Journal of Clinical Gastroenterology and Treatment
Effect of Biologics on the Outcomes of Inflammatory Bowel Disease: A Retrospective Study on a Large Pediatric Cohort
Hoda M. Malatyr1,2*, Bincy Abraham1, Mina S. Emil1, George D. Ferry2,3 and Elizabeth A. Garnett4
1Department of Medicine, Baylor College of Medicine, Houston, Texas, USA
2Department of Pediatrics, Baylor College of Medicine, Houston, Texas, USA
3Texas Children's Hospital, Baylor College of Medicine, Houston, Texas, USA
4University of California, San Francisco, California, USA
*Corresponding author:
Hoda M. Malaty, M.D, Ph.D, Veterans Affairs Medical Center (111D), 2002 Holcombe Blvd, Houston, Texas 77030, USA, Tel: 713-795-0232, Fax: 713-790-1040, E-mail: hmalaty@bcm.tmc.edu
J Clin Gastroenterol Treat, JCGT-1-003, (Volume 1, Issue 1), Research Article; ISSN: 2469-584X
Received: August 18, 2015 | Accepted: September 09, 2015 | Published: September 11, 2015
Citation: Malaty HM, Abraham B, Emil MS, Ferry GD, Garnett EA (2015) Effect of Biologics on the Outcomes of Inflammatory Bowel Disease: A Retrospective Study on a Large Pediatric Cohort. J Clin Gastroenterol Treat 1:003. 10.23937/2469-584X/1510003
Copyright: © 2015 Malaty HM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The incidence of inflammatory bowel disease (IBD) in children has increased over the past 20 years and treatment with biologics is increasingly used.
Objective: We assessed the associations of biologics to IBD outcomes in children.
Methods: A retrospective investigation was conducted on a cohort of children with IBD between 1988/2008 who were enrolled in the Pediatric IBD registry. The diagnosis of IBD was based on clinical, radiological, endoscopic, and/or histological examinations. Clinical outcome measures were grouped into surgical, extra intestinal manifestations (EIMs), and frequency of clinical visits. Patients were categorized into two groups based on receiving biologics (exposed) and (non-exposed). The dates of each in/out patient visit and/or hospitalization were recorded.
Results: Of 335 patients, 73 children received biologics. There was no difference between the two groups in respect to age (mean = 11.6 ± 4.1 years) and gender. The exposed group had more number of surgeries than the non-exposed (OR = 2.5; 95% CI = 1.4-4.7, p = 0.004). The same trends were observed in musculoskeletal EIMS (OR = 3.7; 95% CI = 2.1-6.3, p ≤ 0.0001) and having any type of abscess and/fistula (OR = 3.0; 95% CI = 2.0-5.6, p = 0.002). However, the incidence of dermatologic and oral EIMS decreased in the exposed group than the non-exposed, 3/73 (4.1%) vs. 23/309 (7.4%) respectively, (OR = 0.5; 95% CI = 0.2-1.8, p = 0.44). This trend continued among those with renal or hepatobiliary complications (OR = 0.2; 95% CI = 0.03-1.7, p = 0.14). The 335 children had 3387 visits with a median number of visits significantly dropped from 1988-1994 to 2001-2008 (16.3 ± 6.6 vs. 3.7 ± 1.9, respectively, p = 0.0001).
Conclusions: Biologic therapy offers protective effect to several EIMs in children diagnosed with IBD and it is recommended in severe cases. Moreover, the outpatient clinical visits significantly dropped as did the frequency of hospitalization despite the increase of the number of IBD cases that may reflect the effect of biologic therapy.
Keywords
Inflammatory Bowel Disease (IBD), Children, Biologics, Outcomes
Introduction
Inflammatory bowel disease (IBD) refers to a group of diseases, which includes Crohn's disease (CD) and ulcerative colitis (UC). IBD is a life-long disease [1-5] and is ranked among the five most prevalent gastrointestinal diseases in the United States, with healthcare costs exceeding $1.7 billion annually [2]. There seems to be a worldwide trend toward increasing incidence rates of pediatric IBD [6-10]. The management of children with inflammatory bowel disease can be difficult and it requires knowledge of current advances in the field of IBD. Therefore, gastroenterologists caring for these children must develop an optimal regimen of therapies, nutritional management, and psychological support that optimize IBD remission and minimize disease and drug-induced adverse effects.
Over the past several years, there have been a number of achievements and progress made in the care and management of this disorder. The therapeutic treatment has greatly expanded especially with the evolution of biologics in the treatment and management of IBD in adults and children. In pediatric IBD, biologics is the only anti-TNF drug that has been approved by the FDA, and only for use in refractory and complicated CD [11,12] and in UC. In recent years, long-term follow-up data on biologics use in pediatric CD and increasing data on the drug use in pediatric UC have become available. The use of biologic therapy produces excellent remission rates in adults and children; however there is less evidence in children than in adults.
Therefore, we performed the current study to assess the long term outcomes of biologics therapy use in children diagnosed with IBD.
Material and Methods
Design/Patient population
A retrospective investigation was conducted on a cohort of children diagnosed with IBD between 1988 and 2008 who were enrolled in the Pediatric Inflammatory Bowel registry at Texas Children's Hospital (TCH) [6]. TCH is located in the Greater Houston Metropolitan Area llocated along the Texas Gulf Coast region of the U.S. It is a 10-county metropolitan area, the sixth largest metropolitan area in the U.S. with a population of 5.5 million, highly diverse with respect to race and ethnic backgrounds [13,14]. Moreover, TCH has many health centers of pediatric practices strategically located throughout the area that serve as a referral mechanism to the IBD center. The IBD registry was in compliance with the Health Insurance Portability and Accountability Act and obtained institutional review board approval for the protocol. Informed consent and assent were obtained from parents and patients before entry of patient's information into the registry.
Data collection
The data used for this study was gathered from children with confirmed diagnosis of IBD and retrieved from the local IBD registry database. A manual of procedure was created for all the variables in the registry, including, but not limited to, year of diagnosis, age at diagnosis, race/ethnicity, date of initial IBD diagnosis, sub-type of IBD, and medications prescribed. The data was examined in depth and coded for each variable. The diagnosis of IBD was based on clinical, radiological, endoscopic, and histological examinations.
Outcome measures
The outcome measures of the current study were grouped into surgery (small bowel resection/colectomy), occurrence of abscess/fistula, diabetes, infections, pancreatitis, stricture/bowel obstruction, and extra intestinal manifestations (EIMs). EIMS were subsequently classified into musculoskeletal (arthritis, bone fractures and osteopenia/osteoporosis), ophthalmologic (uveitis, iritis), dermatologic/oral (erythema nodosum, psoriasis, pyoderma gangrenosums and aphthous stomatitis), and renal/hepatobiliary (renal calculi, primary sclerosing cholangitis, autoimmune hepatitis).
Hospital visits in relation to the medication prescribed
The date of each in/out visit for each patient and the date of hospitalization were recorded. The total number of visits and the interval between visits for each child was estimated starting from the date of diagnosis of IBD and ended at the last follow-up visit. We calculated the number of visits/patient/year.
Statistical Analysis
Each patient's initial visit was logged as their enrollment visit and all subsequent visits were logged as follow-ups. Patients were categorized into two groups based on receiving biologics at any time during follow-up (exposed group) and patients who did not receive biologics during this period (non-exposed group). The study outcomes were examined at the end of the follow period. The study time interval was grouped into the 3 periods based on the introduction and use of biologics in children. Between the years 1988 and 1994, there was no use of Biologics, between the years1995-2000, there was the introduction of biologics and in the last period between 2001 and 2008, there was a dramatic use of biologics for treatment IBD in children. Groups were compared using Fisher's exact and t-tests. The cumulative risk of colectomy was estimated, and risk factors for colectomy with Cox hazards proportional models. The 95% Confidence Intervals (CIs) were calculated.
Results
There were 335 children diagnosed with IBD; 231 diagnosed with CD and 104 with UC. Overall, there were 73 children received biologics (exposed group). At the end of follow up period, there was no difference between the exposed and the non-exposed groups in respect to age (mean age at diagnosis was 11.6 ± 4.1 years for each group (range 0.4 - 22 years), gender, or the mean length of follow-up (3.3 years vs. 3.1 years, respectively).
Among the 73 children in the exposed group, the initial date of diagnosis of EIMs occurred after the initial date of diagnosis of IBD in 63 children (85%). The results of Fisher's exact comparing the exposed and the non-exposed groups at the end of follow-up period, in relation to the study clinical outcome measures; surgical, and EIMs is presented in (Table 1). Children who received biologics were more likely to have surgery (OR = 2.5; 95% CI = 1.4-4.7, p = 0.004) of who 60% (12/20) had at least one surgery before receiving biologics. The same trends were observed in respect to musculoskeletal EIMS (OR = 3.7; 95% CI = 2.1-6.3, p ≤ 0.0001) and having any type of abscess and/fistula (OR = 3.0; 95% CI = 2.0-5.6, p = 0.002). The incidence of dermatologic and oral EIMS decreased in the biologics group than the non-exposed group, 3/73 (4.1%) vs. 23/309 (7.4%) respectively, (OR = 0.5; 95% CI = 0.2-1.8, p = 0.44) but was not significant due to the small sample size. This trend also continued in those with renal or hepatobiliary complications of their disease (OR = 0.2; 95% CI = 0.03-1.7, p = 0.14). There was no significant difference between the groups regarding infection (OR = 1.9; 95% CI = 0.5-6.4, p = 0.2).
![]()
Table 1: Univariate analysis of the comparing of the exposed and the non-exposed groups at the end of follow-up period in relation to the study outcomes
View Table 1
Among the 73 children in the exposed group, 62 children had CD and 8 had UC. When compared the same analysis for UC and CD independently, the same trend was maintained among CD patients and not among the UC patients due to the small number in the exposed group children.
Hospital visits/surgery in relation to the medication prescribed
The 335 children had a total of 3387 visits, the average number of visits for exposed group was 9.8 visits and 8.7 for the control p group; P = 0.4. The median number of the overall number of visits significantly dropped from 1988-1994 to 2001-2008 (16.3 ± 6.6 vs. 3.7 ± 1.9, respectively, p = 0.0001) and the median number of visits/patient/year dropped (4.3 ± 2.6 vs. 2.8 ± 1.5), respectively; p = 0.04) (Table 2), (Figure 1). Moreover, the median number of follow-up years dropped from 6.2 ± 2.4 during the period 1988-1994 to 1.4 ± 0.6 during 2001-2008 (P = 0.0001). The overall number of patients seen and diagnosed with IBD was four times higher in the last study period compared to the first period (59 vs.227 patients), respectively. During the first study period, 16 patients out of the 59 were hospitalized while there was 47 of the 227 patients were hospitalized during the last period (30% vs. 21%) during the later period (OR = 2.1, 95% CI = 1.0-5.5, p = 0.042). IBD subtype, age, gender, ethnicity did not have an effect on the median duration visits or hospitalization. When we repeated the analysis for the overall numbers of clinical visits and in-patients visits independently, the same findings emerged.
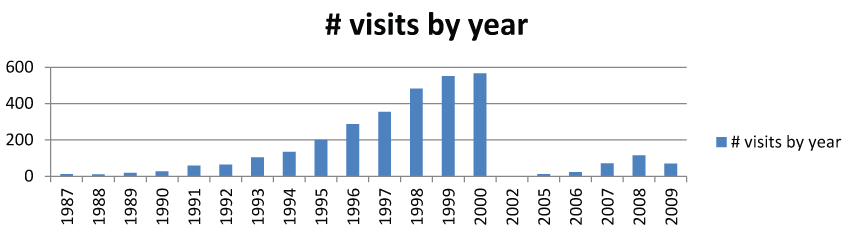
.
Figure 1: The total number of visits for the studied children diagnosed with IBD between the years1988-2008.
View Figure 1
![]()
Table 2: The total of 3387 visits for the 382 studied children diagnosed with IBD between the years1988-2008
View Table 2
Discussion
For more than a decade, treatment with biologics has been of great benefit to many pediatric IBD patients. Several important observations have emerged from our study. First, children received biologic therapy had less EIMs of dermatologic, oral, renal and hepatobiliary manifestations than those who did not receive biologics. Of interest these children (78%) received the biologics therapy before the first date of their EIMs were diagnosed. We were not able to compare our results to other studies as Anti-TNF treatment for children with extra-intestinal symptoms has only been described in case reports and small case series. However, biologics seemed to be effective in cases of pyoderma gangrenosum, orofacial involvement, erythema nodosum, cutaneous metastatic CD of the penile and scrotal skin, pancreatitis, and clavicular osteomyelitis [11-15]. In the current study, we found that children with musculoskeletal symptoms were three times more likely to receive biologics than those with no musculoskeletal symptoms. One published article reported a case of chronic recurrent clavicular osteomyelitis in association with CD [15]. The study reported that steroid therapy resulted in partial remission and an enlarging clavicular mass subsequently recurred, while biologics therapy resulted in significant improvement and resolution of the sclerotic clavicular lesion [15]. Treatment of EIMs of pediatrics IBD is of great importance as approximately 30% of children with IBD develop at least one EIM after diagnosis [16,17]. Moreover, evidence from adult studies suggests that biologics can be effective for the treatment of EIMs [18].
The second important finding of our study is the observed significant use of biologics in patients with severe complicated cases such as those with abscess/fistula, or underwent surgery. Although the presence of fistulas is often an indication to start biologic therapy, data on the efficacy of biologics in children with fistulizing CD are based on a small number of patients [19-23]. Crandall et al performed post-hoc analyses on the effect of biologics on concurrent perianal disease in a subpopulation of 31 pediatric CD patients of the REACH study and concluded that biologics rapidly reduced concurrent perianal disease signs and symptoms in their cohort [23]. Biologics therapy for children with entero-vesicular fistula has been also described two case series [24,25]. Despite closure of the entero-vesicular fistulas, surgery of severe underlying bowel disease was still required in the two studied patients. Our current data did not specify the type of fistula, however, the overall published data suggest that biologic therapy is effective for fistulizing CD and it decreases the risk for surgery. We found an increase in the number of surgeries among the studied IBD children who received biologics. This was due to the severity of these cases and these children received biologics before they went under surgeries.
Of interest, our results did not show difference between the two groups in relation to infection. There is marked variation between studies in the association between biologics therapy (anti-TNF). It has been reported that one of the adverse effects of receiving anti-TNF is the increasing number of infection, especially the granulomatous such as tuberculosis (TB) [26,27]. Other studies were in agreement with our current findings, as a recent study reported the course of TB disease in children who are on biologic therapy and found that during the 3 years of follow-up, none of these patients developed TB disease [28]. Another study published that the rate of serious infection was significantly lower among pediatric patients with IBD treated with anti-TNF agents than those treated with steroids after evaluating 5528 patients with 9516 patient-years of follow-up [29].
The third interesting finding of our study is the significant drop of the outpatient clinical visits per patient/year and the frequency of hospitalization despite the significant increase the number of children diagnosed with IBD during the same period. Such finding is a reflection of the effect of treatment with biologics and adds more emphasis on the increased incidence of IBD in children [7-10].
The utilization of data for constructing a retrospective cohort has some shortcomings. First, the studied population presented to a tertiary care from a single center and the possibilities of missing some IBD cases exists; however, TCH is one of the largest children's hospitals in the nation offering a diverse population at large that closely reflects that of any given state population. However, missing some cases from other practices should not have influenced the validity of our results. The second limitation is that we were not able to describe which type of biologic was used for the children by IBD-subtype. However, there were only 73 children received biologics during the time period of the study that and by dividing them into IBD subtypes and biologic type would limit the power of the analysis. However, the hypothesis of our current study was not based on type of medication, but rather focused on the outcome of biologics use in children with IBD. Moreover, the majority of children received biologics in the current study were prescribed to Infliximab. The third limitation of the study emerged from its retrospective nature that limited the availability of the data on the status or the condition of EIMs among the exposed group, however we were able to examine and compare the study outcomes at the end of the follow period between the exposed and non-exposed group.
In conclusion, this study adds emphasis that biologics offer some protective effect to the several EIMs in children diagnosed with IBD and it is recommended in severe cases with complication. Moreover, the outpatient clinical visits significantly dropped as did the frequency of hospitalization despite a significant increase of children diagnosed with IBD during the same period that may reflect the effect of treatment with biologics.
References
-
Baldassano RN, Piccoli DA (1999) Inflammatory bowel disease in pediatric and adolescent patients. Gastroenterol Clin North Am 28: 445-458.
-
Sandler RS, Everhart JE, Donowitz M, Adams E, Cronin K, et al. (2002) The burden of selected digestive diseases in the United States. Gastroenterology 122: 1500-1511.
-
Marx G, Seidman EG, Martin SR, Deslandres C (2002) Outcome of Crohn's disease diagnosed before two years of age. J Pediatr 140: 470-473.
-
Hyams JS (1996) Crohn's disease in children. Pediatr Clin North Am 43: 255-277.
-
Hyams JS, Davis P, Grancher K, Lerer T, Justinich CJ, et al. (1996) Clinical outcome of ulcerative colitis in children. J Pediatr 129: 81-88.
-
Kugathasan S, Judd RH, Hoffmann RG, Heikenen J, Telega G, et al. (2003) Epidemiologic and clinical characteristics of children with newly diagnosed inflammatory bowel disease in Wisconsin: a statewide population-based study. J Pediatr 143: 525-531.
-
Malaty HM, Fan X, Opekun AR, Thibodeaux C, Ferry GD (2010) Rising incidence of Inflammatory Bowel Disease among children: A 12-years Study. JPGN 50: 27-31.
-
Jakobsen C, Paerregaard A, Munkholm P, Faerk J, Lange A, et al. (2011) Pediatric inflammatory bowel disease: increasing incidence, decreasing surgery rate, and compromisednutritional status: A prospective population-based cohort study 2007-2009. Inflamm Bowel Dis 17: 2541-2550.
-
Sawczenko A, Sandhu BK, Logan RF, Jenkins H, Taylor CJ, et al. (2001) Prospective survey of childhood inflammatory bowel disease in the British Isles. Lancet 357: 1093-1094.
-
Turunen P, Kolho KL, Auvinen A, Iltanen S, Huhtala H, et al. (2006) Incidence of Inflammatory Bowel Disease in Finnish Children, 1987-2003. Inflamm Bowel Dis 12: 677-683.
-
Kugathasan S, Miranda A, Nocton J, Drolet BA, Raasch C, et al. (2003) Dermatologic manifestationsof Crohn disease in children: response to infliximab. J Pediatr Gastroenterol Nutr 37: 150-154.
-
Dretzke J, Edlin R, Round J, Connock M, Hulme C, et al. (2011) A systematic review and economic evaluation of the use of tumour necrosis factor-alpha (TNF-a) inhibitors, adalimumab and infliximab, for Crohn's disease. Health Technol Assess 15: 1-244.
-
Cezard JP, Nouaili N, Talbotec C, Hugot JP, Gobert JG, et al. (2003) A prospective study of the efficacy and tolerance of a chimeric antibody to tumor necrosis factors (remicade) in severe pediatric Crohn disease. J Pediatr Gastroenterol Nutr 36: 632-636.
-
Lionetti P, Bronzini F, Salvestrini C, Bascietto C, Canani RB, et al. (2003) Response to infliximab is related to disease duration in paediatric Crohn's disease. Aliment Pharmacol Ther 18: 425-431.
-
Carpenter E, Jackson MA, Friesen CA, Scarbrough M, Roberts CC (2004) Crohn's-associated chronic recurrent multifocal osteomyelitis responsive to infliximab. J Pediatr 144: 541-544.
-
Jose FA, Garnett EA, Vittinghoff E, Ferry GD, Winter HS, et al. (2009) Development of extraintestinal manifestations in pediatric patients with inflammatory bowel disease. Inflamm Bowel Dis 15: 63-68.
-
Dotson JL, Hyams JS, Markowitz J, LeLeiko NS, Mack DR, et al. (2010) Extraintestinal manifestations of pediatric inflammatory bowel disease and their relation to disease type and severity. J Pediatr Gastroenterol Nutr 51: 140-145.
-
Barrie A, Regueiro M (2007) Biologic therapy in the management of extraintestinal manifestations of inflammatory bowel disease. Inflamm Bowel Dis 13: 1424-1429.
-
Krishnan S, Banquet A, Newman L, Katta U, Patil A, et al. (2006) Lung lesions in children with Crohn's disease presenting as nonresolving pneumonias and response to infliximab therapy. Pediatrics 117: 1440-1443.
-
Silbermintz A, Krishnan S, Banquet A, Markowitz J (2006) Granulomatous pneumonitis, sclerosing cholangitis, and pancreatitis in a child with Crohn disease: response to infliximab. J Pediatr Gastroenterol Nutr 42: 324-326.
-
Ruemmele FM, Lachaux A, Cezard JP, Morali A, Maurage C, et al. (2009) Efficacy of infliximab in pediatric Crohn's disease: a randomized multicenter open-label trial comparing scheduled to on demand maintenance therapy. Inflamm Bowel Dis 15: 388-394.
-
de Ridder L, Escher JC, Bouquet J, Schweizer JJ, Rings EH, et al. (2004) Infliximab therapy in 30 patients with refractory pediatric Crohn disease with and without fistulas in The Netherlands. J Pediatr Gastroenterol Nutr 39: 46-52.
-
Crandall W, Hyams J, Kugathasan S, Griffiths A, Zrubek J, et al. (2009) Infliximab therapy in children with concurrent perianal Crohn disease: observations from REACH. J Pediatr Gastroenterol Nutr 49: 183-190.
-
Teitelbaum JE, Saeed S, Triantafyllopoulou M, Daum F (2007) Infliximab in pediatric Crohn disease patients with enterovesicular fistulas. J Pediatr Gastroenterol Nutr 44: 279-282.
-
Afzal NA, Shenoy MU, Haque S, Wilcox D, Shah N (2010) Recognition and treatment of genitourinary complications in paediatric Crohn's disease using Infliximab. Acta Paediatr 99: 1042-1046.
-
Murdaca G, Spanò F, Contatore M, Guastalla A, Penza E, et al. (2015) Infection risk associated with anti-TNF-a agents: a review. Expert Opin Drug Saf 14: 571-582.
-
Borekci S, Atahan E, Demir Yilmaz D, Mazican N, Duman B, et al. (2015) Factors Affecting the Tuberculosis Risk in Patients Receiving Anti-Tumor Necrosis Factor-a Treatment. Respiration.
-
Atikan BY, Cavusoglu C, Dortkardesler M, Sozeri B (2014) Assessment of tuberculosis infection during treatment with biologic agents in a BCG-vaccinated pediatric population. Clin Rheumatol 67: 2842-2845.
-
Dulai PS, Thompson KD, Blunt HB, Dubinsky MC, Siegel CA (2014) Risks of serious infection or lymphoma with anti-tumor necrosis factor therapy for pediatric inflammatory bowel disease: a systematic review. Clin Gastroenterol Hepatol 12: 1443-1451.





