Journal of Rheumatic Diseases and Treatment
Association of Markers of Oxidative Stress, Medication Therapy and Life Habits in Fibromyalgia
João Eduardo Torrecillas Sartori1#, Luiz Fernando Cireia1#, Denise Poltronieri Martins1*, Gabriela do Prado Rocha1, Juliany Roberta Cintra1, Ana Luiza de Aquino Oliveira1, Juliana Gonçalves Yogolare1, Lucas Romagnolli1, Alexandre de Andrade Budin1, Sabrina Mayara Cezario1, Marcela Augusta de Souza Pinhel2, Camila Ive Ferreira Oliveira1, Dorotéia Rossi da Silva Souza1, Gerardo Maria de Araújo Filho2 and Lazslo Antônio Ávila2
1Research Center for Biochemistry and Molecular Biology at the Medical School of São José do Rio Preto - FAMERP, Brazil
2Medical School of Ribeirão Preto, USP-FMRP, Ribeirão Preto, SP, Brazil
3Department of Medical Psychology and Psychiatry at the Medical School of São José do Rio Preto - FAMERP, São José do Rio Preto, SP, Brazil
#These authors contributed equally to this work
*Corresponding author: Denise Poltronieri Martins, Research Center for Biochemistry and Molecular Biology at the Medical School of São José do Rio Preto - FAMERP, Av. Brigadeiro Faria Lima, 5416, Postal Code: 15090-000, São José do Rio Preto-SP, Brazil, Tel: +55 173201-5864, E-mail: denise-martins@outlook.com
J Rheum Dis Treat, JRDT-2-035, (Volume 2, Issue 2), Research Article; ISSN: 2469-5726
Received: March 07, 2016 | Accepted: May 07, 2016 | Published: May 10, 2016
Citation: Sartori JET, Cireia LF, Martins DP, Rocha GP, Cintra JR, et al. (2016) Association of Markers of Oxidative Stress, Medication Therapy and Life Habits in Fibromyalgia. J Rheum Dis Treat 2:035. 10.23937/2469-5726/1510035
Copyright: © 2016 Sartori JET, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To evaluate the association among genetic polymorphisms of glutathione S-transferase, oxidative stress, lifestyle habits and medication therapy in patients with fibromyalgia (FM).
Patients and methods: A total of 187 women (22-84 years) were studied: 81 patients with fibromyalgia (G1) and 106 individuals without the disease (G2). They had their peripheral blood sample submitted for analysis of GSTM1 and GSTT1 polymorphisms, serum levels of thiobarbituric acid reactive substances (TBARS) and trolox equivalent antioxidant capacity (TEAC). All subjects were asked to complete a questionnaire for data registry.
Results: M1/T1 prevalent genotype in G1 (50.6%) and G2 (45.3%; P = 0.564). Serum TBARS values were similar between the groups (G1 = 218.0 nmol/L; G2 = 233.44 nmol/L, P = 0.9939), whereas TEAC showed reduced values in G1 (2.1 nmol/L) compared with G2 (2.31 nmol/L, P = 0.0005). There was a positive correlation between TBARS and TEAC only among patients (r = 0.49; P = 0.0019). High levels of TEAC stood out among controls, regardless of smoking or drinking habits, compared with G1 (P < 0.0001 for both). There was no association between medication and levels of TBARS and TEAC among the analysed patients (p > 0.05). However, TEAC, when analysed separately, was lower among patients (P = 0.0005).
Conclusions: There was no association of medication and the GSTM1 and GSTT1 polymorphisms with FM. However, reduced TEAC levels in patients reflect the inflammatory effect of the disease, with possible implications in the pathophysiology of FM.
Keywords
Fibromyalgia, Oxidative stress, Reactive oxygen species (ROS), Medication therapy
Introduction
Fibromyalgia (FM) is a syndrome characterized by chronic diffuse musculoskeletal pain and other symptoms that have a negative impact on the quality of life of patients [1,2]. FM predominantly affects women, accounting for approximately 85% of cases [3].
Recently, oxidative stress has been implicated as a relevant event in the pathogenesis of FM [4,5]. Stress is associated with overload of reactive oxygen species (ROS), which damage biomolecules and cellular components [5,6], they trigger the oxidation process [7].
In this context, increased ROS levels have been observed in blood mononuclear cells in patients with FM, followed by reduction by the action of antioxidants, which reflects in changes in markers of oxidative stress, including thiobarbituric acid reactive substances (TBARS) and the Trolox equivalent antioxidant capacity (TEAC), whose level in plasma, serum or cell lysate have been widely used in the assessment of lipid peroxidation [8]. This mechanism leads to oxidative destruction of polyunsaturated fatty acids, which are constituents of cellular membranes [8]. Additionally, glutathione S-transferases, represented by genetic polymorphisms (GSTM1 and GSTT1), act in the detoxification of numerous products of oxidative reactions that damage nucleic acids, lipids and proteins [8]. Glutathione enzyme becomes inactive towards nullity of these genes and with increased exposure of their carriers to oxidative stress [8].
However, there are still controversies over the relationship of FM symptoms with oxidative stress and lipid peroxidation, confirmed by some authors [4,8,10], and denied by others [1]. Thus, genetic factors and their interaction with environmental factors should be identified [2]. This study aimed to evaluate the association of genetic polymorphisms GSTM1 and GSTT1, serum levels of TBARS and TEAC, lifestyle habits and medication therapy in patients with FM.
Patients and Methods
Patients
A total of 187 mixed-race women were studied, divided into two groups: G1- 81 women aged 22-72 years (47.0 ± 25.0 years) with FM diagnosis according to ACR [11]; G2 - 106 women aged 22-84 years (53.0 ± 31.0 years) without FM, chronic and systemic diseases under medication treatment. G1 were examined and selected at the Outpatient Pain Clinic and the G2 at the Blood Centre, both in the Hospital de Base University Medical Centre (HB) at the Medical School of São José do Rio Preto (FAMERP). All men and those women under psychiatric medication treatment (antidepressants, benzodiazepines and others) and with chronic disease were excluded. All participants were informed of the study and signed an Informed Consent Document.
Methods
All subjects completed a questionnaire about clinical data and life habits (smoking and alcohol consumption). Peripheral blood samples were collected from all subjects for analysis of genetic polymorphisms (GSTM1 and GSTT1). Genomic DNA was extracted from whole blood (5 mL), collected with EDTA using the salting out method [12]. Amplification of polymorphic fragment was performed by PCR-RFLP (polymerase chain reaction- restriction fragment length polymorphism). Each reaction included 0.5 μL of each deoxynucleotide (0.8 mM); 2.5 μL of 10 X PCR buffer; 2.5 μL of 10% dimethyl sulfoxide; 2.5 μL of each primer (2.5μM); 0.2 μL of Taq polymerase (5U/μL); 11 μL of Milli Q water; 2 μL of dilution of genomic DNA (0.2 μg).The primers used were: GSTM1- P1: 5' AAC TCC CTA AAA GCT AAA C 3'; P2: 5' GTT GGG CTC AAA TAT ACG GTG G 3'; GSTT1- P1: 5' TTC CTT ACT GGT CCT CAC ATC TC 3'; P2: 5' TCA CCG ATC ATG GCC AGC A 3'. In the same reaction, the CYP1A1gene was used as control, with the following primers: P1: 5' GAA CTG CAG CTT CTG TCT CCA 3'; P2: 5 'CAG CTG CAT TTG GAA GTG CTC 3'. The initial DNA denaturation was obtained at 94°C for 4 minutes, followed by 39 cycles at 94°C for 2 minutes, 59°C for 1 minute and extension at 72°C for 1 minute and a final cycle at 72°C for 10 minutes [13]. The post-PCR product was subjected to electrophoresis on 1.5% agarose gel under a constant current of 150 volts for 45 minutes, separating fragments of 423 base pairs (bp) (GSTT1), 230 bp (GSTM1) and 310 bp (CYP control) (Figure 1). Individuals were classified as non-null genotype (presence of one or two polymorphisms) and null genotype (absence of polymorphisms).
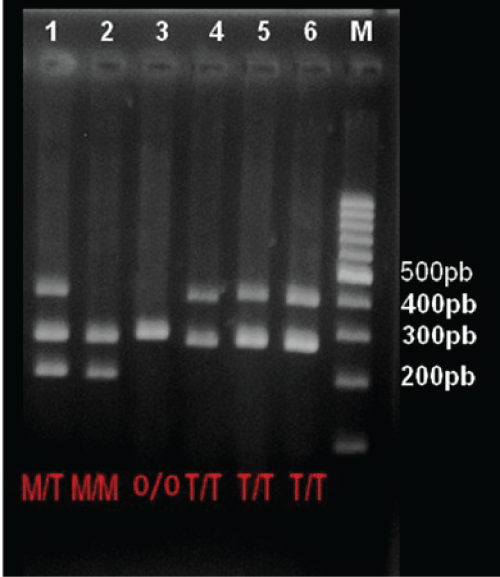
.
Figure 1: Photo of 1.5% agarose gel with standard M/T genotypes (Column 1), M/M (Column 2), 0/0 (Column 3) and T/T (Column 4 to 6). A sample of 100-1000pb (M) molecular weight marker was applied in the last column (ladder); bp = base pairs.
View Figure 1
Biochemical analyses of TBARS and TEAC were performed in 40 subjects of G1 and 54 subjects of G2 at random. The TBARS assay was performed in blood plasma, using the method based on the reaction of malondialdehyde (MDA) with thiobarbituric acid (TBA), according to Khon and Liversedge [14] and modified by Percário [15]. TEAC level was determined by its equivalence to potent antioxidant Trolox (6-hydroxy-2,5,7,8- tetramethylchroman-2-carboxylic acid; Aldrich Chemical Co. 23881-3), a synthetic water soluble vitamin E- analogue, according to Miller [16] modified by Re. [17] Reference values for TBARS were set between 0-440 nmol/mL [18] and TEAC between 1.32-1.60 nmol/mL [19].
Details about medication used for pain relief in FM were registered for 72 subjects of G1 using digital medical records of the hospital HB/FAMERP. The data about medication found in the remaining 9 patients was considered insufficient. For the purpose of clinical evaluation and comparison, the 72 patients of G1 were divided into two groups: one group used one drug and the other used two or more drugs. Classes of medications used in this study included: muscle relaxants, anti-inflammatories and analgesics.
Statistical analysis
The Mann Whitney test was used for quantitative variables (age, TBARS and TEAC values, with median values (due to the absence of normal distribution) and the Fisher's exact test or chi-square test was used for qualitative variables. GSTM1 and GSTT1 polymorphisms were analysed according to the presence or absence of the M1 and T1 alleles (M1/_, M1/T1, T1/_ and _/_). The correlation between the levels of TBARS and TEAC was calculated using the Sperman’s correlation coefficient. Level of significance was set at P < 0.05. Stats Direct, MiniTab14 and GraphPad3 programs were used.
Results
In the analysis of GSTM1 and GSTT1 polymorphisms, the M1/T1 genotype prevailed in G1 (50.6%) and G2 (45.3%; P = 0.5646), whereas nullity showed lower frequency in both groups (8.6 and 8.4, respectively; P = 1.098; Table 1). Furthermore, there was similarity in the distribution of genotypes considering the number of medications administered to relieve pain (P > 0.05; Table 2).
![]()
Table 1: Distribution of Glutathione S-transferase M1 and T1 polymorphisms (GSTT1 and GSTM1) in patients with fibromyalgia (G1) and controls (G2).
View Table 1
![]()
Table 2: Distribution of GSTM1 and GSTT1 polymorphisms considering the number of medications used for the treatment of patients with fibromyalgia.
View Table 2
Table 3 shows the analysis of serum with similar TBARS values between G1 (median = 218.0 nmol/L) and G2 (233.44 nmol/L, P = 0.9939). TEAC levels were lower among G1 (median = 2.1 nmol/L), compared with G2 (median = 2.3 nmol/L, P = 0.0005). Figure 2 shows correlation values between TEAC and TBARS, being positive among G1 (r = 0.49; P = 0.0019) and negative among G2 (r = -0.26; P = 0.05).
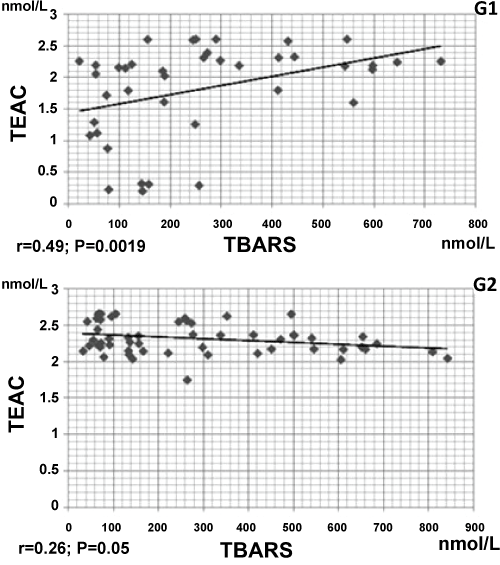
.
Figure 2: Correlation coefficient between serum thiobarbituric acid reactive substances (TBARS) and trolox equivalent antioxidant capacity (TEAC) in patients with fibromyalgia (G1) and individuals without the disease (G2).
View Figure 2
![]()
Table 3: Values of thiobarbituric acid reactive substances (TBARS) and Trolox equivalent antioxidant capacity (TEAC) in patients with fibromyalgia (G1), and controls (G2).
View Table 3
Serum levels of TBARS and TEAC were distributed according to the number of medications (1 and 2 or more) used by G1 (Table 4). Although there was increased TBARS level in the group under one medication (median = 251.4 nmol/L), compared with group under two or more medications (183.2 nmol/L), no significant difference (P = 0.345) could be observed.
![]()
Table 4: Values of thiobarbituric acid reactive substances (TBARS) and Trolox equivalent antioxidant capacity (TEAC) in patients with fibromyalgia (G1), considering the number of medications used in the treatment of disease (1 and 2 or more).
View Table 4
The analysis of biochemical profile and lifestyle habits (smoking and alcohol consumption) showed similar TBARS values between both groups (P > 0.05), as well as significantly increased levels of TEAC among non-smoking G2 (2.3 nmol/L) and non-drinkers G2 (2.3 nmol/L), compared with G1 (2.0 nmol/L and 2.1 nmol/L; respectively, P < 0.0001 for both; Figure 3).
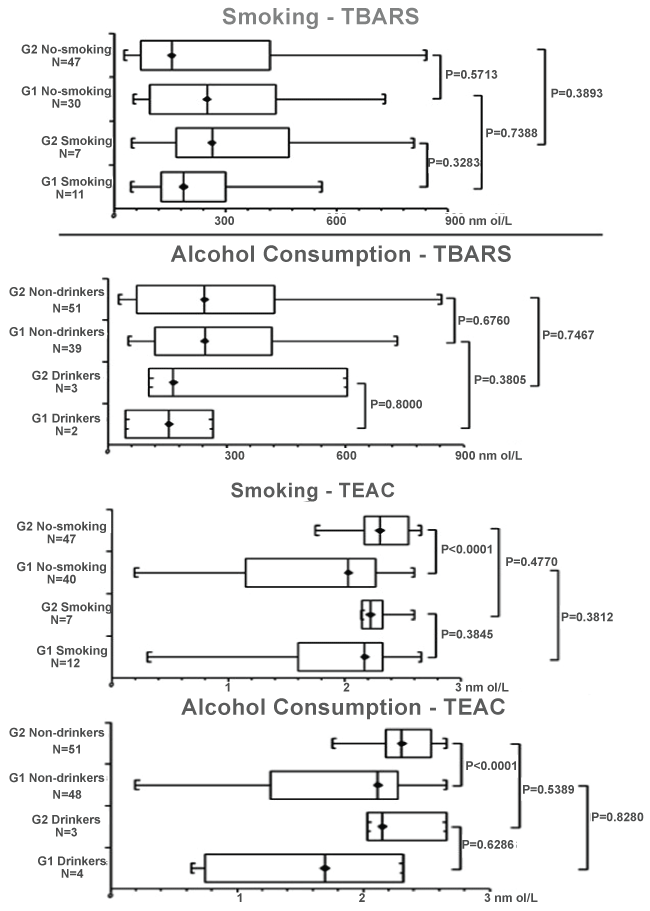
.
Figure 3: Box plot representing median and quartile values for serum levels of thiobarbituric acid reactive substances (TBARS) and trolox equivalent antioxidant capacity (TEAC) in patients with fibromyalgia (G1) and controls (G2), considering lifestyle habits (smoking and alcohol consumption); P = level of significance < 0.05.
View Figure 3
Discussion
In this study, GSTs polymorphisms (M1 and T1) involved in biotransformation and elimination of xenobiotics and protection of cells against oxidative stress [20] showed similar distribution between G1 e G2. It is noteworthy that the null genotype, related to higher exposure to oxidative stress due to homozygous deletion of GSTT1 and GSTM1, showed low frequency in both groups, as observed in studies about cancer in Brazilian populations [21]. The same results were found in studies about multiple sclerosis [22] and endometriossis [23,24] with other populations.
Admittedly, oxidative stress depends on the balance between the production of free radicals and the action of antioxidants [25,26]. In this sense, the antioxidant capacity was analysed by means of serum levels of TEAC, which were reduced in G1 compared with G2. This may be a reflection of the FM physiopathological process itself, which produces increased oxidative stress, thereby reducing the levels of TEAC and TBARS, as observed in the positive correlation between TBARS and TEAC among G1. In contrast, in a similar study of osteoarthritis (another rheumatologic condition associated with inflammatory process), increased levels of TBARS and reduced TEAC [27] were observed. Despite conflicting results, reduced oxidative capacity, whether absolute (genetically determined) or relative (secondary to increased free radical production due to the inflammatory process) is likely to participate in FM pathophysiology [4,26,27].
Medication therapy for pain in FM, although lacking systematised clinical management protocols, includes the use of several classes of medications, namely anti-inflammatory, analgesics and muscle relaxants [28]. These drugs relieve pain and often improve the functional capacity of the patient. The possible antioxidant role of these medications or an association between them, however, is disputable [24-27]. This study has not shown statistically significant association between medications used and serum levels of TBARS and TEAC, possibly due to the small sample size. However, studies have observed the influence of analgesics on levels of TBARS [25]. Furthermore, this study also found that lifestyle habits, such as smoking and alcohol consumption, were similar among G1 and G2, in agreement with current literature data [29]. However, higher levels of TEAC in non-smokers and non-drinkers among G2 suggest greater protection against oxidative stress in these individuals.
In conclusion, GSTM1 and GSTT1 polymorphisms were not associated with FM patients in this study. There was no association between medications and genetic variants or TBARS and TEAC levels among the analysed patients. However, lower levels of TEAC in G1 (even among non-smokers and non-drinkers) suggest increased exposure to oxidative stress in this group, with possible implication in the pathophysiology of the disease. Due to the lack of studies on oxidative stress in FM in Brazilian population, as well as the need to clarify the underlying physiopathological process of the disease, further studies with larger sample size are needed.
Acknowledgements
The authors thank all patients who contributed to this study.
Financing and Support
Institutional Scientific Initiation Scholarship Program/Institutional Scientific Initiation Scholarship Program (PIBIC/CNPQ) and Medical School of São José do Rio Preto (PIBIC/FAMERP).
Ethical Statement
This study was approved by the Research Ethics Committee of the Medical School of São José do Rio Preto /SP (CAEE: 14187413.2.0000.5415).
References
-
Cassisi G, Sarzi-Puttini P, Casale R, Cazzola M, Boccassini L, et al. (2014) Pain in fibromyalgia and related conditions. Reumatismo 66: 72-86.
-
Becker RM, da Silva VK, Machado Fda S, dos Santos AF, Meireles DC, et al. (2010) Association between environmental quality, stress and APOE gene variation in fibromyalgia susceptibility determination. Rev Bras Reumatol 50: 617-624.
-
Feng J, Zhang Z, Wu X, Mao A, Chang F, et al. (2013) Discovery of potential new gene variants and inflammatory cytokine associations with fibromyalgia syndrome by whole exome sequencing. PLoS One 8: e65033.
-
Fatima G, Das SK, Mahdi AA (2015) Some oxidative and antioxidative parameters and their relationship with clinical symptoms in women with fibromyalgia syndrome. Int J Rheum Dis. 2015.
-
Akbas A, Inanir A, Benli I, Onder Y, Aydogan L (2014) Evaluation of some antioxidant enzyme activities (SOD and GPX) and their polymorphisms (MnSOD2 Ala9Val, GPX1 Pro198Leu) in fibromyalgia. Eur Rev Med Pharmacol Sci 18: 1199-1203.
-
Sánchez A, Calpena AC, Clares B (2015) Evaluating the Oxidative Stress in Inflammation: Role of Melatonin. Int J Mol Sci 16: 16981-17004.
-
Bast A, Haenen GR, Doelman CJ (1991) Oxidants and antioxidants: state of the art. Am J Med 91: 2S-13S.
-
Cordero MD, Alcocer-Gómez E, Cano-García FJ, De Miguel M, Carrión AM, et al. (2011) Clinical symptoms in fibromyalgia are better associated to lipid peroxidation levels in blood mononuclear cells rather than in plasma. PloS One 6: e26915.
-
Uzunoglu S, Acar H, Okudan N, Gökbel H, Mevlitoglu I, et al. (2006) Evaluation of the association between null genotypes of glutathione-S-transferases and Behcet's disease. Arch Dermatol Res 297: 289-293.
-
Doshi SB, Agarwal A (2013) The role of oxidative stress in menopause. J Midlife Health 4: 140-146.
-
Wolfe F, Clauw DJ, Fitzcharles MA, Goldenberg DL, Katz RS, et al. (2010) The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res (Hoboken) 62: 600-610.
-
Salazar LA, Hirata MH, Cavalli SA, Machado MO, Hirata RD (1998) Optimized procedure for DNA isolation from fresh and cryopreserved clotted human blood useful in clinical molecular testing. Clin Chem 44: 1748-1750.
-
Rossini A, Rapozo DC, Amorim LM, Macedo JM, Medina R, et al. (2002) Frequencies of GSTM1, GSTT1, and GSTP1 polymorphisms in a Brazilian population. Genet Mol Res 1: 233-240.
-
Kohn HI, Liversedge M (1944) On a New Aerobic Metabolite Whose Production by Brain is Inhibited By Apomorphine, Emetine, Ergotamine, Epinephrine, and Menadione. J Pharmacol and Exp Therap 82: 292.
-
Percário S (1993) Dosagem das LDLs modificadas através da peroxidação lipídica: correlacão com risco aterogênico. AMHFCMSCSP 13: 7-9.
-
Miller NJ, Rice-Evans C, Davies MJ, Gopinathan V, Milner A (1993) A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. ClinSci (Lond) 84: 407-412
-
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, et al. (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26: 1231-1237.
-
Ondei Lde S, Estevão Ida F, Rocha MI, Percário S, Souza DR, et al. (2013) Oxidative stress and antioxidant status in beta-thalassemia heterozygotes. Rev Bras Hematol Hemoter 35: 409-413.
-
Rice-Evans C, Miller NJ (1994) Total antioxidant status in plasma and body fluids. Methods Enzymol 234: 279-293.
-
De Luca C, Raskovic D, Pacifico V, Thai JC, Korkina L (2011) The search for reliable biomarkers of disease in multiple chemical sensitivity and other environmental intolerances. Int J Environ Res Public Health 8: 2770-2797.
-
Sa RA, Moreira Ados S, Cabello PH, Ornellas AA, Costa EB, et al. (2014) Human glutathione S-transferase polymorphisms associated with prostate cancer in the Brazilian population. Int Braz J Urol 40: 463-473.
-
Lee YH, Seo YH, Kim JH, Choi SJ, Ji JD, et al. (2015) Meta-analysis of associations between MTHFR and GST polymorphisms and susceptibility to multiple sclerosis. Neurol Sci 36: 2089-2096.
-
Zhu H, Bao J, Liu S, Chen Q, Shen H (2014) Null genotypes of GSTM1 and GSTT1 and endometriosis risk: a meta-analysis of 25 case-control studies. PLoS One 9: e106761.
-
Eisinger J, Zakarian H, Pouly E, Plantamura A, Ayavou T (1996) Protein peroxidation, magnesium deficiency and fibromyalgia. Magnes Res 9: 313-316.
-
Bellato E, Marini E, Castoldi F, Barbasetti N, Mattei L, et al. (2012) Fibromyalgia syndrome: etiology, pathogenesis, diagnosis, and treatment. Pain Res Treat 2012: 426130.
-
Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M (2006) Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact 160: 1-40.
-
Suantawee T, Tantavisut S, Adisakwattana S, Tanavalee A, Yuktanandana P, et al. (2013) Oxidative stress, vitamin e, and antioxidant capacity in knee osteoarthritis. J Clin Diagn Res 7: 1855-1859.
-
Arnold LM, Clauw DJ (2010) Fibromyalgia syndrome: practical strategies for improving diagnosis and patient outcomes. Am J Med 123: S2.
-
Campos C, Guzmán R, López-Fernández E, Casado A (2011) Urinary biomarkers of oxidative/nitrosative stress in healthy smokers. Inhal Toxicol 23: 148-156.





