International Journal of Stem cell Research and Therapy
Prospect of Cell Therapy for Treating Perianal Fistula, Including Crohn's Disease
Guadalajara H1,4*, García-Arranz M1,2, Georgiev-Hristov T1,4, Cortés D1,2 and García-Olmo D1,2,3
1Instituto de Investigación Sanitaria-Fundación Jiménez Díaz, (IIS-FJD), Madrid, Spain
2Surgery Department, Hospital Universitario Fundación Jiménez Díaz, Madrid, Spain
3Surgery Department, Universidad Autónoma de Madrid, Madrid, Spain
4Surgery Department, Hospital de Villalba, Collado-Villalba, Spain
*Corresponding author: Hector Guadalajara, PhD, MD, Hospital General de Villalba Ctra Alpedrete a Moralzarzal, Km 41 28400-Collado Villalba, Madrid, Spain, Tel: 910908102; E-mail: h.guadalajara@idcsalud.es
Int J Stem Cell Res Ther, IJSCRT-3-028, (Volume 3, Issue 1), Short Review; ISSN: 2469-570X
Received: February 27, 2016 | Accepted: March 26, 2016 | Published: March 31, 2016
Citation: Guadalajara H, García-Arranz M, Georgiev-Hristov T, Cortés D, García-Olmo D (2016) Prospect of Cell Therapy for Treating Perianal Fistula, Including Crohn's Disease. Int
J Stem Cell Res Ther 3:028. 10.23937/2469-570X/1410028
Copyright: © 2016 Guadalajara H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Complex perianal fistula continues to be a real surgical challenge due to the high rate of recurrence and the possibility of fecal incontinence after surgery. In the case of treatment of fistulas in patients with Crohn's disease, the results produced to date have been disappointing in spite of the administration of biological drugs.
Areas Covered: Novel methods are needed for this condition, and cells appear to have potential to improve fistula healing. Mesenchymal stromal cells (MSCs) have been used in several clinical trials, including phase-III studies. After an analysis of the published papers we can conclude that MSCs have evident anti-inflammatory and immune-regulatory properties, MSCs are safe for clinical practice and have decent results considering the challenging conditions surrounding the procedure and the safety profile of the intervention. These trials can also raise important practical issues regarding cell source, dose levels, and method of implantation. The results of ongoing large clinical trials are pending.
Conclusion: According to current data, we can say that cell therapy may become a clinical reality for the treatment of complex perianal fistula and currently can be used as a support for the surgical maneuvers during perianal fistula surgery.
Keywords
Perianal fistula, Crohn's disease, Cell therapy, Outcomes review
Introduction
The incidence of perianal fistula among patients with Crohn's disease (CD) is 28% [1], while in patients without CD, only 2 cases per 10 000 people are detected each year [2]. Recurrent perianal fistulas pose a notoriously difficult surgical challenge and very often surgical attempts fails in these kind of patients. Even simple fistulas are difficult to treat, because the surgeon must access the anal sphincter, which may compromise the functioning of this structure and lead to fecal incontinence. In a cohort of 537 patients with low perianal fistula of cryptoglandular etiology treated by fistulotomy, major incontinence was reported in 28% at 5 years of follow-up [3].
Mucosal or full-thickness advancement flaps and other surgical techniques are less advantageous because post-surgical healing must take place in a septic and inflamed environment. This situation becomes more and more complicated with successive surgical attempts, causing further perianal scarring and distortion of the perianal area and making it difficult to correctly identify possible areas of sepsis. The inevitable result is that these individuals are progressively more difficult to treat, making both patient and surgeon more and more frustrated. Moreover, continuous suppuration in the anal region in unhealed or recurrent fistulas leaves the patient at risk of acute infection with abscess formation, requiring urgent surgical drainage.
Limited surgical treatment for perianal fistula in the context of CD often results in high rates of recurrence, whereas extensive surgical treatment may cause fecal incontinence. Treatments other than surgery are far from ideal due to high recurrence rates and morbidity. Biologic agents such as infliximab do not generally reach the goal of treatment [4-6].
The aim of this review is to discuss the use of cell therapy to treat complex perianal fistula in patients with or without CD, approaching this issue from a practical and surgical point of view.
The Rationale: Why Cells for Perianal Fistula Treatment?
Perianal fistula is very difficult to treat, and even small fistula tracts can be challenging to heal [3,7] due to the impairment of the healing process caused by this condition. In healthy patients, the healing processes is well known, and always consists of the following phases in the same order regardless of the tissue: i) inflammatory phase, ii) proliferative, regenerative, or reparative phase, and iii) maturational phase.
The inflammatory phase is perpetuated due to fecal impaction in the tract, constant contact with bacterial antigens, and infection, thus leading to the release of more pro-inflammatory mediators and subsequent deep tissue destruction and perpetuation of the cycle. This inflammatory phase is even more aggressive in CD [8,9]. During phase 2, myofibroblasts play an important role in coordinating the process [10]. Healing processes that are not well orchestrated may lead to epithelization of the deepest part of the wound [11]. The epithelium acts as a barrier, though in this case the barrier prevents the fistula tract from healing [12,13]. Consolidation of the scar occurs in the third phase, though in this area it may cause fibrosis of the anal sphincter [14], deformation of anal shape leading to incontinence, and further epithelization of tracts leading to perpetuation of the fistula [12,13].
This situation of fistula perpetuation is exacerbated in the case of CD due to an uncontrolled inflammatory response. Improper response to intraluminal intestinal flora causes disproportionate activation of CD4 lymphocytes and the release of a huge amount of cytokines, both of which are potentiated by the loss of apoptosis of these CD4 lymphocytes and the elevation of intestinal permeation [8]. In CD, we have also seen that the pattern of cells seen in the perianal fistula tracks are different from those of patients without CD who develop perianal fistula; in particular, fistulas in CD patients reveal myofibroblasts scattered along a fragmented underlying basement membrane, while non-CD fistulas have a well-ordered distribution, fewer CD68-positive macrophages, and more CD45RO-positive T cells [11]. These changes increase the difficulty of controlling the inflammation and healing processes.
In the scenario of perianal fistula, the use of cells has emerged as a novel means of improving wound healing. Cell transplantation provides a way of increasing the number of cells locally in these critical phases with the aim of restoring normal wound healing. In addition, a special kind of cells named mesenchymal stromal cells (MSCs) have become a potent partner in wound healing. MSCs were initially described as a population of bone-marrow-derived mononuclear cells with a fibroblast-like morphology. They later have been found in a wide range of tissues, such as fat. The immunomodulatory activities of MSCs described to date include: a) suppression of naive and memory CD4+; b) suppression of CD8+ T-cell proliferation and differentiation; c) promotion of regulatory T-cell expansion and enhancement of their immunosuppressive activity; d) impairment of dendritic cells; f) and secretion of nitric oxide, prostaglandin E2, hepatocyte growth factor, and indoleamine 2,3-dioxygenase [15]; g) Finally, MSCs have the ability to migrate to the site of injury, directing these immunomodulatory and regenerative properties to the exact place where they are needed [16].
Practical Issues in the Use of Cells to Treat Perianal Fistula
Sources of cells
MSCs have been obtained from numerous sources (e.g., placenta, [17] umbilical cord) [18] though in clinical settings, those obtained from bone marrow and fat have been used to treat perianal fistulas.
MSCs derived from bone marrow and fat have certain shared characteristics, such as plastic adherence, marker expression, and differentiation properties. Some differences between the 2 sources, however, make adipose-derived MSCs more advantageous because of the following reasons: a) more studies using fat have been published (13 clinical trials conducted with adipose-derived stromal cells, compared to only 3 with bone marrow); b) the number of MSCs in fat is 500 fold higher than in the bone marrow [15]; c) cells are easily isolated in raw form, making it possible to obtain enough cells for a treatment on the same day using liposuction (Figure 1), while the volume of cells obtained from bone marrow aspiration is lower and expansion of the cells is needed before treatment can be performed; d) in other clinical settings (osteonecrosis), adipose-derived stromal cells are phenotypically superior for regeneration [19]; e) ASCs show greater angiogenic potential [20]; f) compared to umbilical cord and adipose tissue-derived stromal cells, bone marrow MSCs had the lowest proliferative capacity [21]; g) and finally ASCs in long term culture are more stable genetically and morphologically [22]. On the other hand ASCs may not produce the expected result due to contamination by other cells types, especially fibroblast [23].
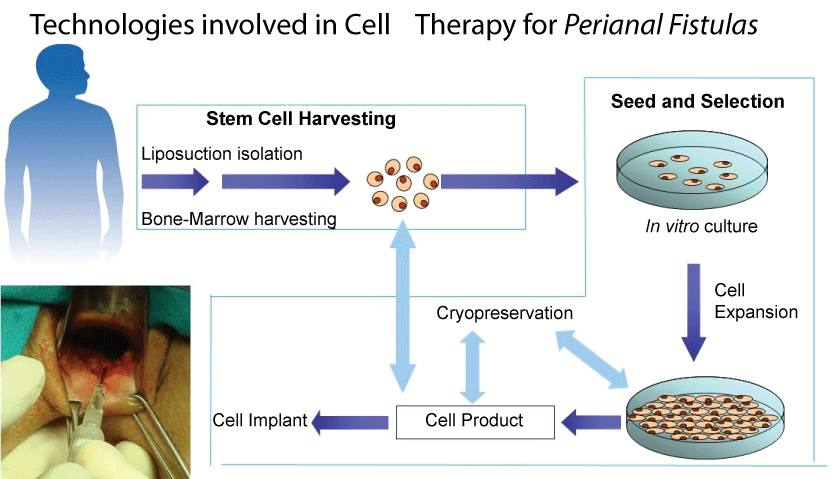
.
Figure 1: Technologies involved in cell therapy for perianal fistulas. Cells can be harvested by isolation from a product of liposuction or by harvesting from bone-marrow aspiration. This product can be injected in raw form after little manipulation. Later, these cells can be seeded in order to select specific MSCs based on their plastic adherent properties. Using "in vitro" culture technologies, we can expand cells until they achieve the adequate quantity (millions). The final cell product can be cryopreserved or administered directly. Moreover, from a cryopreserved product we can culture new doses for patient injection.
View Figure 1
Routes of cell administration to treat perianal fistula
Two routes of administration have been tried: intravenous (systemic) and intralesional. The clinical trial NCT NCT00482092 (data not published) is currently studying intravenous injection of allogeneic MSCs from bone marrow in patients with CD is being studied; however, treatment of the perianal fistula is a secondary endpoint.
To our knowledge, all other studies have been designed to deliver the cells inside the fistula track.
The search for suitable doses
The ideal dose of cells to treat fistula has yet to be determined. The dose used in published studies ranges from 1 ± 10e7 to 9 ± 10e7. A higher dose does not ensure better results. Moreover, the latest publication by Molendijk et al. has revealed that the group with the higher dose exhibits the worst results [24]; nonetheless, some of the trials currently in the recruitment phase or ongoing are using even higher numbers of cells.
Use of cells mixed with scaffolds
Many different scaffolds have been used in an attempt to regenerate tissues, and perianal fistula is one examples. Some studies have already been published using fibrin glue as a scaffold for the treatment of perianal fistula [25,26]. As published elsewhere, our experience is better when cells are combined with fibrin glue [27], and this observation has been confirmed by researchers from other fields such as orthopedic surgery [28].
These improved results may be explained because in large fistula tracks and big cavities, cells are better distributed all along the area. It is true, however, that fistula track are sometimes very small, making straight injection more effective in terms of cell distribution.
Is important to remark that in our experience cells must be suspended in the fibrin component because of the potential for toxicity posed by thrombin.
Finally, a clinical trial has recently been registered on the siteclinicaltrials.gov (NCT01915927) to determine the safety and toxicity of using autologous MSC-coated fistula plug in patients with fistulizing CD.
Safety profile of MSCs
The biosafety of MSCs has been tested by several studies. To date we can assume at least the following: a) no oncological events reported in long-term studies, even in patients with long course of perianal CD, which in itself confers higher risk of cancer [29]; b) genetic stability studies have been performed, expanding MSCs from the bone marrow of sarcoma patients, without revealing any evidence of malignant transformation [30,31]; c) Studies of the newborns of patients treated with cells did not show any influence on fertility, course of pregnancy, newborn weight, or physical condition of newborns [32].
Regulatory affairs
In Europe, cells, which are regulated by the European Medicines Agency, can only be used in 2 scenarios: approved clinical trials or compassionate use (Regulation (EC) No 1394/2009 and Directive 2001/83/EC). In the United States, this issue is regulated by the Evaluations and Research division of the FDA, which has only approved minimally manipulated cell products for homologous use, although this situation is about to change toward another status that is similar to that of the European Union.
Review of Published Clinical Data
Despite the lack of results derived from long-term outcomes of cell therapy in perianal fistula, and the challenges of regulatory affairs adding more difficulties to the trials with cells, since our last review of this issue in 2014 [33], 5 new trials have been registered and 6 new studies have been published.
Published papers
The first publications to come out of cell-based trials for fistula treatment came from a single publication in 2003 and appeared as a proof-of-concept paper concerning a patient with recto-vaginal fistula and CD [34]. The treatment was successful, and since then many trials have been published, most of which have been open-label studies performed on a small number of patients. Nonetheless, the good results obtained are encouraging other researchers to explore this medical condition in larger trials, as seen in Table 1 and Table 2 [25,34-41]. The first multicenter phase-III trial, with an enrollment of 214 patients, was published in 2012 [27]. Although results were not entirely bad with a healing rate of 40% in complex fistulas with minimal intervention the results were similar between patients treated with fibrin glue and the experimental arm (ASC therapy). The authors argued that a plausible explanation for this situation is that the research groups lacked experience manipulating cells, and could have deteriorated the cells during the injection. In addition, significant differences were found in the complexity of patients enrolled in different hospitals. After this experience, some long-term results have been published with modest results [42-44], though others in very complex clinical settings have yielded sufficient results with limited and harmless surgery [45-47]. In conclusion, overall healing rates of patients treated with ASCs range from 30% to 60%. This may be considered a poor result, though it would nonetheless be worth the effort if we achieve one healing in a patient with a complex perianal fistula sometimes avoiding a permanent ostomy.
![]()
Table 1: Published clinical experiences of cell treatments of anal fistula (Part 1). An update of the tables taken from the section "Stem Cell Application in Fistula Disease" [33].
View Table 1
![]()
Table 2: Published clinical experiences of cell treatments of anal fistula (Part 2). An update of the tables taken from the section "Stem Cell Application in Fistula Disease" [33].
View Table 2
Ongoing clinical trials
There are 16 ongoing trials registered on the clinicaltrials.com database (Table 3 and Table 4). Nine are indicated as "unknown status" and are excluded from the analysis. Seven are in ongoing status and one has been completed and is pending of publication. Since the last review [33], 5 new trials have been registered. Three of these new clinical trials use adipose-derived stromal cells, 2 delivered in situ and one using a cell-coated plug. The other 2cells derived from bone marrow and allogeneic umbilical cord.
![]()
Table 3: Ongoing clinical trials using cells for treatment of anal fistula (Part 1). An update of the tables taken from the section "Stem Cell Application in Fistula Disease". [33] Source: Clinicaltrials.gov
View Table 3
![]()
Table 4: Ongoing clinical trials using cells for treatment of anal fistula (Part 2). An update of the tables taken from the section "Stem Cell Application in Fistula Disease". [33] ASCs: adult stem cells. Source: Clinicaltrials.gov
View Table 4
Discussion
Wound healing is the basis of surgery, and to orchestrate this process, MSCs could be helpful due to their potent anti-inflammatory and immunomodulatory effects [31]. Their ability to migrate to the site of injury has also been described [16], facilitating these immunomodulatory and regenerative properties in the exact place where they are required.
MSCs were initially described as a bone-marrow-derived mononuclear cell population with a fibroblast-like morphology. Later, MSCs were reported to be found in many different tissues including fat. Although the mechanisms underlying the effects of MSCs have not been clearly defined, MSCs interact with several steps of inflammation during the healing process [15].
Even a simple perianal fistula remain poses a real surgical challenge due to the high rate of recurrence and the possibility of fecal incontinence after surgery [3]. In a more complex setting, multiple operations and incontinence is the rule, and permanent ostomy is eventually needed.
The frustrating outcomes seen in perianal fistula treatment can be explained in part by a deficient and disorganized cell supply during the wound-healing process, and also by the chronic inflammation of the fistula tract. In the case of perianal fistula associated with CD, the problem becomes worse due to the inflammatory basis of the disease, making healing more inefficient [8].
Correct epithelization of the surface of the wound is vital in this case. Suboptimal epithelization may result in the following: i) the inflammation may destroy deeper tissues, thereby expanding the fistula tracks; ii) inflammation may epithelize the fistula track itself leading to perpetuation of the fistula; and iii) fibrosis and scars may deform the shape of the anus, leading to incontinence [14]. In this scenario, cells could be helpful in orchestrating the healing process.
Preclinical studies conducted by different groups in animal disease models have already demonstrated that MSCs are efficient in improving the healing process. All these studies have demonstrated that MSCs area safe in vivo therapy [30].
Cells have yielded promising results in preclinical studies directed at treating perianal fistula, some approaches have begun clinical development and, and the number of registered clinical trials has been increasing every year since 2003. In 2015, the most advanced programs, which involve CD and fistulous disease, have reached phase III of development.
The results in phase-III randomized clinical studies have shown, however, that the therapeutic efficacy of MSCs in humans is still modest (40%) [27]; we are currently awaiting the results of ongoing large-scale clinical trials. The safety profile evidenced in these trials and long-term analyses have once again demonstrated the safety of MSCs in clinical settings [31]. Nevertheless, a deeper analysis of this result reveals that no other treatment achieves better results in complex perianal fistula with or without CD, and successful treatment of even one patient merits recognition.
In our experience we prefer adipose-derived MSCs due to their quality and quantity. One liposuction treatment obtains enough cells to make the first injection in raw form on the same day of the extraction, and the second injection may use expanded MSCs if needed; if not, they are cryopreserved.
Combination with any other surgical (limited or more reconstructive) and/or other medical treatments may potentiate both approaches.
According to current data we can say that MSC therapy may become a clinical reality for the treatment of complex perianal fistula in the near future.
Moreover, in the field of cell therapy a new generation of cell-based drugs is being developed to improve the therapeutic efficacy of conventional cell-based treatments such as the engineered cells or cells mixed with scaffolds [48].
Conclusions
Surgical wound healing is the most fundamental aspect of surgery. MSCs could be helpful in orchestrating this process due to their potent anti-inflammatory and immunomodulatory effects. Successful treatment of perianal fistula is exceedingly difficult, and even small tract fistulas may have difficulty healing. This difficulty can be explained due to the impairment of the healing process provoked by this condition. This situation is exacerbated in CD. Multiple failed operations are common. Although published results may seem discrete, no other techniques have shown better results nor have the same positive safety profile. Combination with any other surgical (limited or more reconstructive) or medical treatments may potentiate both types. According to current data, we can say that MSC therapy may become a clinical reality for the treatment of complex perianal fistula in the near future.
Financial and Competing Interests Disclosure
UAM and Cellerix SL/Tygenix TAU, share patents rights in cell products. García-Olmo is a member of the scientific advisory board of Tygenix. Damian García-Olmo and Mariano García-Arranz are inventors in two patents related to cell products entitled "Identification and isolation of multipotent cells from non-osteochondral mesenchymal tissue" (10157355957US) and "Use of adipose tissue-derived stem cells in treating fistula" (US11/167061).
Acknowledgement
Oliver Shaw for his expert, deep review of medical style in English.
References
-
Eglinton TW, Barclay ML, Gearry RB, Frizelle FA (2012) The spectrum of perianal Crohn's disease in a population-based cohort. Dis Colon Rectum 55: 773-777.
-
Zanotti, Martinez-Puente, Pascual, Pascual, Herreros, et al. (2007) An assessment of the incidence of fistula-in-ano in four countries of the european union. Int J Colorectal Dis 22: 1459-1462.
-
Göttgens, Janssen, Heemskerk, van Dielen, Konsten, Lettinga, et al. (2014) Long-term outcome of low perianal fistulas treated by fistulotomy: A multicenter study. International Journal of Colorectal Disease 30: 21321-21329.
-
Bouguen, Siproudhis, Gizard, Wallenhorst, Billioud, Bretagne, et al. (2013) Long-term outcome of perianal fistulizing crohn's disease treated with infliximab. Clin Gastroenterol Hepatol 11: 981.e4.
-
Nunes J, Santos PM, Tavares L (2010) Complete resolution of enterocolic fistulas with infliximab. BioDrugs 24 Suppl 1: 28-30.
-
Uchino M, Ikeuchi H, Bando T, Matsuoka H, Takesue Y, et al. (2011) Long-term efficacy of infliximab maintenance therapy for perianal Crohn's disease. World J Gastroenterol 17: 1174-1179.
-
Göttgens, Smeets, Stassen, Beets, Breukink (2014) Systematic review and meta-analysis of surgical interventions for high cryptoglandular perianal fistula. International Journal of Colorectal Disease 30: 583-593.
-
Barnich N, Darfeuille-Michaud A (2007) Role of bacteria in the etiopathogenesis of inflammatory bowel disease. World J Gastroenterol 13: 5571-5576.
-
Rath HC, Schultz M, Freitag R, Dieleman LA, Li F, et al. (2001) Different subsets of enteric bacteria induce and perpetuate experimental colitis in rats and mice. Infect Immun 69: 2277-2285.
-
Baird, Lloyd, Lawrance (2015) Prostaglandin E and polyenylphosphatidylcholine protect against intestinal fibrosis and regulate myofibroblast function. Dig Dis Sci 60: 1603-1616.
-
Bataille F, Klebl F, Rümmele P, Schroeder J, Farkas S, et al. (2004) Morphological characterisation of Crohn's disease fistulae. Gut 53: 1314-1321.
-
Bataille, Rohrmeier, Bates, Weber, Rieder, Brenmoehl, et al. (2008) Evidence for a role of epithelial mesenchymal transition during pathogenesis of fistulae in crohn's disease. Inflamm Bowel Dis 14: 1514-1527.
-
van Koperen PJ, ten Kate FJ, Bemelman WA, Slors JF (2010) Histological identification of epithelium in perianal fistulae: a prospective study. Colorectal Dis 12: 891-895.
-
Dudukgian H, Abcarian H (2011) Why do we have so much trouble treating anal fistula? World J Gastroenterol 17: 3292-3296.
-
Strioga M, Viswanathan S, Darinskas A, Slaby O, Michalek J (2012) Same or not the same? comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem cells and development 21: 2724-2752.
-
Chapel, Bertho, Bensidhoum, Fouillard, Young, Frick, et al. (2003) Mesenchymal stem cells home to injured tissues when co-infused with hematopoietic cells to treat a radiation-induced multi-organ failure syndrome. J Gene Med 5: 1028-1038.
-
Oliveira MS, Barreto-Filho JB (2015) Placental-derived stem cells: Culture, differentiation and challenges. World J Stem Cells 7: 769-775.
-
Li T, Xia M, Gao Y, Chen Y, Xu Y (2015) Human umbilical cord mesenchymal stem cells: an overview of their potential in cell-based therapy. Expert Opin Biol Ther 15: 1293-1306.
-
Wyles, Houdek, Crespo-Diaz, Norambuena, Stalboerger, Terzic, et al. (2015) Adipose-derived mesenchymal stem cells are phenotypically superior for regeneration in the setting of osteonecrosis of the femoral head. Clin Orthop Relat Res 473: 3080-3090.
-
Kim Y, Kim H, Cho, Bae, Suh, Jung (2007) Direct comparison of human mesenchymal stem cells derived from adipose tissues and bone marrow in mediating neovascularization in response to vascular ischemia. Cell Physiol Biochem 20: 867-876.
-
Kern S, Eichler H, Stoeve J, Kluter H, Bieback K (2006) Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 24: 1294-1301.
-
Izadpanah R, Trygg C, Patel B, Kriedt C, Dufour J, et al. (2006) Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue. J Cell Biochem 99: 1285-1297.
-
Brohem CA, de Carvalho CM, Radoski CL, Santi FC, Baptista MC, et al. (2013) Comparison between fibroblasts and mesenchymal stem cells derived from dermal and adipose tissue. Int J Cosmet Sci 35: 448-457.
-
Molendijk I, Bonsing BA, Roelofs H, Peeters KC, Wasser MN, et al. (2015) Allogeneic Bone Marrow-Derived Mesenchymal Stromal Cells Promote Healing of Refractory Perianal Fistulas in Patients With Crohn's Disease. Gastroenterology 149: 918-927.
-
Garcia-Olmo D, Herreros D, Pascual I, Pascual JA, Del-Valle E, et al. (2009) Expanded adipose-derived stem cells for the treatment of complex perianal fistula: a phase II clinical trial. Dis Colon Rectum 52: 79-86.
-
Garcia-Olmo D, Herreros D, Pascual M, Pascual I, De-La-Quintana P, et al. (2009) Treatment of enterocutaneous fistula in Crohn's Disease with adipose-derived stem cells: a comparison of protocols with and without cell expansion. Int J Colorectal Dis 24: 27-30.
-
Herreros MD, Garcia-Arranz M, Guadalajara H, De-La-Quintana P, Garcia-Olmo D (2012) FATT Collaborative Group. Autologous expanded adipose-derived stem cells for the treatment of complex cryptoglandular perianal fistulas: A phase III randomized clinical trial (FATT 1: Fistula advanced therapy trial 1) and long-term evaluation. Dis Colon Rectum 55: 762-772.
-
Kim YS, Choi YJ, Suh DS, Heo DB, Kim YI, et al. (2015) Mesenchymal stem cell implantation in osteoarthritic knees: is fibrin glue effective as a scaffold? Am J Sports Med 43: 176-185.
-
Beaugerie L (2014) [Natural history of intestinal lesions in inflammatory bowel disease]. Rev Prat 64: 1226-1229.
-
Lucarelli, Bellotti, Mantelli, Avanzini, Maccario, Novara, et al. (2014) In vitro biosafety profile evaluation of multipotent mesenchymal stem cells derived from the bone marrow of sarcoma patients. J Transl Med 12.
-
García-Gómez I, Elvira G, Zapata AG, Lamana ML, Ramírez M, et al. (2010) Mesenchymal stem cells: Biological properties and clinical applications. Expert Opin Biol Ther 10: 1453-1468.
-
Sanz-Baro, Garcia-Arranz, Guadalajara, de la Quintana, Herreros, Garcia-Olmo (2015) First-in-human case study: Pregnancy in women with crohn's perianal fistula treated with adipose-derived stem cells: A safety study. Stem Cells Translational Medicine 4: 598-602.
-
Damian Garcia-Olmo HG (2014) Stem cell application in fistula disease. In: Abcarian H, editor. Anal Fistula - Principles and Management. Springer New York 129-138.
-
García-Olmo D, García-Arranz M, García LG, Cuellar ES, Blanco IF, et al. (2003) Autologous stem cell transplantation for treatment of rectovaginal fistula in perianal Crohn's disease: a new cell-based therapy. Int J Colorectal Dis 18: 451-454.
-
Borowski, Gill, Agarwal, Bhaskar (2012) Autologous adipose-tissue derived regenerative cells for the treatment of complex cryptoglandular fistula-in-ano: A report of three cases. Case Reports 2012: bcr2012006988.
-
Lee WY, Park KJ, Cho YB, Yoon SN, Song KH, et al. (2013) Autologous adipose tissue-derived stem cells treatment demonstrated favorable and sustainable therapeutic effect for crohn's fistula. Stem Cells 31: 2575-2581.
-
de la Portilla F, Alba F, Garcia-Olmo D, Herrerias JM, Gonzalez FX, Galindo A (2013) Expanded allogeneic adipose-derived stem cells (eASCs) for the treatment of complex perianal fistula in crohn's disease: Results from a multicenter phase I/IIa clinical trial. Int J Colorectal Dis 28: 313-323.
-
Cho YB, Lee WY, Park KJ, Kim M, Yoo HW, et al. (2013) Autologous adipose tissue-derived stem cells for the treatment of Crohn's fistula: a phase I clinical study. Cell Transplant 22: 279-285.
-
Ciccocioppo, Bernardo, Sgarella, Maccario, Avanzini, Ubezio, et al. (2011) Autologous bone marrow-derived mesenchymal stromal cells in the treatment of fistulising crohn's disease. Gut 60: 788-798.
-
García-Olmo D, Herreros D, De-La-Quintana P, Guadalajara H, Trébol J, et al. (2010) Adipose-derived stem cells in Crohn's rectovaginal fistula. Case Rep Med 2010: 961758.
-
García-Olmo D, García-Arranz M, Herreros D, Pascual I, Peiro C, et al. (2005) A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum 48: 1416-1423.
-
Guadalajara, Herreros, De-La-Quintana, Trebol, Garcia-Arranz, Garcia-Olmo (2011) Long-term follow-up of patients undergoing adipose-derived adult stem cell administration to treat complex perianal fistulas. International Journal of Colorectal Disease 27: 595-600.
-
Cho YB, Park KJ, Yoon SN, Song KH, Kim do S, et al. (2015) Long-term results of adipose-derived stem cell therapy for the treatment of Crohn's fistula. Stem Cells Transl Med 4: 532-537.
-
Ciccocioppo, Gallia, Sgarella, Kruzliak, Gobbi, Corazza (2015) Long-term follow-up of crohn disease fistulas after local injections of bone Marrowâ€"Derived mesenchymal stem cells. Mayo Clinic Proceedings 90: 747-755.
-
Garcia-Olmo D, Guadalajara H, Rubio-Perez I, Herreros MD, de-la-Quintana P, et al. (2015) Recurrent anal fistulae: limited surgery supported by stem cells. World J Gastroenterol 21: 3330-3336.
-
Borowski DW, Gill TS, Agarwal AK, Tabaqchali MA, Garg DK, et al. (2015) Adipose Tissue-Derived Regenerative Cell-Enhanced Lipofilling for Treatment of Cryptoglandular Fistulae-in-Ano: The ALFA Technique. Surg Innov 22: 593-600.
-
Park KJ, Ryoo SB, Kim JS, Kim TI, Baik SH, et al. (2015) Allogeneic adipose-derived stem cells for the treatment of perianal fistula in Crohn's disease: a pilot clinical trial. Colorectal Dis .
-
Kim IG, Ko J, Lee HR, Do SH, Park K (2016) Mesenchymal cells condensation-inducible mesh scaffolds for cartilage tissue engineering. Biomaterials 85: 18-29.





