International Journal of Cancer and Clinical Research
Feasibility and Efficacy of Definitive Hypofractionated High-Dose Radiotherapy for Cutaneous Angiosarcoma of the Scalp
Emiko Shimoda1, Kazuya Inoue1, Nobuhide Wakai1, Yoko Morimoto1, Isao Asakawa1, Nobumasa Fujitani1,2, Tadashi Yoshimine1,2, Tetsuro Tamamoto1, Maiko Takeda3, Kohei Ogawa4, Hideo Asada4 and Masatoshi Hasegawa1*
1Department of Radiation Oncology, Nara Medical University, Kashihara, Japan
2Central Division of Radiology, Nara Medical University Hospital, Kashihara, Japan
3Department of Diagnostic Pathology, Nara Medical University, Kashihara, Japan
4Department of Dermatology, Nara Medical University, Kashihara, Japan
*Corresponding author:
Masatoshi Hasegawa, M.D., Ph.D., Department of Radiation Oncology, Nara Medical University, 840 Shijo-cho Kashihara, Nara 634-8522, Japan, Tel: +81-744-29-8908, Fax: +81-744-25-3434, E-mail: hasegawa@naramed-u.ac.jp
Int J Cancer Clin Res, IJCCR-2-032, (Volume 2, Issue 5), Original Article; ISSN: 2378-3419
Received: October 02, 2015 | Accepted: October 31, 2015 | Published: November 03, 2015
Citation: Shimoda E, Inoue K, Wakai N, Morimoto Y, Asakawa I, et al. (2015) Feasibility and Efficacy of Definitive Hypofractionated High-Dose Radiotherapy for Cutaneous Angiosarcoma of the Scalp. Int J Cancer Clin Res 2:032. 10.23937/2378-3419/2/5/1032
Copyright: © 2015 Shimoda E, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Cutaneous angiosarcoma is a rare but highly aggressive vascular tumor resistant to all treatment modalities available. The aim of this study was to analyze the treatment outcomes of patients who received definitive hypofractionated high-dose radiotherapy (RT) for angiosarcoma of the scalp. Between April 2008 and December 2014, 11 patients with histologically proven cutaneous angiosarcoma of the scalp visited our Department of Radiation Oncology, because dermatologists suggested that there was no indication for surgery in those cases. One patient rejected all radical treatments and the other 10 patients were treated by RT with curative intent along with chemotherapy or immunotherapy. Eight patients were treated with 6 - 12 MeV electron beams and the other 2 patients were treated with 4 MV X-ray Intensity Modulated Radiation Therapy (IMRT) and electron beams. The total irradiated dose was 63 - 75 Gy (median: 72.5 Gy) in 26 - 30 fractions, and the fraction size was 2.5 Gy in principle. The median age of the patients treated with RT was 80 years old (range: 73 - 91) and the median follow-up time was 16.5 months (range: 5.6 - 86.3). Four patients are still alive. A complete response (CR) was achieved in 10 patients (100%) and only one patient suffered local relapse 20 months after RT. Medians of overall survival (OS), progression-free survival (PFS), and local relapse-free survival (LRFS) were 38.7, 13.4, and 19.8 months, respectively. Local control rates were 100 and 75% at 1 and 2 years, respectively. Skin ulceration was CTCAE grade 2 in 5 patients (50%) and grade 3 in 5 (50%), alopecia was grade 2 in all patients (100%), but no patient developed grade 4 or more severe adverse events after RT. Hypofractionated high-dose RT was feasible and achieved excellent local control of cutaneous angiosarcoma in the elderly patients.
Keywords
Cutaneous angiosarcoma, Definitive radiation therapy, Hypofractionated high-dose radiotherapy, Electron beam, Linear-quadratic model
Abbreviations
RT: Radiotherapy, VMAT: Volumetric Modulated Arc Therapy, IMRT: Intensity Modulated Radiation Therapy, ELF: Extended Local Field, LF: Local Field, GTV: Gross Tumor Volume, CTV: Clinical Target Volume, PTV: Planning Target Volume, OS: Overall Survival, PFS: Progression-Free Survival, LRFS: Local Relapse-Free Survival, FFLR: Freedom from Local Relapse, CTCAE: Common Toxicity Criteria for Adverse Events, LQ Model: Linear-Quadratic Model, BED: Biologically Effective Dose, Fr: Fractions, CR: Complete Response
Introduction
Cutaneous angiosarcoma of the scalp is a rare but highly aggressive malignant vascular tumor [1-3]. The majority of the previous papers reported that the probability of loco-regional control was very low, that of hematogenous metastasis was relatively high, and the 2- and 5 -year overall survival (OS) rates ranged from 10 to 40% [1-5]; however, recent papers reported that combined modality therapy, surgery followed by radiotherapy (RT), often resulted in a relatively more favorable outcome. The optimal treatment for this tumor has been suggested to be surgery followed by RT and chemotherapy, but local control and survival rates remain unsatisfactory, and so more effective treatment strategies have to be established [6-9]. It is sometimes difficult to remove the tumors completely by surgery because the majority of the patients are elderly and tumors frequently spread extensively. More effective RT for this tumor is expected, but the role of RT without surgery has not been established [4]. This study was performed to analyze the feasibility and efficacy of definitive hypofractionated high-dose RT without surgery for cutaneous angiosarcoma of the scalp.
Methods
Patients and tumors
Between April 2008 and December 2014, 11 patients with histologically proven cutaneous angiosarcoma of the scalp visited the Department of Radiation Oncology of our institution for RT, because dermatologists suggested that there was no indication for surgery in those cases. Patients' selection criteria are as follows: Patients with histologically proven cutaneous angiosarcoma of the scalp suggested to be inappropriate for surgery due to "large size (10 cm or more in diameter)" or "80 years of age or older". These patients were more than 70 years old and had no distant metastasis. One of them rejected all radical treatments after obtaining sufficient information from dermatologists and radiation oncologists, and the other 10 patients were treated by RT with curative intent with chemotherapy or interleukin-2 immunotherapy.
Each patient gave written informed consent after receiving in-depth explanations, including information on the radiation exposure from RT and adverse events associated with the treatment, especially the risk of severe skin ulceration and alopecia by high-dose RT.
Tumor types in this study were classified as "infiltrative" or "nodular" according to the macroscopic findings because the RT methods had to be changed depending on the type and thickness of tumor. Tumors showing nodular shape or evident swelling are defined as "nodular" and tumors growing infiltratively without evident swelling are defined as "infiltrative".
In the present study, data from these 10 patients treated by RT were analyzed.
Treatments
Eight patients were treated with electron beams using a linear accelerator (Primus KD2; Siemens, Munich, Germany). The other two patients were treated with 4 MV X-rays (TrueBeam STx; Varian, Palo Alto, U.S.A.) and electron beams (Trilogy; Varian, Palo Alto, U.S.A.). The energy of the electron beams (6, 9, or 12 MeV) and the thickness of the bolus (5 or 10 mm) were decided by radiation oncologists considering the thickness of each tumor estimated by CT or MRI. Volumetric modulated arc therapy (VMAT), a type of intensity modulated radiation therapy (IMRT), was utilized for the latter two patients considering the dose distribution of X-rays.
Wide-field RT, total scalp irradiation or extended local field (ELF) irradiation, was performed for patients with extensive disease according to the target volume of RT. The clinical target volume (CTV)-1 or planning target volume (PTV)-1 for ELF irradiation was defined as the gross tumor volume (GTV) plus 50 mm or greater margins; however, margins in some cases were partially smaller than 50 mm but greater than 30 mm considering the severe adverse effects on high-risk organs like the eyes. CTV-2 or PTV-2 for local field (LF) irradiation was defined as GTV plus margins of 30 mm or smaller.
The fraction size of RT was 2.5 Gy and the dose for ELF irradiation (PTV-1) was at least 55 Gy in principle, being followed by LF irradiation (PTV-2) as a boost. The total dose, PTV-1 and PTV-2, for GTV was decided by the radiation oncologists on evaluating the tumor response to irradiation and acute adverse events in each case. Chemotherapy or immunotherapy was performed by the dermatologists.
Clinical response and toxicity evaluation
Overall survival (OS), progression-free survival (PFS), local relapse-free survival (LRFS), and freedom from local relapse (FFLR) of the 10 patients treated by RT were estimated from the day RT started using the Kaplan-Meier method (StatMate version 4.0: ATMS, Tokyo, Japan). OS is defined as the length of time from the first day of RT to death from any cause. Patients lost to follow-up were assumed to have died for survival estimation. Local relapse was defined as recurrence within the radiation field. Toxicities following RT were graded using the Common Toxicity Criteria for Adverse Events (CTCAE) v.4.03 (Table 1) [10].
![]()
Table 1: Common Terminology Criteria for Adverse Events (CTCAE) v4.03
View Table 1
The biologically effective dose (BED) in linear-quadratic (LQ) model was utilized to estimate the altered fractionation schedules compared with the conventional schedules [11,12]. BED-2, BED-3, and BED-10 were calculated by the LQ model using assumed alpha/beta ratios of 2, 3, and 10, respectively.
Results
The patient characteristics and treatments of the 10 patients treated with radiation in this series are summarized in tables 2 and 3, respectively. The median age was 80 years old (range: 73 - 91). No patients had distant metastases and all were treated by RT with a curative intent with chemotherapy or immunotherapy.
![]()
Table 2: Patient and tumor characteristics
View Table 2
![]()
Table 3: Treatment methods and BED
View Table 3
The median follow-up time was 16.5 months (range 5.6 - 86.3). Two patients lost to follow-up after admission to a hospice were recorded as deaths for survival estimation.
In the eight patients treated with electron beams, the number of beam ports to cover the wide-field, total scalp or ELF irradiation (PTV-1), ranged from 1 to 4. Three of them were total scalp irradiation performed by 3 or 4 field ports, and the other five were smaller than the total scalp and performed by 1 or 2 beam ports.
The other two patients were treated with 4 MV X-rays and electron beams. The VMAT by X-rays was utilized for total scalp or ELF irradiation (PTV-1), and then the electron beams were used for LF irradiation (PTV-2) as a boost.
In principle, 55 Gy in 22 fractions (fr) -65 Gy in 26 fr of ELF irradiation and 5 Gy in 2 fr-12.5 Gy in 5 fr of LF irradiation as a boost were performed for each case (Table 3). The total dose was 63-75 Gy in 26-30 fr (median 72.5 Gy in 29 fr). Calculated BED-2, BED-3, and BED-10 ranged from 169 to 78 Gy according to the fractionated radiation schedules and assumed alpha/beta ratios.
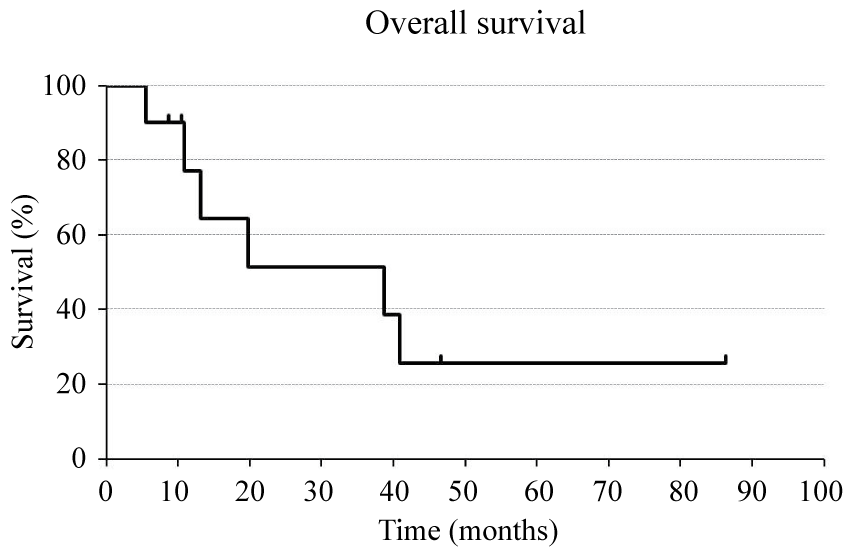
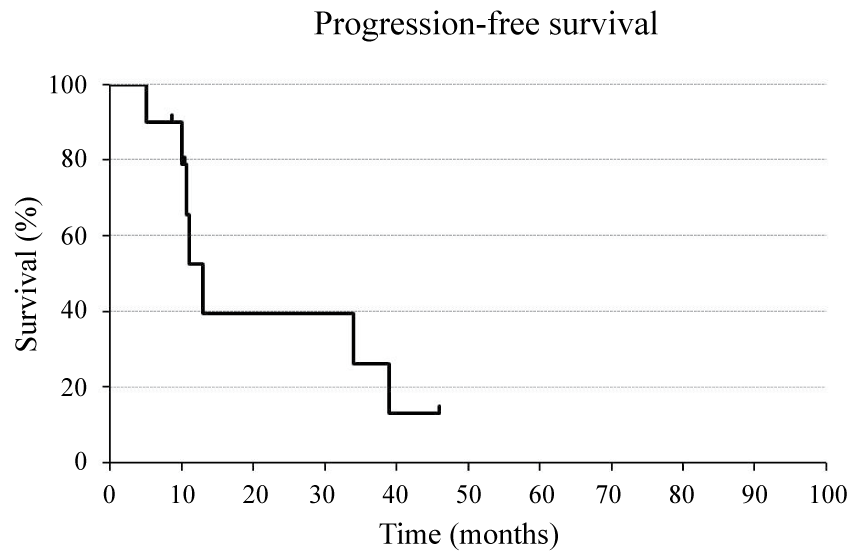
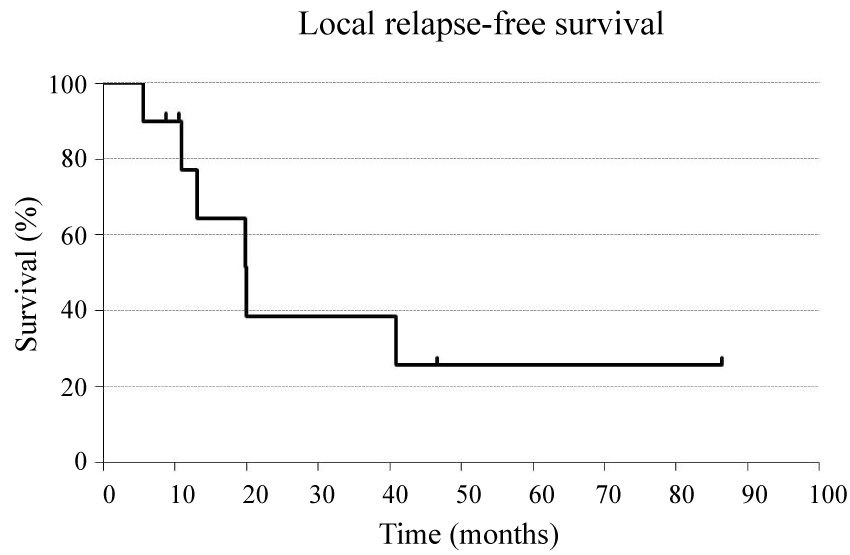
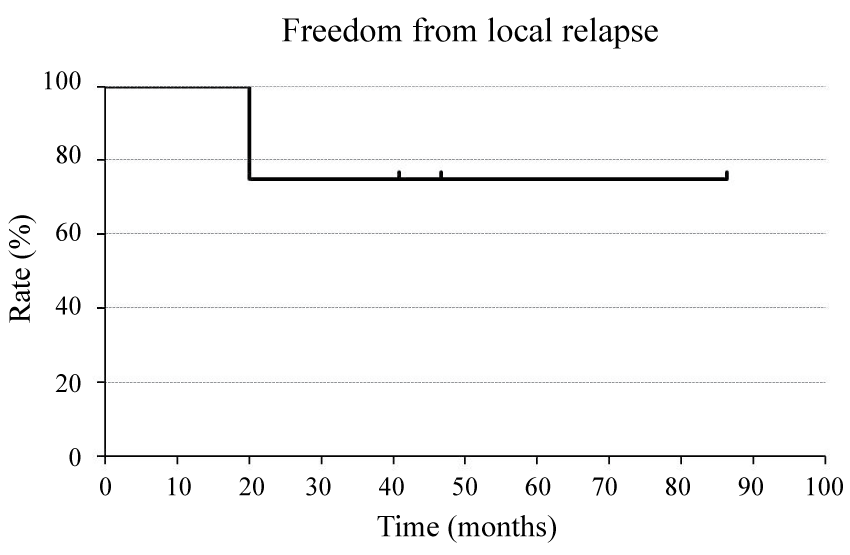
A complete response (CR) was achieved in all patients (100%) and no local relapse was found in the 9 patients during the follow-up. Only 1 patient suffered local relapse 20 months after RT. The radiation dose for the patient was 63 Gy in 26 fr, being the lowest in the series, and the calculated BED-2, BED-3, and BED-10 were 140, 114, and 78 Gy, respectively. In contrast, the highest dose irradiated for 3 patients was 75 Gy in 30 fractions and the calculated BED-2, BED-3, and BED-10 were 169, 138, and 94 Gy, respectively. Metastases were found in 7 patients after the RT and the lung was the most common site (n = 3). The other sites were the cervical lymph nodes, bone, brain, leg, and liver. One patient suffered both distant metastasis and local relapse (Table 4). One patient died of gastric cancer 19.8 months after the RT.
![]()
Table 4: Tumor responses and adverse events
View Table 4
Four patients were still alive without local relapse at 8.7 - 86.3 months and three of them remained disease-free at the last check-up. The patient surviving for 86.3 months suffered brain metastases that were treated by stereotactic radiosurgery using 6 MV X-rays (Novalis; Brainlab, Feldkirchen, Germany).
Kaplan-Meier survival curves are shown in figure 1, figure 2 and figure 3. Medians of OS, PFS, and LRFS were 38.7, 13.4, and 19.8 months, respectively. FFLR are shown in figure 4 and local control rates were 100 and 75% at 1 and 2 years, respectively.
In the skin and subcutaneous tissue of the irradiated scalp, skin ulceration was CTCAE grade 2 in 5 patients (50%) and grade 3 in 5 (50%), and alopecia was grade 2 in all patients (100%), but no patient developed grade 4 or more severe adverse events after RT. It took a few weeks or months for the skin ulcer to heal. No other adverse events of grade 2 or more severe have been observed in the scalp thus far.
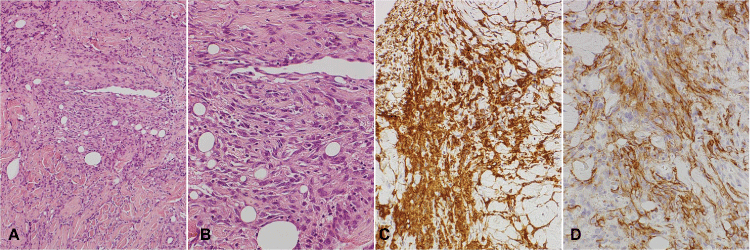
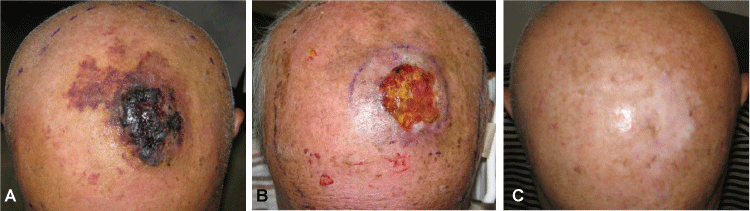
The microscopic and macroscopic features of case No. 4 are shown in figures 5 and figure 6, respectively. Atypical or spindle-shaped tumor cells in the biopsy specimen are CD31- and CD34-positive. Disappearance of the tumor and healing of the skin ulcer was observed following RT.
.
Figure 5: Microscopic features of angiosarcoma (case No. 4). (A) Hematoxylin and eosin staining (original magnification X40). (B) Hematoxylin and eosin staining (original magnification X100). (C) Immunohistochemical staining for CD31 (original magnification X100). (D) Immunohistochemical staining for CD34 (original magnification X100).
View Figure 5
.
Figure 6: Macroscopic appearance of angiosarcoma (case No. 4). (A) Angiosarcoma before treatment. (B) Skin ulcer after radiotherapy (70 Gy/28 fractions). (C) Disappearance of the tumor and healing of the skin ulcer 5 years after treatment.
View Figure 6
Discussion
Cutaneous angiosarcoma of the scalp is a highly aggressive and radio-resistant vascular tumor and the 5-year local-regional control rates by RT have been reported to be less than 50% [4,5]. Optimal treatments with RT for the tumor have not been established. The efficacy of RT is limited and the local control rate by RT alone was suggested to be much lower than that by surgical resection followed by RT [3-9]. Morrison WH et al. reported that the actuarial 5-year control rates for patients irradiated with and without clinical disease were 24 and 40%, respectively and the 5-year actuarial survival rates for patients irradiated with and without clinical disease were 13 and 50%, respectively [3]. In another report, the median survival time for 21 patients treated with radiation without surgery was 8 months [13]. Ohguri T, et al. reported that fifteen cases were treated with curative RT and recombinant interleukin-2 (rIL-2) but without surgery, and the median overall (disease-specific) and local recurrence-free survival were 26.1 and 12.7 months, respectively [14]. Compared with these reports, the present study suggests that the hypofractionated high-dose RT might become a candidate as an optimal RT for inoperable cases. Furthermore, efficacy of anti-mitotic taxane drug, anti-angiogenesis drug, and immunotherapy for angiosarcoma has been suggested [13-15].
In the present study, all patients treated with RT without resection showed a favorable initial response and achieved CR, but only the first patient had local recurrence. He had been treated with the lowest dose in this series. Therefore, we would recommend 70 Gy in 28 fr (2.5 Gy per fr) or more as an optimal dose fractionation schedule to the tumor. This curative RT without surgery might be as effective as the surgery followed by RT even for resectable disease, but the number of the patients in this series was limited because of the rarity of the disease.
Alpha/beta ratios of the LQ model for most malignancies are generally higher than those of late responding normal tissues, but the ratios have been suggested to be relatively lower in some malignancies like prostate cancer and malignant melanoma [11,12]. If the ratios for angiosarcoma are as low as those for prostate cancer, hypofractionated RT might be more effective than conventionally fractionated RT, although the potential doubling time of angiosarcoma might not be as long as that of prostate cancer. If the alpha/beta ratio is 10.0, the BED of 70 Gy/28 fr is 87.5 Gy and that of 70 Gy/35 fr is 84 Gy; however, if the ratio is 2.0, the BED of 70 Gy/28 fr is 157.5 Gy and that of 70 Gy/35 fr is 140 Gy. The larger fraction size might contribute to the greater efficacy of RT for a tumor with a low alpha/beta ratio than that with a high ratio. Furthermore, this hypofractionated high-dose RT makes the overall treatment time of RT shorter because of the smaller fraction numbers, and this might be useful for suppressing tumor repopulation during the treatment period.
Conclusions
This study showed the feasibility and efficacy of hypofractionated high-dose RT for cutaneous angiosarcoma of the scalp. Grade 2 and 3 adverse events were acceptable and the local control was excellent; however, the number of the patients was limited due to the rarity of the tumor, and the effects of chemotherapy and immunotherapy had to be considered. A multi-institutional prospective study will be necessary to establish effective and feasible treatment for unresectable cutaneous angiosarcoma.
Ethical Statement
This study was approved by the institutional ethics committee of Nara Medical University.
References
-
Maddox JC, Evans HL (1981) Angiosarcoma of skin and soft tissue: a study of forty-four cases. Cancer 48: 1907-1921.
-
Holden CA, Spittle MF, Jones EW (1987) Angiosarcoma of the face and scalp, prognosis and treatment. Cancer 59: 1046-1057.
-
Morrison WH, Byers RM, Garden AS, Evans HL, Ang KK, et al. (1995) Cutaneous angiosarcoma of the head and neck. A therapeutic dilemma. Cancer 76: 319-327.
-
Mark RJ, Poen JC, Tran LM, Fu YS, Juillard GF (1996) Angiosarcoma. A report of 67 patients and a review of the literature. Cancer 77: 2400-2406.
-
Mendenhall WM, Mendenhall CM, Werning JW, Reith JD, Mendenhall NP (2006) Cutaneous angiosarcoma. Am J Clin Oncol 29: 524-528.
-
Guadagnolo BA, Zagars GK, Araujo D, Ravi V, Shellenberger TD, et al. (2011) Outcomes after definitive treatment for cutaneous angiosarcoma of the face and scalp. Head Neck 33: 661-667.
-
Ogawa K, Takahashi K, Asato Y, Yamamoto Y, Taira K, et al. (2012) Treatment and prognosis of angiosarcoma of the scalp and face: a retrospective analysis of 48 patients. Br J Radiol 85: e1127-1133.
-
Scott MT, Portnow LH, Morris CG, Marcus RB Jr, Mendenhall NP, et al. (2013) Radiation therapy for angiosarcoma: the 35-year University of Florida experience. Am J Clin Oncol 36: 174-180.
-
Mullins B, Hackman T1 (2015) Angiosarcoma of the Head and Neck. Int Arch Otorhinolaryngol 19: 191-195.
-
U.S. Department of Health and Human Services, National Institutes of Health, National Cancer Institute (2010) Skin and subcutaneous tissue disorders. Common Terminology Criteria for Adverse Events (CTCAE) 4: 179-187.
-
Fowler JF (1984) The first James Kirk memorial lecture. What next in fractionated radiotherapy? Br J Cancer Suppl 6: 285-300.
-
Fowler JF (2005) The radiobiology of prostate cancer including new aspects of fractionated radiotherapy. Acta Oncol 44: 265-276.
-
Sasaki R, Soejima T, Kishi K, Imajo Y, Hirota S, et al. (2002) Angiosarcoma treated with radiotherapy: impact of tumor type and size on outcome. Int J Radiat Oncol Biol Phys 52: 1032-1040.
-
Ohguri T, Imada H, Nomoto S, Yahara K, Hisaoka M, et al. (2005) Angiosarcoma of the scalp treated with curative radiotherapy plus recombinant interleukin-2 immunotherapy. Int J Radiat Oncol Biol Phys 61: 1446-1453.
-
Fuller CK, Charlson JA, Dankle SK, Russell TJ (2010) Dramatic improvement of inoperable angiosarcoma with combination paclitaxel and bevacizumab chemotherapy. J Am Acad Dermatol 63: e83-84.