International Journal of Cancer and Clinical Research
Trastuzumab Emtansine in Patients with HER2-Positive Metastatic Breast Cancer: a Multicenter Japanese Retrospective Study
Masako Sato1, Masato Takahashi1, Kenichi Watanabe1, Nobumoto Tomioka1, Mitsugu Yamamoto1, Yoshiaki Narita2, Hiroaki Kato2, Masaru Abe2, Yukiko Tabata2, Masaya Kawada3, Motoshi Tamura4, Mitsuchika Hosoda5, Motoi Baba5, Kazuhiro Ogasawara6, Tomohiro Oshino6, Hiromitsu Akabane7, Yasuhiro Suzuki8, Masatoshi Kadoya9, Kazuhiro Iwai10, Yumi Okawa11, Kenjiro Misu12, Kazuomi Ichinokawa13 and Hiroko Yamashita5*
1Department of Breast Surgery, NHO Hokkaido Cancer Center, Japan
2Department of Surgery, Teine Keijinkai Hospital, Japan
3Department of Surgery, Tonan Hospital, Japan
4Department of Surgery, KKR Sapporo Medical Center, Japan
5Department of Breast Surgery, Hokkaido University Hospital, Japan
6Department of Surgery, Kushiro Rosai Hospital, Japan
7Department of Surgery, Asahikawa-Kosei General Hospital, Japan
8Department of Surgery, Keiyukai Sapporo Hospital, Japan
9Department of Surgery, Ebetsu City Hospital, Japan
10Department of Surgery, Oji General Hospital, Japan
11Department of Surgery, Sapporo City General Hospital, Japan
12Department of Surgery, Kushiro Red Cross Hospital, Japan
13Department of Surgery, NTT East Sapporo Hospital, Japan
*Corresponding author:
Hiroko Yamashita, Department of Breast Surgery, Hokkaido University Hospital, Kita 14, Nishi 5, Kita-ku, Sapporo, 060-8648, Japan, Tel: +81-11-706-7381, Fax: +81-11-706-7384, E-mail: hirokoy@huhp.hokudai.ac.jp
Int J Cancer Clin Res, IJCCR-4-076, (Volume 4, Issue 1), Original Article; ISSN: 2378-3419
Received: December 23, 2016 | Accepted: January 26, 2017 | Published: January 30, 2017
Citation: Sato M, Takahashi M, Watanabe K, Tomioka N, Yamamoto M, et al. (2017) Trastuzumab Emtansine in Patients with HER2-Positive Metastatic Breast Cancer: a Multicenter Japanese Retrospective Study. Int J Cancer Clin Res 4:076. 10.23937/2378-3419/1410076
Copyright: © 2017 Sato M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Trastuzumab emtansine (T-DM1) is currently approved for the treatment of patients with HER2-positive metastatic breast cancer after failure of prior anti-HER2 therapies. However, the efficacy of T-DM1 in patients who received pertuzumab, and in those patients with brain metastases, is currently unclear.
Methods: A total of 63 women with HER2-positive metastatic breast cancer were treated with T-DM1 between April 2014 and April 2016. The efficacy and safety of T-DM1 therapy was investigated.
Results: There were 43 (67%) patients with visceral metastases and 14 (22%) patients with brain metastases. As adjuvant or neoadjuvant therapy in 45 patients with recurrent breast cancer, 19 (30%) patients had received trastuzumab. In the metastatic setting, all but two patients (97%) had undergone trastuzumab, 41 (65%) patients had received pertuzumab and 21 (33%) patients had received lapatinib prior to T-DM1. Patients had received a median of three regimens prior to T-DM1 for metastatic breast cancer. The response rate of patients on T-DM1 was 35%, the clinical benefit rate was 49%, and median time to treatment failure (TTF) was 4.0 months. In 41 patients pretreated with pertuzumab, the response rate was 29%, the clinical benefit rate was 46%, and median TTF was 5.0 months. In 14 patients with brain metastases, median TTF was 6.0 months, although none achieved CR or PR for brain tumors. The most commonly reported grade 3 or 4 adverse event was thrombocytopenia, which was experienced by 13 (21%) patients, although this was not associated with severe bleeding. Treatment termination was necessary in 5 (8%) patients because of side effects.
Conclusion: T-DM1 is an effective and well-tolerated treatment for patients with HER2-positive metastatic breast cancer that had progressed after previous anti-HER2 therapies. T-DM1 could be used for patients who have experienced progression following prior treatment with pertuzumab.
Keywords
Trastuzumab emtansine, Pertuzumab, Metastatic breast cancer, HER2-positive, Clinical experience
Abbreviations
T-DM1: Trastuzumab emtansine; HER2: Human epidermal growth factor receptor type 2; ER: Estrogen receptor; PgR: Progesterone receptor; CR: Complete response; PR: Partial response; SD: Stable disease; PD: Progressive disease; TTF: Time to treatment failure; HR: Hazard ratio
Introduction
Trastuzumab emtansine (T-DM1) is an antibody-drug conjugate incorporating the HER2-targeting antitumor properties of trastuzumab with the cytotoxic activity of the microtubule-inhibitory agent, DM1 [1,2]. Trastuzumab and DM1 are conjugated via a stable linker. T-DM1is the most recently approved (2014) anti-HER2 agent in Japan, and has been used for the treatment of HER2-positive metastatic breast cancer after failure of prior anti-HER2 therapies.
Results from phase III studies have confirmed the efficacy and safety of T-DM1 in patients with HER2-positive metastatic breast cancer [3,4]. The pivotal "EMILIA" study was a phase III randomized international trial that evaluated the efficacy and safety of T-DM1 compared to the combination of lapatinib plus capecitabine in patients with metastatic or locally advanced HER2-positive breast cancer, these patients had been previously treated with trastuzumab and taxanes [3]. A total of 991 patients participated, and median progression-free survival (PFS) was 9.6 months with T-DM1 vs. 6.4 months with lapatinib plus capecitabine (hazard ratio (HR) = 0.65, 95% confidence interval (CI), 0.55 to 0.77; P < 0.001). Overall survival (OS) was also significantly longer in patients treated with T-DM1 (median OS was 30.9 months with T-DM1 vs. 25.1 months with lapatinib plus capecitabine, HR = 0.68; 95% CI, 0.55 to 0.85; P < 0.001). Another randomized phase III trial, the TH3RESA trial, has compared T-DM1 with a treatment of the physician's choice in patients with progressive disease after two or more HER2-directed regimens for metastatic breast cancer [4]. PFS was significantly improved with T-DM1 compared with physician's choice (median 6.2 months [95% CI 5.59-6.87] vs. 3.3 months [2.89-4.14]; stratified HR 0.528 [95% CI 0.422-0.661]; P < 0.0001). Interim overall survival analysis showed a trend favoring T-DM1 (stratified HR 0.552 [95% CI 0.369-0.826]; P = 0.0034), but the stopping boundary was not crossed. T-DM1 had a lower incidence of grade 3 or worse adverse events (32%) when compared to the treatment of the physician's choice (43%), and was also associated with a favorable toxicity profile. Furthermore, a single arm phase II study (JO22997) in Japanese patients who had been intensively pretreated for locally advanced or metastatic breast cancer has been conducted [5]. In 73 patients treated with 3.6 mg/kg T-DM1 every 3 weeks, the objective response rate was 38.4% and median PFS was 5.6 months.
Although controlled clinical trials provide the highest levels of evidence, they do not always reflect clinical reality. For patients with HER2-positive metastatic breast cancer who have progressed during or after first-line HER2-targeted therapy with the combination of trastuzumab, pertuzumab, and a taxane, T-DM1 therapy is now a standard second-line treatment [6]. However, neither the EMILIA nor other T-DM1 trials included patients who had received pertuzumab and there is no published clinical trial that addresses this issue. Moreover, few data are currently available regarding the efficacy and safety of T-DM1 in patients with brain metastases, since most clinical trials have excluded patients with these complications, or have only included highly selected patients.
A retrospective exploratory analysis in the Emilia trial suggested that T-DM1 may confer a survival advantage over lapatinib-capecitabinein patients with treated, asymptomatic central nervous system (CNS) metastases and previously treated HER2-positive metastatic breast cancer, without increasing the risk for CNS progression [7]. The estimated median OS with CNS metastases at baseline was 26.8 months versus 12.9 months in the T-DM1 and lapatinib-capecitabine arms, respectively. This finding was consistent with the OS result among all randomized patients in the EMILIA trial, with a median OS of 30.9 months in the T-DM1 arm and 25.1 months in the lapatinib-capecitabine arm (HR = 0.68; P < 0.001).
In the present study, we report our experience with T-DM1 in patients with HER2-positive metastatic breast cancer that had progressed after previous anti-HER2 therapies. Of 63 patients in our cohort, 41 patients (65%) had received pertuzumab prior to T-DM1 therapy. The effectiveness and safety of T-DM1 treatment were retrospectively investigated.
Patients and Methods
Patients and treatment
A retrospective review was carried out on a total of 63 women with HER2-positive metastatic breast cancer who were treated with T-DM between April 2014 and April 2016 in 13 hospitals (Table 1). Most patients were heavily pretreated prior to T-DM1 therapy (Table 2). Patients were given T-DM1 at a dose of 3.6 mg/kg intravenously every 21 days. If a patient needed a dose reduction, the dose was reduced first from 3.6 mg/kg to 3.0 mg/kg and then from 3.0 mg/kg to 2.4 mg/kg. Echocardiography was performed prior to T-DM1 therapy and during anti-HER2 therapies (including T-DM1) approximately every three months.
Clinical responses were evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST version 1.1). CT, MRI, ultrasonography, and/or bone scintigraphy were used to evaluate the response to T-DM1 for patients without measurable lesions or for patients with bone metastasis only and the results were included in this study. The evaluation of brain metastases was performed using contrast-enhanced MRI or CT. Radiologists in each hospital assessed the existence of metastases and tumor response to T-DM1 therapy. Clinical benefit rate was defined as the sum of all patients experiencing complete response (CR), partial response (PR) or stable disease (SD) lasting 6 months or more. Time to treatment failure (TTF) and safety were also retrospectively analyzed. TTF was defined as the time from the date of T-DM1 treatment commencement to discontinuation of treatment for any reason, including disease progression, treatment toxicity, and death. Adverse events were evaluated using the National Cancer Institute Common Toxicity Criteria, version 4.0. This study was done in accordance with the guidelines of the 1996 Declaration of Helsinki.
Statistical analysis
Estimation of survival was performed using the Kaplan-Meier method. Patients who were still receiving T-DM1, ended T-DM1 therapy because of side effects, or died for other reasons at the time of analysis were included as censored cases. Univariate analysis with Cox proportional hazards regression models was used to identify factors predicting TTF during T-DM1 treatment. P values < 0.05 were considered to be significant.
Results
Patients' characteristics
Of 63 patients, 45 (71%) had recurrent breast cancer and 18 (29%) had stage IV disease (Table 1). Median age at the time of the start of T-DM1 therapy was 64 years (range, 40-80 years). There were 35 (56%) patients with ER or progesterone receptor (PgR)-positive and HER2-positive tumors, and 28 (44%) patients with tumors that were ER-and PgR-negative and HER2-positive. Regarding the estimation of HER2 status, 56 (89%) patients were evaluated as 2 + by immunohistochemistry (IHC), and 7 (11%) patients were evaluated as 2 + by IHC and positive by in situ hybridization. Forty-three (67%) patients had visceral metastases. There were 28 (44%) patients with lung metastases, 21 (33%) patients with liver metastases, 25 (40%) patients with bone metastases and 14 (22%) patients with brain metastases.
![]()
Table 1: Characteristics of patients.
View Table 1
Prior treatments before T-DM1 therapy
The previous treatments, in the adjuvant and metastatic setting, and the numbers of previous regimens for metastatic breast cancer prior to treatment with T-DM1 are listed in Table 2. As adjuvant or neoadjuvant therapy in 45 patients with recurrent breast cancer, 26 (41%) patients had received anthracyclines, 22 (35%) patients had received taxanes, and 19 (30%) patients had received trastuzumab. In the metastatic setting, all but two patients (97%) had undergone trastuzumab therapy, 41 (65%) patients had received pertuzumab and 21 (33%) patients had received lapatinib prior to T-DM1 therapy. Various chemotherapeutic drugs were combined with anti-HER2 agents prior to T-DM1, these included docetaxel, paclitaxel, vinorelbine, eribulin, gemcitabine, and capecitabine. Patients had received a median of three regimens prior to T-DM1 for metastatic breast cancer. Seventeen (27%) patients received T-DM1 as the second-line therapy, 8 (13%) patients as the third-line, 12 (19%) patients as the forth-line, and 26 (41%) patients as the fifth line or more. The median time since first-line treatment of metastatic breast cancer was 29 months (range, 6-217 months).
![]()
Table 2: Prior treatments.
View Table 2
![]()
Table 3: Response to T-DM1.
View Table 3
Response to T-DM1 therapy and survival
There were 3 (5%) patients with complete response (CR), 19 (30%) patients with partial response (PR), 9 (14%) patients with long stable disease (SD) ( ≥ 6 months), 9 (14%) patients with SD (< months), and 21 (33%) patients with progressive disease (PD) with T-DM1 therapy, so that the objective response rate was 35%, and the clinical benefit rate was 49% (Table 3). The median TTF of patients who received T-DM1 therapy was 4.0 months (range, 0.7-26 months, Figure 1). In 41 patients pretreated with pertuzumab, the response rate was 29%, the clinical benefit rate was 46%, and median TTF was 5.0 months (Table 4). In 14 patients with brain metastases, median TTF was 6.0 months, although none achieved CR or PR for brain tumors (Table 5). There were 5 patients with SD and 5 patients with PD for brain metastases.
![]()
Table 4: Details in patients pre-treated with pertuzumab.
View Table 4
![]()
Table 5: Details in patients with brain metastases.
View Table 5
At the time of the analysis, 21 (33%) patients were still receiving T-DM1. As anti-HER2 therapies and chemotherapies after T-DM1 treatment, 15 patients received trastuzumab with chemotherapeutic agents such as vinorelbine, eribulin and gemcitabine, 8 patients received both trastuzumab and pertuzumab with chemotherapeutic agents such as vinorelbine, paclitaxel and eribulin, and 9 patients received lapatinib plus capecitabine. The range of overall survival spanned 1.0-26 months (Figure 2).
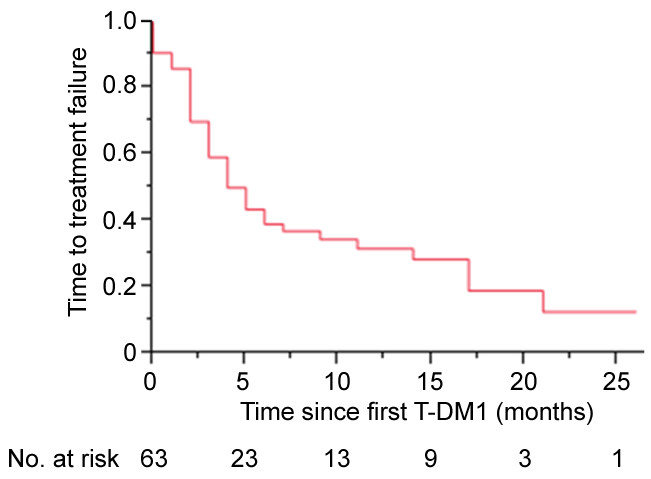
.
Figure 1: Time to treatment failure (TTF) Kaplan-Meier curve in 63 patients treated with T-DM1 therapy. The median TTF was 4.0 months (range, 0.7-26 months).
View Figure 1
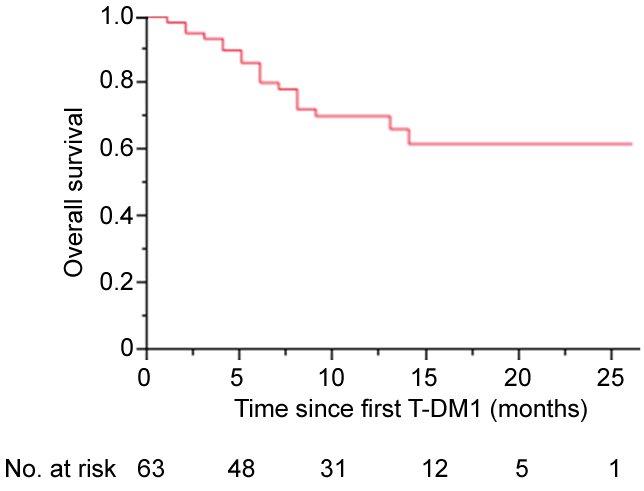
.
Figure 2: Overall survival Kaplan-Meier curve in 63 patients treated with T-DM1 therapy (range, 1.0-26 months).
View Figure 2
We analyzed factors predicting TTF during T-DM1 treatment with Cox proportional hazards regression models. Univariate analysis did not show any factor that was correlated with TTF (Table 6).
![]()
Table 6: Univariate analysis of factors predicting time to progression during T-DM1 treatment.
View Table 6
Adverse events
The most commonly reported adverse events were thrombocytopenia (n = 48, 76%) and increased ALT and/or AST (n = 42, 67%) (Table 7). Grade 3 thrombocytopenia was seen in 11 (18%) patients, and grade 4 thrombocytopenia was observed in 2 (3%) patients. Of these patients with thrombocytopenia, hemorrhage (nasal, gingival, or subcutaneous bleeding) was seen in 9 (14%) patients (grade 1 in 8 patients and grade 2 in 1 patient). None of the patients required a platelet transfusion. Increased ALT and/or AST of grade 3 were seen in 3 (5%) patients. Other serious adverse events were observed in one patient with rash (grade 3) and in one patient with neutropenia (grade 3). Fatigue (grade 1 and 2) was seen in 24 (38%) patients. No case of febrile neutropenia was documented. There were 5 (8%) patients with dose delays, and 11 (17%) patients with dose reductions because of side effects. Treatment termination was necessary in 5 (8%) patients because of side effects. None of the patients experienced cardiac toxicity during or after T-DM1 treatment.
![]()
Table 7: Adverse events.
View Table 7
Discussion
Here we report our clinical experience with T-DM1 in patients with HER2-positive metastatic breast cancer pretreated with anti-HER2 therapies and chemotherapies. The response rate in our study was 35%, which was similar to that of previous phase II trials [1,2] including the Japanese phase II JO22997 study (38.4%) [5], although central radiology review was not done in this study. Most patients in our study were heavily treated prior to T-DM1 therapy. Moreover, 41 (65%) patients had received pertuzumab prior to T-DM1 treatment. The combination of trastuzumab, pertuzumab, and a taxane is the standard of care in first-line treatment for patients with advanced HER2-positive breast cancer and T-DM1 is recommended as the second-line treatment in this setting [6]. However, clinical trials available with T-DM1 didn't accrued patients treated with pertuzumab because the drug was not approved when the EMILIA and Th3RESA trials were designated.
Dzimitrowicz and colleagues recently reported a retrospective study on the efficacy of T-DM1 in routine contemporary clinical practice in patients that had received pertuzumab any time before T-DM1 treatment [8]. Among the 78 patients who received T-DM1 as a predominantly second-line or later treatment regimen, one third received therapy with T-DM1 for ≥ 6 months, although tumor response rates were 17.9%. This study also reported that the median duration of therapy was 4.0 months, which is the same as the median TTF in our analysis. However, our median TTF is poorer than those of PFS reported in the EMILIA (9.6 months), TH3RESA (6.2 months), and JO22997 (5.6 months) trials [3-5]. Because all of the patients in Dzimitrowicz's study and 65% of patients in this study received pertuzumab prior to T-DM1 treatment, we infer that prior pertuzumab therapy might modulate the efficacy of shorter duration T-DM1 therapy. However, we could not find predictive factors including neither prior pertuzumab treatment nor other factors that were significantly predictive of the response to T-DM1 therapy.
The EMILIA study investigated whether tumor biomarkers, such as HER2, EGFR, and HER3 mRNA expression, PTEN protein expression and PIK3CA mutations could predict the efficacy of T-DM1 [9]. None of the factors were predictive, and T-DM1 appeared to be effective in both PIK3CA-mutated and wild-type tumors, although other standard HER2-directed therapies are less effective in tumors with PIK3CA-mutations. On the other hand, the ZEPHIR trial found that pretreatment HER2 imaging using HER2-positron emission tomography/computed tomography with 89Zr-trastuzumab showed intra-/interpatient heterogeneity in HER2 mapping of metastatic disease, and that this may be a useful predictor of the T-DM1 response [10]. Although the equipment for HER2 imaging is limited, we may suggest that imaging biomarkers will be an increasingly important tool for predicting HER2-targeting therapies.
Our study showed that the most commonly reported grade 3 or 4 adverse event was thrombocytopenia. We observed this in 21% of patients experienced, which is consistent the JO22997 study (22%), but more frequent than observed in the EMILIA (12.9%), TH3RESA (4%) and previously reported trials (8-9%) [5]. Also similar to the JO22997 study in Japanese patients, we found that grade 3/4 thrombocytopenia was not associated with severe bleeding, and none of patients received a platelet transfusion. An integrated safety analysis of 884 patients treated with T-DM1 in six studies demonstrated that decreases in platelet count were more common in Asian than non-Asian patients irrespective of platelet count values at baseline [11]. According to the ethnic sensitivity assessment of T-DM1 in Japanese patients and the global population, pharmacokinetics was comparable across ethnic groups [12]. T-DM1 is used at a dose of 3.6 mg/kg every 21 days in Japan and other countries. A Japanese phase II trial also reported that the pharmacokinetic profile of T-DM1 3.6 mg/kg every 21 days in Japanese patients showed no relevant differences from the predictable and well-characterized profile consistently demonstrated in phase II studies in non-Japanese populations [5]. Modeling of T-DM1-associated thrombocytopenia suggests that this side effect is caused by partial depletion of the platelet pool [13] and is typically transient [14]. This may explain why grade 3/4 thrombocytopenia has not been associated with clinically serious symptoms in Japanese patients receiving T-DM1.
Fourteen (22%) patients had brain metastases in our study, and none of them responded to T-DM1 treatment. On the other hand, the presence of brain metastases did not affect TTF. A direct correlation between quantitative HER2 expression and the risk for advanced breast cancer patients to develop brain metastases has been reported [15]. Jacot and colleagues recently reported a retrospective study on the efficacy and safety of T-DM1 in patients with brain metastases [16]. Of 39 patients, clinical benefit was 59%, and median PFS was 6.1 months. Their study indicated that treatment was well tolerated and free of unexpected toxicities, treatment delay, or dose reduction. Recently, expansion hematoma in delayed cerebral radiation necrosis was observed in two individuals who were treated with T-DM1 [17]. A potential enhancement of radiation necrosis in the brain appears to be one of the most significant adverse events associated with T-DM1 in patients that have received stereotactic radiosurgery for brain metastases.
In conclusion, our study demonstrates that T-DM1 is an effective and well-tolerated treatment for patients with HER2-positive metastatic breast cancer that had progressed after previous anti-HER2 therapies including pertuzumab and trastuzumab therapies.T-DM1 could be used for patients who have experienced progression following prior pertuzumab treatment.
References
-
Martinez MT, Perez-Fidalgo JA, Martin-Martorell P, Cejalvo JM, Pons V, et al. (2016) Treatment of HER2 positive advanced breast cancer with T-DM1: A review of the literature. Crit Rev Oncol Hematol 97: 96-106.
-
Bo Ma, Qianqian Ma, Hongqiang Wang, Guolei Zhang, Huiying Zhang, et al. (2016) Clinical efficacy and safety of T-DM1 for patients with HER2-positive breast cancer. Onco Targets Ther 9: 959-976.
-
Verma S, Miles D, Gianni L, Krop IE, Welslau M, et al. (2012) Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med 367: 1783-1791.
-
Krop IE, Kim SB, Gonzalez-Martin A, LoRusso PM, Ferrero JM, et al. (2014) Trastuzumab emtansine versus treatment of physician's choice for pretreated HER2-positive advanced breast cancer (TH3RESA): a randomised, open-label, phase 3 trial. Lancet Oncol 15: 689-699.
-
Kashiwaba M, Ito Y, Takao S, Doihara H, Rai Y, et al. (2016) A multicenter Phase II study evaluating the efficacy, safety and pharmacokinetics of trastuzumab emtansine in Japanese patients with heavily pretreated HER2-positive locally recurrent or metastatic breast cancer. Jpn J Clin Oncol 46: 407-414.
-
Giordano SH, Temin S, Kirshner JJ, Chandarlapaty S, Crews JR, et al. (2014) Systemic therapy for patients with advanced human epidermal growth factor receptor 2-positive breast cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 32: 2078-2099.
-
Krop IE, Lin NU, Blackwell K, Guardino E, Huober J, et al. (2015) Trastuzumab emtansine (T-DM1) versus lapatinib plus capecitabine in patients with HER2-positive metastatic breast cancer and central nervous system metastases: a retrospective, exploratory analysis in EMILIA. Ann Oncol 26: 113-119.
-
Dzimitrowicz H, Berger M, Vargo C, Hood A, Abdelghany O, et al. (2016) T-DM1 Activity in Metastatic Human Epidermal Growth Factor Receptor 2-Positive Breast Cancers That Received Prior Therapy With Trastuzumab and Pertuzumab. J Clin Oncol.
-
Baselga J, Lewis Phillips GD, Verma S, Ro J, Huober J, et al. (2016) Relationship between Tumor Biomarkers and Efficacy in EMILIA, a Phase III Study of Trastuzumab Emtansine in HER2-Positive Metastatic Breast Cancer. Clin Cancer Res 22: 3755-3763.
-
Gebhart G, Lamberts LE, Wimana Z, Garcia C, Emonts P, et al. (2016) Molecular imaging as a tool to investigate heterogeneity of advanced HER2-positive breast cancer and to predict patient outcome under trastuzumab emtansine (T-DM1): the ZEPHIR trial. Ann Oncol 27: 619-624.
-
Dieras V, Harbeck N, Budd GT, Greenson JK, Guardino AE, et al. (2014) Trastuzumab emtansine in human epidermal growth factor receptor 2-positive metastatic breast cancer: an integrated safety analysis. J Clin Oncol 32: 2750-2757.
-
Li C, Wang B, Lu D, Jin JY, Gao Y, et al. (2016) Ethnic sensitivity assessment of the antibody-drug conjugate trastuzumab emtansine (T-DM1) in patients with HER2-positive locally advanced or metastatic breast cancer. Cancer Chemother Pharmacol 78: 547-558.
-
Bender BC, Schaedeli-Stark F, Koch R, Joshi A, Chu YW, et al. (2012) A population pharmacokinetic/pharmacodynamic model of thrombocytopenia characterizing the effect of trastuzumab emtansine (T-DM1) on platelet counts in patients with HER2-positive metastatic breast cancer. Cancer Chemother Pharmacol 70: 591-601.
-
Sara A Hurvitz, Reva Kakkar (2012) The potential for trastuzumab emtansine in human epidermal growth factor receptor 2 positive metastatic breast cancer: latest evidence and ongoing studies. Ther Adv Med Oncol 4: 235-245.
-
Renata Duchnowska, Wojciech Biernat, Barbara Szostakiewicz, Jeff Sperinde, Fanny Piette, et al. (2012) Correlation between quantitative HER-2 protein expression and risk for brain metastases in HER-2+ advanced breast cancer patients receiving trastuzumab-containing therapy. Oncologist 17: 26-35.
-
Jacot W, Pons E, Frenel JS, Guiu S, Levy C, et al. (2016) Efficacy and safety of trastuzumab emtansine (T-DM1) in patients with HER2-positive breast cancer with brain metastases. Breast Cancer Res Treat 157: 307-318.
-
Koichi Mitsuya, Junichiro Watanabe, Yoko Nakasu, Nakamasa Hayashi, Hideyuki Harada, et al. (2016) Expansive hematoma in delayed cerebral radiation necrosis in patients treated with T-DM1: a report of two cases. BMC Cancer 16: 391.





