Journal of Rheumatic Diseases and Treatment
Hypercoagulation in Patients with Rheumatoid Arthritis Correlates with Activation of Act1/NF-κb Signaling Pathway
Pingheng Zhang, Jian Liu* and Bing Tan
Department of Rheumatism Immunity, The First Affiliated Hospital, Anhui University of Chinese Medicine, Hefei (230031), China
*Corresponding author: Jian Liu, Department of Rheumatism Immunity, The First Affiliated Hospital, Anhui University of Chinese Medicine, Hefei (230031), China, E-mail: liujianahzy@126.com
J Rheum Dis Treat, JRDT-1-024, (Volume 1, Issue 4), Research Article; ISSN: 2469-5726
Received: August 19, 2015 | Accepted: November 13, 2015 | Published: November 16, 2015
Citation: Zhang P, Liu J, Tan B (2015) Hypercoagulation in Patients with Rheumatoid Arthritis Correlates with Activation of Act1/NF-κb Signaling Pathway. J Rheum Dis Treat 1:024. 10.23937/2469-5726/1510024
Copyright: © 2015 Zhang P, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To explore the role of the Act1/NF-κB signaling pathway in the development of hypercoagulation states in patients with rheumatoid arthritis (RA).
Methods: Peripheral blood samples were taken from 30 RA patients and 20 healthy volunteers, as controls. Ex vivo correlates of disease severity, such as C-reactive protein, erythrocyte sedimentation rate, anti-cyclic citrullinated peptide and rheumatoid factor, and immunological activation, such as interleukin (IL)-10, IL-17 and IL-6 were measured biochemically. Factors derived from the coagulation fibrinolytic system were also determined, such as the number of platelets, platelet activating factor, platelet activating factor-acetylhydrolase, D-dimer, thrombin time, prothrombin time, partial thromboplastin time, and fibrinogen levels. Furthermore, analysis of NF-κB signaling molecules Act1, p65, p50, IκBα and IκB kinase α (IKKα) was performed using semi-quantitative reverse transcription and western blotting.
Results: Compared with the healthy control group, the levels of D-dimer, fibrinogen, and the number of platelets were significantly increased in the peripheral blood of RA patients, whereas partial thromboplastin time and thrombin time were decreased. IL-4, IL-10 and PAF-AH were significantly decreased in the serum of RA patients, while IL-6, IL-17, Act1, p50, p65, IκBα and PAF were significantly increased. Multiple regression analysis showed that coagulant and fibrinolytic indexes correlated significantly with cytokines, NF-κB, activity indexes and clinical symptoms.
Conclusion: Hypercoagulation state was prevalent in patients with RA and was related to inflammatory factors, activity indexes and activation of NF-κB. The results suggested that abnormal activation of the NF-κB signaling pathway and an imbalance of pro- and anti-inflammatory cytokines was the result of a disturbance in the coagulation-fibrinolytic system.
Keywords
Nuclear factor κ B (NF-κB), Rheumatoid arthritis, Cytokines, Platelet activating factor/platelet activating factor-platelet-activating factor acetylhydrolase (PAF/PAF-AH), Hypercoagulable state
Introduction
Rheumatoid Arthritis (RA) is a chronic, systemic, autoimmune disease, characterized by joint synovitis, with inflammatory cell infiltrates and bone and cartilage matrix destruction caused by activated synovial fibroblasts [1,2]. In China, the incidence of RA is about 0.33%; the incidence for females is three times that of males. The pathogenesis of RA is linked to immune dysfunction and an imbalance of T helper cells is associated with disease progression [3]. Effector T cells are responsible for promoting the production of autoantibodies and they release a variety of pro-inflammatory cytokines, initiating and perpetuating chronic inflammation in RA [4,5].
Nuclear factor kappa B (NF-κB) is an essential transcription factor, which regulates the expression of inflammatory cytokines such as IL-17 and IL-6 [6]. NF-κB plays a key role in the pathogenesis of RA; it has been shown to regulate the inflammatory response, promote synovial cell hyperplasia and reconstruction of the joint architecture. Recent studies have shown that the development of RA is accompanied by a disturbance of the coagulation fibrinolysis system with clinical manifestations that include high blood coagulation [7-9]. Additionally, platelet activating factor (PAF), a potent phospholipid activator, was shown to be an important mediator of leukocyte function and inflammation [10,11]. PAF plays a key role in the infiltration of inflammatory cells to the joints, thus, PAF and a high coagulation state, are closely related to the processes of synovitis, tissue destruction, angiogenesis, and induced platelet aggregation [12].
The present study explores the role of a high coagulation state in the pathogenesis of RA and its regulation by the NF-κB pathway.
Materials and Methods
Materials
Patients:The study cohort comprised 30 RA inpatients at the department of rheumatology of the First Affiliated Hospital of Anhui University of Traditional Chinese Medicinea and was carried out from January 2014 to December 2014. Patients had active disease, DAS28 score > 2.6, and were defined using the RA classification standards proposed by the 2010 American Rheumatism Association joint European League against rheumatism [13]. There were 20 females and 10 males, aged 28-70 years (mean 50.80 ± 11.01 years). The duration of illness was 0.5-20 years (mean 9.20 ± 5.75 years). The control group comprised 20 healthy individuals, consisting of 15 females and 5 males, aged 20-58 years (mean 38.63 ± 9.13 years). They were recruited from the Anhui Province hospital medical center, and were not suffering from any known diseases. The distribution of age and sex were comparable between the two groups and there was no statistically significant difference (P > 0.05). The study protocol was approved by the ethics committee of the First Affiliated Hospital of Anhui University of Traditional Chinese Medicine (Anhui Hefei, China; 2012AH-020-01). The study participants all provided informed consent.
Methods
Determination of immunological disease correlates in peripheral blood:IL-10, IL-6, IL-4, IL-17, Act1, p50, p65, IκBa, PAF and PAF-AH levels were detected with enzyme-linked immunosorbent assays (ELISA; R&D Systems, USA). Five ml of venous blood was taken from each patient, during the morning following fasting, and centrifuged for 15 min at 3000 rpm. Supernatants were stored at -80°.
Determination of biochemical disease correlates in peripheral blood:Platelets (PLT) were detected using a Sysmex XT-2000i automated hematology analyzer (Sysmex, Hyogo, Japan). D-dimer (D-D), thrombin time (TT), prothrombin time (PT), partial thromboplastin time (APTT) and fibrinogen (FBG) levels were detected using a Sysmex CA-1500 automatic coagulation analyzer. Erythrocyte sedimentation rate (ESR) was detected using the Westergren method. C-reactive protein (Hs-CRP) and rheumatoid factor (RF) were detected using a Hitachi 7060 automatic biochemical analyzer (Hitachi Chemical Co, Tokyo, Japan).
mRNA isolation and PCR:mRNA encoding Act1, p65, p50, IκBα and IKKα were detected using semi-quantitative PCR. The primer sequences are shown in table 1. Total RNA was extracted from patients' blood using TRIzol (ThermoFisher Scientific) and the instructions of the primer manufacturer (Invitrogen). Reverse transcription was performed using a reverse transcription kit (Thermo Scientific). PCR reaction conditions used were as follows: 5 min of initial denaturation at 95°; 35 cycle programmed with 30 s of denaturation at 95°, 30 s of annealing at 55° and 50 s of extension at 72°; and finally 10 min extension at 72°. PCR products were detected by electrophoresis on 1.5% agarose gels, which included 0.1 μg/mL ethidium bromide. The gels were photographed and analyzed using Image Pro Plus software to calculate the integrated optical density where the integrated optical density of mRNA/β-actin was the relative amount of mRNA.
![]()
Table 1: Semi-quantitative PCR primer sequence and amplification length.
View Table 1
Protein isolation and western blotting:Total protein was extracted from peripheral blood using a preparation of modified radioimmunoprecipitation buffer (Blue Skies Technology) and quantified using a Coomassie brilliant blue protein quantitative kit (Abcam). p50, p65 and IκBa protein levels were detected by western blot. The integral absorbance value of the bands was detected with BandScan software, and the ratios, p50/β-actin, p65/β-actin and IκBa/β-actin determined the relative amount of p50, p65 and IκBa protein expression level, respectively.
Statistical analyses
Statistical analyses were carried out using SPSS v17.0 (IBM, Chicago, IL, USA). Measurement data are the mean ± standard deviation (SD) of normally distributed data. The independent-samples t-test was used to compare measurement data between groups. The rank and inspection test was used for non-normally distributed data. Multiple regression analysis was carried out for relevant data. P < 0.05 was considered significant.
Results
Coagulation index levels in RA patients
Firstly, we analyzed coagulation parameters and found that, compared with the control group, D-D, FBG and PLT were significantly increased in the RA patients' peripheral blood (P < 0.01). Abnormal cases were 26, 17, and 12, respectively, accounting for 86.67%, 56.67%, and 40%. APTT and TT were shortened (P < 0.05); abnormal cases were 5 and 1, respectively, accounting for 16.67% and 3.33%. PT was not significantly different between the two groups (P > 0.05); however, a difference was apparent; abnormal cases were 2, accounting for 6.67% (Table 2, Figure 1).
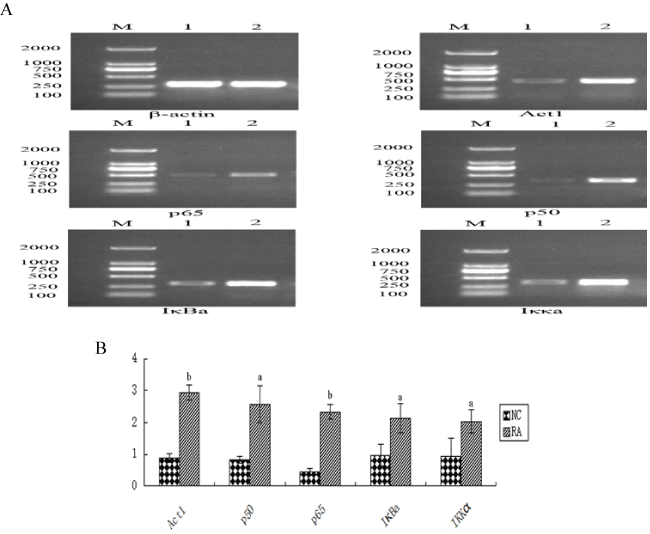
.
Figure 1: The expression of NF-κB mRNA level in two groups. A: The representation image that the PCR was detected Act1, p65, p50, IκBa, IKKa mRNA expression. B: The semi-quantitative analysis of Act1, p65, p50, IκBa, IKKa mRNA 1: NC group; 2: RA group; aP < 0.05, bP < 0.01 vs. 1.
View Figure 1
![]()
Table 2: Coagulation index levels in RA patients compared with healthy controls
Compared with the healthy control group, aP < 0.05, bP < 0.01.
View Table 2
Analysis of cytokines, NF-κB and PAF/PAF-AH in RA patient serum
Analysis of RA patient serum showed that IL-4, IL-10 and PAF-AH levels were significantly decreased, compared with healthy controls (P < 0.05). Conversely, IL-6, IL-17, Act1, p50, p65, IκBα and PAF were significantly increased (P < 0.05; Table 3).
![]()
Table 3: Cytokines, NF-κB and PAF/PAF-AH levels (
)
Compared with the NC group, aP < 0.05, bP < 0.01
View Table 3
Analysis of signaling molecule mRNA in RA patients
Compared with the healthy control group, mRNA encoding Act1, p65, p50, IκBα and Iκκα levels were significantly increased in RA patient samples (P < 0.05; Figure 1).
Analysis of p50, p65 and IκBα protein levels in RA patients
Compared with the healthy control group, p50, p65 and IκBα protein levels were significantly increased in RA patient samples (P < 0.05; Figure 2).
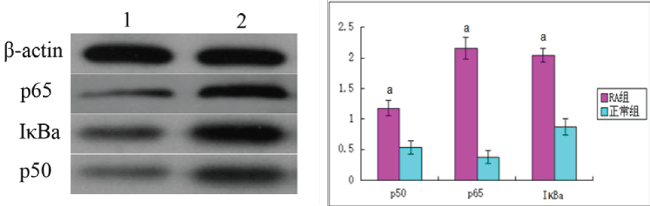
.
Figure 2: The expression of the protein of p50, p65 and IκBa in two groups. A: The representation image that the Western blot was detected Act1, p65, p50, IκBa, IKKa mRNA expression. B: The semi-quantitative analysis of p65, p50, IκBa expression of protein 1: NC group; 2: RA group; aP < 0.05 vs. NC group
View Figure 2
Correlations between coagulation indicators, cytokines, and NF-κB
Multiple regression analysis showed that, in patients with RA, D-D positively correlated with ESR, IL-17, IL-6 and p50. FBG positively correlated with ESR and IL-6; PLT positively correlated with DAS28, CCP, CRP, IL-17 and p50, and negatively with IL-4; PT positively correlated with IκBα; PAF positively correlated with RF, CRP, CCP, IL-17, IL-6, p65 and p50, and negatively with IL-4; PAF-AH negatively correlated with p50 and positively with IL-4 (P < 0.05, P < 0.05; Table 4).
![]()
Table 4: Correlations between coagulation indicators, cytokines, and NF-κB (r).
aP < 0.05, bP < 0.01
View Table 4
Discussion
RA is a chronic autoimmune inflammatory disease; however its precise etiology and pathogenesis are unclear. Basic pathological changes include synovitis, joint synovial hyperplasia and pannus formation [14,15]. The incidence of RA is 0.5-1.0%, with a high disability rate for patients with RA [16]. Existing studies have shown that the coagulation fibrinolytic indexes, FBG and D-D, were significantly higher in RA patients than in healthy controls [17,18]. FBG is synthesized by the liver and forms an essential component of blood clots. D-D is a product of cross linked fibrin degradation. It serves as a marker of high coagulation and fibrinolytic hyper function, with alterations in D-D concentration indicating dysfunction of the coagulation fibrinolytic system [19]. PLT are another essential element in blood coagulation, inflammation and thrombosis [20]. Our results suggested that alterations in the blood coagulation fibrinolytic system in patients with RA had occurred. Therefore, a high coagulation state may be a major clinical manifestation of RA. At present, the high coagulation state in RA patients is poorly understood in theory and clinical practice. To gain insight into the relationship between the level of coagulation and disease state, we performed statistical analyses on blood coagulation fibrinolytic abnormalities in 30 patients with RA. Our results showed that D-D, FBG and PLT were significantly increased in RA patients compared with healthy controls. Furthermore, APTT and TT were decreased, whereas PT showed no significant changes, despite showing some abnormalities. The results of this study are consistent with previous studies [18,21] thus suggesting that blood coagulation fibrinolytic system abnormalities may be of importance to the disease state in RA.
The abnormal change of coagulation fibrinolytic indexes in patients with RA was closely related to immune inflammation indexes. However, coagulation fibrinolytic system disorders were closely related to an imbalance of pro- and anti-inflammatory cytokines. Our results showed that IL-10 was negatively correlated with D-D [22]. IL-4 has been shown to produce antiplatelet antibodies, which led to a decrease in platelet number [23]. Additionally, IL-4 can co-operate with IL-10 [24]. IL-6 has been shown to up-regulate fibrinogen production to promote high blood coagulation, and is correlated with the degree of vascular inflammation [25]. IL-17 is a proinflammatory cytokine capable of stimulating fibroblasts to secrete IL-6 and other inflammatory factors. IL-17 signaling can lead to the activation of IKKα by Actl, which leads to the phosphorylation of IκBα and its eventual degradation. Degradation of IκBα enables the nuclear translocation of NFκB, in which it functions as a transcription factor that promotes the transcription of pro-inflammatory-related genes. Chang et al. [26] showed that Actl-mediated activation of NF-κB was dependent on IL-17, constituting an important signal transduction regulator. IL-17 is secreted extracellularly, activating NF-κB, and leading to increased secretion of IL-6 [27]. IL-6, in turn, induces neutrophils and other granulocytes to produce PAF, potentiating the inflammatory reaction, and stimulating synovial cell proliferation [12] and platelet aggregation [28], thus, contributing to the occurrence of high coagulation states in RA patients. PAF-AH is capable of inactivating PAF, thus, the dynamic balance between PAF and PAF-AH plays an important role in maintaining the normal activity of PAF. As a result, the change in PAF-AH could also lead to a high blood coagulation state. Our study found that IL-4, IL-10 and PAF-AH levels were significantly decreased in RA patients; however, IL-6, IL-17, PAF, Act1, p50, p65, IκBα and IKKα were significantly increased. Collectively, this suggests that abnormal activation of NF-κB occurs, with a resulting imbalance of cytokines and abnormal changes in blood coagulation fibrinolysis indicators in patients with RA.
Additionally, the results demonstrate that D-D positively correlated with ESR, IL-17, IL-6 and p50. FBG positively correlated with ESR and IL-6, whereas PLT positively correlated with DAS28, CCP, CRP, IL-17 and p50, and negatively with IL-4. Furthermore, PT positively correlated with IκBa; PAF positively correlated with RF, CRP, CCP, IL-17, IL-6, p65 and p50, and negatively with IL-4; PAF-AH was negatively correlated with p50, and positively with IL-4 (Table 4). Collectively, these results suggest that the changes in coagulation indexes were closely related to clinical symptoms and inflammatory factors, activity indexes and abnormal activation of NF-κB in RA patients.
In conclusion, the occurrence of high coagulation state in RA patients might be a result of abnormal activation of NF-κB, thereby up-regulating the expression of D-D, FBG, PLT and PAF, and down-regulating the expression of APTT, PT, TT and PAF-AH. It is also possible that the occurrence of a high blood coagulation state may be related to increased IL-6 and IL-17, and decreased IL-4 and IL-10. However, the mechanism of developing into a high coagulation state requires further study to determine possible targets for therapeutic intervention.
Acknowledgements
I gratefully acknowledge the Department of Rheumatism Immunity, The First Affiliated Hospital, Anhui University of Chinese Medicine, and Professor He Liu jian and my research team for their cooperation.
This work was supported by the Key Projects in the National Science & Technology Pillar Program in the Twelth Five-Year Plan Period (NO: 2012BAI26B02). Clinical Trials. gov Identifier: NCT01774877.
References
-
Juhasz P, Mester a, Biro AJ, Hejj G, Poor G (2014) Clinical and radiological dissociation of anti-TNF plus methotrexate treatment in early rheumatoid arthritis in routine care: Results from the ABRAB study. BMC Musculoskeletal Disorders 15: 251.
-
Haraoui B, Pope J (2011) Treatment of early rheumatoid arthritis: concepts in management. Semin Arthritis Rheum 40: 371-388.
-
Poosarla A, D N R, Athota RR, Sunkara VG (2011) Modulation of T cell proliferation and cytokine response by Plumbagin, extracted from Plumbago zeylanica in collagen induced arthritis. BMC Complement Altern Med 11: 114.
-
Paradowska-Gorycka A, Grzybowska-Kowalczyk A, Wojtecka-Lukasik E, Maslinski S (2010) IL-23 in the pathogenesis of rheumatoid arthritis. Scand J Immunol 71: 134-145.
-
Tang C, Chen S, Qian H, Huang W (2012) Interleukin-23: as a drug target for autoimmune inflammatory diseases. Immunology 135: 112-124.
-
Ogura H, Arima Y, Kamimura D, Murakami M (2013) The gateway theory: how regional neural activation creates a gateway for immune cells via an inflammation amplifier. Biomed J 36: 269-273.
-
Ruikai Z, Jian L (2012) The research progress: The role of platelet activation products in RA and the intervention effect of traditional Chinese medicine. World Journal of Integrated Traditional and Western Medicine 8: 725-730.
-
Yuting Z, Baikelimu G, Rong W (2014) The relationship between clinical stages, TCM syndrome type and markers of prethrombotic state in RA patients. Journal of Xinjiang Medical University, 37: 1308-1311.
-
Yonghua C, Haihen Q, Gaoli (2010) Clinical Sinificance of Changes of Plasma Prethrombotic State Markers Levels in Patients with Rheumatoid Arthritis. J Radioimmunol 23: 207-208.
-
Suri R, Mallia P, Martin JE, Footitt J, Zhu J, et al. (2014) Bronchial platelet-activating factor receptor in chronic obstructive pulmonary disease. Respir Med 108: 898-904.
-
Dongmei Z, Jianxin Y, Junshan M (2015) The correlation research between Acute ischemic stroke severity and sCD40L, PAF in serum. Clin J Med Omc 43: 374-376.
-
Mazereeuw G, Herrmann N, Xu H, Blanchard AP, Figeys D, et al. (2015) Platelet activating factors are associated with depressive symptoms in coronary artery disease patients: a hypothesis-generating study. Neuropsychiatr Dis Treat 11: 2309-2314.
-
Radner H, Neogi T, Smolen JS, Aletaha D (2014) Performance of the 2010 ACR/EULAR classification criteria for rheumatoid arthritis: a systematic literature review. Ann Rheum Dis 73: 114-123.
-
Pope RM, Shahrara S (2013) Possible roles of IL-12-family cytokines in rheumatoid arthritis. Nat Rev Rheumatol 9: 252-256.
-
Tamai M, Kawakami A, Uetani M, Fukushima A, Arima K, et al. (2012) Magnetic resonance imaging (MRI) detection of synovitis and bone lesions of the wrists and finger joints in early-stage rheumatoid arthritis: comparison of the accuracy of plain MRI-based findings and gadolinium-diethylenetriamine pentaacetic acid-enhanced MRI-based findings. Mod Rheumatol 22: 654-658.
-
Scott DL, Wolfe F, Huizinga TW (2010) Rheumatoid arthritis. Lancet 376: 1094-1108.
-
Hong-xia X, Hua-dong C, Dong-mei L (2011) The Clinical Significance of D-dimer and Fibrinogen in Rheumatoid Arthritis. Journal of China Medical University 40: 1004-1006.
-
Wei Z (2014) The clinical significance of detection of D-dimmer and fibrinogen in rheumatoid arthritis. Hainan Medical Journal 25: 55-57.
-
Guang'en JNG, Naizhen Z, Zhiqing C (2012) Association of coagulation and fibrinolysis with microalbuminuria in patients with essential hypertensive. J Clin Cardiol (China) 28: 587-589.
-
Ming M, Lei Z, Weimin G (2012) Experimental research: the effect of activated platelets on synovial inflammation and hyperplasia in RA. Chin J Cell Mol Immunol 28: 828-829.
-
Ming M, Lei Z, Weiming G (2012) Activated platelets effect on rheumatoid arthritis synovial inflammation and hyperplasia of experimental research. Chin J Cell Mol Immunol 28: 828-833.
-
Guoyu W, Li Y, Hong Z (2013) Clinical Significance of Measurement on Serum IL-2, IL-10, IL-18 and D-D Levels after Treatment in Pediatric Patients with Mycoplasma Pneumonia. J Radioimmunol 26: 720-722.
-
Qiongyu W, Qingping G, Youhua C (2004) The influence of IL-4 and IFN-γ on the pathogenesis of idiopathic thrombocytopenic purpura. J Clin Intern Med 21: 809-810.
-
Fernandes JC, Martel-Pelletier J, Pelletier JP (2002) The role of cytokines in osteoarthritis pathophysiology. Biorheology 39: 237-246.
-
Lin CY, Yang YH, Lee CC, Huang CL, Wang LC, et al. (2006) Thrombopoietin and interleukin-6 levels in Henoch-Schonlein purpura. J Microbiol Immunol Infect 39: 476-482.
-
Chang SH, Park H, Dong C (2006) Act1 adaptor protein is an immediate and essential signaling component of interleukin-17 receptor. J Biol Chem 281: 35603-35607.
-
Yao Z, Fanslow WC, Seldin MF, Rousseau AM, Painter SL, et al. (2011) Herpesvirus saimiri encodes a new cytokine, IL-17, which binds to a novel cytokine receptor. J Immunol 187: 4392-4402.
-
Zheng HL, Wen HX, Liu GY, Ni J (2008) Role of platelet-activating factor in progesterone synthesis and vascular endothelial growth factor expression in rat luteal cells. Sheng Li Xue Bao 60: 275-278.
Special issue title: Rheumatoid Arthritis
Handling Editor: Pedro Santos-Moreno
Chief and Scientific Director
Department of Rheumatology
Universidad La Sabana, Bogota Colombia
USA





