Journal of Sleep Disorders and Management
Perioperative Management of Patients with Obstructive Sleep Apnea
Cigdem Akyol Beyoglu*
Department of Anesthesiology and Reanimation, Istanbul University, Istanbul, Turkey
*Corresponding author:
Dr. Cigdem Akyol Beyoglu, MD, Istanbul University, Cerrahpasa Medical School Department of Anesthesiology and Reanimation, Istanbul University, Istanbul, Turkey, E-mail: cigdem-akyoll@hotmail.com
J Sleep Disord Manag, JSDM-2-009, (Volume 2, Issue 1), Review Article
Received: December 05, 2015: Accepted: February 22, 2016: Published: February 25, 2016
Citation: Beyoglu CA (2016) Perioperative Management of Patients with Obstructive Sleep Apnea. J Sleep Disord Manag 2:009
Copyright: © 2016 Beyoglu CA. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The aim of this manuscript is to determine perioperative management of patients with obstructive sleep apnea. Obstructive sleep apnea (OSA) may cause postoperative complications related to upper airway collapse and cardiopulmonary system. Treatment with continue positive airway pressure (CPAP) may offer high standards of living to the patients and may protect them against postoperative complications. Long-acting and potent opioids may cause postoperative airway collapse in this population. Patients should be monitored and closely observed after surgery and anesthesia. In conclusion preoperative and postoperative CPAP administrations reduce the risk of postoperative complications significantly in OSA patients. Opioids should be avoided if possible intraoperatively.
Keywords
Obstructive sleep apnea, Perioperative management, Continue positive airway pressure
Introduction
Obstructive Sleep Apnea (OSA) is the intermittent and repetitive, partial (hypopnea) or complete (apnea) blockade of upper airway during sleep. The obstruction is removed upon waking up and reconstruction of muscle tonus. Episodes of apnea, hypopnea and waking occur successively throughout the sleep, resulting in certain medical problems associated with episodes of hypoxia due to segmented sleep of poor quality and apneas. Hypoxia triggers symptomatic stimulation; which, in turn, increases levels of stress hormones (ACTH; cortisol) in circulation, causes dyslipidemia, inflammatory system activation, endothelial injury and platelet aggregation [1]. These effects might predispose OSA patients for hypertension (HT), arrhythmia, stroke, myocardial infarction (MI), pulmonary HT, congestive heart failure (CHF), delayed healing of wounds and mental dysfunction [1]. The gold standard method for diagnosis is polysomnography performed at sleep laboratories. The prevalence of OSA is 3% to 28% in the adult population [2]. It is estimated that the prevalence of OSA is greater in surgical patients than in healthy population and associated with the type of surgery [3]. The prevalence of OSA is found to be > 70% in the bariatric surgery population [4,5].
Treatment of the syndrome involves the use of positive airway pressure (PAP) to prevent upper airway collapse in the majority of patients [6]. Although continuous PAP (CPAP) is the most commonly used and effective treatment modality, medical, behavioral and surgical treatment might also be an alternative in selected patients [6]. The risk of cardiac and cerebrovascular events is decreased with long term use of CPAP [1].
Preoperative clinical course-perioperative risk
Majority of patients with OSA are undiagnosed before surgery [7]. Patients who are suspected to have OSA should be questioned with regard to clinical symptoms meticulously. The physical examination should include upper airway anatomy, respiratory, cardiovascular, and neurologic systems [8]. Risk factors for OSA are obesity, congestive heart failure, atrial fibrillation, treatment refractory hypertension, type 2 diabetes mellitus, stroke, nocturnal dysrhythmias, pulmonary hypertension, high-risk driving populations (such as commercial truck drivers) and bariat¬ric surgery [6] (Table 1).
![]()
Table 1: Risk factors for OSA.
View Table 1
Patients who have risk factors for OSA and those provide suggestive findings in clinical examination and anamnesis should be seeked for OSA. Polysomnography is not recommended in preoperative period due to its time-consuming and high cost effect [9]. Therefore anesthesiologists need to use some practical screening tools to identify patients with OSA who were undiagnosed before surgery. As well as there are several ones STOP-BANG and Berlin questionnaires are well defined and proved to be validated to predict patients who have true OSA [3,10-12]. Berlin questionnaire (BQ) is consisted of totally 10 questions which are about snoring in category 1, daytime symptoms in category 2 and hypertension or body mass index (BMI) in category 3. Abrisami et al. [10] reported that the sensitivity of BQ in determining the patients carrying high risk for OSA is up to 95%. The STOP-BANG questionnaire (snoring, tiredness, observed apnea, high blood pressure, body mass index, age, neck circumference, gender) is divided into 2 categories including 4 questions about OSA symptoms and 4 questions about physical characteristics of the patient. Chung et al. [13] found the STOP-BANG questionnaire with the highest sensitivity in predicting OSA patients. It is suggested to use in predicting OSA patients preoperatively as the most useful one [12]. Also American Society of Anesthesiologists (ASA) taskforce on OSA developed a checklist for identifying patients with OSA. It is a 14-item checklist categorized into physical characteristics, history of airway obstruction during sleep, and complaints of somnolence with a sensitivity of 79% [9].
Another tool to screen OSA in surgical patients is nocturnal pulse oxymetry. The strong correlation between oxygen desaturation index (ODI) from nocturnal oxymetry and apnea-hypopnea index (AHI) from PSG has been shown recently [14]. Nocturnal oxymetry and home sleep testing are recommended as alternative screening tools in surgical population [3,9].
Patients with OSA are in increased risk of perioperative complications [15-17]. The most common complications have been reported to be associated with airway security and pulmonary system that may cause brain damage even death if unmonitored postoperatively [15,18]. Postoperative cardiac events such as dysrhythmias, atrial fibrillation are the secondly major complications after surgery in OSA patients [18]. Besides cardio respiratory complications patients with OSA had been shown to have higher incidence of encephalopathy and postoperative infections (mediastinitis) [19]. Patients with OSA have been shown to have higher rate of transfer to intensive care unit (ICU) and increased length of hospital stay when compared to control subjects [18]. Patients with OSA who are not compliant with their CPAP therapy preoperatively are at highest risk of postoperative complications [17]. The following factors play major roles in the development of perioperative complications in OSA patients:
1. Upper airway anatomy
2. Disease course and severity of cardiopulmonary effects
3. Decreased FRC and oxygen reserve due to obesity
In the postoperative period, anxiety, postoperative pain, release of stress hormones, and impaired sleep due to disruption of circadian rhythm become more severe in patients with sleep disorders. Anesthetics and opioid analgesics might cause the collapse of upper airways and impairment of arousal mechanisms via central mechanisms [20]. As a result of these effects existing obstructive apneas might become more severe postoperatively. Also, supine position might lead to upper airway collapse.
Anesthesiologists should evaluate the cardiopulmonary effects of OSA meticulously in the preoperative period, and arrange for CPAP treatment until stability is established, or for approximately 3 months, in patients with congestive heart failure and hypercarbia (Pco2 > 6.5 Kpa) [1].
Preoperative management of suspected OSA patients
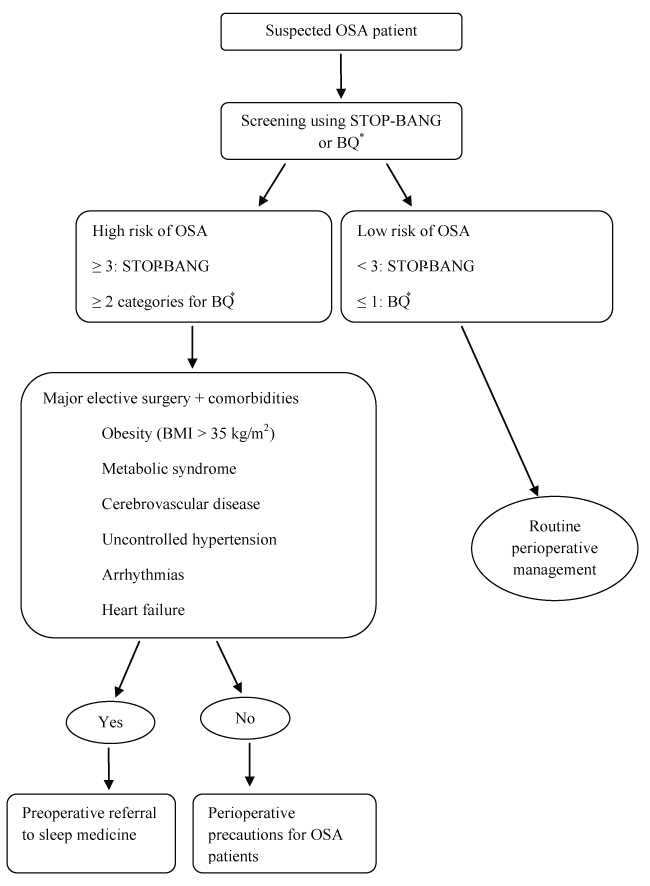
In case of a patient is not diagnosed of OSA, but has high risk of OSA with reference to the questionnaires (≥ 3 for STOP-BANG and ≥ 2 categories + for BQ), then urgent surgery must be allowed. If the high risk patient will undergo major elective surgery and has comorbidities, the anesthesiologist should refer the patient to a sleep physician. Comorbidities include uncontrolled hypertension, heart failure, arrhythmias, cerebrovascular disease, morbid obesity, and metabolic syndrome (Figure 1) [9]. Also there maybe patients of high risk of OSA but without any comorbidities, then minor surgery should be allowed. On such an occasion, the anesthesiologist must show a great caution for the patient peroperatively as he is an OSA patient. If perioperative experiences such as difficult airway or postoperative oxygen desaturation suggest high risk of OSA, then the anesthesiologist should refer the patient to a sleep physician postoperatively.
Preoperative pap
Anesthesiologists should evaluate the results of sleep laboratory tests preoperatively and generate a notion on the course and severity of the disease. Consultations should be obtained to evaluate the test results when necessary. Patients should be re-evaluated by the related specialist when clinical symptoms persist despite treatment, or no treatment has been initiated.
When a patient is diagnosed with OSA, it should be decided if treatment is required and if required which type of therapy is needed. Positive airway pressure treatment is often first choice in majority of patients [6]. There are two ways of implementing PAP therapy; CPAP or bi-level PAP (BPAP). Fixed CPAP is usually the first choice as it its usefulness is well established and easy to use [6]. Auto CPAP is taking more common place in treatment with the advantage of lower pressure level requirements in supervision of home sleep apnea testing. If OSA is complicated with chronic respiratory failure or central sleep apnea, then BPAP maybe more useful [21]. Physicians should note that changes in device parameters in the post-anesthetic period in patients treated with CPAP might warrant oxygen or humidifier addition to the device.
Berlin questionnaire
Continue positive airway pressure is an effective method to maintain upper airway opening following postoperative extubation in patients with possible OSA and no previous history of treatment. Toleration of CPAP might be reduced in case of patients who are using CPAP for the first time or in the presence of inexperienced healthcare staff. The use of “auto-CPAP” under close follow-up and monitorization might be an option in the latter case [20].
Intraoperative management
Benzodiazepines and opioids might aggravate OSA symptoms by causing relaxation of pharyngeal muscles. Sedative premedication should be avoided as much as possible in these patients; and should be administered under close follow-up and monitorization if essential. However, anti-reflux and anti-sialagogue premedications are recommended. Pre-oxygenation should be administered routinely against the risks of difficult airway and prolonged intubation to protect patients from potential hypoxia.
Intubation
Induction planning in OSA patients should encompass the possibilities of difficult mask ventilation and difficult intubation. Factors that might be associated with difficult airway including obesity, wide neck circumference, and Mallampati should be assessed carefully [5]. Difficult mask ventilation is an expected incident in obese OSA patients, and therefore the necessary equipment should be ready during induction.
Selection of the anesthetic agent
Local anesthesia or regional anesthesia techniques should be preferred in non-sedative surgical procedures of OSA patients. Intravenous (IV) sedation should not be administered in these patients, and airway security should always be the priority. Airway should be secured via general anesthesia with endotracheal intubation in case of surgery cannot be performed under regional anesthesia. Intravenous benzodiazepines and opioids cause respiratory depression through the central mechanisms. These agents might lead to upper airway collapse by causing relaxation of pharyngeal muscles and preventing tonic contractions. They also block the reflex respiratory response to hypoxia and hypercarbia [2]. Therefore, IV benzodiazepines and opioids are not used in pre-induction premedication. Long-term effects of these agents might exacerbate the upper airway obstruction following extubation, and suppress arousal and respiratory responses triggered by apnea and hypoxia, in intraoperative administrations. These effects might lead to deep and prolonged apnea and even respiratory arrest in OSA patients. Therefore, long-acting benzodiazepines and opioids should be avoided intraoperatively in OSA patients, and postoperative pain treatment should crucially include regional methods, less potent and short-acting opioids, and multimodal analgesia modalities to reduce the amount of opioid to be used.
Although the use of opioids as part of regional techniques does not have any proven disadvantages in OSA patients, postoperative respiratory depression potential of epidural opioids should be kept in mind [22].
Extubation
The extubation period is a critical process in OSA patients when airway opening is in risk of collapse. Extubation should be performed when patient has adequate muscle tonus, active airway reflexes, and is completely awake. It should be noted that upper airway obstruction might occur meanwhile, and oral or nasal airways of proper size should be ready to maintain airway opening when necessary. Upper airway collapse might lead to negative pressure pulmonary edema due to respiration effort despite closed glottis.
Termination of anesthesia and extubation are risky periods when stress hormone levels peak and hemodynamic fluctuations often occur. Anesthesiologists should be alert in this period when OSA patients might often be hypertensive or arrhythmic and close hemodynamic follow-up should be pursued to intervene when necessary.
Postoperative management
Postoperative first 24 hours are the most critical period in OSA patients. Physicians should note that decreased pharyngeal muscle tonus, upper airway obstruction, and respiratory depression might potentially occur in this period as a result of intraoperative IV benzodiazepine and opioid administrations, and suppress reflex respiratory response to hypoxia and hypercarbia. Patients should be followed-up with standard monitorization including peripheral oxygen (O2) saturation, electrocardiography and non-invasive arterial pressure in the early postoperative period.
Postoperative nasal CPAP should particularly be used in OSA patients who received intravenous opioids and need to remain in supine position. Non-invasive mechanical ventilation support might often be provided with the preoperative devices and parameters; however, increased pressure support or additional O2 supplementation might also be needed. Full-face mask rather than nasal mask might be tried to better tolerate the leakages in case the patient has nasogastric tube.
Oxygen support should be avoided if postoperative CPAP will not be used in OSA patients. Hypoxia of the apneic period triggers reflex respiratory response in these patients, and additional O2 support might prevent arousals by removing hypoxia of this period. Combined effect of intraoperatively administered intravenous opioids and apneas not met by waking and deep respiration might result in respiratory arrest.
OSA is also a risk factor for postoperative delirium [23]. Although tissue hypoxia and neuronal injury appear to be responsible; the exact causes, results and treatment options should be investigated in clinical studies.
Conclusion
Obstructive sleep apnea is a challenge in anesthetic management due to difficulties in airway management and cardiopulmonary comorbidities. Preoperative and postoperative CPAP administrations reduce the risk of complications significantly. Opioids should be avoided if possible intraoperatively and postoperative pain management should be attained with regional techniques or multimodal analgesia with the lowest possible dosages and short-acting opioids. Patients should be followed-up closely in the first 24 hours of postoperative period, and SpO2 and ECG monitorization should be obtained if possible. Perioperative anesthesia management of OSA patients remains to be a poorly understood topic, and further clinical studies are worthwhile.
References
-
Martinez G, Faber P (2011) Obstructive sleep apnoea. Contin Educ Anaesth Crit Care Pain 11: 5-8.
-
Marty M, Lacaze O, Arreto CD, Pierrisnard L, Bour F, et al. (2015) Snoring and Obstructive Sleep Apnea: Objective Efficacy and Impact of a Chairside Fabricated Mandibular Advancement Device. J Prosthodont.
-
Vasu TS, Grewal R, Doghramji K (2012) Obstructive sleep apnea syndrome and perioperative complications: a systematic review of the literature. J Clin Sleep Med 8: 199-207.
-
Frey WC, Pilcher J (2003) Obstructive sleep-related breathing disorders in patients evaluated for bariatric surgery. Obes Surg 13: 676-683.
-
O'Keeffe T, Patterson EJ (2004) Evidence supporting routine polysomnography before bariatric surgery. Obes Surg 14: 23-26.
-
Epstein LJ, Kristo D, Strollo PJ Jr, Friedman N, Malhotra A, et al. (2009) Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med 5: 263-276.
-
Chung F, Ward B, Ho J, Yuan H, Kayumov L, et al. (2007) Preoperative identification of sleep apnea risk in elective surgical patients, using the Berlin questionnaire. J Clin Anesth 19: 130-134.
-
Kushida CA, Littner MR, Morgenthaler T, Alessi CA, Bailey D, et al. (2005) Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep 28: 499-521.
-
Seet E, Chung F (2010) Obstructive sleep apnea: preoperative assessment. Anesthesiol Clin 28: 199-215.
-
Abrishami A, Khajehdehi A, Chung F (2010) A systematic review of screening questionnaires for obstructive sleep apnea. Can J Anaesth 57: 423-438.
-
Bouloukaki I, Komninos ID, Mermigkis C, Micheli K, Komninou M, et al. (2013) Translation and validation of Berlin questionnaire in primary health care in Greece. BMC Pulm Med 13: 6.
-
El Sayed I (2012) Comparison of four sleep questionnaires for screening obstructive sleep apnea. Egyptian Journal of Chest Diseases and Tuberculosis 61: 433-441.
-
Chung F, Yegneswaran B, Liao P, Chung SA, Vairavanathan S, et al. (2008) STOP questionnaire: a tool to screen patients for obstructive sleep apnea. Anesthesiology 108: 812-821.
-
Chung F, Liao P, Sun F, Shapiro C, Elsaid H (2009) Nocturnal oximeter: a sensitive and specific tool to detect the surgical patients with moderate and severe OSA. Anesthesiology 111: A480.
-
Fouladpour N, Jesudoss R, Bolden N, Shaman Z, Auckley D (2016) Perioperative Complications in Obstructive Sleep Apnea Patients Undergoing Surgery: A Review of the Legal Literature. Anesth Analg. 122: 145-151.
-
Gupta RM, Parvizi J, Hanssen AD, Gay PC (2001) Postoperative complications in patients with obstructive sleep apnea syndrome undergoing hip or knee replacement: a case-control study. Mayo Clin Proc 76: 897-905.
-
Liao P, Yegneswaran B, Vairavanathan S, Zilberman P, Chung F (2009) Postoperative complications in patients with obstructive sleep apnea: a retrospective matched cohort study. Can J Anaesth 56: 819-828.
-
Kaw R, Chung F, Pasupuleti V, Mehta J, Gay PC, et al. (2012) Meta-analysis of the association between obstructive sleep apnoea and postoperative outcome. Br J Anaesth 109: 897-906.
-
Kaw R, Golish J, Ghamande S, Burgess R, Foldvary N, et al. (2006) Incremental risk of obstructive sleep apnea on cardiac surgical outcomes. J Cardiovasc Surg (Torino) 47: 683-689.
-
Meoli AL, Rosen CL, Kristo D, Kohrman M, Gooneratne N, et al. (2003) Upper airway management of the adult patient with obstructive sleep apnoea in the perioperative period- avoiding complications. Sleep. 26: 1060-1065.
-
Kakkar RK, Berry RB (2007) Positive airway pressure treatment for obstructive sleep apnea. Chest 132: 1057-1072.
-
Knill RL, Clement JL, Thompson WR (1981) Epidural morphine causes delayed and prolonged ventilatory depression. Can Anaesth Soc J 28: 537-543.
-
Mirrakhimov AE, Brewbaker CL, Krystal AD, Kwatra MM (2014) Obstructive sleep apnea and delirium: exploring possible mechanisms. Sleep Breath 18: 19-29.