International Journal of Cancer and Clinical Research
Systematic Review and Meta-Analysis to Determine the Optimum Staging modality for Primary Breast Cancer
Berney M1, Manning A2, Carter M1, Sacchini V2 and Kell M R. Barry M1*
1Department of Breast Surgery, Mater Misericordiae University Hospital, Dublin 7, Ireland
2Breast Service, Memorial Sloan-Kettering Cancer Center, New York, USA
*Corresponding author:
Mitchel Barry, Breast Service, Mater Misericordiae University Hospital, Dublin 7, Ireland, Tel: 00353-87-9608910, E-mail: mbarry@mater.ie
Int J Cancer Clin Res, IJCCR-2-026, (Volume 2, Issue 4), Original Article; ISSN: 2378-3419
Received: July 13, 2015 | Accepted: September 22, 2015 | Published: September 24, 2015
Citation: Berney M, Manning A, Carter M, Sacchini V, Kell M R. Barry M (2015) Systematic Review and Meta-Analysis to Determine the Optimum Staging modality for Primary Breast Cancer. Int J Cancer Clin Res 2:026. 10.23937/2378-3419/2/4/1026
Copyright: © 2015 Berney M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Staging of primary breast cancer can radically alter the management if stage IV disease is accurately detected. A systematic review and meta-analysis were performed to determine whether conventional CT TAP or PET-CT provides the optimum staging modality. A meta-analysis involving 158 patients with primary breast cancer showed that PET-CT was more sensitive than CT-TAP for detection of metastatic disease (OR 3.28, 95% CI 2.37 - 4.53, p < 0.0001). A systematic review of 1780 patients undergoing PET-CT was compared with 709 patients having a CT TAP for staging. The sensitivity/specificity of PET-CT was 75-100%/91-98%. Sensitivity/specificity of CT TAP was 83-97.7%/85-100%. PET-CT is therefore the optimum staging imaging modality to detect metastatic breast cancer. Large multicentre randomized controlled trials comparing PET-CT with conventional CT TAP are required to support the findings of this study. However, in the absence of such robust data, clinicians may be guided by the results of this analysis.
Introduction
Breast cancer is the leading cause of cancer death in women in Western countries [1]. For patients with primary breast cancer, preoperative staging is used to determine the most appropriate disease management for each patient and provides important prognostic information. The presence of distant metastatic disease is a major prognostic factor since women with metastases have a 5-year survival rate of approximately 25% compared with approximately 80% survival rate for women with localized disease [2].
ESMO guidelines for staging recommend chest radiograph with abdominal ultrasound or CT and bone scan for the detection of metastatic disease in patients with clinically positive axillary nodes, large tumours (eg. > 5cm), or clinical signs, symptoms or laboratory values suggesting the presence of metastatic disease [3].
In recent years, more advanced investigative modalities have been used for breast cancer staging including computed tomography (CT), positron emission tomography (PET), and combined PET-CT. These modalities have been shown to improve the accurate staging and management of patients with advanced disease when compared to traditional modalities (eg. chest radiograph, ultrasound, bone scintigraphy) [4,5]. While the indications for advanced imaging have been enumerated in the NCCN and ESMO guidelines, there is still no clear consensus about which modality is the most appropriate investigation for patients with suspected metastatic disease.
Since appropriate staging can have a significant impact on patient management, it is important that the correct modality is selected to optimize treatment. This meta-analysis and systematic review was performed to determine whether conventional CT TAP or PET-CT is the optimum staging technique for patients with primary breast cancer.
Methods
This meta-analysis was conducted according to the PRISMA guidelines for meta-analysis reporting (2009) [4]. An electronic search using the Medline database was performed. Studies comparing PET-CT with CT TAP in the staging of breast cancer were identified and a meta-analysis was conducted of the pooled trial data to determine the optimum staging modality. A systematic review of trials evaluating either PET-CT or CT TAP was also selected for analysis. Studies were excluded based on the following criteria: non-English language, patients with recurrent breast cancer, and patients with inflammatory breast cancer.
Investigator (M.B) performed a systematic literature search of the Medline database to identify relevant studies, using the following combination of search terms; breast cancer, distant metastases OR staging, CT OR computed tomography, PET-CT OR positron emission tomography. Data was extracted from the studies, including authors, year of publication, age, sample size, study design (prospective or retrospective), sensitivity and specificity for each modality.
General variance methods were used to combine data across studies with fixed and random effects models. The fixed effects analysis weighted the natural logarithm of each study's OR by the inverse of its variance plus an estimate for the between-study variance in the presence of between-study heterogeneity. Analyses were conducted using Stats direct version 2.5.6 (Stats Direct Limited, Cheshire, UK) and SPSS version 12.0 (SPSS, Inc., Chicago, IL). All statistical tests were two-tailed.
Results
Three trials were identified which directly compared the use of PET-CT and CT-TAP in breast cancer patients undergoing staging for metastatic disease (Table 1). Both imaging modalities were used in 158 patients. The sensitivity and specificity of PET-CT were superior to that of CT-TAP for the detection of distant metastases in these patients (sensitivity 91 - 97.8%, specificity 91-93.5% for PET-CT versus sensitivity 77-87.6%, specificity 42-93% for CT-TAP). Overall the meta-analysis showed that PET-CT was more sensitive than CT-TAP in the detection of metastatic disease (OR 3.28, 95% CI 2.37-4.53, p < 0.0001) (Figure 1).
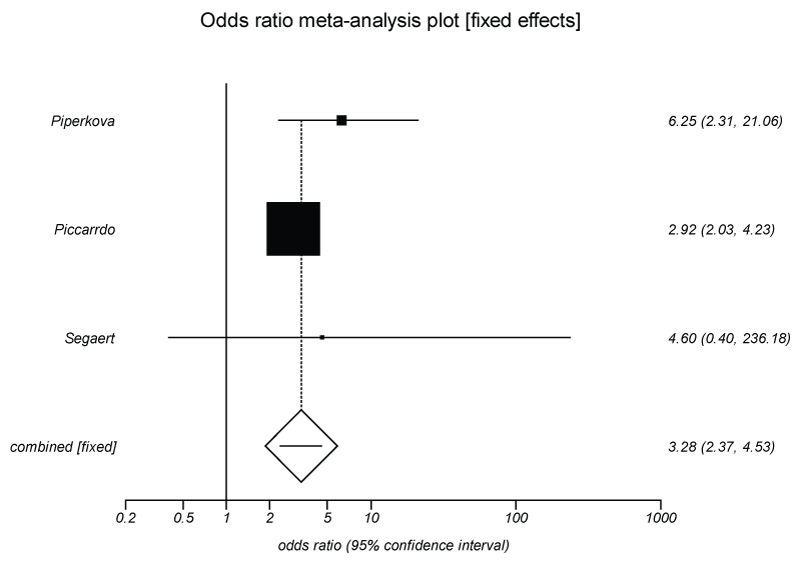
.
Figure 1: Forest box plot comparing PET/CT and CT TAP sensitivity for the staging of breast cancer.
View Figure 1
![]()
Table 1: Studies that compared PET-CT and CT-TAP in the detection of metastatic disease in breast cancer patients
View Table 1
The systematic review examined the role of either PET-CT or CT TAP in the diagnosis of metastatic breast cancer. Eleven studies in which 1710breast cancer patients underwent PET-CT and 3 studies in which 619 patients underwent CT-TAP were selected (Table 2 and Table 3). These studies involved comparisons with conventional imaging techniques such as chest x-ray, liver ultrasound and bone scan and included breast cancer patients with both early and advanced disease. The sensitivity of PET-CT was 75-100% and specificity was 91-98%. Sensitivity of CT TAP was 83-97.7% and specificity was 85-100%.
![]()
Table 2: Studies comparing PET-CT to conventional imaging for detection of metastatic breast cancer.
View Table 2
![]()
Table 3: Studies comparing CT-TAP to conventional imaging for detection of metastatic disease in breast cancer patients.
View Table 3
Discussion
In the meta-analysis, the PET-CT is clearly shown to be the superior imaging modality (OR 3.28, 95% CI 2.37-4.53, p < 0.0001) (Figure 1). The systematic review confirmed that PET-CT is superior to conventional imaging (Table 2). However the systematic review failed to demonstrate any significant difference between PET-CT and CT TAP in terms of sensitivity/specificity however there was significant heterogeneity across the studies involved regarding patient selection and disease stage. This may explain the discordance in findings between the meta-analysis and the systematic review.
Although many studies have compared both PET-CT and CT-TAP to conventional imaging, few have compared these modalities with each other and it is not surprising that such variety exists. The lack of consensus on which staging investigation(s) should be performed at such a critical point in the patient's care is likely a reflection of the lack of available evidence.
Indications for imaging and stage of disease differed between studies in this meta-analysis; Piperkova et al, in a retrospective review, reported on 49 patients who underwent imaging for staging, restaging or assessment of treatment response [5]. Piccardo reported prospectively on 39 patients with stage I-IV breast cancer who were referred for skeletal survey due to symptoms suggestive of, or with known bone metastases [6]. Finally Segaert et al. reported retrospectively on 70 newly diagnosed breast cancer patients with stage IIB and III disease [7]. Despite such variety between studies the question of which imaging modality can more reliably detect metastatic disease applied similarly to each study group and it is unlikely that this would be affected by individual patient or tumour factors. This meta-analysis supports the conclusions found in each of the papers individually; that PET-CT is superior to CT alone in staging breast cancer. This meta-analysis then provides valuable information for decision-makers.
In the systematic literature review there were 11 studies comparing PET-CT to conventional imaging and all demonstrated that PET-CT was superior in the detection of metastatic disease. Koolen et al. report a sensitivity of 100% as none of 129 patients with negative scans were subsequently shown to have metastatic disease [8]. The lowest sensitivity of 75% reported by Riegger et al. was still significantly better than the sensitivity of 50% achieved with conventional imaging in that series [9]. Niikura and colleagues report a sensitivity and specificity of 97.4% and 91.2% respectively for PET-CT, compared to 85.9% and 67.3% with conventional imaging which included CT TAP in some but not all patients [10]. A direct comparison between PET-CT and abdominal CT (performed in 84% of patients) for the detection of liver metastases in that study revealed that all 20 cases were detected by both modalities [10]. Choi et al reported sensitivity and specificity of 100% and 96.4% respectively for PET-CT, compared to 61.5% and 99.2% with conventional imaging for detection of distant metastases [11].
The most common site for metastases to occur is bone and the primary diagnostic tool for diagnosis of bone metastases is bone scintigraphy. It has been reported that PET and PET-CT are superior to bone scintigraphy for detection of bone metastases [3,12]. Koolen et al report that bone scintigraphy showed suspicious lesions in 20 patients, however following further investigations only 4 were confirmed as metastatic disease (16 false positives) and 3 more patients were found to have bone metastases not detected with bone scintigraphy [8]. PET-CT detected metastases in all 7 patients with only 2 additional false positives. Koolen et al reported that PET-CT was superior for both sclerotic and lytic lesions [8]. In the study by Fuster et al it was reported that bone scintigraphy identified only 2 of 6 bone metastases with 4 false negative lytic lesions [13]. PET-CT identified all metastatic bone lesions in that series. Niikura et al. report a sensitivity and specificity of 98% and 96% respectively for PET-CT compared to 76% and 86% for bone scintigraphy for detecting bone metastases in 56 of 225 patients [10]. It is suggested that PET-CT could replace bone scintigraphy as the initial imaging modality for detection of bone metastases in the staging of newly diagnosed breast cancer and the findings of this systematic review add further support to this.
The prognostic impact of PET-CT findings in patients with stage II and stage III disease was assessed by Groheuxet al. [14]. Clinical stage was changed in 77 of 254 patients based on PET-CT findings with increasingly higher percentage change seen from stage IIA to IIIC [14]. Only 2.3% of stage IIA patients were upstaged based on PET-CT, 10.7% of stage IIB, increasing to 47.1% of patients with stage IIIC disease. Tanaka et al. examined the use of contrast enhanced CT in clinical staging of asymptomatic breast cancer patients with stage I, II and III disease [15]. Although 65 of 483 patients had abnormal CT results, only 26 patients (5.4%) were found to have true metastatic disease. Similar to the study by Groheux et al., the impact of CT differed significantly with clinical stage. None of 155 patients with stage I disease were upstaged based on CT TAP, 5 of 261 patients (1.9%) with stage II disease and 21 of 67 (31.3%) of stage III patients were upstaged. Groheux et al. suggest commencement of staging in patients with stage IIB disease or higher, which is not very different from the current NCCN guidelines which suggest staging at stage IIIA and higher [14,16]. Similarly, Gunalp et al. assessed the clinical impact of PET-CT on initial staging of 141 patients where PET-CT modified staging for 26% of stage I patients, 29% of stage IIA patients, 46% of stage IIB patients, 58% of stage IIIA patients and 100% of stage IIIB patients [17]. Garami et al. also found upstaging of TNM classification in a significant number of patients compared to conventional imaging (54 of 115 patients, 47%) [18].
Incorrect diagnosis of stage IV disease based on false positive staging may result in a change from curative to palliative intent, ultimately affecting patients' survival and the importance of confirming metastatic disease, histologically where possible, is emphasized. Increased costs associated with PET-CT may be reduced by eliminating other unnecessary staging investigations and providing more efficient cost-effective interventions for metastatic disease detected at an earlier stage. This meta-analysis and literature review suggests that PET-CT is preferable to CT-TAP as the imaging modality of choice for breast cancer staging for detection of metastatic disease. This was the conclusion seen in each of the studies in the meta-analysis. However, in the absence of large multicentre randomized controlled trials comparing PET-CT with conventional CT TAP, this study provides clinicians with up-to-date analysis of the literature comparing PET-CT and CT TAP for staging of breast cancer.
References
-
American Cancer Society (2007) Cancer Facts & Figures 2007. Atlanta GA: American Cancer Society.
-
Ries LAG, Harkins D, Krapcho M, Mariotto A, Miller BA, et al. (2006) SEER Cancer Statistics Review, 1975-2003, Bethesda, MD: National Cancer Institute.
-
Mahner S, Schirrmacher S, Brenner W, Jenicke L, Habermann CR, et al. (2008) Comparison between positron emission tomography using 2-[fluorine-18] fluoro-2-deoxy-D-glucose, conventional imaging and computed tomography for staging of breast cancer. Ann Oncol 19: 1249-1254.
-
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, et al. (2009) The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evalulate Health Care Interventions: Explanations and Elaboration. PLoS Med 6: 1-28.
-
Piperkova E, Raphael B, Altinyay ME, Castellon I, Libes R, et al. (2007) Impact of PET/CT in comparison with same day contrast enhanced CT in breast cancer management. Clin Nucl Med 32: 429-434.
-
Piccardo A, Altrinetti V, Bacigalupo L, Puntoni M, Biscaldi E, et al. (2012) Detection of metastatic bone lesions in breast cancer patients: fused (18)F-Fluoride-PET/MDCT has higher accuracy than MDCT. Preliminary experience. Eur J Radiol 81: 2632-2638.
-
Segaert I, Mottaghy F, Ceyssens S, De Wever W, Stroobants S, et al. (2010) Additional value of PET-CT in staging of clinical stage IIB and III breast cancer. Breast J 16: 617-624.
-
Koolen BB, Vrancken Peeters MJ, Aukema TS, Vogel WV, Oldenburg HS, et al. (2012) 18F-FDG PET/CT as a staging procedure in primary stage II and III breast cancer: comparison with conventional imaging techniques. Breast Cancer Res Treat 131: 117-126.
-
Riegger C, Herrmann J, Nagarajah J, Hecktor J, Kuemmel S, et al. (2012) Whole-body FDG PET/CT is more accurate than conventional imaging for staging primary breast cancer patients. Eur J Nucl Med Mol Imaging 39: 852-863.
-
Niikura N, Costelloe CM, Madewell JE, Hayashi N, Yu TK, et al. (2011) FDG-PET/CT compared with conventional imaging in the detection of distant metastases of primary breast cancer. Oncologist 16: 1111-1119.
-
Choi YJ, Shin YD, Kang YH, Lee MS, Lee MK, et al. (2012) The Effects of Preoperative (18)F-FDG PET/CT in Breast Cancer Patients in Comparison to the Conventional Imaging Study. J Breast Cancer 15: 441-448.
-
Morris PG, Lynch C, Feeney JN, Patil S, Howard J, et al. (2010) Integrate positron emission tomography/computed tomography may render bone scintigraphy unnecessary to investigate suspected metastatic breast cancer. J clinOncol 28: 3154-3159.
-
Fuster D, Duch J, Paredes P, Velasco M, Muñoz M, et al. (2008) Preoperative staging of large primary breast cancer with [18F]fluorodeoxyglucose positron emission tomography/computed tomography compared with conventional imaging procedures. J Clin Oncol 26: 4746-4751.
-
Groheux D, Hindié E, Delord M, Giacchetti S, Hamy AS, et al. (2012) Prognostic impact of (18)FDG-PET-CT findings in clinical stage III and IIB breast cancer. J Natl Cancer Inst 104: 1879-1887.
-
Tanaka S, Sato N, Fujioka H, Takahashi Y, Kimura K, et al. (2012) Use of contrast-enhanced computed tomography in clinical staging of asymptomatic breast cancer patients to detect asymptomatic distant metastases. Oncol Lett 3: 772-776.
-
Aebi S, Davidson T, Gruber G, Cardoso F; ESMO Guidelines Working Group (2011) Primary breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 22 Suppl 6: vi12-24.
-
Gunalp B, Ince S, Karacalioglu AO, Ayan A, Emer O, et al. (2012) Clinical impact of (18)F-FDG PET/CT on initial staging and therapy planning for breast cancer. Exp Ther Med 4: 693-698.
-
Garami Z, Hascsi Z, Varga J, Dinya T, Tanyi M, et al. (2012) The value of 18-FDG PET/CT in early stage breast cancer compared to traditional diagnostic modalities with an emphasis on changes in disease stage designation and treatment plan. EJSO 38: 31-37.





