International Journal of Stem cell Research and Therapy
Neural Stem Cells and their Niches in Neuroregeneration
Jong-Hang Chen1, Mei-Shu Chen1, Su-Liang Chen1 and Ing-Ming Chiu1,2,3,*
1Institute of Cellular and System Medicine, National Health Research Institutes, Miaoli 350, Taiwan
2Department of Life Sciences, National Chung Hsing University, Taichung 402, Taiwan
3Department of Internal Medicine, The Ohio State University, 480 Medical Center Drive Columbus, Ohio 43210, U.S.A.
*Corresponding author: Ing-Ming Chiu, Institute of Cellular and System Medicine, National Health Research Institutes, 35, Keyan Rd, Miaoli 350, Taiwan, Tel: +886-37-246166; Fax: +886-2-2363-0495; E-mail address: ingming@nhri.org.tw
Int J Stem Cell Res Ther, IJSCRT-2-015, (Volume 2, Issue 2), Review Article; ISSN: 2469-570X
Received: September 17, 2015 | Accepted: December 16, 2015 | Published: December 21, 2015
Citation: Jong-Hang C, Mei-Shu C, Su-Liang C, Ing-Ming C (2015) Neural Stem Cells and their Niches in Neuroregeneration. Int J Stem Cell Res Ther 2:015. 10.23937/2469-570X/1410015
Copyright: © 2015 Jong-Hang C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Stem cell-based therapies in alleviating symptoms or reversing the progression of neurodegenerative diseases and nerve injuries have been investigated extensively. In this review, we first summarize our current understanding of the ontogeny of neural stem cells (NSCs) and their mobilization in vivo. We then focus on the cellular reprogramming in generation of iP-NSCs, iNeurons and iNSCs, and applications of NSCs and iNeurons in cell-based therapy, disease modeling and drug discovery. NSCs belong to the adult stem cells, which can self-renew and differentiate into neurons, astrocytes and oligodendrocytes. NSCs reside in specialized niches of the adult mammalian brain, which support their self-renew and differentiation throughout life. Two regions continuously generate new neurons, including subventricular zone (SVZ) of the lateral ventricles and subgranular zone (SGZ) of the hippocampus. For adult neurogenesis, sensitizing to the stress and physical stimulations or exposure to an enriched environment increases adult neurogenesis in animal models. Therefore, mobilizations of NSCs could improve the symptoms of neuronal degeneration disorders of the central nervous system. In peripheral nerve injury, transplantation of NSCs provided effective therapies for many neurological diseases and injuries. Mechanisms of peripheral neuroregeneration by NSCs administration include differentiation into Schwann cells, secretion of neurotrophic factors, and promoting myelination. Moreover, differentiated cells that have been genetically reprogrammed to an embryonic stem cell-like state by forced gene expression and/or factors treatment could maintain the defining properties of embryonic stem cells or functional neurons. Processes of cellular reprogramming were illustrated in the generation of induced pluripotent stem cells-derived NSCs (iP-NSCs), induced neuronal cells (iNeurons) and induced NSCs (iNSCs). The primary advantage in the use of cellular reprogramming is that adult cells are more easily obtained and with less ethical concerns. Thus, iP-NSCs, iNeurons and iNSCs are considered as a good cell source for cell-based therapies, disease modeling and drug discovery in neurodegenerative diseases.
Keywords
Neural stem cells, Neuroregeneration, Mobilizations of NSCs, Cellular reprogramming, iP-NSCs, iNeurons, iNSCs
Introduction
The prevalence of neurodegenerative diseases such as Alzheimer's disease, Parkinson's disease, Huntington's disease, amyotrophic lateral sclerosis (ALS), and spinal muscular atrophy (SMA) is increasing rapidly. Stem cell-based therapies in alleviating symptoms or reversing the progression of neurodegenerative diseases and nerve injuries, including stroke, spinal cord injury, and peripheral nerve injury (PNI), have been investigated extensively [1-4]. In many tissues, stem cells serve as an internal repair system, dividing essentially without limit to replace damaged cells. Stem cells are classified into two categories, embryonic stem cells and adult stem cells (also called somatic stem cells). Embryonic stem cells are pluripotent, which can become all cell types of the body, and adult stem cells are thought to be limited to differentiate into different cell types of their tissue of origin [5-8]. Adult stem cells have been identified in many tissues and organs, including brain, bone marrow, peripheral blood, blood vessels, skeletal muscle, skin, teeth, heart, gut, liver, ovarian epithelium and testis. It resides in specific areas of the tissue (called stem cell niche), which remain quiescent (non-dividing) for long periods of time until they are activated by a need for more cells to maintain tissues, or by disease or tissue injury. Therefore, scientists believe that if the activation and differentiation of stem cells can be controlled, these cells may become the basis of cell-based therapies.
Neural stem cells (NSCs) belong to the adult stem cells, which reside in the brain, and can self-renew and differentiate into neurons, astrocytes and oligodendrocytes [9]. NSCs were proposed as a promising cellular source for the treatment of diseases in nervous systems [10,11]. Two strategies were suggested in the cell therapy of nervous system disorders: 1) to transplant exogenous NSCs into the injury sites; 2) to stimulate the functional activity of endogenous NSCs in vivo [12,13]. Scientists reported that exposure to an enriched environment had been shown structural and functional changes in the brain and significant increase in neurogenesis in vivo [14,15]. Moreover, it has been demonstrated that pluripotency can be restored to adult somatic cells through ectopic co-expression of defined transcription factors, thus proving that the fate of somatic cells is not immutable and paving the way for modeling human diseases and personalized cell therapies. In recent years, scientists develop cell reprogramming methods in the generating of induced pluripotent stem cells-derived NSCs (iP-NSCs), induced NSCs (iNSCs) and induced neuronal cells (iNeurons or iNs) for cell-based therapy in neurodegenerative diseases and nerve injuries, disease modeling and drug discovery [16-19].
In this review, we first summarize our current understanding of the ontogeny of NSCs (including characterization, isolation and differentiation) and their mobilization in vivo. We then focus on the cellular reprogramming in generation of iP-NSCs, iNeurons and iNSCs, and applications of NSCs and iNeurons in cell therapy, disease modeling and drug discovery.
NSCs and Generation of Neurons
Permanent loss of neurons with no possibility of cellular regeneration has been challenged recently by extensive evidence that certain brain areas retain the capability to generate new neurons into adulthood in rodents [20-23], nonhuman primates [24] and human [25]. New neurons are added to the olfactory bulb and the hippocampus of dentate gyrus in mammals, and to the neostriatum, paraolfactory and parahippocampal regions in birds [26]. Previous studies reported that implantation of NSCs promotes neuroregeneration in the animal models of nerve injury [27,28]. NSCs are highly desirable for clinical applications, which are based on two reasons: 1) NSCs are well-studied and well-characterized cells; 2) NSCs are capable of potentially unlimited self-renewal, and can be differentiated into neurons, astrocytes and oligodendrocytes both in vitro under defined conditions and in vivo after transplantation [29].
Characterization of NSCs
NSCs are characterized by two properties, self-renewal and neuronal differentiation (multipotency). Self-renewal and differentiation of NSCs were regulated by the specialized microenvironment, or niche, in which these cells reside. Different signaling pathways have been implicated to play a role in the process [30,31]. Neurosphere formation is frequently used as a standard for evaluating self-renewal ability in NSCs. The mechanisms of neurosphere formation are related to the cell proliferation and cell-cell adhesion, which means that neurospheres can form from the clonal progeny of single NSCs, and/or from the adhered cells by cell-cell interactions initiated between NSCs [32]. The potential for neuronal differentiation of neurosphere-isolated cells upon withdrawal of growth factors was used to determine multipotency of NSCs [33,34].
Isolation of NSCs
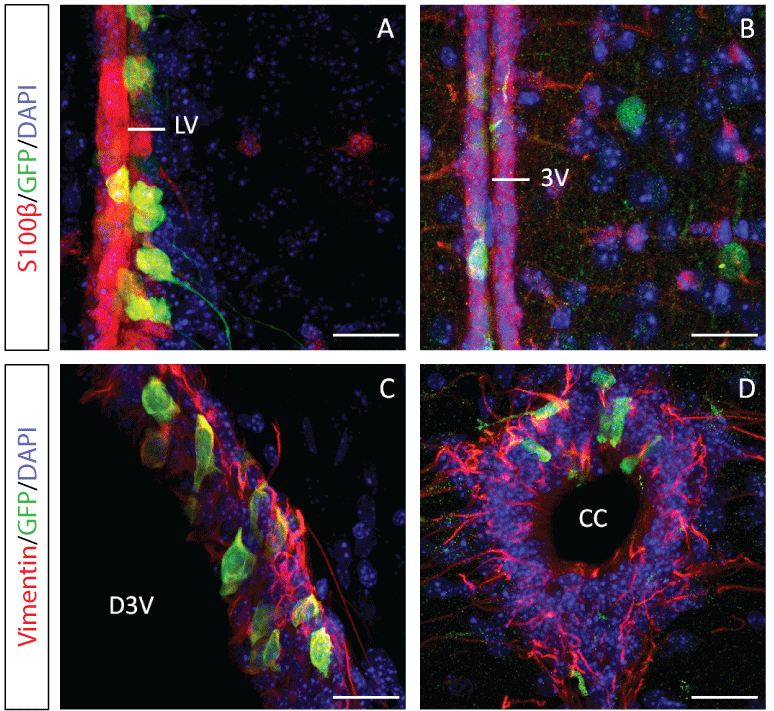
Fluorescence-activated cell sorting (FACS) has been applied in isolation of different precursor and progenitor populations from tissues. NSCs can be isolated from brain tissues by cell-surface markers, such as CD133 or CD24, or by Sox1, Sox2 or Nestin promoter-driven GFP reporter. The cells cultured in the presence of growth factors and examined to determine whether they could expand to form neurospheres. Notably, fibroblast growth factor 1 (FGF1) is expressed in ventral cochlear neurons, olfactory bulbs, and hippocampal neurons. The brain-specific FGF-1B promoter is active only in the brain, and is active in ependymal cells of ventricles and dopaminergic neurons in the brains of FGF-1B promoter-driven GFP (F1B-GFP) transgenic mice (Figure 1) [35]. Brain-specific 1B promoter of FGF1 gene facilitates the isolation of NSCs with self-renewal and multipotent capacities [36]. Implantations of F1B-GFP-selected NSCs from mouse brains were able to repair the damaged sciatic nerve of rats [36,37]. Taken together, F1B-GFP could be used to directly isolate NSCs from mouse brain, and the F1B-GFP-selected NSCs could be used in neuroregeneration.
.
Figure 1: The F1B-GFP+ cells were ependymal cells and distributed in the entire ventricular system of F1B-GFP transgenic mouse brain. The brains of F1B-GFP transgenic mice were stained with ependymal cell markers, S100β [(A) and (B)] and Vimentin [(C) and (D)] antibodies. Scale bar: 25 μm. Abbreviations: LV, lateral ventricle; 3V, third ventricle; D3V, dorsal third ventricle; CC, central canal [35].
View Figure 1
Differentiation of NSCs
Several neurotrophic factors, such as leukemia inhibitory factor (LIF), FGF1, platelet-derived growth factor (PDGF), nerve growth factor (NGF)/brain-derived neurotrophic factor (BDNF)/neutrophin-3 (NT-3), glial cell line-derived neurotrophic factor (GDNF), and retinoic acid (RA), promote differentiation of NSCs in mammalian neural development [1]. Previous studies also reported that levels of neurotrophic factors are changed in some neurodegenerative diseases, including Alzheimer's disease, Parkinson's disease, Huntington's disease and amyotrophic lateral sclerosis [38,39]. For examples, LIF is a member of the family of interleukin-6 cytokines, and mediated through a cell surface complex composed of LIF receptor β (LIFRβ) and gp130. LIF induced differentiation of NSCs into astrocytes, and the LIF knock-out mice display reduced astrocyte and microglial activation in the hippocampus [40,41]. FGF1 is expressed predominantly in neural tissues, and presents its effect through high-affinity cell surface receptors. FGF1 has been shown to promote mitogenesis of neuroblasts, to enhance both neurite initiation and elongation of retinal ganglion cells, and to stimulate neuronal differentiation and neurite outgrowth of PC12 cells [42,43]. The presence of FGF1 in the adult brain suggests a role for FGF1 in neural maintenance in addition to neurogenesis [1]. Therefore, Identification of external signals involved in the regulation of NSCs differentiation may allow for improved transplantation methods and augment the treatment of neurodegenerative disorders.
Mobilization of NSCs
NSCs resided in specialized niches of the adult mammalian brain that support their self-renewal and differentiation throughout life. Two regions continuously generate new neurons, which include subventricular zone (SVZ) of the lateral ventricles, and subgranular zone (SGZ) of the hippocampus. NSCs are largely quiescent in vivo, and occasionally become activated to proliferate. In SVZ, NSCs divide under both homeostasis and during regeneration to give rise to transit amplifying progenitors, which in turn generate neuroblasts that migrate to the olfactory bulb and give rise to olfactory bulb neurons [44-46]. NSCs that touch the ventricles are quiescent and are directly exposed to the cerebrospinal fluid (CSF). These cells contact ependymal cells via adherens junctions and thus anchor to the niche [47]. Vascular cell adhesion molecule 1 (VCAM1) receptors of the adherent junctions are located to the apical surface of NSCs in SVZ. Quiescent NSCs, upon disruption, could exit from the ependymal/CSF niche and result in neuron genesis in the SVZ [45,48]. In SGZ, new neurons are bred and functionally integrated into the granular cell layer of the dentate gyrus. Four types of neural progenitors (Type 1-4) are located in the SGZ during neurogenesis. Type 1 cells are likely the quiescent NSCs by the constant cell population size and proliferation rate. Types 2, 3 and 4 are daughter cells from Type 1 cells. Type 2 cells generally referred to as neural progenitor cells, while Type 3 cells are called neuroblasts. During the Type 3 stage, cells shift from the horizontal to vertical and begin radial migration into the granule cell layer. Type 4 cells cease mitosis, extend axons and lead to the development of mature granule cells [49]. In birds, new neuronal precursors were born in the ventricular zone, migrate into high vocal center (HVC) where they differentiated into neurons, which projected to robust nucleus of the arcopallium or became interneurons [26]. Sensitization of NSCs had been demonstrated in vivo through singing, exercise, stress and physical stimulations.
Learning (Singing):As shown in adult canaries, a learning behavior model, they modify their songs every year and continue to learn new songs in adulthood. Song syllables are delivered in a very stereotyped manner during the breeding season, and become less frequent and stereotypy after the breeding season ends. In the observations of song change results, the most dramatic change that occurs in the song circuits of adult canaries is neurogenesis and neuronal replacement in the HVC of brain region, an important nucleus in the brain that controls this learning behavior of canaries [50-52]. Adult neurogenesis is thought to provide neural plasticity used in forming and storing new memories. New neurons continue to be added to the HVC of adult canaries, where they replace older neurons that have died [26,53-56]. Therefore, the neurogenesis of adult canaries is responsible for the seasonal plasticity of learning behavior. The activating mechanisms of neuronal replacement in songbirds may contribute to the understanding of the repair of human brain damages and neuronal degeneration diseases.
Exercise:Physical activity showed activation of neural precursors in the adult neurogenic niches [57-59], and enhanced the learning and memory function in both mice [60,61] and human [62]. Comparison of hidden-platform water-maze learning, forced-exercise (yoked-swim controls), voluntary-exercise (running wheel) or standard-living (control) conditions revealed that running increases NSCs proliferation and neurogenesis. Voluntarily physical activity was sufficient for enhanced neurogenesis in the dentate gyrus of hippocampus of adult mice [14]. Several signaling pathways have been implicated in the exercise-mediated activation of neurogenesis, including hippocampal PKC activity, vascular endothelial growth factor (VEGF), insulin-like growth factoe 1 (IGF-1), bone morphogenetic protein (BMP) and growth hormone pathway [57]. In therapeutic strategy, physical exercise can rescue the psychiatric illness, aging dementia and alcoholic-reduced adult neurogenesis [57,58,63].
Response to stress (Stroke/ Muse):Stroke results from the occlusion of a cerebral artery leading to brain infarction, which is the most common cause of death and long-term disability in adulthood. Stroke is followed by activating of NSCs in the SVZ, and newborn neuroblasts migrate towards the injury sites of brain [64-67]. The most obvious site for post-stroke neurogenesis is in the SVZ. Stroke interacts with the SVZ adult neurogenesis through several signaling pathways, including erythropoietin [68], stromal derived factor 1 (SDF1), angiopoietin 1 [69] and calpains [67]. However, scientists revealed that most neuroblasts die before differentiating into functional mature neurons, which fail to integrate into the neuronal circuitry [66,7,71]. Therefore, discovery of efficient strategies in promoting post-stroke neurogenesis for improvement of patient recovery after brain injury is needed.
The multilineage-differentiating stress-enduing (Muse) cells are stress-tolerant and nontumoroigenic pluripotent stem cells, and show the ability to generate multiple cell types of the three germ layers. The Muse cells could be found in adult human mesenchymal population and adipose tissue without introducing exogenous genes [72-74]. Human Muse cells, intravenously injected, were able to integrate and differentiate into damaged sites including skin, muscle and liver tissues of mice [72,75]. Transplantation of human Muse cells into infarct brain of mice showed that Muse cells integrated into the injury sites and differentiated into neuronal cells in replacement of the lost neurons [76]. As shown in human adipose tissue-derived Muse cells (Muse-ATs), Muse-ATs reside within the adipocyte and stromal vascular fractions. Interactions of surounding cells with Muse-ATs resulted in their lineage plasticity, adipose tissue differentiation and repair, and the production and recruitment of signaling molecules in times of cellular stress [73,75]. Therefore, understanding of mechanisms in activating Muse and applications of Muse in replacement of the lost neurons are worthy for further investigations.
Physical stimulation (Magnetism/Ultrasound):Repetitive transcranial magnetic stimulation (rTMS) is a noninvasive approach of brain stimulation, which has been developed and demonstrated to be effective for Parkinson's disease and neuropsychiatric disorders [77-79]. Chronic rTMS promotes adult hippocampal neurogenesis and alleviates stress-related behaviors in mice with neuropsychiatric disorders. The studies provided further evidence that proliferation and neuronal differentiation of SVZ-derived NSCs of adult mice were increased after rTMS treatment [79-81].
BDNF is the neurotrophic factor involved in synaptic plasticity and neuron generation. Major depressive disorder patients showed lower BDNF levels in serum, and constitutively low rate of adult hippocampal cell proliferation [82]. Infusion of BDNF into the midbrain of rats showed antidepressant-like results [83]. Rats emit ultrasonic vocalizations in appetitive situations, like juvenile play or when tickled playfully. This tickling-induced ultrasound positively correlated with adult hippocampal cell proliferation as well as BDNF levels in mice [84-86]. Therefore, ultrasound stimulation might be an alternative strategy for treatment of major depressive disorder and Alzheimer's disease by increasing brain BDNF levels and neurogenesis in the hippocampus.
Cellular Reprogramming in Generation of iP-NSCs, iNeurons and iNSCs
Cellular reprogramming was used in the generation of iP-NSCs, iNeurons and iNSCs. Cellular reprogramming rendered as adult cells to an embryonic stem cell-like state by forced gene expression and/or factors treatment to maintain the properties of stem cells (such as iP-NSCs and iNSCs) or functional neurons (such as iNeurons) [87-90]. The primary advantage in the uses of cellular reprogramming is that adult cells are easily obtained from living animals. Different cell sources for cellular reprogramming were developed in previous studies, including blood cells [91-94], skin-derived fibroblasts [95], urine [96,97] and hair follicles [98]. Two different reprogramming processes, including indirect and direct methods, had been explored in the induction of neural progenitor cells [29]. Indirect reprogramming uses the same iPSCs factors (such as Sox2, c-Myc, Klf4 and Oct-4) for cellular reprogramming, designated iP-NSCs. In this method, addition of specific growth factors to the reprogramming medium can overcome fibroblasts to go through an intermediate unstable state. Direct reprogramming uses the neural-specific reprogramming factors for cellular reprogramming, allowing direct obtaining of neural progenitor cells (iNSCs) from fibroblasts. Both iP-NSCs and iNSCs showed neuronal cell differentiation and functional recovery in the transplantation of rodents [87,99].
iPSCs-derived neural stem cells (iP-NSCs)
The discovery of the technique that induces pluripotency in somatic cells (iPSCs) raised great hopes in regenerative medicine [100]. In the iPSCs studies, Kim et al. [101] first reported the cellular reprogramming of fibroblasts into induced neural precursor cells by using the same reprogramming factors of iPSCs. These transdifferentiated NSCs have the distinct advantage of being expandable in vitro and retaining the ability to give rise to multiple neuronal subtypes and glial cells. However, theses NSCs-like colonies lose their self-renewal capacity after 3-5 passages in vitro. They did not observe oligodendrocytes differentiation [101]. Their et al. [87] developed neurosphere-like colonies that could be expanded for more than 50 passages and do not depend on sustained expression of the reprogramming factors, which were generated by constitutively inducing Sox2, Klf4 and c-Myc while strictly limiting Oct4 activity. The authors transplanted their iP-NSCs into the brain of neonatal myelin-deficient rats. The results revealed that grafted iP-NSCs survive and give rise to differentiated neural cells in vivo [87]. Neuronal lineage-specific reprogramming factors (Ascl1, Brn2 and Myt1l) also have been shown to induce iPSCs into neural cells [90].
In clinical applications, iP-NSCs were thought as a good cell source for cell therapies and/or disease modeling and drug discovery in ALS, SMA and Alzheimer's disease. For example, recent animal study demonstrated that human iP-NSCs transplantation prolong the lifespan in ALS mouse model. ALS is a progressive, fatal, neurodegenerative disease caused by the death of motor neurons in the cortex, brain stem and spinal cord. The progressive neurodegeneration of ALS resulted in muscle atrophy and weakness, and respiratory failure. Astrocytes become dysfunctional during the progression of ALS [102,103]. As shown in Kondo et al. [104], transplantation of human iP-NSCs into the spinal cord of ALS mice improves lifespan of the treated mice. The patient-derived iPSCs of SMA and Alzheimer's disease were also successfully used in disease modeling [105,106].
The concerns about iP-NSCs were due to viral infection with overexpression of c-Myc oncogene, which have shown increased tumorigenicity [17,107-110]. Therefore, to explore alternative stem cell sources that do not confer the disadvantage of iPSCs but are widely multipotent for clinical applications are needed in the future.
iNeurons
To resolve the clinical hurdle of the iPSC derivations, the iNeurons were developed in overcoming the disadvantage of teratoma formation by the pluripotent cells. Recent studies showed that the generation of iNeurons is fast, efficient and devoid of tumorigenic pluripotent stem cells [89], which provide a novel and powerful tool that were chosen in studying neurological disease, drug discovery and regeneration medicine. Vierbuchen et al. [89] revealed the direct conversion of fibroblasts to functional neurons by defined factors. The results reported that most efficient conversions were achieved when Ascl1 and Brn2 were combined with Myt1l (termed BAM pool). In the BAM infection, iNeurons showed the expressions of neuronal markers, and were capable in forming functional synapses by co-cultured with astrocytes [89]. Moreover, hepatocytes-derived iNeurons by the BAM factors were developed and have acquired functional properties of mature neurons [111].
Encouraged by findings in mouse cells, human fibroblasts could also be directly induced to become functional neurons [90,112-114]. The direct conversion of human fibroblasts into functional neurons using BAM factors had been reported [115]. NeuroD1 co-infected with BAM (BAMN) factors generated the most mature neuronal cells in human fibroblasts by morphological and functional characterization [90]. The miRNAs (particularly the miR-124) showed significant contribution in reprogramming of iNeurons. Expression of miR-9/9* and miR-124 in human fibroblasts induces their conversion into neurons. The miR-9/9*-124-DAM-(miR-9/9*-124 together with NeuroD2, Ascl1 and Myt1l)-converted iNeurons showed positive action potentials of functional evaluation [112,116]. Combination of miR-124, Brn2 and Myt1l (termed IBM pool) factors also reprogram human postnatal and adult primary dermal fibroblasts into functional neurons [113]. Small-molecule cocktails could convert mouse and human fibroblasts into functional neurons without exogenous genetic factors [117,118].
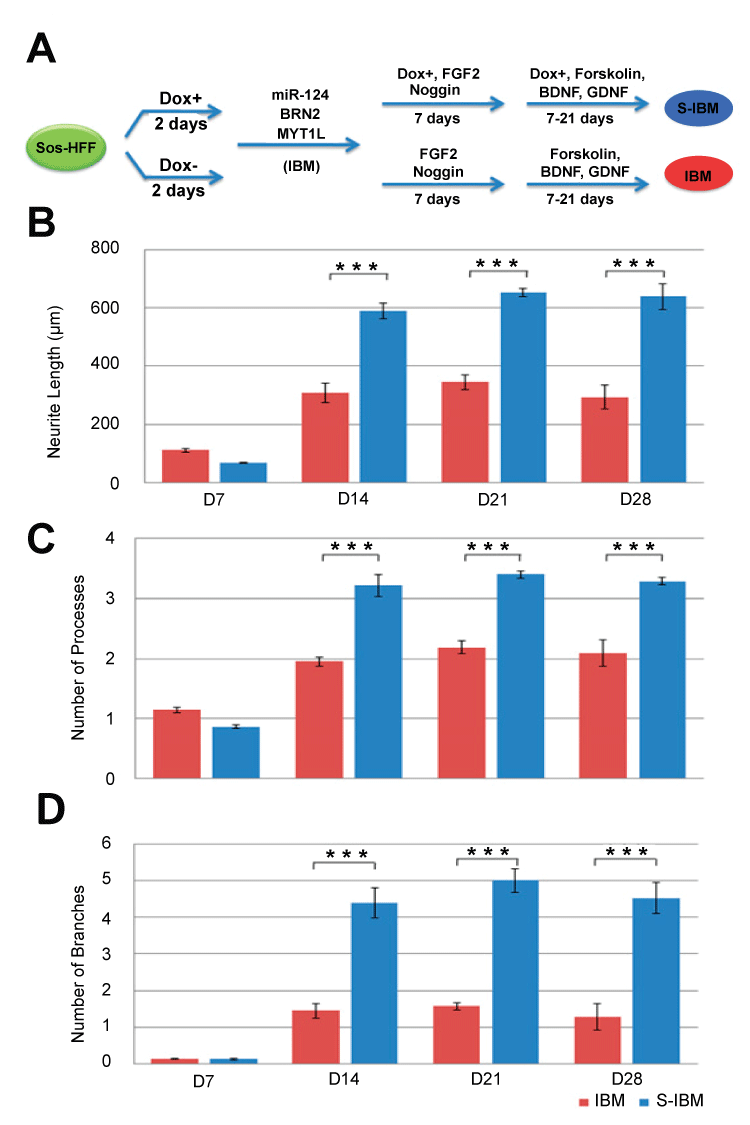
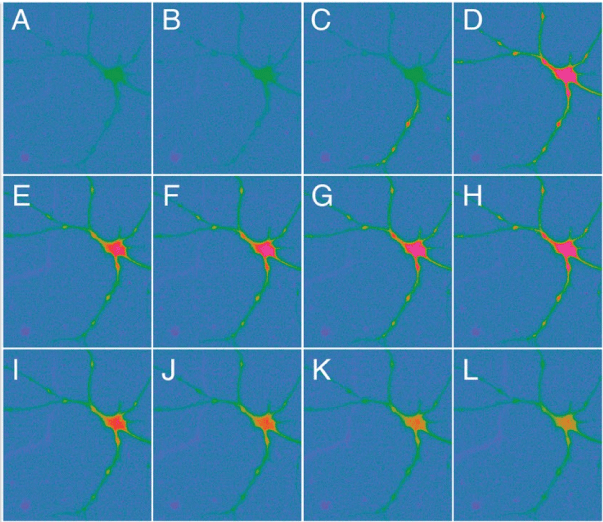
In particular, our finding demonstrated that SH2B adaptor protein 1β (SH2B1) can enhance neurite outgrowth of iNeurons which were reprogrammed from human foreskin fibroblasts, when combined with IBM factors under defined conditions. These SH2B1-enhanced iNeurons (termed SIBM-iNeurons) showed canonical morphology and expressing multiple neural markers and functional proteins for neurotransmitter release and exhibited action potentials. As shown in figure 2, SH2B1 enhances the length of neurite outgrowth and the numbers of processes and branches of human iNeurons induced by IBM factors. SIBM-iNeurons also showed calcium influx activity upon glutamate stimulation (Figure 3). This study demonstrated that the combination of SIBM factors in reprogramming of human fibroblasts is 10 times more efficiency than other combinations and can exhibit action potential as early as day 14 [114].
.
Figure 2: SH2B1 enhanced the length of neurite outgrowth and the numbers of processes and branches of human iNeurons induced by IBM factors. The schema showed the experimental protocol for reprogramming fibroblast cells into neurons (A). Within 28 days of infection, SIBM-iNeurons showed twofold more total neurite outgrowth than IBM-iNeurons at days 14, 21, and 28. n = 3; ***, p < 0.001 (B).Within 28 days of infection, SIBM-iNeurons showed significantly more neuronal processes (C) and branches (D) than IBM-iNeurons at days 14, 21, and 28. n = 3; ***, p < 0.001. Abbreviations: SIBM comprises SH2B1, miR124, Brn2 and Myt1l [114].
View Figure 2
.
Figure 3: SIBM-iNeurons showed Ca2+ influx activity upon glutamate stimulation at Day 28. SIBM-iNeurons were labeled with Fura2-AM, which is an intracellular Ca2+ indicator. Images were taken at consecutive intervals of 3.1 seconds after glutamate injection. The red color indicates the uptake intensity of Fura2-AM [114].
View Figure 3
iNSCs
Unlike iNeurons, iNSCs are capable of self-renewing, as well as differentiating into neurons and astrocytes, which means that iNSCs (Table 1) would provide an unlimited source of neuronal cells for clinical applications [99,119]. Lujan et al. [88] reported that Sox2 and FoxG1 are capable of generating clonal self-renewing, bipotent iNSCs that gave rise to astrocytes and functional neurons. When Brn2 was added to Sox2 and FoxG1, tripotent iNSCs that could be differentiated not only into neurons and astrocytes but also into oligodendrocytes were obtained [88]. In vivo assessment, iNSCs labeled with GFP were transplanted into the SVZ of adult mice. The results demonstrated that iNSCs exhibit cell morphology, gene expression, epigenetic features, differentiation potential and self-renewing capacity, as well as in vitro and in vivo functionality similar to those of wild-type NSCs. These iNSCs did not generate teratomas after injection into immunosuppressed mice, and the expression levels of the endogenous NSC factors were similar to those of control NSCs [99]. Furthermore, iNSCs achieve long-term survival and functional integration in the adult mouse brain. The results demonstrated that iNSCs long-term transplantation is a safe procedure and might be a valuable tool for cell therapies of neuroregeneration [120]. Moreover, the use of one transcription factor (such as Sox2, Oct4 or Nanog) can also reprogram the mature cells into iNSCs [121,122]. In Corti et al. [122], ectopic expression of the single reprogramming factor (Sox2, Oct4 or Nanog) into human cortical astrocytes gave rise to colonies of small cells resembling NSCs as well as neurospheres. The number of colonies obtained was significantly greater with Nanog than with Sox2 or Oct4. In addition, Sheng et al. [123] showed that direct reprogramming of Sertoli cells into iNSCs by nine transcription factors (Sox2, Klf4, c-Myc, Brn2, Pax6, Id1, Hex1, Ngn2 and Ascl1) [123]. These iNSCs could differentiate into neurons, astrocytes and oligodendrocytes in vivo.
![]()
Table 1: Generation of iNSCs by various neuronal-specific transcription factors from somatic cells.
View Table 1
The iP-NSCs-like colonies were generated with low efficiency and could not self-renew, and the cells apparently lacked the ability to differentiate into oligodendrocytes [101]. Moreover, both undifferentiated iP-NSCs harbor potential tumorigenic risks [29,107,108,124]. iNSCs that were induced directly from fibroblasts using neuronal-specific transcription factors bypassed a partial or complete pluripotent state. Therefore, iNSCs reduced the risk of teratoma formation due to the persistent undifferentiated cells. Thus, it would be desirable to convert fibroblasts into proliferative neural precursor cells instead of neurons and iPSCs-derived cells.
Applications of NSCs and iNeurons in Cell Therapy, Disease Modeling and Drug Discovery
Cell-based therapy could aid in alleviating symptoms or even reversing the progression of neurodegenerative diseases and nerve injuries. Neurodegeneration can disrupt molecular pathways, synapses, neuronal subpopulations, and local circuits in specific brain regions, as well as higher-order neural networks. Abnormal network activities may result in a vicious cycle in impairing the integrity and functions of neurons and synapses.
NSCs treatment in PNI repair
Neuroregeneration of PNI is a complex process with Wallerian degeneration. After injury, the axonal skeleton disintegrates, and the axonal membrane breaks apart. The axonal degeneration is followed by degradation of the myelin sheath and infiltration by macrophages which serve to clear the debris from the degeneration [125,126]. Transplantation of NSCs provide effective therapies for many neurological diseases and injuries, which clearly explain the benefits observed after cell therapy [127]. The paracrine action of growth factors, cytokines, and hormones that are secreted or released by transplanted cells has been shown to provide most of the benefits after stem/progenitor cell administration. Mechanisms of peripheral neuroregeneration in NSCs administration include differentiation into Schwann cells, secretion of neurotrophic factors and promoting myelination [128]. Our studies showed that F1B-GFP-selected NSCs from mouse brain were able to repair the damaged sciatic nerve [9,37]. These NSCs were differentiated into Schwann cells, which contribute to the development of microenvironment that stimulates axonal regeneration in repairing of PNI (unpublished results).
iNeurons in disease modeling and drug discovery
iNeurons generated via direct conversion of neuron do not pass through a pluripotent state, are not tumorigenic, and may serve as an intresting alternative to iPSC-derived cells for generating patient- and/or disease-specific neurons. In recent years, iNeurons have been utilized in modeling of neurodegenerative diseases such as Parkinson's disease [115,129,130], ALS and SMA [131], Dravet syndrome and mild febrile seizures [132], Glaucoma [133], Frontotemporal lobar degeneration (FTLD) [134], autism spectrum disorder [135] and Alzheimer's disease [118]. The results are been summarized in table 2.
![]()
Table 2: Applications of iNeurons in disease modeling and drug discovery.
View Table 2
A key feature of Parkinson's disease is progressive degeneration and death of dopaminergic neuron (DAs) in the substantia niagra region of the midbrain. Mouse and human fibroblast-derived DA showed positive DA markers, dopamine release and DA electrophysiology [115,129,130]. Injection of induced DA in rats presented attenuated rotational behavior at 8 weeks post injection. Long-term engraftment revealed that induced DA retained their DA neuron-like properties in vivo for up to 16 weeks post injection [130]. Therefore, directly reprogrammed DA are a promising source of cell-replacement therapy for PD.
In ALS and SMA, the motor neurons control the contraction of muscle fibers actuating movement, which are selective targets of degeneration in ALS ans SMA. The induced motor neurons showed gene expression signature, electrophysiological characteristics of motor neurons and forming functional synapses with muscle and sensitivity to disease stimuli of ALS. In vivo functional tests demonstrated that induced motor neurons were able to engraft, migrate to appropriate sites of integration, and correctly respond to guidance cues, projecting their axons out of the central nervous system in the developing chick spinal cord [131]. Therefore, this induced motor neuron is a suitable substrate for in vitro studies of motor neuron function, disease, and injury.
The Dravet syndrome and mild febrile seizures arise due to genetic mutation of SCN1A, the gene encoding α1 pore-forming subunit of the Nav 1.1 voltage-gated sodium channel. The patient-specific induced-glutamatergic neurons (GAs) showed delayed inactivation of sodium channels, indicating that theses neurons can be potentially used for disease modeling and drug screening [132].
Glaucoma is one of the neurodegerative disease and was characterized by accelerated death of retinal ganglion cells (RGC), which ultimately leads to progressive visual field loss and eventual blindness. Adenovirus-mediated transduction of three reprogramming factors (Table 2) can directly convert mouse fibroblasts to RGC-like neurons which expressed RGC markers and exhibited membrane action potential of functional RGC neurons. The cells showed great potential in retinal degenerative disease modeling [133].
Mobilization of NSCs as a means for neuroregeneration
A milestone is marked in our understanding of the brain with the recent awareness that the adult nervous system can generate new neurons. Table 3 summaries that neurogenesis in the adult brain can be mobilized via different ways, and the NSC mobilizations may improve the symptoms of neurodegenerative disorders. For adult brain neurogenesis, sensitization through stress and physical stimulations, and exposure to enriched environment can increase neurogenesis in animal models. Enriched environment consists of expanded learning opportunities, increased social interactions, more physical activity and larger housing [14,136,137]. As shown in table 3, learning, stroke and magnetism stimulation induced SVZ neurogenesis, and exercise and ultrasound stimulation induced hippocampal neurogenesis. For clinical investigations, exercise enhanced learning and memory function in both mice and human with neurodegenerative diseases [60-62]. Treatment of rTMS improves the symptoms of Parkinson's disease, and induced endogenous dopamine release in the ipsilateral dorsal striatum [78]. Ultrasound increased BDNF levels and cell proliferation in adult hippocampus of mice. Ultrasound might be used in treatment of major depressive disorder [138]. Therefore, mobilizations of NSCs could be a worthwhile strategy for regeneration of neurons in neurodegenerative diseases in vivo.
![]()
Table 3: Mobilization of NSCs in neurogenesis of the adult brain.
View Table 3
In spite of the rapid progress in stem cell research, several critical challenges of stem cell-based therapy in repairing neurodegenerative diseases remain to be resolved. These challenges include (1) the source of NSCs should be personalized; (2) the protocols to activate human NSCs should be properly established; (3) the isolation methods of human NSCs should be standardized; (4) the clinical efficacy of activations or transplants of NSCs should be evaluated in more adequate animal models; and (5) the mechanism of intrinsic repairing of neurodegeneration diseases needs to be better characterized. Mobilizations of NSCs by different means can provide unlimited cell sources in the adult brain. The mobilized NSCs could in turn regenerate neurons in repairing of neurodegenerative diseases in vivo. Moreover, patient-derived cells are ideal NSC sources for autologous cell transplantation because they can avoid immune rejection. In particular, generation of iNeurons from patient is fast, efficient and devoid of tumorigenicity that might be conferred by iP-NSCs. Therefore, mobilizations of NSCs in vivo and cellular reprogramming for cell transplantation are expected to alleviate symptoms or even reverse the progression of various neurodegenerative diseases.
References
-
Hsu YC, Lee DC, Chiu IM (2007) Neural stem cells, neural progenitors, and neurotrophic factors. Cell Transplant 16: 133-150.
-
Lindvall O, Kokaia Z (2005) Stem cell therapy for human brain disorders. Kidney Int 68: 1937-1939.
-
Melone MA, Jori FP, Peluso G (2005) Huntington's disease: new frontiers for molecular and cell therapy. Curr Drug Targets 6: 43-56.
-
Schultz SS (2005) Adult stem cell application in spinal cord injury. Curr Drug Targets 6: 63-73.
-
MacDonald SC, Fleetwood IG, Hochman S, Dodd JG, Cheng GK, et al. (2003) Functional motor neurons differentiating from mouse multipotent spinal cord precursor cells in culture and after transplantation into transected sciatic nerve. J Neurosurg 98: 1094-1103.
-
Murakami T, Fujimoto Y, Yasunaga Y, Ishida O, Tanaka N, et al. (2003) Transplanted neuronal progenitor cells in a peripheral nerve gap promote nerve repair. Brain Res 974: 17-24.
-
Aquino JB, Hjerling-Leffler J, Koltzenburg M, Edlund T, Villar MJ, et al. (2006) In vitro and in vivo differentiation of boundary cap neural crest stem cells into mature Schwann cells. Exp Neurol 198: 438-449.
-
Heine W, Conant K, Griffin JW, Hoke A (2004) Transplanted neural stem cells promote axonal regeneration through chronically denervated peripheral nerves. Exp Neurol 189: 231-240.
-
Hsu SH, Su CH, Chiu IM (2009) A novel approach to align adult neural stem cells on micropatterned conduits for peripheral nerve regeneration: a feasibility study. Artif Organs 33: 26-35.
-
Lindvall O, Kokaia Z (2006) Stem cells for the treatment of neurological disorders. Nature 441: 1094-1096.
-
Englund U, Björklund A, Wictorin K (2002) Migration patterns and phenotypic differentiation of long-term expanded human neural progenitor cells after transplantation into the adult rat brain. Brain Res Dev Brain Res 134: 123-141.
-
Kulbatski I, Mothe AJ, Nomura H, Tator CH (2005) Endogenous and exogenous CNS derived stem/progenitor cell approaches for neurotrauma. Curr Drug Targets 6: 111-126.
-
Martens DJ, Seaberg RM, van der Kooy D (2002) In vivo infusions of exogenous growth factors into the fourth ventricle of the adult mouse brain increase the proliferation of neural progenitors around the fourth ventricle and the central canal of the spinal cord. Eur J Neurosci 16: 1045-1057.
-
van Praag H, Kempermann G, Gage FH (1999) Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci 2: 266-270.
-
Kempermann G, Kuhn HG, Gage FH (1997) Genetic influence on neurogenesis in the dentate gyrus of adult mice. Proc Natl Acad Sci U S A 94: 10409-10414.
-
Maherali N, Sridharan R, Xie W, Utikal J, Eminli S, et al. (2007) Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell 1: 55-70
-
Okita K, Ichisaka T, Yamanaka S (2007) Generation of germline-competent induced pluripotent stem cells. Nature 448: 313-317.
-
Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, et al. (2007) In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448: 318-324.
-
Yamanaka S, Takahashi K (2006) [Induction of pluripotent stem cells from mouse fibroblast cultures]. Tanpakushitsu Kakusan Koso 51: 2346-2351.
-
Altman J, Das GD (1965) Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol 124: 319-335.
-
Kuhn HG, Dickinson-Anson H, Gage FH (1996) Neurogenesis in the dentate gyrus of the adult rat: age-related decrease of neuronal progenitor proliferation. J Neurosci 16: 2027-2033.
-
Kempermann G, Kuhn HG, Gage FH (1997) More hippocampal neurons in adult mice living in an enriched environment. Nature 386: 493-495.
-
Lois C, Alvarez-Buylla A (1993) Proliferating subventricular zone cells in the adult mammalian forebrain can differentiate into neurons and glia. Proc Natl Acad Sci U S A 90: 2074-2077.
-
Gould E, Tanapat P, McEwen BS, Flügge G, Fuchs E (1998) Proliferation of granule cell precursors in the dentate gyrus of adult monkeys is diminished by stress. Proc Natl Acad Sci U S A 95: 3168-3171.
-
Eriksson PS, Perfilieva E, Björk-Eriksson T, Alborn AM, Nordborg C, et al. (1998) Neurogenesis in the adult human hippocampus. Nat Med 4: 1313-1317.
-
Barnea A, Pravosudov V (2011) Birds as a model to study adult neurogenesis: bridging evolutionary, comparative and neuroethological approaches. Eur J Neurosci 34: 884-907.
-
Zhang H, Wei YT, Tsang KS, Sun CR, Li J, et al. (2008) Implantation of neural stem cells embedded in hyaluronic acid and collagen composite conduit promotes regeneration in a rabbit facial nerve injury model. J Transl Med 6: 67.
-
Shi Y, Zhou L, Tian J, Wang Y (2009) Transplantation of neural stem cells overexpressing glia-derived neurotrophic factor promotes facial nerve regeneration. Acta Otolaryngol 129: 906-914.
-
Ruggieri M, Riboldi G, Brajkovic S, Bucchia M, Bresolin N, et al. (2014) Induced neural stem cells: methods of reprogramming and potential therapeutic applications. Prog Neurobiol 114: 15-24.
-
Ghods AJ, Irvin D, Liu G, Yuan X, Abdulkadir IR, et al. (2007) Spheres isolated from 9L gliosarcoma rat cell line possess chemoresistant and aggressive cancer stem-like cells. Stem Cells 25: 1645-1653.
-
Shi Y, Sun G, Zhao C, Stewart R (2008) Neural stem cell self-renewal. Crit Rev Oncol Hematol 65: 43-53.
-
Chen JH, Lee DC, Chen MS, Ko YC, Chiu IM (2015) Inhibition of neurosphere formation in neural stem/progenitor cells by acrylamide. Cell Transplant 24: 779-796.
-
Johe KK, Hazel TG, Muller T, Dugich-Djordjevic MM, McKay RD (1996) Single factors direct the differentiation of stem cells from the fetal and adult central nervous system. Genes Dev 10: 3129-3140.
-
Suh H, Consiglio A, Ray J, Sawai T, D'Amour KA, et al. (2007) In vivo fate analysis reveals the multipotent and self-renewal capacities of Sox2+ neural stem cells in the adult hippocampus. Cell Stem Cell 1: 515-528.
-
Chen MS, Lin HK, Chiu H, Lee DC, Chung YF, et al. (2015) Human FGF1 promoter is active in ependymal cells and dopaminergic neurons in the brains of F1B-GFP transgenic mice. Dev Neurobiol 75: 232-248.
-
Hsu YC, Lee DC, Chen SL, Liao WC, Lin JW, et al. (2009) Brain-specific 1B promoter of FGF1 gene facilitates the isolation of neural stem/progenitor cells with self-renewal and multipotent capacities. Dev Dyn 238: 302-314.
-
Lin YL, Jen JC, Hsu SH, Chiu IM (2008) Sciatic nerve repair by microgrooved nerve conduits made of chitosan-gold nanocomposites. Surg Neurol 70 Suppl 1: S1:9-18.
-
Lai HC, Wu MJ, Chen PY, Sheu TT, Chiu SP, et al. (2011) Neurotrophic effect of citrus 5-hydroxy-,6,7,8,3',4'-hexamethoxyflavone: promotion of neurite outgrowth via cAMP/PKA/CREB pathway in PC12 cells. PLoS One 6: e28280.
-
Levy YS, Gilgun-Sherki Y, Melamed E, Offen D (2005) Therapeutic potential of neurotrophic factors in neurodegenerative diseases. BioDrugs 19: 97-127.
-
Holmberg KH, Patterson PH (2006) Leukemia inhibitory factor is a key regulator of astrocytic, microglial and neuronal responses in a low-dose pilocarpine injury model. Brain Res 1075: 26-35.
-
Koblar SA, Turnley AM, Classon BJ, Reid KL, Ware CB, et al. (1998) Neural precursor differentiation into astrocytes requires signaling through the leukemia inhibitory factor receptor. Proc Natl Acad Sci U S A 95: 3178-3181.
-
Chang YJ, Chen KW, Chen CJ, Lin MH, Sun YJ, et al. (2014) SH2B1β interacts with STAT3 and enhances fibroblast growth factor 1-induced gene expression during neuronal differentiation. Mol Cell Biol 34: 1003-1019.
-
Lin WF, Chen CJ, Chang YJ, Chen SL, Chiu IM, et al. (2009) SH2B1beta enhances fibroblast growth factor 1 (FGF1)-induced neurite outgrowth through MEK-ERK1/2-STAT3-Egr1 pathway. Cell Signal 21: 1060-1072.
-
Codega P, Silva-Vargas V, Paul A, Maldonado-Soto AR, Deleo AM, et al. (2014) Prospective identification and purification of quiescent adult neural stem cells from their in vivo niche. Neuron 82: 545-559.
-
Silva-Vargas V, Crouch EE, Doetsch F (2013) Adult neural stem cells and their niche: a dynamic duo during homeostasis, regeneration, and aging. Curr Opin Neurobiol 23: 935-942.
-
Pastrana E, Silva-Vargas V, Doetsch F (2011) Eyes wide open: a critical review of sphere-formation as an assay for stem cells. Cell Stem Cell 8: 486-498.
-
Mirzadeh Z, Merkle FT, Soriano-Navarro M, Garcia-Verdugo JM, Alvarez-Buylla A (2008) Neural stem cells confer unique pinwheel architecture to the ventricular surface in neurogenic regions of the adult brain. Cell Stem Cell 3: 265-278.
-
Kokovay E, Wang Y, Kusek G, Wurster R, Lederman P, et al. (2012) VCAM1 is essential to maintain the structure of the SVZ niche and acts as an environmental sensor to regulate SVZ lineage progression. Cell Stem Cell 11: 220-230.
-
Hanson ND, Owens MJ, Nemeroff CB (2011) Depression, antidepressants, and neurogenesis: a critical reappraisal. Neuropsychopharmacology 36: 2589-2602.
-
Goldman SA, Nottebohm F (1983) Neuronal production, migration, and differentiation in a vocal control nucleus of the adult female canary brain. Proc Natl Acad Sci U S A 80: 2390-2394.
-
Paton JA, Nottebohm FN (1984) Neurons generated in the adult brain are recruited into functional circuits. Science 225: 1046-1048.
-
Nottebohm F (1985) Neuronal replacement in adulthood. Ann N Y Acad Sci 457: 143-161.
-
Scharff C, Adam I (2013) Neurogenetics of birdsong. Curr Opin Neurobiol 23: 29-36.
-
Doupe AJ (1994) Songbirds and adult neurogenesis: a new role for hormones. Proc Natl Acad Sci U S A 91: 7836-7838.
-
Nottebohm F, O'Loughlin B, Gould K, Yohay K, Alvarez-Buylla A (1994) The life span of new neurons in a song control nucleus of the adult canary brain depends on time of year when these cells are born. Proc Natl Acad Sci U S A 91: 7849-7853.
-
Kirn J, O'Loughlin B, Kasparian S, Nottebohm F (1994) Cell death and neuronal recruitment in the high vocal center of adult male canaries are temporally related to changes in song. Proc Natl Acad Sci U S A 91: 7844-7848.
-
Vukovic J, Blackmore DG, Jhaveri D, Bartlett PF (2011) Activation of neural precursors in the adult neurogenic niches. Neurochem Int 59: 341-346.
-
Yang TT, Lo CP, Tsai PS, Wu SY, Wang TF, et al. (2015) Aging and Exercise Affect Hippocampal Neurogenesis via Different Mechanisms. PLoS One 10: e0132152.
-
Jun H, Mohammed Qasim Hussaini S, Rigby MJ, Jang MH (2012) Functional role of adult hippocampal neurogenesis as a therapeutic strategy for mental disorders. Neural Plast 2012: 854285.
-
van Praag H, Shubert T, Zhao C, Gage FH (2005) Exercise enhances learning and hippocampal neurogenesis in aged mice. J Neurosci 25: 8680-8685.
-
Van der Borght K, Havekes R, Bos T, Eggen BJ, Van der Zee EA (2007) Exercise improves memory acquisition and retrieval in the Y-maze task: relationship with hippocampal neurogenesis. Behav Neurosci 121: 324-334.
-
Erickson KI, Voss MW, Prakash RS, Basak C, Szabo A, et al. (2011) Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci U S A 108: 3017-3022.
-
Crews FT, Nixon K, Wilkie ME (2004) Exercise reverses ethanol inhibition of neural stem cell proliferation. Alcohol 33: 63-71.
-
Gage FH (2000) Mammalian neural stem cells. Science 287: 1433-1438.
-
Jin K, Wang X, Xie L, Mao XO, Zhu W, et al. (2006) Evidence for stroke-induced neurogenesis in the human brain. Proc Natl Acad Sci U S A 103: 13198-13202.
-
Arvidsson A, Collin T, Kirik D, Kokaia Z, Lindvall O (2002) Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat Med 8: 963-970.
-
Machado VM, Morte MI, Carreira BP, Azevedo MM, Takano J, et al. (2015) Involvement of calpains in adult neurogenesis: implications for stroke. Front Cell Neurosci 9: 22.
-
Tsai PT, Ohab JJ, Kertesz N, Groszer M, Matter C, et al. (2006) A critical role of erythropoietin receptor in neurogenesis and post-stroke recovery. J Neurosci 26: 1269-1274.
-
Ohab JJ, Fleming S, Blesch A, Carmichael ST (2006) A neurovascular niche for neurogenesis after stroke. J Neurosci 26: 13007-13016.
-
Kaneko N, Sawamoto K (2009) Adult neurogenesis and its alteration under pathological conditions. Neurosci Res 63: 155-164.
-
Ma DK, Bonaguidi MA, Ming GL, Song H (2009) Adult neural stem cells in the mammalian central nervous system. Cell Res 19: 672-682.
-
Kuroda Y, Kitada M, Wakao S, Nishikawa K, Tanimura Y, et al. (2010) Unique multipotent cells in adult human mesenchymal cell populations. Proc Natl Acad Sci U S A 107: 8639-8643.
-
Heneidi S, Simerman AA, Keller E, Singh P, Li X, et al. (2013) Awakened by cellular stress: isolation and characterization of a novel population of pluripotent stem cells derived from human adipose tissue. PLoS One 8: e64752.
-
Ogura F, Wakao S, Kuroda Y, Tsuchiyama K, Bagheri M, et al. (2014) Human adipose tissue possesses a unique population of pluripotent stem cells with nontumorigenic and low telomerase activities: potential implications in regenerative medicine. Stem Cells Dev 23: 717-728.
-
Simerman AA, Dumesic DA, Chazenbalk GD1 (2014) Pluripotent muse cells derived from human adipose tissue: a new perspective on regenerative medicine and cell therapy. Clin Transl Med 3: 12.
-
Yamauchi T, Kuroda Y, Morita T, Shichinohe H, Houkin K, et al. (2015) Therapeutic effects of human multilineage-differentiating stress enduring (MUSE) cell transplantation into infarct brain of mice. PLoS One 10: e0116009.
-
Kammer T, Spitzer M (2012) Brain stimulation in psychiatry: methods and magnets, patients and parameters. Curr Opin Psychiatry 25: 535-541.
-
Arias-Carrión O (2008) Basic mechanisms of rTMS: Implications in Parkinson's disease. Int Arch Med 1: 2.
-
Zhang Y, Mao RR, Chen ZF, Tian M, Tong DL, et al. (2014) Deep-brain magnetic stimulation promotes adult hippocampal neurogenesis and alleviates stress-related behaviors in mouse models for neuropsychiatric disorders. Mol Brain 7: 11.
-
Abbasnia K, Ghanbari A, Abedian M, Ghanbari A, Sharififar S, et al. (2015) The effects of repetitive transcranial magnetic stimulation on proliferation and differentiation of neural stem cells. Anat Cell Biol 48: 104-113.
-
Ueyama E, Ukai S, Ogawa A, Yamamoto M, Kawaguchi S, et al. (2011) Chronic repetitive transcranial magnetic stimulation increases hippocampal neurogenesis in rats. Psychiatry Clin Neurosci 65: 77-81.
-
Karege F, Perret G, Bondolfi G, Schwald M, Bertschy G, et al. (2002) Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Res 109: 143-148.
-
Siuciak JA, Lewis DR, Wiegand SJ, Lindsay RM (1997) Antidepressant-like effect of brain-derived neurotrophic factor (BDNF). Pharmacol Biochem Behav 56: 131-137.
-
Wöhr M, Kehl M, Borta A, Schänzer A, Schwarting RK, et al. (2009) New insights into the relationship of neurogenesis and affect: tickling induces hippocampal cell proliferation in rats emitting appetitive 50-kHz ultrasonic vocalizations. Neuroscience 163: 1024-1030.
-
Yamamuro T, Senzaki K, Iwamoto S, Nakagawa Y, Hayashi T, et al. (2010) Neurogenesis in the dentate gyrus of the rat hippocampus enhanced by tickling stimulation with positive emotion. Neurosci Res 68: 285-289.
-
Tufail Y, Matyushov A, Baldwin N, Tauchmann ML, Georges J, et al. (2010) Transcranial pulsed ultrasound stimulates intact brain circuits. Neuron 66: 681-694.
-
Thier M, Wörsdörfer P, Lakes YB, Gorris R, Herms S, et al. (2012) Direct conversion of fibroblasts into stably expandable neural stem cells. Cell Stem Cell 10: 473-479.
-
Lujan E, Chanda S, Ahlenius H, Südhof TC, Wernig M (2012) Direct conversion of mouse fibroblasts to self-renewing, tripotent neural precursor cells. Proc Natl Acad Sci U S A 109: 2527-2532.
-
Vierbuchen T, Ostermeier A, Pang ZP, Kokubu Y, Sudhof TC, et al. (2010) Direct conversion of fibroblasts to functional neurons by defined factors. Nature 463: 1035-1041.
-
Pang ZP, Yang N, Vierbuchen T, Ostermeier A, Fuentes DR, et al. (2011) Induction of human neuronal cells by defined transcription factors. Nature 476: 220-223.
-
Tan HK, Toh CX, Ma D, Yang B, Liu TM, et al. (2014) Human finger-prick induced pluripotent stem cells facilitate the development of stem cell banking. Stem Cells Transl Med 3: 586-598.
-
Okita K, Yamakawa T, Matsumura Y, Sato Y, Amano N, et al. (2013) An efficient nonviral method to generate integration-free human-induced pluripotent stem cells from cord blood and peripheral blood cells. Stem Cells 31: 458-466.
-
Geti I, Ormiston ML, Rouhani F, Toshner M, Movassagh M, et al. (2012) A practical and efficient cellular substrate for the generation of induced pluripotent stem cells from adults: blood-derived endothelial progenitor cells. Stem Cells Transl Med 1: 855-865.
-
Staerk J, Dawlaty MM, Gao Q, Maetzel D, Hanna J, et al. (2010) Reprogramming of human peripheral blood cells to induced pluripotent stem cells. Cell Stem Cell 7: 20-24.
-
Yoshikawa K, Naitoh M, Kubota H, Ishiko T, Aya R, et al. (2013) Multipotent stem cells are effectively collected from adult human cheek skin. Biochem Biophys Res Commun 431: 104-110.
-
Zhou T, Benda C, Dunzinger S, Huang Y, Ho JC, et al. (2012) Generation of human induced pluripotent stem cells from urine samples. Nat Protoc 7: 2080-2089.
-
Bharadwaj S, Liu G, Shi Y, Wu R, Yang B, et al. (2013) Multipotential differentiation of human urine-derived stem cells: potential for therapeutic applications in urology. Stem Cells 31: 1840-1856.
-
Wang Y, Liu J, Tan X, Li G, Gao Y, et al. (2013) Induced pluripotent stem cells from human hair follicle mesenchymal stem cells. Stem Cell Rev 9: 451-460.
-
Han DW, Tapia N, Hermann A, Hemmer K, Höing S, et al. (2012) Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell 10: 465-472.
-
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126: 663-676.
-
Kim J, Efe JA, Zhu S, Talantova M, Yuan X, et al. (2011) Direct reprogramming of mouse fibroblasts to neural progenitors. Proc Natl Acad Sci U S A 108: 7838-7843.
-
Haidet-Phillips AM, Maragakis NJ (2015) Neural and glial progenitor transplantation as a neuroprotective strategy for Amyotrophic Lateral Sclerosis (ALS). Brain Res.
-
Haidet-Phillips AM, Hester ME, Miranda CJ, Meyer K, Braun L, et al. (2011) Astrocytes from familial and sporadic ALS patients are toxic to motor neurons. Nat Biotechnol 29: 824-828.
-
Kondo T, Funayama M, Tsukita K, Hotta A, Yasuda A, et al. (2014) Focal transplantation of human iPSC-derived glial-rich neural progenitors improves lifespan of ALS mice. Stem Cell Reports 3: 242-249.
-
Kondo T, Asai M, Tsukita K, Kutoku Y, Ohsawa Y, et al. (2013) Modeling Alzheimer's disease with iPSCs reveals stress phenotypes associated with intracellular Abeta and differential drug responsiveness. Cell Stem Cell 12: 487-496.
-
Boza-Moran MG, Martinez-Hernandez R, Bernal S, Wanisch K, Also-Rallo E, et al. (2015) Decay in survival motor neuron and plastin 3 levels during differentiation of iPSC-derived human motor neurons. Sci Rep 5: 11696.
-
Fong CY, Gauthaman K, Bongso A (2010) Teratomas from pluripotent stem cells: A clinical hurdle. J Cell Biochem 111: 769-781.
-
Miura K, Okada Y, Aoi T, Okada A, Takahashi K, et al. (2009) Variation in the safety of induced pluripotent stem cell lines. Nat Biotechnol 27: 743-745.
-
Nakagawa M, Koyanagi M, Tanabe K, Takahashi K, Ichisaka T, et al. (2008) Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nat Biotechnol 26: 101-106.
-
Ieda M, Fu JD, Delgado-Olguin P, Vedantham V, Hayashi Y, et al. (2010) Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell 142: 375-386.
-
Marro S, Pang ZP, Yang N, Tsai MC, Qu K, et al. (2011) Direct lineage conversion of terminally differentiated hepatocytes to functional neurons. Cell Stem Cell 9: 374-382.
-
Yoo AS, Sun AX, Li L, Shcheglovitov A, Portmann T, et al. (2011) MicroRNA-mediated conversion of human fibroblasts to neurons. Nature 476: 228-231.
-
Ambasudhan R, Talantova M, Coleman R, Yuan X, Zhu S, et al. (2011) Direct reprogramming of adult human fibroblasts to functional neurons under defined conditions. Cell Stem Cell 9: 113-118.
-
Hsu YC, Chen SL, Wang YJ, Chen YH, Wang DY, et al. (2014) Signaling adaptor protein SH2B1 enhances neurite outgrowth and accelerates the maturation of human induced neurons. Stem Cells Transl Med 3: 713-722.
-
Pfisterer U, Kirkeby A, Torper O, Wood J, Nelander J, et al. (2011) Direct conversion of human fibroblasts to dopaminergic neurons. Proc Natl Acad Sci U S A 108: 10343-10348.
-
Yoo AS, Staahl BT, Chen L, Crabtree GR (2009) MicroRNA-mediated switching of chromatin-remodelling complexes in neural development. Nature 460: 642-646.
-
Li X, Zuo X, Jing J, Ma Y, Wang J, et al. (2015) Small-Molecule-Driven Direct Reprogramming of Mouse Fibroblasts into Functional Neurons. Cell Stem Cell 17: 195-203.
-
Hu W, Qiu B, Guan W, Wang Q, Wang M, et al. (2015) Direct Conversion of Normal and Alzheimer's Disease Human Fibroblasts into Neuronal Cells by Small Molecules. Cell Stem Cell 17: 204-212.
-
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255: 1707-1710.
-
Hemmer K, Zhang M, van Wullen T, Sakalem M, Tapia N, et al. (2014) Induced neural stem cells achieve long-term survival and functional integration in the adult mouse brain. Stem Cell Reports 3: 423-431.
-
Ring KL, Tong LM, Balestra ME, Javier R, Andrews-Zwilling Y, et al. (2012) Direct reprogramming of mouse and human fibroblasts into multipotent neural stem cells with a single factor. Cell Stem Cell 11: 100-109.
-
Corti S, Nizzardo M, Simone C, Falcone M, Donadoni C, et al. (2012) Direct reprogramming of human astrocytes into neural stem cells and neurons. Exp Cell Res 318: 1528-1541.
-
Sheng C, Zheng Q, Wu J, Xu Z, Wang L, et al. (2012) Direct reprogramming of Sertoli cells into multipotent neural stem cells by defined factors. Cell Res 22: 208-218.
-
Yamanaka S (2009) A fresh look at iPS cells. Cell 137: 13-17.
-
Dubový P (2011) Wallerian degeneration and peripheral nerve conditions for both axonal regeneration and neuropathic pain induction. Ann Anat 193: 267-275.
-
Gaudet AD, Popovich PG, Ramer MS (2011) Wallerian degeneration: gaining perspective on inflammatory events after peripheral nerve injury. J Neuroinflammation 8: 110.
-
Trounson A, Thakar RG, Lomax G, Gibbons D (2011) Clinical trials for stem cell therapies. BMC Med 9: 52.
-
Ren Z, Wang Y, Peng J, Zhao Q, Lu S (2012) Role of stem cells in the regeneration and repair of peripheral nerves. Rev Neurosci 23: 135-143.
-
Caiazzo M, Dell'Anno MT, Dvoretskova E, Lazarevic D, Taverna S, et al. (2011) Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature 476: 224-227.
-
Liu X, Li F, Stubblefield EA, Blanchard B, Richards TL, et al. (2012) Direct reprogramming of human fibroblasts into dopaminergic neuron-like cells. Cell Res 22: 321-332.
-
Son EY, Ichida JK, Wainger BJ, Toma JS, Rafuse VF, et al. (2011) Conversion of mouse and human fibroblasts into functional spinal motor neurons. Cell Stem Cell 9: 205-218.
-
Jiao J, Yang Y, Shi Y, Chen J, Gao R, et al. (2013) Modeling Dravet syndrome using induced pluripotent stem cells (iPSCs) and directly converted neurons. Hum Mol Genet 22: 4241-4252.
-
Meng F, Wang X, Gu P, Wang Z, Guo W (2013) Induction of retinal ganglion-like cells from fibroblasts by adenoviral gene delivery. Neuroscience 250: 381-393.
-
Iovino M, Pfisterer U, Holton JL, Lashley T, Swingler RJ, et al. (2014) The novel MAPT mutation K298E: mechanisms of mutant tau toxicity, brain pathology and tau expression in induced fibroblast-derived neurons. Acta Neuropathol 127: 283-295.
-
Yoshimizu T, Pan JQ, Mungenast AE, Madison JM, Su S, et al. (2015) Functional implications of a psychiatric risk variant within CACNA1C in induced human neurons. Mol Psychiatry 20: 284.
-
Johansson BB, Ohlsson AL (1996) Environment, social interaction, and physical activity as determinants of functional outcome after cerebral infarction in the rat. Exp Neurol 139: 322-327.
-
Fordyce DE, Wehner JM (1993) Physical activity enhances spatial learning performance with an associated alteration in hippocampal protein kinase C activity in C57BL/6 and DBA/2 mice. Brain Res 619: 111-119.
-
Tsai SJ (2015) Transcranial focused ultrasound as a possible treatment for major depression. Med Hypotheses 84: 381-383.





