Journal of Dermatology Research and Therapy
Folliculotropic Mycosis Fungoides: A Clinicopathological Study of a Series of 10 Cases
V Vergote1*, P Wolter2, E Hauben3 and AM Busschots4
1Department of internal medicine, University Hospitals, UZ Leuven, Belgium
2Department of medical oncology, University Hospitals, UZ Leuven, Belgium
3Department of pathology, University Hospitals, UZ Leuven, Belgium
4Department of dermatology, University Hospitals, UZ Leuven, Belgium
*Corresponding author:
Vibeke Vergote, Department of internal medicine, University Hospitals, Herestraat 49, UZ Gasthuisberg, 3000 Leuven, Belgium, Tel: 016-346880, E-mail: Vibeke.Vergote@uzleuven.be
J Dermatol Res Ther, JDRT-2-022, (Volume 2, Issue 2), Case Report; ISSN: 2469-5750
Received: February 16, 2016 | Accepted: April 11, 2016 | Published: April 13, 2016
Citation: Vergote V, Wolter P, Hauben E, Busschots AM (2016) Folliculotropic Mycosis Fungoides: A Clinicopathological Study of a Series of 10 Cases. J Dermatol Res Ther 2:022. 10.23937/2469-5750/1510022
Copyright: © 2016 Vergote V, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Folliculotropic mycosis fungoides (FMF) is a rare variant of cutaneous T-cell lymphoma, characterizedby the infiltration of atypical lymphocytes into hair follicles (folliculotropism).
Design: A single centre retrospective cohort analysis.
Objective: To provide additional data regarding clinicopathological features, response to treatment, prognosis and disease course of FMF.
Methods: We searched cutaneous lymphoma database for patients with FMF. FMF was defined by both clinical and histological features, including immunohistochemical studies and molecular genetic analysis of T-cell receptor genes.
Results: Ten patients (8 male, 2 female) with a median age at diagnosis of 45 years were included. The median duration of the presence of skin lesions before diagnosis was 11 years. The most common sites of involvement were head and neck (80%), trunk and extremities (80-90%). Clinical features included patches and plaques, often associated with follicular prominence and alopecia, or comedo-like lesions, nodules and cysts. Fifty percent complained of pruritus. Histological findings included folliculotropism and/or follicular mucinosis (present in 80%). Initial therapy consisted of psoralen plus UV-A (PUVA) treatment in 50% of our patients. Only 2 out of 10 patients achieved complete remission. At the conclusion of this study 3 patients had died of lymphoma progression.
Conclusions: Our findings support the current classification of folliculotropic mycosis fungoides as a distinct entity in the WHO-EORTC classification of cutaneous lymphomas, with distinct clinicopathological features, survival and therapeutic responses.
Introduction
Cutaneous T-cell lymphoma (CTCL) is a malignancy of skin-homing T-cells and can have great variability in its clinical presentation. The overall incidence of CTCL is 6,4 cases per million [1,2]. Mycosis fungoides (MF) is the most common type of CTCL and accounts for almost 50% of all primary cutaneous lymphomas. Histologically, it is characterized by epidermal infiltration (epidermotropism) of small to medium sized T-lymphocytes with cerebriform nuclei. Sometimes these atypical T-lymphocytes form a collection in the epidermis, called Pautrier microabscesses. MF typically presents as erythematous skin patches evolving into infiltrated plaques and tumors [3,4].
Apart from this so-called classic Alibert-Bazin type of MF, many clinical and histological subtypes have been reported, including hypo pigmented, bullous, pustular and other types of MF. Since these variant types of MF have the same clinical behavior and require the same therapeutic approach as the classic type of MF they are not considered separate entities [3,5].
Folliculotropic mycosis fungoides (FMF) is a rare form of CTCL that represents less than 10% of patients with MF [6]. It is characterized by the infiltration of atypical lymphocytes into hair follicles, often with sparing of the epidermis, and preferential involvement of the head and neck area. Most cases show mucinous degeneration of the hair follicles (follicular mucinosis). Distinct clinical and histological features, survival outcome, and therapeutic responses have been identified in patients with FMF. In light of its unique clinical and histopathological features, FMF has been designated as a distinct variant of CTCL in the new WHO-EORTC classification system for cutaneous lymphomas in 2005 [3,7].
In this study, we report a series of 10 patients with folliculotropic MF, providing additional information regarding its clinicopathological features, the response to treatment, prognosis and the disease course of this unusual form of CTCL.
Materials and Methods
We searched our database of cutaneous lymphoma patients (total 198 patients) for patients with FMF in the period between 1976 and 2010. We defined FMF by both clinical and histological features, including immunohistochemical studies and molecular genetic analysis of T-cell receptor genes. The initial and predominant clinical features consisted of patches or plaques with follicular prominence, comedonal or cystic lesions and alopecia. Histological criteria included the presence of a perifollicular dermal infiltrate with variable numbers of atypical T-cells with cerebriform nuclei infiltrating the follicular epithelium (folliculotropism) with or without the presence of mucinous degeneration of the follicular epithelium (follicular mucinosis). Furthermore, molecular genetic analysis of T-cell receptor genes and immunohistochemical studies for CD3, CD4, CD5, CD7 and CD8 were performed. All representative histopathological sections were reviewed by a dermatopathologist experienced in cutaneous lymphomas. In each patient the diagnosis has been made by an expert panel of dermatologists, oncologists and dermatopathologists.
A total of 10 patients fulfilled the inclusion criteria of this retrospective review and were extensively reviewed. All data available regarding clinical presentation, histology, staging, treatments, response to therapy and follow-up were recorded.
Time of onset was defined as the time from initial characteristic clinical features. Disease progression was defined as the development of histologically documented tumoral skin lesions or generalized erythroderma in patients previously having patch-plaque stage skin disease (T1-2); the development of biopsy-proven lymph nodal involvement in patients with previously skin-limited disease; the development of visceral involvement in patients with prior skin- or lymph node-limited disease, and death due to lymphoma.
Results
Clinical features
A total of 10 patients were included in the study. The main clinical characteristics, therapy and follow-up have been summarized in table 1 and table 2. Among the 10 patients, there were 8 males and 2 females, resulting in a male to female ratio of 4:1. The median age of onset of skin lesions was 29.5 years (ranging from 17 to 68), while the median age at diagnosis of FMF was 45 years (ranging from 18 to 71). The median duration of skin lesions before diagnosis was 11 years. At the time of staging, 9 out of 10 patients had disease limited to patches or plaques (stage IA-B), whereas only one patient had advanced disease with tumor-stage skin lesions and lymph node involvement. The clinical characteristics of folliculotropic mycosis fungoides are variable and patients frequently show a combination of features. The most common presentation at the time of diagnosis consisted of patches and plaques, often associated with follicular prominence and alopecia. In fact 8 of our 10 patients presented with alopecia at the time of diagnosis. Another major form of presentation was a combination of comedo-like lesions, nodules and cysts. Half of our patients complained of pruritus.
![]()
Table 1: Clinical findings, therapy and follow-up in 10 patients with folliculotropic mycosis fungoides.
View Table 1
![]()
Table 2: Summary of clinical characteristics.
View Table 2
Eight of our patients presented with lesions involving the neck, scalp or face. However, the location of the skin lesions in folliculotropic mycosis fungoides is quite variable, and often also includes trunk and extremities. Four patients had lesions on the buttocks and 3 patients had lesions in the genital area.
Histopathological features
The main histopathological features are summarized in table 3. A total of 47 skin biopsy specimens were obtained from our 10 patients included in the study. Characteristically, the diagnostic specimens showed perifollicular and perivascular to diffuse dermal infiltrates with variable infiltration of the follicular epithelium by medium-sized to large atypical T-cells with cerebriform nuclei [5] (Figure 1). In 8 out of 10 cases the characteristic infiltration of the follicular epithelium by atypical T-cells (folliculotropism) was found. In 2 other cases these characteristics could not be found, and the diagnosis of folliculotropic MF was based on combined clinical signs and histological, immunohistochemical and/or molecular evidence of CTCL. Only a small proportion of patients (2 cases) showed formation of Pautrier microabscesses and in 3 cases epidermotropism was found. Notably, 6 cases showed mucinous degeneration of the follicular epithelium (follicular mucinosis), as assessed with Alcian blue staining. Eccrine gland infiltration could be seen in 2 cases, but none of the biopsy specimens showed epidermal cysts and comedones.
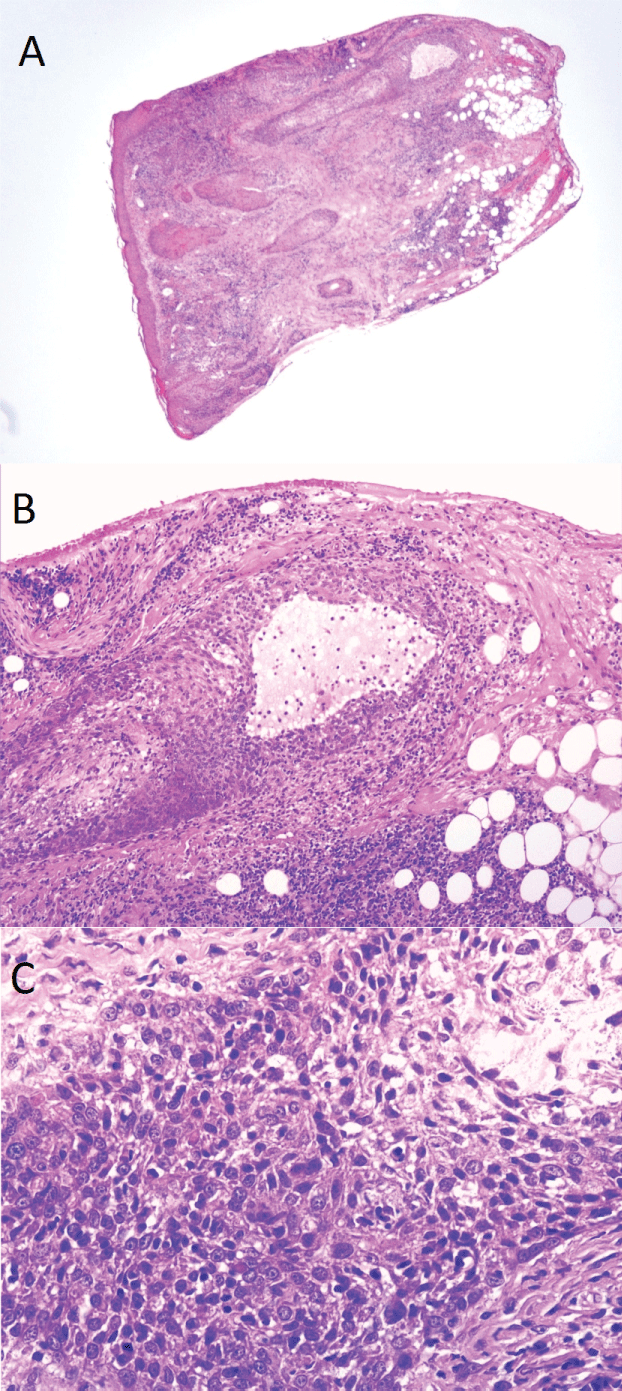
.
Figure 1: Characteristic histological features of folliculotropic MF. (All on Hematoxylin-eosin stain). A) Perifollicular infiltrates by atypical T-lymphocytes with infiltration of follicular epithelium (folliculotropism); B) Mucinous degeneration of the follicular epithelium (follicular mucinosis) with mucin lake; C) Detail of perifollicular infiltrate showing atypical cerebriform, hyperchromatic T-lymphocytes.
View Figure 1
![]()
Table 3: Histological features.
View Table 3
Immunohistochemical analysis demonstrated a mature T-helper phenotype with CD3+, CD4+, CD8- staining of the neoplastic T-cells in most cases. Loss of CD7 and/or CD5 was observed in 7 cases. T-cell receptor clonality was found in 8 out of 10 biopsy specimens, whereas TCR clonality in blood analysis was only found in 5 cases.
Therapy and outcome
Detailed information regarding initial treatment, other therapies and outcome are outlined in table 1 and table 4. Initial therapy consisted of psoralen plus UV-A (PUVA) treatment in 50% of our patients. Two patients were initially treated with topical chlormustine (BCNU) and one patient received a combination of PUVA and BCNU. Furthermore, one patient received isotretinoin and one other patient received combination chemotherapy (cyclophosphamide, doxorubicin, teniposide and prednisone with bleomycin and vincristine or CHVmP-BV) as initial treatment.
![]()
Table 4: Treatment and follow-up.
View Table 4
In most cases, initial clinical responses were observed with this therapy, but only 2 patients achieved complete remission. Other therapies as second-line treatment included local and total body skin irradiation, topical steroids, interferon-alpha, prednisone, retinoids (acitretin, isotretinoin, bexarotene) and chemotherapy.
The follow-up period varied between 1 and 34 years, with a median follow-up of 9, 5 years. Progression was reported in 50% of our cases, including development of tumoral skin lesions (3 cases), nodal involvement (3 cases), visceral involvement (1 case) and development of erythrodermic MF (1 case). At the conclusion of the study 3 patients had died from lymphoma progression, 5 patients were alive with stable disease, 1 patient lived with progressive disease and only 1 patient lived without disease.
Discussion
Clinical features
Folliculotropic MF is a rare form of CTCL that represents 4% of primary cutaneous lymphomas and less than 10% of patients with MF [3,6]. Patients with FMF demonstrate a significantly higher male to female ratio (4-5:1), as was confirmed in our study (4:1) [8]. Previous literature reported an average age at diagnosis of 53 years, which seems to be similar to that of classic MF (median age at diagnosis of 55-60 years) [3,5,7,9]. The age at diagnosis in our patients was lower, with a median age of 45 years, though this number might be biased due to the low number of patients included in our study. Notably, the median duration of skin lesions before diagnosis was 11 years. In comparison, previous studies reported a duration of lesions before diagnosis of 48 to 50 months [5,9]. However, in our study the median duration of skin lesions was calculated based on the patients history, not the date of first consultation. This might explain the difference with previous studies. In FMF, multiple biopsy specimens are often required before a definitive diagnosis can be made. Hence, familiarity with the clinical and histological presentation and a high degree of suspicion accompanied by multiple biopsies may help reach the proper diagnosis [7].
Clinically, this type of MF manifests itself as acneiform lesions, with formation of comedo-like plugs and epidermal cysts, follicular papules, follicular keratosis, erythematous patches, infiltrated plaques and sometimes tumors. The skin lesions are often associated with alopecia. Some patients may even develop mucorrhea (discharge of mucinous substance from follicular orifices) [5,8,10-14].
Unlike classical MF, pruritus seems to be a rather frequent clinical feature in folliculotropic MF. Pruritus is often severe, requiring special medical attention, and may represent a good characteristic of disease progression [3,5,9,15].
Patients with FMF tend to present with lesions localized to the head and neck area. This presentation is in contrast with the common bathing suit distribution of conventional MF, which rarely involves the face. Other predilection sites include upper extremities and torso [3,7,15].
Histopathology
As with the clinical presentation, the histological features in FMF are distinctive from classical MF [7,16]. FMF is characterized by dense lymphocytic infiltrates surrounding and infiltrating the hair follicles. In most cases the epidermis is spared (folliculotropism instead of epidermotropism). The neoplastic lymphocytes are small to medium-sized cells with irregular, cerebriform nuclei. In some cases, Pautrier's microabscesses can be seen within the follicular epithelium. Infiltration of eccrine glands may be seen in some cases [5]. There is often a considerable admixture of eosinophils and sometimes plasma cells [3].
The follicles often show mucinous degeneration of the follicular epithelium (follicular mucinosis), varying from focal spots of mucin deposition to complete destruction of follicle and mucin lakes [5,8,10-14]. Previous studies reported follicular mucinosis in 73% of the FMF cases (ranging from 51 to 96%) [5,7,9]. This number was confirmed in our study as 6 out of 10 cases showed follicular mucinosis. In recent studies no differences in clinical presentation and clinical behavior were observed between cases of FMF with or without associated follicular mucinosis. Therefore, it was proposed that the concept of folliculotropic MF should be applied to all cases of MF showing perifollicular or intrafollicular lymphomatous infiltration regardless of the presence of follicular mucinosis [3,5,17,18].
In most cases the neoplastic T-cells have a mature T-helper phenotype (CD3+, CD4+, CD8-) as in classical MF. In many cases, there is loss of one or more lymphocyte antigen such as CD7, CD5 or CD2. Loss of this pan T-cell antigens is often seen as an important adjunct in the diagnosis of MF, but can also be nonspecific. Furthermore, clonal T-cell receptor gene rearrangements are detected in most cases [3,15,19]. In our study T-cell receptor clonality was found in 8 out of 10 biopsy specimens.
The difficulty in histologic recognition of FMF is further characterized by frequent nondiagnostic biopsies. In these cases, multiple biopsy specimens are often required to make the definitive diagnosis.
Treatment and outcome
The importance of distinction between MF and FMF is not an academic exercise but is emphasized by the differences in prognosis and therapeutic response in these 2 groups [7]. Previous publications from the Dutch Cutaneous Lymphoma Group [5] and from Northwestern University in Chicago [7] conclude that folliculotropic MF has a more aggressive behavior and a worse prognosis than classic MF [5,7,9]. In a study of 51 FMF patients, published by Van Doorn et al., overall survival data demonstrated a 5-year survival of 64% and a 10-year survival of only 14%. These data were later confirmed by the study of Lehman et al, which showed similar 5-year survival rates (62%) in 50 FMF patients [20]. Comparison of these data with classic MF indicate that patients with folliculotropic MF have a similar (at 5 years) or worse (at 10 years) survival than patients with tumor-stage MF. The largest series to date (189 FMF patients), published by Agar et al. [21] identified FMF as an independent risk factor for survival and progression outcome. Furthermore, the calculated risk of disease progression in the study of Van Doorn et al. was 37% at 5 years and 66% at 10 years, which is higher than in classical MF patients with plaque and tumor stage. However, these results could not be confirmed by the study of Gerami et al. They found an overall survival after 5, 10 and 15 years of respectively 87%, 82% and 41% for early-stage FMF (≤ IIA) and 83%, 67% and 27% for late-stage FMF (≥ IIB) [5,7,8]. The WHO-EORTC cutaneous lymphoma group reports a disease specific 5-year survival of 80% [3]. In our study 3 out of 10 patients died of lymphoma progression (1, 3 and 8 years after diagnosis, respectively). More studies are needed to evaluate the exact overall survival in folliculotropic MF.
These findings raised the question of the usefulness of applying the standard clinical staging system for CTCL to patients with FMF, and the Dutch group proposed considering patients with FMF as having the tumor-stage disease, regardless of the clinical Because of the perifollicular appearance of the skin lesions [5,8].
localization of the dermal infiltrates, folliculotropic MF is often less responsive to skin-targeted therapies, such as PUVA and topical nitrogen mustard, than classical plaque stage MF [3,20]. Other investigators have hypothesized that the lymphocyte biologic characteristics that determine pilotropism also contribute to therapy resistance [20]. In 50% of our patients PUVA was given as initial treatment, with none of these patients achieving complete remission. Other treatments included topical chlormustine, topical steroids, retinoids, prednisolone, interferon-alpha, local and total skin irradiation and combination chemotherapy.
In the Dutch study by Van Doorn et al., many patients responded to local radiotherapy. Therefore, they suggest that radiotherapy should be the first treatment of choice in patients presenting with a single plaque or tumor or a few clustered skin lesions, but without patches or follicular papules at other sites. Only in selected cases with superficial lesions at multiple sites, PUVA is recommended as first treatment. Alternatively, total skin electron beam irradiation or a combination of PUVA with interferon-alpha or retinoids are effective treatments. Persistent tumors can be treated effectively with local radiotherapy [3,5].
A more recent study by Gerami et al. suggested that initial treatment for early-stage FMF (≤ IIA) should include phototherapy, preferably PUVA in combination with a retinoid or interferon alpha [7]. A good response to bexarotene has also been reported in several other studies, suggesting that bexarotene given alone or in combination with PUVA might have a role in the treatment of patients with FMF [9,22-26]. Prognosis is poor in advanced disease (≥ IIB) and aggressive therapy is required. Poor responses were observed on conventional chemotherapies such as CHOP (Cyclophosphamide, Hydroxydaunorubicin or doxorubicin, Oncovin or vincristine, and Prednisone), liposomal doxorubicin and gemcitabine. Partial or complete remissions were seen with alemtuzumab (anti-CD52 monoclonal antibody), irradiation and allogeneic stem cell transplantation [7,27].
Conclusion
Our findings support the current classification of folliculotropic mycosis fungoides as a distinct entity in the WHO-EORTC classification of cutaneous lymphomas, with distinct clinicopathological features, survival, and therapeutic responses. As confirmed in previous studies, folliculotropic MF has a more aggressive behavior and a worse prognosis than classic MF. Appreciating these differences between FMF and conventional MF is important in order to optimize therapy and management of this rare form of CTCL.
References
-
Hwang ST, Janik JE, Jaffe ES, Wilson WH (2008) Mycosis fungoides and Sézary syndrome. Lancet 371: 945-957.
-
Criscione VD, Weinstock MA (2007) Incidence of cutaneous T-cell lymphoma in the United States, 1973-2002. Arch Dermatol 143: 854-859.
-
Willemze R, Jaffe ES, Burg G, Cerroni L, Berti E, et al. (2005) WHO-EORTC classification for cutaneous lymphomas. Blood 105: 3768-3785.
-
Yost JM, Do TT, Kovalszki K, Su L, Anderson TF, et al. (2009) Two cases of syringotropic cutaneous T-cell lymphoma and review of the literature. J Am Acad Dermatol 61: 133-138.
-
van Doorn R, Scheffer E, Willemze R (2002) Follicular mycosis fungoides, a distinct disease entity with or without associated follicular mucinosis: a clinicopathologic and follow-up study of 51 patients. Arch Dermatol 138: 191-198.
-
van Doorn R, Van Haselen CW, van Voorst Vader PC, Geerts ML, Heule F, et al. (2000) Mycosis fungoides: disease evolution and prognosis of 309 Dutch patients. Arch Dermatol 136: 504-510.
-
Gerami P, Rosen S, Kuzel T, Boone SL, Guitart J (2008) Folliculotropic mycosis fungoides: an aggressive variant of cutaneous T-cell lymphoma. Arch Dermatol 144: 738-746.
-
Kazakov DV, Burg G, Kempf W (2004) Clinicopathological spectrum of mycosis fungoides. J Eur Acad Dermatol Venereol 18: 397-415.
-
Muniesa C, Estrach T, Pujol RM, Gallardo F, Garcia-Muret P, et al. (2010) Folliculotropic mycosis fungoides: clinicopathological features and outcome in a series of 20 cases. J Am Acad Dermatol 62: 418-426.
-
Fraser-Andrews E, Ashton R, Russell-Jones R (1999) Pilotropic mycosis fungoides presenting with multiple cysts, comedones and alopecia. Br J Dermatol 140: 141-144.
-
Pereyo NG, Requena L, Galloway J, Sangüeza OP (1997) Follicular mycosis fungoides: a clinicohistopathologic study. J Am Acad Dermatol 36: 563-568.
-
Mehregan DA, Gibson LE, Muller SA (1991) Follicular mucinosis: histopathologic review of 33 cases. Mayo Clin Proc 66: 387-390.
-
Nickoloff BJ, Wood C (1985) Benign idiopathic versus mycosis fungoides-associated follicular mucinosis. Pediatr Dermatol 2: 201-206.
-
Cerroni L, Fink-Puches R, Bäck B, Kerl H (2002) Follicular mucinosis: a critical reappraisal of clinicopathologic features and association with mycosis fungoides and Sézary syndrome. Arch Dermatol 138: 182-189.
-
Mantaka P, Helsing P, Gjersvik P, Bassarova A, Clausen OP, et al. (2013) Clinical and histopathological features of folliculotropic mycosis fungoides: a Norwegian patient series. Acta Derm Venereol 93: 325-329.
-
Gerami P, Guitart J (2007) The spectrum of histopathologic and immunohistochemical findings in folliculotropic mycosis fungoides. Am J Surg Pathol 31: 1430-1438.
-
Flaig MJ, Cerroni L, Schuhmann K, Bertsch HP, Kind P, et al. (2001) Follicular mycosis fungoides. A histopathologic analysis of nine cases. J Cutan Pathol 28: 525-530.
-
Ke MS, Kamath NV, Nihal M, Mikkola DL, Koc ON, et al. (2003) Folliculotropic mycosis fungoides with central nervous system involvement: demonstration of tumor clonality in intrafollicular T cells using laser capture microdissection. J Am Acad Dermatol 48: 238-243.
-
Smoller BR (2008) Mycosis fungoides: what do/do not we know? J Cutan Pathol 35 Suppl 2: 35-39.
-
Lehman JS, Cook-Norris RH, Weed BR, Weenig RH, Gibson LE, et al. (2010) Folliculotropic mycosis fungoides: single-center study and systematic review. Arch Dermatol 146: 607-613.
-
Agar NS, Wedgeworth E, Crichton S, Mitchell TJ, Cox M, et al. (2010) Survival outcomes and prognostic factors in mycosis fungoides/Sézary syndrome: validation of the revised International Society for Cutaneous Lymphomas/European Organisation for Research and Treatment of Cancer staging proposal. J Clin Oncol 28: 4730-4739.
-
Drugeon C, Charlat I, Boulinguez S, Viraben R (2007) [Bexarotene therapy in folliculotropic cutaneous T-cell lymphoma]. Ann Dermatol Venereol 134: 639-643.
-
Mitteldorf C, Stadler R, Bertsch HP, Neumann C (2007) Folliculotropic mycosis fungoides with CD30+ large-cell transformation in a young woman: beneficial effect of bexarotene. Br J Dermatol 156: 584-586.
-
Leverkus M, Rose C, Bröcker EB, Goebeler M (2005) Follicular cutaneous T-cell lymphoma: beneficial effect of isotretinoin for persisting cysts and comedones. Br J Dermatol 152: 193-194.
-
Shistik G, Scalf LA, Fenske N, Glass LF (2004) Follicular mycosis fungoides: successful treatment with oral bexarotene. J Drugs Dermatol 3: 301-304.
-
Apisarnthanarax N, Ha CS, Duvic M (2003) Mycosis fungoides with follicular mucinosis displaying aggressive tumor-stage transformation: successful treatment using radiation therapy plus oral bexarotene combination therapy. Am J Clin Dermatol 4: 429-433.
-
Shin J, Monti S, Aires DJ, Duvic M, Golub T, et al. (2007) Lesional gene expression profiling in cutaneous T-cell lymphoma reveals natural clusters associated with disease outcome. Blood 110: 3015-3027.





