International Journal of Critical Care and Emergency Medicine
Glycemic Disturbances on Admission as a Predictor of Inpatient Mortality
Daniel Sáenz-Abad1*, José Antonio Gimeno-Orna2, María del Carmen Lahoza-Pérez1, Elena Rivero-Sanz1, Marta Jordán-Domingo1, Artur Juan-Arribas3, María del Mar Becerra-Mayor4 and Cristina Baquer-Sahún1
1Emergency Department, Hospital Clínico Universitario Lozano Blesa, Spain
2Department of Endocrinology and Metabolism, Hospital Clínico Universitario. Lozano Blesa, Spain
3Emergency Department, Hospital Sant Jaume in Calella, Spain
4Emergency Department, Hospital Carlos Haya, Spain
*Corresponding author:
Daniel Sáenz-Abad, Emergency Department, Hospital Clínico Universitario Lozano Blesa, Juan Carlos I 59, 3aB, Zaragoza, Spain, Tel: 0034-676-844-8144, E-mail: danielsaenzabad@hotmail.com
Int J Crit Care Emerg Med, IJCCEM-1-007, (Volume 1, Issue 2), Original Research; ISSN: 2474-3674
Received: September 12, 2015 | Accepted: October 28, 2015 | Published: October 30, 2015
Citation: Sáenz-Abad D, Gimeno-Orna JA, Lahoza-Pérez MC, Rivero-Sanz E, Jordán-Domingo M, et al. (2015) Glycemic Disturbances on Admission as a Predictor of Inpatient Mortality. Int J Crit Care Emerg Med 1:007. 10.23937/2474-3674/1510007
Copyright: © 2015 Sáenz-Abad D. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Our aim was to determine the influence of glycemic disturbances on admission in the emergency department (ED) on hospital mortality.
Methods: This is a case-control retrospective analytical study. The cases were patients deceased during hospitalization and controls where those discharged in the same time period. Patients were age-matched and the final outcome of the study was hospital mortality. Rapid Emergency Medicine Score (REMS) and Charlson comorbidity index as well as blood analysis (full blood count, glucose, renal function, ions) were determined. Abnormal glucose levels (dysglycemia) were those considered to be < 70 or > 200 mg/dl. We used logistic regression, integrated discrimination improvement (IDI) index and ROC curves to determine the predictive mortality capacity.
Results: 1153 patients were included in the study. Hyperglycemia was detected in 171 (14.8%) patients whilst hypoglycemia in 19 (1.6%) patients, with a prevalence of dysglycemia at 16.4%. The best model to predict hospital mortality included REMS (OR1point = 1.09; CI 95% 1.033-1.15; p = 0.001), Charlson comorbidity index (OR1point = 1.42; CI 95% 1.31-1.55; p < 0.001), hemoglobin (OR1gr/dl = 0.89; CI 95% 0.84-0.95; p < 0.001), thrombocytopenia (OR = 2.95; CI 95% 1.71-5.11; p < 0.001), leucocytosis (OR = 1.94; CI 95% 1.47-2.56; p < 0.001), diabetes mellitus (DM) (OR = 0.5; CI 95% 0.35-0.71; p < 0.001) and presence of dysglycemia (OR = 1.8; CI 95% 1.2-2.8; p = 0.005).
There was a significant improvement in the area under the ROC curve between the best predictive model chosen versus that including only the REMS score (0.522 vs 0.7478; p < 0.0001).IDI index after inclusion of dysglycemia was 0.00678 (p = 0.009).
Conclusions: The inclusion of dysglycemia in the mortality predictive scores increases its discrimination capacity in predicting hospital mortality.
Keywords
Hyperglycemia, Hypoglycemia, Diabetes, Hospital mortality
Introduction
The ED is the first and most important gateway of gravely ill patients with the aim to predict mortality. The REMS score [1,2] is composed of clinical variables, which include age, blood pressure, heart rate, oxygen saturation, respiratory rate, and Glasgow Come Score and is an invaluable tool in predicting mortality in the ED. However, other variables such as blood analyses are not included and may improve in predicting mortality outcome. As an example, the acute physiology and chronic health evaluation (APACHE) score used in patients in intensive care unit (ICU) in our country, incorporated analytical tests and demonstrated to be superior to its predecessor APACHE II [3] in measuring severity of disease and predicting mortality.
Abnormal glucose levels (dysglycemia), both hyper [4] and hypoglycemia [5], have been associated with an increase in hospital mortality. However, it is unclear whether it is an independent risk factor for mortality or a marker for underlying severe disease, as intensive glucose control has not demonstrate to reduce hospital mortality in critically ill patients in ICU [6], as well as those in general wards [7].
The aims of our study were:
1. To determine the importance of dysglycemia on admission as a marker of hospital mortality.
2. To evaluate whether adding glucose levels on admission to mortality predictive scores improves their discrimination capacity.
3. To determine whether the best predictive model for hospital mortality includes glucose levels.
Material and Methods
Study design
Retrospective analytical case-control study.
Patients
All patients (n = 650) who deceased during the hospitalizationin our hospital between the 1st of July and the 31st of December 2014 were collected as cases. 647 patients who were discharged in the same period were used as controls. Patients were age-matched.
Final outcome
Hospital mortality
Predictive variables
We collected the following variables in the ED:
- Clinical characteristics: sex, age, level of consciousness, blood pressure, heart rate, respiratory rate, oxygen saturation and temperature. With this information REMS score was calculated.
- Previous illnesses, including known DM, with Charlson comorbidity index calculation.
- Blood analysis: glucose, creatinine (with glomerular filtration rate determined using the chronic kidney disease epidemiology collaboration (CKD-EPI) equation), ions, full blood count. We defined hyperglycemia as a glucose level > 200 mg/dl, and hypoglycemia as < 70 mg/dl. Dysglycemia was defined as the presence of hyper or hypoglycemia.
Laboratory methods
Serum glucose was determined using hexokinase enzyme method. Other laboratory parameters were determined using standardized auto-analyser.
Statistical method
Mean and standard deviation were determined for describing quantitative variables and frequency distribution was determined for describing qualitative variables. We used student T-test and Chi-square test respectively to compare data.
We calculated potential mortality risk factors using univariate and multivariate logistic regression analysis. We evaluated the contribution of dysglycemia in three models with progressive adjustment:
Model 1: REMS
Model 2: Model 1 + known diabetes, sex, sodium, potassium, glomerular filtration rate, hemoglobin, thrombocytopenia and leucocytosis.
Model 3: Model 2 + Charlson comorbidity index
Using the IDI index, we calculated the improved predictive mortality capacity of each predictive model with the addition of dysglycemia. Finally we determined the best predictive hospital mortality model using sequential backward exclusion procedure. We used the ROC curve analysis to calculate the difference of predictive power between the model including only REMS and the best predictive model. Statistically significant differences were defined as p < 0.05. We used the programs SPSS 22.0 and R 3.2.1 to extrapolate the data.
Results
Patient characteristics according to initial glucose level
We collected data from 1297 patients (650 cases and 647 controls). 144 of these did not have available initial glucose levels in ED resulting in their elimination from the study and leaving 1153 patient data for the analysis.
The patients excluded were younger (70.1 vs 77.3 years; p < 0.001), more frequently men (68.1 vs 55.5%; p = 0.004), they had lower percentage of leucocytosis (29.6 vs 50.4%; p = 0.033), and lower REMS score (8.2 vs 9.3; p < 0.001) and Charlson comorbidity index (1.4 vs 2.3; p < 0.001).
The commonest causes of admission to the ED were dyspnoea (17%) and fever (5.9%). We detected hyperglycemia on admission in 171 (14.8%) patients and hypoglycemia in 19 (1.6%) patients, resulting in a total prevalence of dysglycemia of 16.4%. Table 1 shows the patient characteristics classified by initial serum glucose levels. It is shown that those patients who presented with hypoglycemia had worse Charlson score, had lower hemoglobin levels, and were more likely to be diabetic and thrombocytopenic. Patients with hyperglycemia were more likely to be diabetic and elderly, have worse REMS score, have higher levels of hemoglobin and leucocytosis and have a lower glomerular filtration rate. In table 2 the differences between cases and controls are presented along with the univariate risk associated to each variable. Patients who died had a higher prevalence of hyper, hypo and dysglycemia, higher REMS and Charlson scores, lower glomerular filtration rate, higher potassium and lower sodium levels, and greater alterations in the full blood count.
![]()
Table 1: Patient characteristics according to initial serum glucose level.
View Table 1
![]()
Table 2: Case and control characteristics.
View Table 2
Evaluating dysglycemia as a marker for hospital mortality
In the total sample it was shown that hyperglycemia, as well as hypoglycemia and dysglycemia, were associated with an increased risk of mortality. However, although hyperglycemia and dysglycemia were independent predictors for mortality in the adjusted analysis (Table 3), hypoglycemia lost the statistical significance after the adjustment for variables included in the models 2 and 3.
The interaction between dysglycemia and DM was significant (p = 0.006) so we undertook stratified analysis for this variable (Table 3), which showed that hyperglycemia and dysglycemia were independent predictive mortality markers for non-diabetic patients but not for known diabetics.
![]()
Table 3: Prognostic influence of glycemic disturbances on admission with progressive adjustments for potencial confounding factors.
View Table 3
Discriminative analysis
We used the IDI index to determine whether dysglycemia could improve the power of discrimination in the three models described in the material and methods section. In the total sample of patients and in those without a history of DM, information about the presence of dysglycemia on admission improved the predictive capacity of hospital mortality in all three models (Table 4).
![]()
Table 4: Improved IDI after addition of dysglycemia in the three models.
View Table 4
Selecting the best predictive model
From the total sample we determined that the best model for predicting hospital mortality was that including REMS (OR1point = 1.09; CI 95% 1.033-1.15; p = 0.001), Charlson comorbidity index (OR1point = 1.42; CI 95% 1.31-1.55; p < 0.001), hemoglobin (OR1gr/dl = 0.89; CI 95% 0.84-0.95; p < 0.001), thrombocytopenia (OR = 2.95; CI95% 1.71-5.11; p < 0.001), leucocytosis (OR = 1.94; CI 95% 1.47-2.56; p < 0.001), DM (OR = 0.5; CI 95% 0.35-0.71; p < 0.001) and presence of dysglycemia (OR = 1.8; CI 95% 1.2-2.8; p = 0.005).
There was a significant improvement in the area under the ROC curve between the best predictive model chosen and that including only the REMS score (0.522 vs 0.7478; p < 0.0001) as show in figure 1.
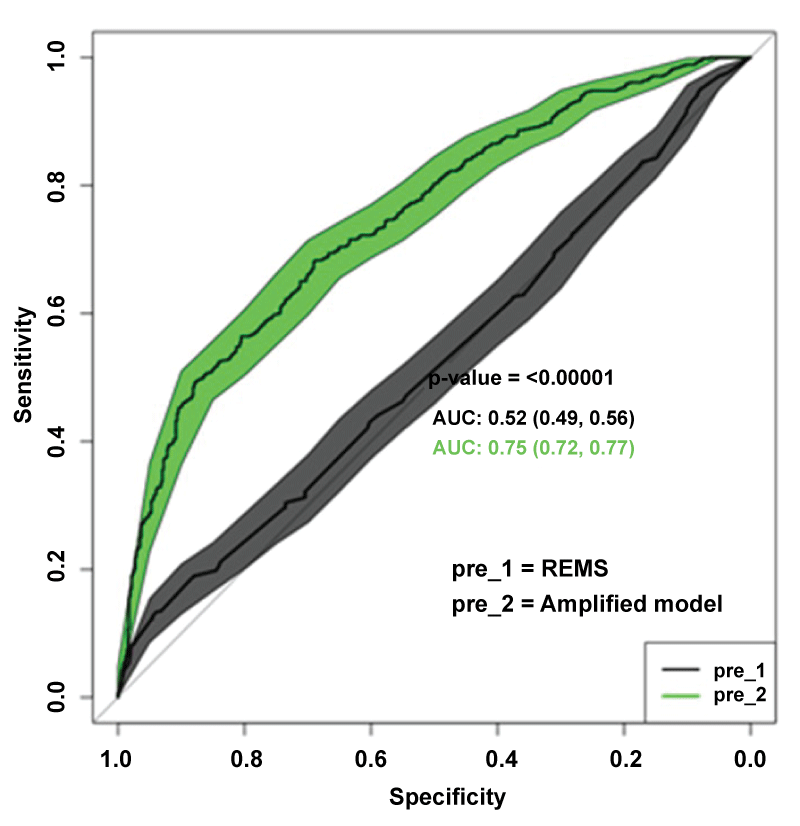
.
Figure 1: Curve ROC comparison predicting hospital mortality between REMS vs amplified model with REMS, Charlson comorbidity index, DM, dysglycemia, and full blood count disturbances.
View Figure 1
Discussion
In this study we have showed that the presence of dysglycemia (hyper or hypoglycemia) on admission in the ED is a marker for increased risk of hospital mortality. It improves the discriminative ability of a clinical validated model (REMS) and of an expanded model with clinical and analytical variables. However, this prognostic marker on arrival to the ED is most significant, clinically and statistically, in those patients without a history of DM. Additionally, we showed that a model with inclusion of clinical and analytical variables (REMS, Charlson comorbidity index, dysglycemia, full blood count, DM), improves the capacity to predict hospital mortality respect to REMS.
The first predictive mortality scales were used in medical and surgical patients within the ICU such as the APACHE [8] with its modifications over the years [9], which is based on clinical variables, age, previous general health and laboratory markers. Further on, other specific scales started to appear for trauma patients [10], for patients with sepsis (Mortality in Emergency Department Sepsis score) [11], acute coronary syndrome [12], asthma [13], and pneumonia [14]. Additionally, these tools expanded for the use in the ED such as the rapid acute physiology score (RAPS) [15] and REMS which improved the RAPS predictive capacity by incorporating oxygen saturation and age into the marking scheme [1]. For this reason we decided to use the REMS scoring scheme for our sample on arrival at the ED and hypothesized that the addition of accessible and rapid analytical tests, including glucose levels, would improve its discrimination capacity as did the APACHE IV scale with its predecessor APACHEII [3].
The onset of hyperglycemia in hospitalized patients is an indicator of a bad prognosis and has been associated to increase the mortality in critically ill patients [16], patients with cardiac ischemia [17], pneumonia [18], and stroke [19]. It is believed that this is due to altered immune, endothelial, and cardiac functions with levels above 200 mg/dl [20]. However, the fact that hyperglycemia is associated with a poorer prognosis in non-diabetic patients [4] in comparison with known diabetics, and the fact that intensive glucose control does not improve the mortality outcome in hospitalized patients [4,7] may indicate that the initial hyperglycemia in the ED is a marker of disease severity rather than a direct cause of mortality. In our study patients with hyperglycemia tended to have signs of more severe disease and the effect of the hyperglycemia was only significant in non-diabetic patients, which would support it as a marker of acute stress.
Similarly, conclusions of increased hospital mortality associated with hypoglycemia [21] have been reported, and there are plausible pathophysiological mechanisms that could explain this relationship [22]. Nevertheless, this effect is less marked in insulin-induced hypoglycemia [5,23] than in spontaneous hypoglycemia, which would again indicate it as a marker of disease severity. In our study, as in others [23], patients with hypoglycemia had worse Charlson scores and an increased risk in mortality. The absence of an independent effect in multivariate analysis and the effect more marked in patients without known DM, would strengthen the role of hypoglycemia as a marker of global health.
Given the fact that both hyper and hypoglycemia have shown in other case-control studies to increase hospital mortality [24] as well as in our own, we evaluated whether this effect exists with the combined effect of dysglycemia i.e. to include both hyper and hypoglycemia. We found that the dysglycemia was included in the best predictive model made up of clinical and analytical variables, which improved significantly the discrimination capacity in predicting hospital mortality.
The strengths of our study were the inclusion of an ample sample of deceased hospital patients, age-matching patients to eliminate confounding factors, and the addition of analytical tests to predictive mortality scales. Weaknesses include the retrospective and observational nature of the study, which may limit the possibility to reach causal inferences. Additionally, the limited number of patients with hypoglycemia on admission, and the lack of other variable may have dampened the predictive importance of hyperglycemia, such as C-reactive protein. Finally, data about the influence of body mass index or previous impaired fasting glucose are not available.
In conclusion, the addition of the Charlson comorbidity index and analytical variables to the REMS score, improves the predictive capacity of hospital mortality. Among the included variables we stress the importance of glucose levels on admission in the ED due to its easy and rapid access which when incorporated into mortality prediction scales improves their discrimination capacity, especially in non-diabetic patients.
References
-
Olsson T, Terent A, Lind L (2004) Rapid Emergency Medicine score: a new prognostic tool for in-hospital mortality in nonsurgical emergency department patients. J Intern Med 255: 579-587.
-
Bulut M, Cebicci H, Sigirli D, Sak A, Durmus O, et al. (2014) The comparison of modified early warning score with rapid emergency medicine score: a prospective multicentre observational cohort study on medical and surgical patients presenting to emergency department. Emerg Med J 31: 476-481.
-
Sánchez-Casado M, Hostiguela-Martín VA, Raigal-Cano A, Labajo L, Gomez-Tello V, et al. (2015) Predictive scoring systems in multiorgan failure: A cohort study. Med Intensiva.
-
Umpierrez GE, Isaacs SD, Bazargan N, You X, Thaler LM, et al. (2002) Hyperglycemia: an independent marker of in-hospital mortality in patients with undiagnosed diabetes. J Clin Endocrinol Metab 87: 978-982.
-
Kosiborod M, Inzucchi SE, Goyal A, Krumholz HM, Masoudi FA, et al. (2009) Relationship between spontaneous and iatrogenic hypoglycemia and mortality in patients hospitalized with acute myocardial infarction. JAMA 301: 1556-1564.
-
Kansagara D, Fu R, Freeman M, Wolf F, Helfand M (2011) Intensive insulin therapy in hospitalized patients: a systematic review. Ann Intern Med 154: 268-282.
-
Murad MH, Coburn JA, Coto-Yglesias F, Dzyubak S, Hazem A, et al. (2012) Glycemic control in non-critically ill hospitalized patients: a systematic review and meta-analysis. J Clin Endocrinol Metab 97: 49-58.
-
Knaus WA, Zimmerman JE, Wagner DP, Draper EA, Lawrence DE (1981) APACHE-acute physiology and chronic health evaluation: a physiologically based classification system. Crit Care Med 9: 591-597.
-
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13: 818-829.
-
Champion HR, Sacco WJ, Copes WS, Gann DS, Gennarelli TA, et al. (1989) A revision of the Trauma Score. J Trauma 29: 623-629.
-
Shapiro NI, Wolfe RE, Moore RB, Smith E, Burdick E, et al. (2003) Mortality in Emergency Department Sepsis (MEDS) score: a prospectively derived and validated clinical prediction rule. Crit Care Med 31: 670-675.
-
Antman EM, Cohen M, Bernink PJ, McCabe CH, Horacek T, et al. (2000) The TIMI risk score for unstable angina/non-ST elevation MI: A method for prognostication and therapeutic decision making. JAMA 284: 835-842.
-
Rodrigo G, Rodrigo C (1997) A new index for early prediction of hospitalization in patients with acute asthma. Am J Emerg Med 15: 8-13.
-
Fine MJ, Auble TE, Yealy DM, Hanusa BH, Weissfeld LA, et al. (1997) A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med 336: 243-250.
-
Rhee KJ, Fisher CJ Jr, Willitis NH (1987) The Rapid Acute Physiology Score. Am J Emerg Med 5: 278-282.
-
Krinsley JS (2003) Association between hyperglycemia and increased hospital mortality in a heterogeneous population of critically ill patients. Mayo Clin Proc 78: 1471-1478.
-
Cabrerizo-García JL, Gimeno-Orna JA, Zalba-Etayo B, Pérez-Calvo JI (2011) Hyperglycaemia as bad prognostic factor in acute coronary syndrome. Rev Clin Esp 211: 275-282.
-
McAlister FA, Majumdar SR, Blitz S, Rowe BH, Romney J, et al. (2005) The relation between hyperglycemia and outcomes in 2,471 patients admitted to the hospital with community-acquired pneumonia. Diabetes Care 28: 810-815.
-
Capes SE, Hunt D, Malmberg K, Pathak P, Gerstein HC (2001) Stress hyperglycemia and prognosis of stroke in nondiabetic and diabetic patients: a systematic overview. Stroke 32: 2426-2432.
-
Inzucchi SE, Rosenstock J (2005) Counterpoint: Inpatient glucose management: a premature call to arms? Diabetes Care 28: 976-979.
-
Turchin A, Matheny ME, Shubina M, Scanlon JV, Greenwood B, et al. (2009) Hypoglycemia and clinical outcomes in patients with diabetes hospitalized in the general ward. Diabetes Care 32: 1153-1157.
-
Desouza CV, Bolli GB, Fonseca V (2010) Hypoglycemia, diabetes, and cardiovascular events. Diabetes Care 33: 1389-1394.
-
Garg R, Hurwitz S, Turchin A, Trivedi A (2013) Hypoglycemia, with or without insulin therapy, is associated with increased mortality among hospitalized patients. Diabetes Care 36: 1107-1110.
-
Bruno A, Gregori D, Caropreso A, Lazzarato F, Petrinco M, et al. (2008) Normal glucose values are associated with a lower risk of mortality in hospitalized patients. Diabetes Care 31: 2209-2210.





