, Anderson SR, Prakash N, Rodriquez EJ, et al. (2015) A MRI Assessment of the Response of Chronic, Occult, Synovial-Based Inflammation of Gout to Serum Urate Lowering Therapy. J Rheum Dis Treat 1:019. 10.23937/2469-5726/1510019
Copyright: © 2015 Carter JD, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: We have previously demonstrated the severity of synovitis in patients with inter-critical gout did not correlate with serum urate levels (sUA). The aim of this sub-study was to determine if serum urate lowering therapy can improve this chronic synovitis.
Methods: All participants had inter-critical gout and received a 3T MRI with and without gadolinium of their index joint (i.e. the joint most often involved with acute attacks). If the subject had a sUA of ≥ 7.0 mg/dL and evidence of synovial pannus on their MRI, they were eligible for enrollment. All were treated with febuxostat with a target sUA of ≤ 6.0 mg/dL. At month 9, the MRI of the index joint was repeated and compared to baseline. The MRI's were read by two musculoskeletal radiologists in a blinded fashion. The primary endpoint was to determine if there was significant improvement in the severity of synovial pannus from baseline to month 9.
Results: 25/32 subjects completed the protocol. The average sUA and synovial pannus score at screening was 9.3 mg/dL (+/- 1.3 SD) and 3.66 (+/- 1.2 SD), respectively. At month 9, the average sUA decreased to 5.36 mg/dL (+/- 1.4 SD; p < 0.0001), but there was no significant change in the severity of synovial pannus with the average score being 3.42 (+/- 1.3 SD; p = 0.34). The inter-reader agreement between the two radiologists was good (kappa = 0.63). However, a post-hoc analysis using a more quantitative synovial pannus scoring scale suggested a significant decrease.
Conclusions: Nine months of serum urate lowering therapy significantly decreases serum urate levels but the effect on the chronic, occult, synovial-based inflammation of gout remains unclear.
Introduction
Clinical gout begins as an acute intermittent and intensely painful arthritis that can progress to a chronic, inflammatory, destructive condition. The initial episodes may last several days and are often followed by pain-free, inter-critical periods that may last months to years. Chronic gouty arthritis is manifested by shorter inter-critical periods, boney destructive changes and possible development of visible tophi. Advanced gout leads to chronic pain, decreased quality of life, and an increased global economic burden [1-5]. Epidemiologic data suggest that the prevalence of gout is increasing [6]. The obvious key to controlling the natural course of this serious disease is an efficacious therapy that treats the root cause.
It has been known for years that hyperuricemia is the primary risk factor for the development of clinical gout. There is also a large breadth of data demonstrating that serum urate lowering therapy (ULT) decreases the likelihood of future clinical attacks of gout [7]. Further, the more aggressive the serum ULT, the less likely the patient will experience future attacks [7]. Similar studies have demonstrated that serum ULT can reduce or even resolve soft-tissue tophi [8].
It is accepted that joint damage can continue to occur during the inter-critical periods of gout if the patient remains hyperuricemic. This is based off data demonstrating the presence of monosodium urate (MSU) crystals and evidence of ongoing inflammation in the synovial fluid of patients during the inter-critical period [9-11].
Surprisingly, there are very few data evaluating the potential long-term radiographic protective effect of serum ULT in patients with gout. The largest study to evaluate the effect of serum ULT on radiographic progression demonstrated that although reduced tophaceous deposition on physical exam correlated with normalization of serum urate levels, no correlation existed between radiographic changes and mean serum urate levels [12]. Conversely, a recent exploratory study of eight patients with gout suggested that profound ULT with pegloticase might lead to improvement in structural damage [13].
We have previously demonstrated that MRI is the best advanced imaging technique at demonstrating the synovial pannus that is present during inter-critical gout [14]. We recently performed a study involving 72 participants with gout demonstrating that synovial pannus is present in the vast majority of patients with inter-critical gout, but the severity of this synovitis did not correlate with serum urate levels [15]. This study involved a pilot sub-study in which we treated 32 of these 72 subjects for nine months with febuxostat with a target serum urate level of less than 6mg/dL. The aim of this sub-study was to determine if serum urate lowering therapy can improve this chronic occult synovial-based inflammation. Improvement in synovial-based inflammation during inter-critical gout likely has a radiographic protective effect.
Patients and Methods
Patients and study design
This was a single-center prospective pilot study performed at the University of South Florida (USF Health) in Tampa, FL. It was a sub-study of the parent study assessing the prevalence of synovial pannus and its correlation with serum urate levels in patients with inter-critical gout [15]. All of the assessments, including MRI's, were performed at the same location. Ethical approval was obtained from the USF Institutional Review Board and all study participants gave informed consent and approval prior to undergoing any study-related procedures. The trial was registered at www.clinicaltrials.gov, identifier: NCT01112982.
Eligible patients were aged 18 years or greater with a known history of gout and able to give informed consent. To be included in the study, the patient's diagnosis of gout was defined by one of three criteria: current or previous documentation of intracellular MSU crystals in synovial fluid, a tophus proved to contain MSU crystals, or at least six of the American College of Rheumatology (ACR) diagnostic criteria for gout [16]. All participants were screened during an inter-critical period and they were excluded if they had an acute attack of gout (any joint) within 4 weeks of screening. Potential participants were also excluded if they had a history of any other type of inflammatory arthritis, including another crystal induced arthritis. Because all subjects had to have a MRI with gadolinium contrast, they were excluded if their serum creatinine was >1.8 mg/dL, or if their estimated glomerular filtration rate (eGFR) was ≤ 30 mL/min/1.73 m2. Because the primary endpoint included documented synovial pannus on MRI, participants were excluded if they received oral corticosteroids (any dose) within 4 weeks, or parenteral or intrarticular corticosteroids within 6 weeks, of their screening visit and MRI. In order to qualify for the sub-study, the participant had to have evidence of synovial pannus (any degree) on the baseline MRI of the index joint (the joint most frequently involved with gouty attacks) and have a serum urate level of ≥ 7.0 mg/dL at their screening visit. All subjects also received a baseline plain radiograph of the index joint to check for the presence of erosions and osteoarthritis.
If the participant met these criteria and were not on a serum ULT or were on a serum ULT therapy other than febuxostat, they were treated in a prospective fashion with febuxostat with the goal of achieving a serum urate level target of < 6.0 mg/dL. All subjects were treated in an open-label fashion for nine months starting at a dose of 40mg by mouth daily of febuxostat and increasing to 80mg daily if the target serum urate level was not achieved at any point after baseline. The serum urate levels (as well as other safety laboratories) were obtained at week 2 and then months 1, 3, 6, and 9. All subjects were also treated with colchicine as a prophylaxis against mobilization flares until month 6. The dose of colchicine was 0.6mg by mouth twice daily (unless renal adjustment was required) until month 3 and then 0.6 mg daily by mouth until month 6 at which point it was discontinued. Corticosteroids (oral, parenteral, or intra-articular) were not allowed at any point. All study participants then had a MRI repeated of their index joint at month 9.
All of the MRI's of the index joints (at baseline and month 9) were 3 Tesla, with and without gadolinium, and included T1, T2, and STIR images. All of these MRI's were assessed independently by two musculoskeletal radiologists (SRA and NP), independent of time, and in a blinded fashion. The MRI's were assessed for the severity of synovial pannus, which was graded on a previously utilized scale of 1 to 6 (with 6 being the most severe) [17]. The six grades of synovial pannus on MRI were defined as follows: Grade 1, no contrast enhancement; Grade 2, focal synovial contrast enhancement; Grade 3, diffuse synovial contrast enhancement; Grade 4, grade 3 plus diffuse synovial thickening; Grade 5, grade 4 plus villonodular synovial thickening; Grade 6, grade 5 plus cartilage and subchondral bone erosions. All of the MRI's were also assessed for bone erosions, intraosseous tophi, soft-tissue tophi, soft-tissue edema, joint effusions, and bone marrow reactive changes. Baseline radiographs were also assessed independently in a blinded fashion for erosions and joint space narrowing. Both radiologists had to agree on the presence of these radiographic findings in order to be counted.
Study objectives
The primary endpoint of this sub-study was to determine if a nine month course of aggressive serum ULT in patients with gout significantly affects the severity of synovial pannus in the index joint as determined by comparing the baseline and month 9 MRI's using the aforementioned grading scale. Secondary endpoints included an assessment of significant change of the subjects' serum CRP and estimated Glomerular Filtration Rate (eGFR) from baseline to month 9. Other endpoints included assessing for significant change of erosive changes, intraosseous tophi, soft tissue tophi, joint effusion, bone marrow edema/lesions, and soft tissue edema on the MRI from baseline to month 9.
Statistical analysis
The target enrollment for this sub-study was 32 subjects. This sample size was determined using an a-priori calculation for a Student's T-test with an alpha level of 0.05, an anticipated effect size of 0.8 (i.e. large effect), and a statistical power of 0.8. This equated to 26 subjects. We enrolled 32 participants to allow for ~20% dropout or lost to follow-up.
The baseline MRI pannus scores were compared to the 9 month MRI pannus scores using a paired t-test. The same statistical method was utilized to compare baseline and month 9 CRP's and eGFR's. Fisher exact test was used to compare the other MRI findings on baseline compared to month 9. In instances where the two radiologists disagreed on the exact grade of synovial pannus, the two grades were averaged. Inter-reader agreement between the two musculoskeletal radiologists with regard to severity of synovial pannus on MRI was assessed by calculating a kappa-coefficient.
Results
Characteristics of the study population
There were 32 participants who signed informed consent and enrolled in this sub-study; 25/32 subjects completed the entire 9 month protocol. Table 1 for the patient baseline demographics of all 32 participants. Of the seven subjects who did not complete the protocol, two subjects withdrew consent and five were lost to follow-up. The participants were primarily males with disease duration of approximately 10 years. The first metatarsophalangeal (MTP) joint was the most common index joint; this was the case in 25/32 (78%) subjects. 9/32 (28%; kappa = 0.79) subjects had erosive changes on their baseline radiographs and 15/32 (47%; kappa = 0.72) had joint space narrowing. The study subjects' mean baseline CRP and eGFR were 0.37 mg/dL (standard deviation (SD) of 0.4) and 80.4 mL/min/1.73 m2 (SD of 19.2), respectively.
![]()
Table 1: Study Subject Characteristics.
View Table 1
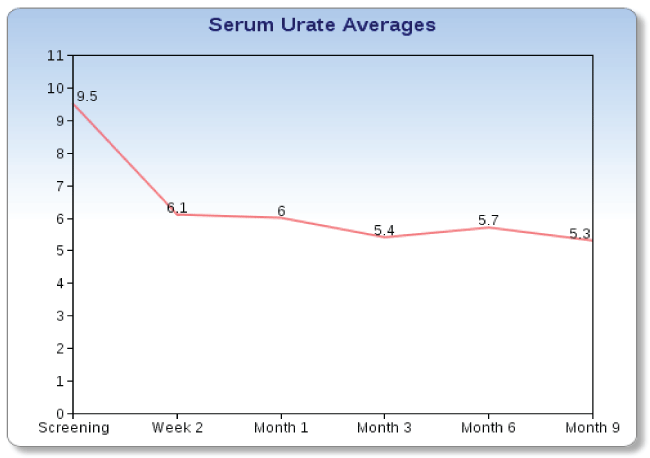
The study subjects' average serum urate level at baseline was 9.54 mg/dL (SD of 1.43; range of 7.2-13.4). After initiating treatment with febuxostat, the subjects experienced a rapid, sustained, and significant decrease in their serum urate levels (Figure 1). The decrease in serum urate level was statistically significant as soon as week 2 (p < 0.0001) and remained significant through month 9 (p < 0.0001). The average serum urate level at month 9 was 5.3 mg/dL (SD of 1.4 mg/dL) and 16/25 (64%) of the subjects who completed the protocol were at their serum urate level target (i.e. < 6.0 mg/dL) at month 9. Only 3 subjects had a serum urate level above 6.8 mg/dL (i.e. the level of saturation) at their month 9 visit (7.0 mg/dL, 7.0 mg/dL and 9.1 mg/dL (the latter required febuxostat dose adjustment because of increased liver function tests)).
Regarding the study participants' synovial pannus on their MRI, the average baseline severity score was 3.66 (SD of 1.2) in the 25/32 subjects who completed the entire protocol. At month 9, the average synovial pannus severity score in these same 25 subjects was 3.42 (SD of 1.3). This was a non-significant change with a p-value of 0.34 (Table 2). None of the 25 subjects had resolution of their synovial pannus on their month 9 MRI of the index joint. The inter-reader agreement for the grading of synovial pannus between the two musculoskeletal radiologists for all of the MRIs was good (kappa = 0.63). Further analysis was performed dividing the study participants into 3 subgroups based on their month 9 serum urate level, i.e. those with a serum urate level of < 5.0 mg/dL (Group 1), 5.0mg/dL to 6.0mg/dL (Group 2), or > 6.0 mg/dL (Group 3). There was no significant change in the synovial pannus score from baseline to month 9 in any group. Group 1 (n = 11) average baseline pannus severity score was 3.78 (SD of 1.06) and 3.41 (SD of 1.09) at month 9 (p-value = 0.51). Group 2 (n = 5) average baseline pannus severity score was 4.0 (SD of 0.79) and 3.6 (SD of 1.19) at month 9 (p-value = 0.10). Group 3 (n = 9) average baseline pannus severity score was 3.33 (SD of 1.44) and 3.33 (SD of 1.62) at month 9 (p-value = 1.0).
![]()
Table 2: Change in the Average Synovial Pannus Grade on the Index Joint from Baseline to Month 9.
View Table 2
There was also no significant change in CRP or eGFR from baseline to month 9. The average CRP was 0.38 mg/dL (SD of 0.4) at month 9 (p = 0.91); and the average month 9 eGFR was 77.7 mL/min/1.73 m2 (SD of 20.0; p = 0.13).
With regard to the other findings on the MRI, there was no significant change in number of subjects with bone erosions, soft tissue tophi, soft tissue edema, joint effusions, or bone marrow edema/lesions (osteitis) from baseline to month 9 according to either musculoskeletal radiologist (data not shown). However, there was an apparent significant decrease in the number of subjects with intraosseous tophi; according to radiologist #1, 12/25 subjects had intraosseous tophi at baseline and this decreased to 4/25 at month 9 (p = 0.03) and these same numbers decreased from 13/25 at baseline to 6/25 at month 9 as determined by radiologist #2 (p = 0.08).
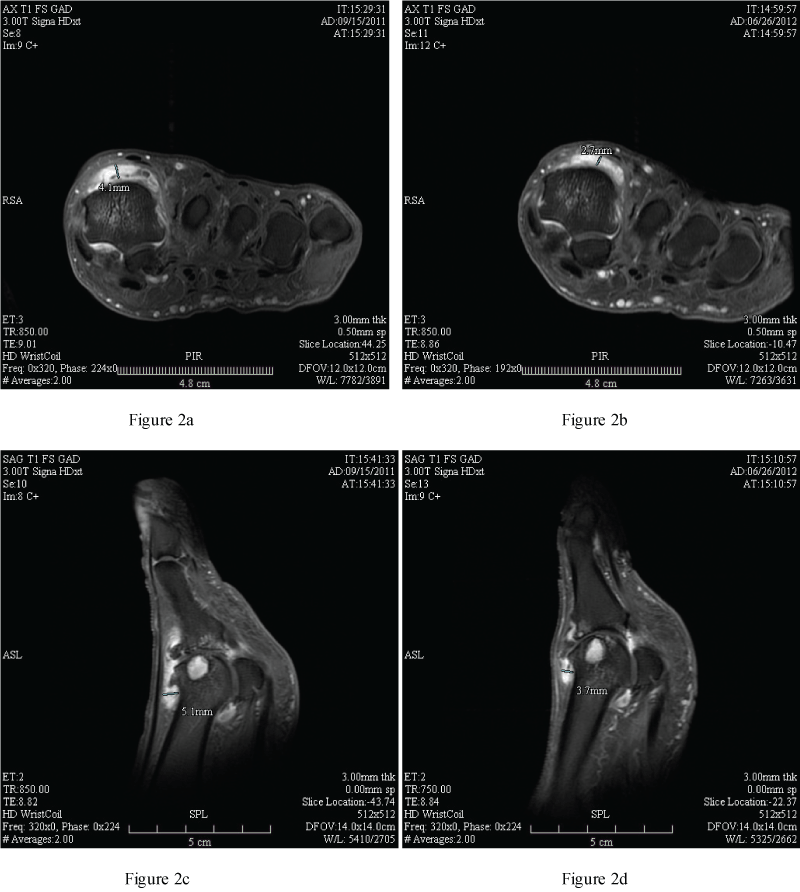
A post-hoc analysis was performed on these same MRI images by one of our MSK radiologists (SRA) in a blinded fashion using a novel more quantitative tool. This novel MRI assessment consisted of a whole-joint quantitative determination of the total synovial membrane volume (SMV). Evaluations were performed on axial, coronal, and sagittal post-contrast T1 with fat saturation images; maximal synovial thickness was measured on the ventral, dorsal, medial and lateral joint surfaces utilizing two planes and the most representative measurement was utilized (Figure 2). Measurements were perpendicular to bone surface to enhance reproducibility. The four surface scores were summed to create a SMV for that index joint. Utilization of this more quantitative scoring system did demonstrate significant change. The mean SMV score at baseline and month 9 was 14.26 mm (+/- 7.94 SD) and 10.58 mm (+/- 7.11 SD), respectively (p < 0.0001). The change in the mean SMV scores was also analyzed according to groups 1, 2 and 3 (described above), i.e. based on the patient's serum urate level at month 9. There was no clear pattern with p-values of 0.001, 0.13, and 0.01 for groups 1, 2 and 3 respectively. However, if the change in the mean SMV scores was assessed according to those with a serum urate level at month 9 of < 6.8 mg/dL (n = 20) compared to those with a serum urate level of ≥ 6.8 mg/dL (n = 5) the p-values were 0.0001 and 0.05, respectively. This same analysis was performed with the synovial pannus scoring system that we utilized as the primary endpoint and the results remained non-significant (data not shown). This further suggests that whole joint quantitative assessment with SMV is a better indicator of synovial-based inflammation during inter-critical gout and a more sensitive tool to detect changes in this inflammation.
.
Figure 2: a: Baseline Dorsal Axial Measurement of Synovial Pannus;
b: 9 Month Dorsal Axial Measurement of Synovial Pannus;
c: Baseline Dorsal Sagittal Measurement of Synovial Pannus;
d: 9 Month Dorsal Sagittal Measurement of Synovial Pannus.
View Figure 2
Safety
The most common adverse event was increased liver function tests (LFT). 8/25 (32%) subjects experienced some degree of LFT elevation during the study, but only 1 (4%) subject experienced increased LFTs of greater than two times the upper limit of normal. This resolved with dose adjustment of the febuxostat. One subject also experienced mild thrombocytopenia without complication that resolved spontaneously. 2/25 participants experienced mild nausea that resolved with supportive care. There was one serious adverse event (exacerbation of congestive heart failure requiring hospitalization).
Discussion
This prospective study analyzed the effect of nine months of serum ULT on the severity of the chronic synovial-based inflammation that is present during inter-critical gout. Thirty-two subjects were enrolled in this prospective open-label trial and 25 successfully completed the entire nine month protocol. The study subjects experienced a significant, rapid, and sustained decrease in their serum urate levels while on febuxostat. These same patients did not demonstrate any improvement in the severity of their synovial pannus, as assessed by MRI, after nine months of effective sULT using a previously described synovial pannus scale (designed for inflammatory monoarthritis, but not gout specifically). However, there is not a validated scale to quantitatively assess synovial-based inflammation of gout. We designed a novel quantitative tool that can easily be applied to MRI images in a retrospective fashion to better quantitate this inflammation. We termed this assessment the synovial membrane volume (SMV) and a blinded assessment of the SMV did demonstrate a significant change after nine months of serum ULT.
Hyperuricemia is the primary risk factor and necessary precursor for gout. Patients with gout have elevated serum urate levels for many years prior to their first attack of gout [18,19]. If untreated, the intervals between acute attacks of gout become shorter and eventually a chronic inflammatory arthritis can develop. There are data demonstrating the presence of synovial-based monosodium urate crystals and occult synovial-based inflammation during inter-critical gout [9-11]. These findings make intuitive sense because of the eventual development of a chronic, clinically-apparent inflammatory arthritis that develops in those patients who are untreated. Therefore, it would also seem logical that effective serum ULT would abrogate this synovial-based inflammation.
The significance of this synovial-based inflammation in patients with gout is not completely understood. There is no question that serum ULT decreases the frequency of clinical attacks of gout and visible tophi. Indeed, in our study there was a decrease in the number of patients with intraosseous tophi after nine months of serum ULT. If we are successful at decreasing serum urate levels below the level of saturation, then patients experience less painful attacks and demonstrate improvement in their tophaceous burden. The only large study performed analyzing the long-term effect of serum ULT on radiographic progression suggested no effect [12]. However, a recent small exploratory study suggests that more profound serum ULT can affect radiographic progression [13]. Our study assessed the synovial-based inflammation that is likely responsible for radiographic progression. The pannus scale we used as the primary endpoint demonstrated no affect, but the more quantitative tool we used in the post-hoc analysis suggested a positive effect of the serum ULT. This highlights the need for a validated assessment tool that can be applied to MRI images allowing a more sensitive detection tool in gout. We feel our SMV assessment protocol deserves further study.
There could be limitations to this study that preclude us from seeing the true long-term effect of serum ULT on the synovial-based inflammation that accompanies gout. The most obvious was the original pannus scoring tool we used. Currently there is no such validated tool for gout and out post-hoc analysis suggests this technique might be better. These results also need to be interpreted with caution due to the relatively small sample size. It is also possible that nine months of therapy was not enough time to demonstrate a significant positive association. We know that the effect of ULT on the total body urate pool lags behind that of serum urate levels [20,21]. It could be the case that this chronic, occult synovial-based inflammation does not improve until the total body urate burden is below a certain level for an extended period of time. This might be particularly true for patients with a heavy tophaceous burden. Currently we have no adequate means of measuring ones total body urate pool/burden. However, recent data assessing prophylaxis therapy against "mobilization" flares when initiating serum ULT demonstrated that flares were common if prophylaxis was withdrawn at week 8 (40%) whereas flare rates were low (3-5%) at the end of 6 months of prophylaxis [22]. This suggests total body urate burden is significantly lower after 6 months versus 8 weeks of serum ULT. It is also possible that our therapeutic serum urate target needs to be lower as suggested by a recent exploratory study [13]. Another limitation is that there was no control population in this prospective trial and contrast-enhanced MRI synovitis has been demonstrated in other common conditions, namely osteoarthritis (OA) [23-25]. However, only 47% of our patients had osteoarthritis at baseline (10/25 (40%) of completers) as evidenced by joint space narrowing on their baseline plain radiograph and all of the patients in this sub-study had contrast-enhanced MRI synovitis at baseline and month 9. Future controlled trials are needed to confirm these findings.
The quest to determine why only ~20% of patients with hyperuricemia will ever develop clinical gout [26] is ongoing. While hyperuricemia is the primary risk factor and a necessary precursor, it is not the sole determinant. While there might have been improvement in the synovial-based inflammation on our study, none of the patients had resolution of their synovial pannus after nine months of therapy regardless of the scale used. These data suggest that the chronic, occult, synovial-based inflammation of gout is, at least in part, uncoupled from serum urate levels. Perhaps the total body urate burden is the driving force behind the chronic inflammatory response and this burden can take years to normalize. We might also need to adjust our therapeutic serum urate target. It is also possible that the chronic, occult, synovial-based inflammation of gout is driven by a second, yet to be described, pathophysiologic mechanism that is, at least in part, unrelated to serum urate levels.
Disclosures
John D. Carter: Grant from Takeda Pharmaceuticals NA, Inc; Grant from AstraZeneca; Grant from Genentech; on Speakers Bureaus of Amgen, Abbvie, and Takeda.
Acknowledgement
This work support provided by Takeda Pharmaceuticals USA, Inc. and AstraZeneca.
References
-
Becker MA, Schumacher HR, Benjamin KL, Gorevic P, Greenwald M, et al. (2009) Quality of life and disability in patients with treatment-failure gout. J Rheumatol 36: 1041-1048.
-
Brixner DI, Ho MJ (2005) Clinical, humanistic, and economic outcomes of gout. Am J Manag Care 11: S459-464.
-
Halpern R, Fuldeore MJ, Mody RR, Patel PA, Mikuls TR (2009) The effect of serum urate on gout flares and their associated costs: an administrative claims analysis. J Clin Rheumatol 15: 3-7.
-
Singh JA, Strand V (2008) Gout is associated with more comorbidities, poorer health-related quality of life and higher healthcare utilisation in US veterans. Ann Rheum Dis 67: 1310-1316.
-
Wu EQ, Forsythe A, Guérin A, Yu AP, Latremouille-Viau D, et al. (2012) Comorbidity burden, healthcare resource utilization, and costs in chronic gout patients refractory to conventional urate-lowering therapy. Am J Ther 19: e157-166.
-
Arromdee E, Michet CJ, Crowson CS, O'Fallon WM, Gabriel SE (2002) Epidemiology of gout: is the incidence rising? J Rheumatol 29: 2403-2406.
-
Shoji A, Yamanaka H, Kamatani N (2004) A retrospective study of the relationship between serum urate level and recurrent attacks of gouty arthritis: evidence for reduction of recurrent gouty arthritis with antihyperuricemic therapy. Arthritis Rheum 51: 321-325.
-
Perez-Ruiz F, Calabozo M, Pijoan JI, Herrero-Beites AM, Ruibal A (2002) Effect of urate-lowering therapy on the velocity of size reduction of tophi in chronic gout. Arthritis Rheum 47: 356-360.
-
Pascual E, Batlle-Gualda E, Martínez A, Rosas J, Vela P (1999) Synovial fluid analysis for diagnosis of intercritical gout. Ann Intern Med 131: 756-759.
-
Pascual E (1991) Persistence of monosodium urate crystals and low-grade inflammation in the synovial fluid of patients with untreated gout. Arthritis Rheum 34: 141-145.
-
Bomalaski JS, Lluberas G, Schumacher HR Jr (1986) Monosodium urate crystals in the knee joints of patients with asymptomatic nontophaceous gout. Arthritis Rheum 29: 1480-1484.
-
McCarthy GM, Barthelemy CR, Veum JA, Wortmann RL (1991) Influence of antihyperuricemic therapy on the clinical and radiographic progression of gout. Arthritis Rheum 34: 1489-1494.
-
Dalbeth N, Doyle AJ, McQueen FM, Sundy J, Baraf HS (2014) Exploratory study of radiographic change in patients with tophaceous gout treated with intensive urate-lowering therapy. Arthritis Care Res (Hoboken) 66: 82-85.
-
Carter JD, Kedar RP, Anderson SR, Osorio AH, Albritton NL, et al. (2009) An analysis of MRI and ultrasound imaging in patients with gout who have normal plain radiographs. Rheumatology (Oxford) 48: 1442-1446.
-
Carter JD, Patelli M, Anderson SR, Prakash N, Rodriquez EJ, et al. (2015) An MRI assessment of chronic synovial-based inflammation in gout and its correlation with serum urate levels. Clin Rheumatol 34: 345-351.
-
Wallace SL, Robinson H, Masi AT, Decker JL, McCarty DJ, et al. (1977) Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum 20: 895-900.
-
Argyropoulou MI, Fanis SL, Xenakis T, Efremidis SC, Siamopoulou A (2002) The role of MRI in the evaluation of hip joint disease in clinical subtypes of juvenile idiopathic arthritis. Br J Radiol 75: 229-233.
-
Campion EW, Glynn RJ, DeLabry LO (1987) Asymptomatic hyperuricemia. Risks and consequences in the Normative Aging Study. Am J Med 82: 421-426.
-
Loeb JN (1972) The influence of temperature on the solubility of monosodium urate. Arthritis Rheum 15: 189-192.
-
Hoskison TK, Wortmann RL (2006) Advances in the management of gout and hyperuricaemia. Scand J Rheumatol 35: 251-260.
-
Schlesinger N (2011) Difficult-to-treat gouty arthritis: a disease warranting better management. Drugs 71: 1413-1439.
-
Wortmann RL, Macdonald PA, Hunt B, Jackson RL (2010) Effect of prophylaxis on gout flares after the initiation of urate-lowering therapy: analysis of data from three phase III trials. Clin Ther 32: 2386-2397.
-
Javaid MK, Kiran A, Guermazi A, Kwoh CK, Zaim S, et al. (2012) Individual magnetic resonance imaging and radiographic features of knee osteoarthritis in subjects with unilateral knee pain: the health, aging, and body composition study. Arthritis Rheum 64: 3246-3255.
-
Haugen IK, Lillegraven S, Slatkowsky-Christensen B, Haavardsholm EA, Sesseng S, et al. (2011) Hand osteoarthritis and MRI: development and first validation step of the proposed Oslo Hand Osteoarthritis MRI score. Ann Rheum Dis 70: 1033-1038.
-
Roemer FW, Guermazi A, Felson DT, Niu J, Nevitt MC, et al. (2011) Presence of MRI-detected joint effusion and synovitis increases the risk of cartilage loss in knees without osteoarthritis at 30-month follow-up: the MOST study. Ann Rheum Dis 70: 1804-1809.
-
Sanders S, Wortmann RL (2007) In: Imboden JB Current Rheumatology Diagnosis & Treatment. (2nd edn) New York, NY: McGraw-Hill, 345-352.