Journal of Rheumatic Diseases and Treatment
Impact of the Addition of Ginger Extract and Copper Sulphate to Glucosamine Sulphate on Il-1β-Stimulated Chondrocytes
Francis Rousset1, Laurent Grange2, Minh Vu Chuong Nguyen1, Clara Pinosa-Zezza1, Philippe Gaudin2, Thierry Conrozier3, Françoise Morel1 and Bernard Lardy1,4*
1Université Grenoble Alpes, GREPI EA 7408, Grenoble, France
2Rheumatology Department, Universitary Hospital A. Michallon, Grenoble, France
3Service de Rhumatologie, Hopital Nord Franche Comté, Belfort, France
4Biochemical Enzymology and protein laboratory, University Hospital A. Michallon, Grenoble, France
*Corresponding author: Bernard Lardy, Biochemical Enzymology and protein laboratory, University hospital A. Michallon, Grenoble, France, E-mail: blardy@chu-grenoble.fr
J Rheum Dis Treat, JRDT-2-038, (Volume 2, Issue 3), Research Article; ISSN: 2469-5726
Received: May 27, 2016 | Accepted: August 31, 2016 | Published: September 02, 2016
Citation: Rousset F, Grange L, Nguyen MVU, Pinosa-Zezza C, Gaudin P, et al. (2016) Impact of the Addition of Ginger Extract and Copper Sulphate to Glucosamine Sulphate on Il-1β-Stimulated Chondrocytes. J Rheum Dis Treat 2:038. 10.23937/2469-5726/1510038
Copyright: © 2016 Rousset F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Oxidative stress is an important component of the pathogenesis of osteoarthritis. NADPH oxidase Nox4 is a reactive oxygen species (ROS) producing enzyme highly expressed in chondrocytes and may be a key contributor of this oxidative component by regulating Matrix metalloproteases (MMPs) expression. In order to optimize the Glucosamine sulphate (GS) efficacy against osteoarthritis, a new therapeutic association (Glucosamine Copper Ginger) has been developed (GSCG). In this study, we compared the effects of GSCG and GS on Nox4 activity, MMPs expression in IL-1β-stimulated C-20/A4 chondrocytes.
Methods: C-20/A4 chondrocytes were pre-treated with GSCG or GS. After IL-1β stimulation, we assessed MMPs expression by Western blot or apoptosis using the activation of caspase 3 as marker. Impact of compounds on Nox4 activity was measured by chemiluminescence on the inducible Nox4 expressing cells line HEK293 T-REx™.
Results: ROS production and related MMP1 expression were significantly decreased (respectively by 30% and 40%) after pre-incubation with GSCG. On the other hand, no significant effect was observed after GS treatment. ADAMTS5 expression was markedly decreased by GS and GSCG. Moreover, results reported a significant decrease in the IL-1β-induced caspase 3 activation in presence of GS and GSCG.
Conclusions: This study provided experimental evidence that glucosamine sulfate decreases ADAMTS5 expression and apoptosis. In addition, ginger root and copper sulphate decreased pro-MMP1 expression by regulating NOX4 activity. Our data suggest the implication of Heme Oxygenase-1 in the molecular mechanisms. These findings emphasize in vitro the potential beneficial effects of therapeutic association of Glucosamine, copper sulphate and Ginger extract.
Keywords
Chondrocyte, NADPH oxidase 4, Interleukin-1, Metalloproteases, Glucosamine sulphate
Abbreviations
Cu: Copper Sulphate; GSCG: Glucosamine-Copper-Ginger; GR: Ginger Root Extract; GS: Glucosamine Sulphate; HO-1: Heme Oxygenase-1; IL: Interleukin; MMP: Matrix Metalloproteases; Nox: NADPH Oxidase; OA: Osteoarthritis; ROS: Reactive Oxygen Species
Introduction
Osteoarthritis (OA) is a common disorder characterized by loss of articular cartilage as a result of degenerative changes in the joint. Interleukin-1β (IL-1β) is the main cytokine involved in OA progression leading to an increase in MMP release by chondrocytes and a decrease in synthesis of extracellular matrix component such as proteoglycans or type II collagen [1]. In response to IL-1β, chondrocytes actively produce reactive oxygen species (ROS) which could play a central role in this matrix homeostasis breakdown [2-4].
The NADPH oxidase enzymes constitute a large enzyme family which function is solely dedicated to the production of cellular ROS. Nox4 is one of the 7 ROS generating Nox members expressed in human [5]. Our recent work showed that Nox4 represents the major source of ROS production in human primary chondrocytes [4,6] and in the C-20/A4 human chondrocyte cell line [3]. In this cell line, Nox4-derived ROS regulate collagenase 1 (MMP-1) expression and chondrocyte death consecutive to IL-1β stimulation [3,4]. In this context, an induction of heme oxygenase-1 (HO-1) expression was shown to prevent MMP1 expression and chondrocyte DNA fragmentation by down regulating Nox4 activity [4]. Therefore, the balance between Nox4 and HO-1 activities in chondrocyte may contribute to cartilage homeostasis.
OA represents a major socio-economic burden in developed countries and available treatments are largely palliative or display weak benefits on the progression of the pathology [7,8]. Glucosamine sulphate (GS), a basic structural element that composes cartilage proteoglycans is a dietary supplement approved as a symptomatic slow-acting drug for osteoarthritis (SYSADOA). This compound showed significant anti-inflammatory and anti-catabolic effects on chondrocyte, in vitro [9]. The anabolic effects of GS were primarily thought to be attributable to its capacity to provide building blocks for the synthesis of glycosaminoglycans (GAGs) by chondrocytes but various additional GS properties were also demonstrated. In vitro, GS has been shown to inhibit expression and/or activity of proteolytic enzymes such as metalloproteinase [10] or aggrecanase [11] and to reverse the effects of Il-1b on the Nuclear Factor Kappa B (NFkB) activation, relaying pro-inflammatory and pro-catabolic processes in articular chondrocytes. However, impact of GS on structural features of OA is still difficult to evidence. This lack of clear proofs is illustrated by discrepancies in GS recommendations by the main OA management institutions. Indeed, Osteoarthritis Research Society International (OARSI) and European League Against Rheumatisms (EULAR) have recommended the use of GS for hip and knee OA management while American College of Rheumatology (ACR) and National Institute for Health and Clinical Excellence (NICE) estimate this recommendation too premature [9].
Beside GS, some other natural compounds less well-known may also display an anti-arthritic activity. For example, ginger root extract (GR) was shown to decrease pro-inflammatory mediators like Prostaglandin E2 and Nitric oxide (NO) production in arthritic and normal human cartilage slides [12]. Also, copper sulphate promotes collagen synthesis by human articular chondrocytes in vitro [13]. In order to optimize Glucosamine sulphate (GS) efficacy, a new therapeutic association (GSCG) of GS (78.9%), copper sulphate (0.105%) and ginger root extract (5.26%) was developed [Cuivramine®, Laboratoire Labhra, Lyon, France] and tested on C-20/A4 chondrocytes and HEK293 T-REx™, a commonly used cell line to study basic feature of Nox4 activity [14,15]. Therefore, the aim of the study was to compare in vitro the potential additional effects of the association of Glucosamine-Copper-Ginger versus Glucosamine alone on the level of ROS production, the MMP expression and chondrocyte apoptosis upon IL-1β stimulation.
Methods
Material
Human C-20/A4 chondrocyte cell line immortalized by SV40 is a generous gift from Dr M.B. Goldring (Harvard Institute of Medicine, Boston, MA, USA). HEK293 Nox4 T-REx™ cells were kindly provided by PATIM laboratory, (Pr KH. Krause, Geneva University, Switzerland). Dulbecco's modified Eagle's medium (DMEM), fetal bovine serum (FBS) and geneticin (G418) were purchased from life technologies (Saint Aubin, France); blasticidin was from Funakoshi Co (Tokyo, Japan); ECL Western Blotting detection reagents and second antibodies-HRP were purchased from GE healthcare (Buckingamshire, England); complete mini EDTA-free protease inhibitor EASYpack, Na4P2O7, Na3VO4, Phenylmethylsulfonile fluoride (PMSF), luminol, Horseradish Peroxidase (HRPO), Triton X-100, protoporphyrin-IX cobalt chloride (CoPP-IX) were purchased from SIGMA (Saint Quentin Fallavier, France); leupeptin, pepstatin, trypsin inhibitor, Tosyl-lysine-chloromethyl-ketone (TLCK) and human interleukin-1β were purchased from Roche (Meylan, France); Diisopropylfluoro-phosphate (DFP) was purchased from Acros Organics (Halluin, France); goat antibodies directed against HO-1 and actin were purchased from Santa Cruz Biotechnologies (Heidelberg, Germany); Pro-MMP1 monoclonal antibody was obtained from R&D Systems (Lille, France); rabbit polyclonal antibodies directed against the metallopeptidase with trombospondin motifs 5 (ADAMTS5) (RP1-ADAMTS5) and active caspase 3 were respectively from Biovalley (Illkirch, France) and Abcam (Paris, France).
Cell culture
All cells were cultured in DMEM containing 4.5 g/L glucose and 0.11 g/L sodium pyruvate, supplemented with 10% (v/v) fetal bovine serum, 100 units/ml penicillin, 100 mg/ml streptomycin and 2 mM glutamine at 37°C in atmosphere containing 5% CO2 [16]. Selecting antibiotics, blasticidin (5 μg/ml) and geneticine (400 μg/ml) were used for HEK293 Nox4 T-REx™ cells. Expression of Nox4 by HEK293 Nox4 T-REx™ cells were induced by the addition of 1 μg/ml tetracycline in the culture media [14]. All experiments were performed within cell passages 3 to 10 at 60-90% confluence.
Experimental conditions
For experiments, C-20/A4 chondrocytes and HEK293 T-REx™ were pre-incubated 96 h with 100 or 500 μg/ml of glucosamine sulphate (GS) or of the therapeutic association (GSCG) composed by glucosamine sulphate 93.75% (w/w) Ginger roots extract 5.26% (w/w) copper sulphate 0.1% (w/w). Cells were also pre-incubated with 5.26 μg/ml, 26.3 μg/ml or 52.6 μg/ml Ginger roots extract (GR) or 0.105 μg/ml, 0.52 μg/ml or 1.04 μg/ml copper sulphate (Cu). For GR and Cu, the concentrations used correspond respectively to the amount contained in 100 μg/ml, 500 μg/ml or 1,000 μg/ml of therapeutic association. After the 96 h of pre-incubation, tetracycline-induced HEK293 Nox4 T-REx™ cells were used for NADPH oxidase activity assessment by chemiluminescence. For the C-20/A4 chondrocytes experiments, after the 96 h of pre-incubation, the cells were incubated in serum free DMEM in presence of the experimental doses of GS, GSCG, GR or Cu and stimulated or not with 2 ng/ml IL-1β during 24 h for MMP1 and ADAMTS5 assessment or 96 h for HO-1 expression and active caspase 3 assay.
Measurement of NADPH oxidase activity in intact cells by luminescence assay
ROS production was measured as described by Grange, et al. [3]. Briefly, cells were detached with trypsin, washed twice with PBS and collected by centrifugation (400 g, 8 min, room temperature). The viability of the suspended cells was over 90%, as determined by the trypan blue exclusion method. In a 96-well plate, 5 × 105 living cells suspended in 50 μl were added per well. Before the start of the assay, 200 μl of a PBS solution containing 20 μM luminol, and 10 units/ml of horseradish peroxidase was added in each well. Relative luminescence unit (RLU) counts were recorded every 30 s for a total of 45 min at 37°C using a Luminoscan® luminometer (Labsystems, Helsinki, Finland).
Cells extract preparation for intracellular protein immunodetection
Cells were treated with 3 mM DFP and lysed in Triton X-100 lysis buffer containing 20 mM Tris-HCl pH7.4, 1% (v/v) Triton X-100, 150 mM NaCl, 1 mM EDTA, 2 μg/ml leupeptin, 2 μg/ml pepstatin, 10 μg/ml trypsin inhibitor, 44 μg/ml PMSF, 10 μM TLCK and complete mini EDTA-free protease inhibitor. After 10 min incubation on ice, the mixture was centrifuged at 1,000 g for 10 min at 4°C. The supernatant was then used for SDS-PAGE and Western blot detection of active caspase-3, HO-1 or Actin.
Secreted protein detection
After incubation under the conditions described above in experimental conditions section, the culture medium was removed, 10 times concentrated with centricon 3 kDa (Merck Millipore, Billerica, USA) then secretion of pro-MMP1 or ADAMTS5 by chondrocytes was assessed by Western blot.
SDS/PAGE and western blotting
Triton X-100 cell extract or centricon 10X concentrated cell culture supernatant was denatured at 60°C for 30 min and loaded on a 10% or 12.5% (p/v) SDS-PAGE for migration and then electro-transfer to nitrocellulose. Immunodetection was performed using primary mAb against pro-MMP1 (1:500), pro-MMP13 (1:1,000) or polyclonal rabbit antibody against the metallopeptidase ADAMTS5 (1:1,000) or active caspase 3 (1:1,000) or goat antibody directed against HO-1 (dilution 1:500) or actin (1:500). Secondary antibody was conjugated to peroxidase (1:5,000). Peroxidase activity was detected using ECL reagents (GE Healthcare). After revelation, densitometry of the bands corresponding to proteins of interest was assessed with the Scion Image software.
Cell survival assay
After the 96 h pre-incubation with therapeutic compounds, C-20/A4 chondrocytes were incubated for 7 days in DMEM without foetal bovine serum, with or without 2 ng/ml IL-1β and GS, GSCG, GR and Cu at the experimental amount. Medium and floating cells were then removed and replaced with complete DMEM (supplemented with 10% foetal bovine serum) allowing alive cell to growth for 7 additional days. Finally chondrocytes are detached with trypsin and living cells were counted by trypan blue assay.
Statistical data
Data are presented as means +/- S.D., significance levels were assessed using one-way analysis of variance (ANOVA) followed by appropriate post hoc analysis. A p-value of 0.05 or less between groups was considered to indicate a statistically significant difference.
Results
Differential impact of the therapeutic association and glucosamine sulphate on NADPH oxidase activity in HEK293 T-REx™ cells
The antioxidant effects glucosamine sulphate (GS) and a therapeutic association (GSCG) were investigated on HEK293 T-REx™ cells, a reproducible and reliable cell line model to study Nox4. In this cell line Nox4 expression can be induced upon the addition of tetracycline leading to a 20 fold increase in the ROS production (Figure 1) [14]. When 100 μg/ml or of GSCG or 500 μg/ml of GS were extemporaneously added to the cells right before the ROS production measurement, no significant impact on Nox4 activity was noticeable showing that GS and GSCG do not have a direct antioxidant properties (Figure 1A). However, Nox4 activity decreased significantly about 30% after 96 h pre-incubation with 500 μg/ml GSCG but not with GS (Figure 1B). To investigate which compound composing the GSCG was responsible for this effect, T-REx™ cells were pre-incubated for 96 h with 5.26, 26.3 or 52.6 μg/ml of ginger root (GR); or with 0.105, 0.525 or 1.05 μg/ml of copper sulphate (Cu) (the equivalent amount contained in 100, 500 or 1,000 μg/ml GSCG) (Figure 1C). Results showed a dose dependant decrease of Nox4 activity in the presence of GR and Cu for the slighter doses. Interestingly, this inhibition was not significant after 48 h, suggesting that the time necessary for the GR- and Cu- induced inhibition of Nox4 activity required more than 48h to be effective (Supplementary Figure 1). Together, our data indicate that the inhibition of Nox4 activity by GSCG is triggered by GR and Cu rather than by GS.
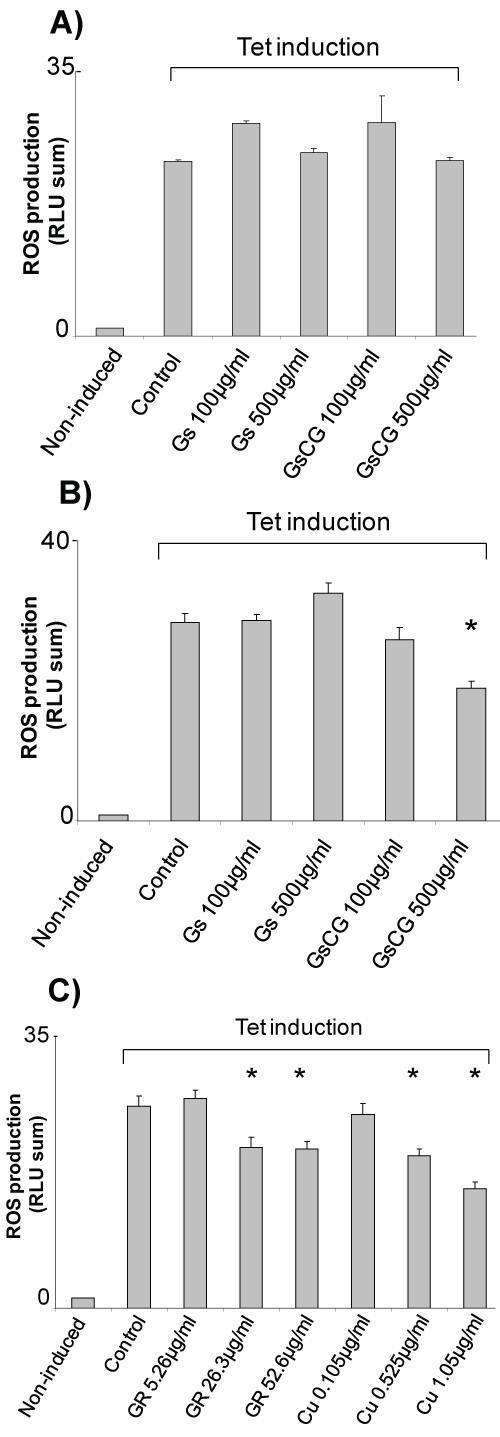
.
Figure 1: Impact of GSCG and GS on NADPH oxidase activity of Nox4 in HEK293 T-REx™ cells.
Nox4 expression in HEK293 Nox4 T-REx™ cells were induced or not induced with 1 μg/ml tetracycline for at least 24 h in complete DMEM medium. Nox4 activity was assessed by chemiluminescence from 5.105 tetracycline-induced HEK293 Nox4 T-REx™ alive cells. A) Effects of extemporaneous addition or; B) 96 h pre-incubation with 100 or 500 μg/ml GS or GSCG on NADPH oxidase 4 activity of HEK293 Nox4 T-REx™ cells; C) Effects of 96 h pre-incubation with 0.105, 0.525 or 1.05 μg/ml Cu or 5.26, 26.3 or 52.6 μg/ml GR** on NADPH oxidase 4 activity of HEK293 Nox4 T-REx™ cells. **Concentrations of GR and Cu are equivalent to the amount contained in 100, 500 or 1000 μg/ml GSCG. In all experiments, results are expressed as the sum of all «Relative Light Units» (RLU) measurements from a 20 min kinetic. Values represent the mean +/- S.E. of three determinations obtained the same day on three independent experiments. *p < 0.05 versus control tetracycline-induced cells alone.
View Figure 1
Impact of the therapeutic association and glucosamine sulphate on IL-1β stimulated pro-MMP1 and ADAMTS5 secretion
We have previously reported that while the pro-MMP1 secretion is regulated by Nox4 activity, the regulation of ADAMTS5 secretion is not redox dependent in C-20/A4 chondrocytes [3,4]. To assess the impact of GSCG and GS on the secretion pattern of these proteases, C-20/A4 chondrocytes were pre-incubated with 100 or 500 μg/ml of GSCG or of GS respectively for 96 h in complete DMEM medium (see section experimental conditions). Medium was then replaced by serum free DMEM and cells were stimulated (24 h) or not with 2 ng/ml IL-1β in presence or not of GSCG or of GS. Results showed an important increase in both pro-MMP1 and ADAMTS5 secretion in the cell culture medium after chondrocyte stimulation by IL-1β (Figure 2A and Figure 2B). Interestingly, under these conditions, GSCG at 500 μg/ml was able to reduce significantly IL-1β-induced pro-MMP1 secretion (50%) while GS had not any significant effects (Figure 2A). On the other hand, both GS and GSCG neutralized IL-1β induced ADAMTS5 secretion (Figure 2B). Based on these data, we can assume that impact of GSCG on MMP1 depends on Cu and/or GR while effects on ADAMTS5 are dependent on GS.
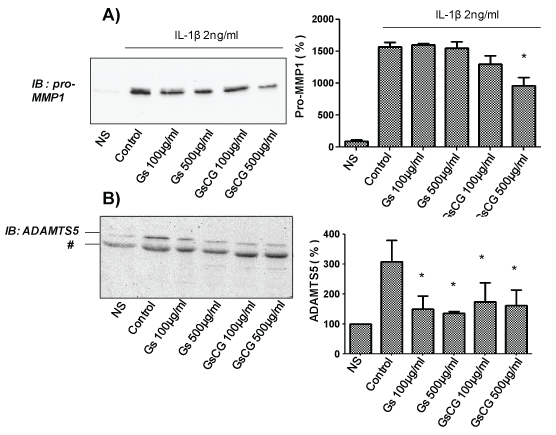
.
Figure 2: Impact of GSCG and GS on pro-MMP1 and ADAMTS5 secretion by C-20/A4 chondrocytes.
Chondrocytes were pre-incubated with 100 or 500 μg/ml GS or GSCG for 96 h in complete DMEM medium and stimulated for 24 supplementary hours with 2 ng/ml IL-1β in serum free DMEM. Supernatant was then collected, concentrated 10 times by centricon and 20 μg of proteins (determined by Bradford method) were loaded for A- pro-MMP1 or B- ADAMTS5 immunodetection by Western blot. Immunoblot (IB) are representative of three independent experiments. Data obtained were quantified by densitometry are indicated as mean +/- SD of three independent experiments. Results are expressed as percentage of non-stimulated condition (NS). *p < 0.05 versus non stimulated cells. # unspecific band.
View Figure 2
In order to evaluate whether GR and/or Cu mediates the effects observed for GSCG, similar experiments were performed with a 96 h pre-incubation with only 5.26 mg/ml or 26.3 mg/ml ginger root (GR); or 0.105 mg/ml or 0.52 mg/ml copper sulphate (Cu) in complete medium (the equivalent amount contained in 100, 500 or 1,000 μg/ml GSCG). We observed that both GR and Cu were able to decrease IL-1β-stimulated pro-MMP1 secretion (Figure 3A) in a dose dependent manner reaching ~40% decreases with the highest dose of GR and Cu studied. These results confirm that the inhibition of GSCG on pro-MMP1 secretion comes from GR and Cu compounds. In contrast, we do not observed significant decrease in ADAMTS5 secretion with both compounds suggesting that GS alone can mediate the inhibition of GSCG on IL-1β-induced ADAMTS5 secretion (Figure 3B).
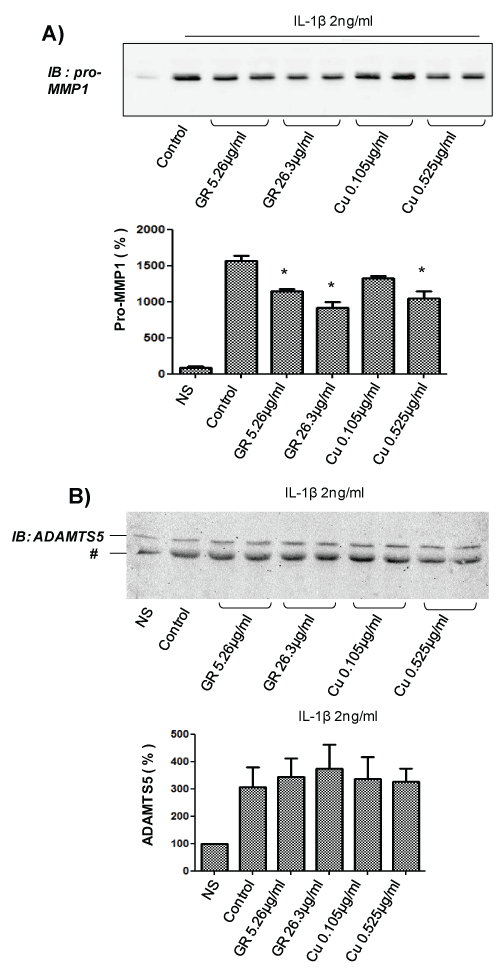
.
Figure 3: Impact of Cu and GR on pro-MMP1 and ADAMTS5 secretion by C-20/A4 chondrocytes.
A- Chondrocytes were pre-incubated with 0.105 or 0.525 μg/ml Cu or 5.26, or 26.3 μg/ml GR** for 96 h in complete DMEM medium and stimulated for 24 supplementary hours with 2 ng/ml IL-1β in serum free DMEM. Supernatant was then collected, concentrated 10 times by centricon and 20 μg of proteins (determined by Bradford method) were loaded for A- pro-MMP1 or B- ADAMTS5 immunodetection by Western blot. ** Concentrations of GR and Cu are equivalent to the amount contained in 100 or 500 μg/ml GSCG. Immunoblot (IB) are representative of three independent experiments. Data obtained were quantified by densitometry are indicated as mean +/- SD of three independent experiments. Results are expressed as percentage of non-stimulated condition (NS). *p < 0.05 versus non stimulated cells. # unspecific band.
View Figure 3
Effect of the therapeutic association and its components on C-20/A4 chondrocytes viability and apoptosis
IL-1β-induced chondrocyte death is a leading cause of osteoarthritis [17]. Nox4 plays a key role in this phenomenon in the C-20/A4 cell line [4]. We therefore next assessed the impact of GSCG and its components on chondrocytes survival and apoptosis in presence of IL-1β (Figure 4). Our results showed a dramatic decrease in chondrocyte survival in presence of IL-1β (Figure 4A). Interestingly, survival was improved in a dose dependant fashion with GSCG and GS treatments but not by GR nor by Cu. To confirm these data, we evaluated the caspase 3 activation by Western blot (Figure 4B). IL-1β-induced caspase 3 activation was significantly prevented by GSCG and GS at 500 μg/ml. In contrast effects of GR and Cu were not statistically significant compared to IL-1β-stimulated cells. Together, these data show that GSCG is able to prevent IL-1β induced chondrocyte death via GS. GR and Cu do not play a similar role despite the fact that they can reduce Nox4-derived ROS generation.
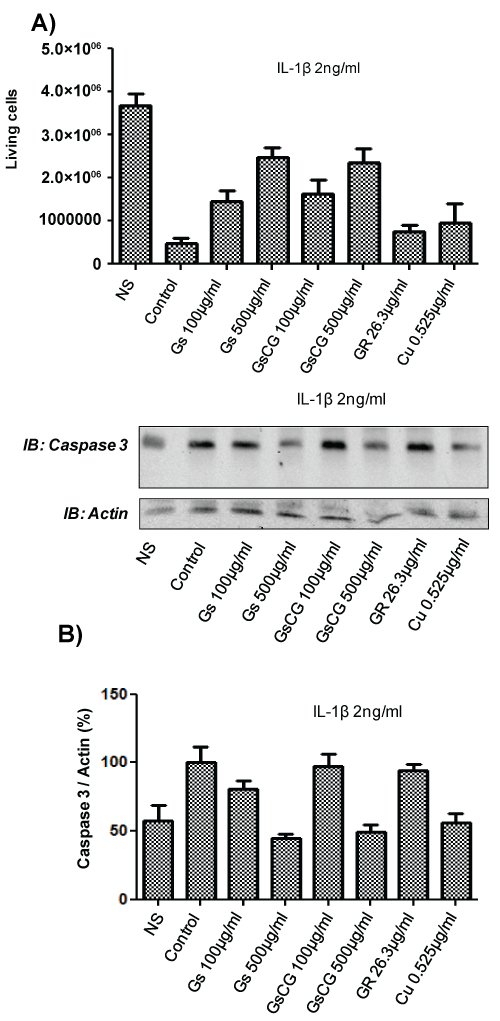
.
Figure 4: Impact of GSCG and its components on chondrocyte viability and caspase 3 activation.
Chondrocytes were pre-incubated with 100 or 500 μg/ml GS or the equivalent amount of GR and Cu contained in GSCG for 96 h in complete DMEM medium. A) Cells were then treated for 7 days with 2 ng/ml IL-1β in presence of GS, GR, Cu or GSCG in serum free DMEM. Medium was then replaced by fresh complete DMEM media allowing cells to growth. After 7 days, cells were detached and living chondrocytes were counted in Malassez cell with 10% (V/V) Trypan blue. Histogram represents the number of living chondrocytes for each condition. Experiment is representative of two independent experiments; B) For active caspase 3 assay, after the 96 h pre-incubation, cells were treated for 4 days with 2 ng/ml IL-1β in presence of GS, GR, Cu or GSCG in serum free DMEM. Caspase 3 and actin expression were then assessed by Western blot on 75 μg of protein (determined by Bradford method) from a Triton X-100 1% cell extract. Immunoblot (IB) are representative of 3 independent experiments. Data obtained were quantified by densitometry are indicated as mean +/- SD of three independent experiments. Ratio of Caspase 3/actin are expressed as percentage of IL-1β-treated chondrocytes (Control). *p < 0.05 versus IL-1β-treated chondrocytes.
View Figure 4
Impact of the therapeutic association and glucosamine sulphate on HO-1 induction
The right balance between NOX4 and Heme Oxygenase-1 activities may contribute to cartilage extracellular matrix homeostasis [4]. Since HO-1 has been shown to be upregulated in chondrocytes upon GS treatment [18], we assessed the impact of GSCG and its components on HO-1 expression in the C-20/A4 cell line. Chondrocytes were cultured 96 h in presence of the compounds and submitted to a Triton X-100 extraction protocol for Western Blotting. As positive control, HO-1 expression was induced by cobalt protoporphyrin-IX (CoPP-IX) in the C-20/A4 cell line (Figure 5 lower panel). We observed an increase of HO-1 expression in C-20/A4 chondrocytes in a dose dependent manner with a GS or with a GSCG treatment (x3.5 with 500 μg/ml). The effect is approximately two times stronger with GSCG suggesting that this increase may be the sum of the effects of GS and Cu given the fact that each one of these compounds led approximately to a 2 fold increase of HO-1 expression. In contrast, there was no upregulation of HO-1 protein when the C-20/A4 cells were treated with GR.
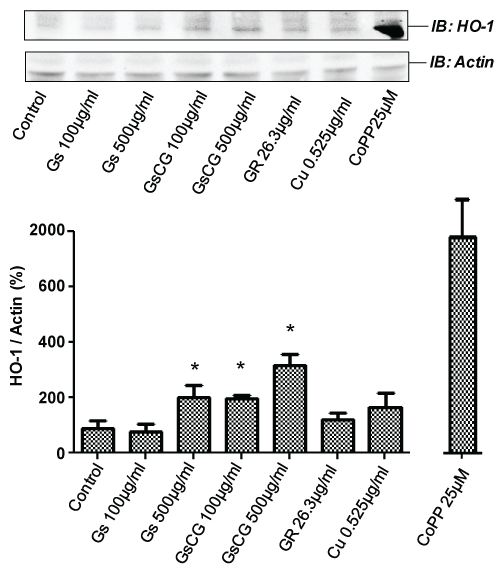
.
Figure 5: Impact of GSCG and its components depends on HO-1 induction.
C-20/A4 chondrocytes were pre-incubated with 100 or 500 μg/ml GS or GSCG or the equivalent amount of GR and Cu contained in 500 μg/ml GSCG for 96 h in complete DMEM medium. The positive control for HO-1 induction was performed by a 24 h incubation with 25 μg/ml CoPP-IX. Cells were then lyzed in a buffer containing 1% Triton X-100 HO-1 and actin were detected by Western blot on 50 μg protein (determined by Bradford method). Immunoblot (IB) are representative of 3 independent experiments. Data obtained were quantified by densitometry are indicated as mean +/- SD of three independent experiments. Ratio of HO-1/actin are expressed as percentage of non-stimulated cells (Control). *p < 0.05 versus non treated chondrocytes.
View Figure 5
Discussion and Conclusion
Various anti-inflammatory properties have been demonstrated for glucosamine in the three main tissues involved in OA: cartilage, synovial membrane and subchondral bone [9,19]. Particularly glucosamine was able to reverse the pro-catabolic and pro-inflammatory effects of IL-1β in articular chondrocytes [10]. IL-1β activates the production of reactive oxygen species (ROS) and secretion of MMPs as well as chondrocyte apoptosis. Those events lead to matrix breakdown and are key features of osteoarthritis (OA). In human C-20/A4 chondrocytes the NADPH oxidase Nox4 is the main source of ROS production upon IL-1β stimulation [3]. Since heme molecules are essential for the NADPH oxidase maturation and activity, induction of HO-1 expression decreased dramatically Nox4 activity in C-20/A4 and HEK293 T-REx™ Nox4 cell lines. In this context, HO-1 led to a significant decrease of the expression of the MMP1, DNA fragmentation and cell death [4].
This study compared properties of glucosamine to that of copper sulphate (Cu) or of Ginger roots (GR) extract and showed that these compounds induced different effects on protein expression or activation when tested alone or in combination (GSCG). A summarized table 1, the increased expression of ADAMTS5 and MMP1 observed upon IL-1β activation was reversed in the presence of GS as well as of GSCG. Interestingly, copper sulphate and Ginger roots extract have a significant effect on MMP1 expression which explain a superior impact of GSCG compared to GS. In contrast, copper sulphate and Ginger roots extract have no effect on the expression of ADAMTS5; this implies that the negative impact of GSCG on ADAMTS5 expression after IL-1β activation results only on GS properties.
![]()
Table 1: Summary of the effects of GSCG and its components on the IL-1β stimulated C-20/A4 chondrocyte cell line.
View Table 1
While ADAMTS5 expression is not linked to an oxidative stress [20], one possible mechanism driving MMP1 expression could be dependent on redox sensitive AP-1 elements present in its promoter [21]. Thus, H2O2 dependent modification of AP-1 DNA binding activity may have profound effects on the extracellular matrix of cartilage by up-regulating metalloproteinases and reducing synthesis of the tissue-specific components [21,22]. In this context, restoring redox homeostasis in joint might be of crucial interest to prevent cartilage degradation in articular diseases.
While neither GSCG nor its components displayed direct antioxidant effects in our hands, copper sulphate and Ginger roots extract required at least 48 hours' time delay to significantly down regulate NOX4 activity. Similar observation has been reported in the literature where phenolic extract from GR inhibited lipopolysaccharide (LPS)-stimulated nitric oxide (NO), prostaglandin E2 (PGE2) and ROS production in murine macrophage cells RAW 264.7 [23]. These anti-inflammatory effects of GR were associated with its strong inhibition of NF-κB nuclear translocation in LPS-stimulated RAW 264.7 cells. The mechanism whereby Cu decreases the quantity of ROS is not well understood yet. Cu is known to provoke an oxidative stress which could possibly lead to lethal consequences. Metal accumulation causes an increase in ROS production, such as H2O2, O2 and OH-, that leads to oxidative stress [24]. Cu related oxidative stress is followed by a phase of stimulation of antioxidant pathways, including SOD, CAT and thioredoxine [25]. Our data indeed show an increase in the ROS production by the HEK293 T-REx™ cell line after 48 h treatment with Cu alone (Supplementary Figure 1). However, after 96 h incubation with Cu, we observed a significant antioxidant effect. Thus we assume that Cu may stimulate antioxidant pathways of chondrocytes, including induction of HO-1.
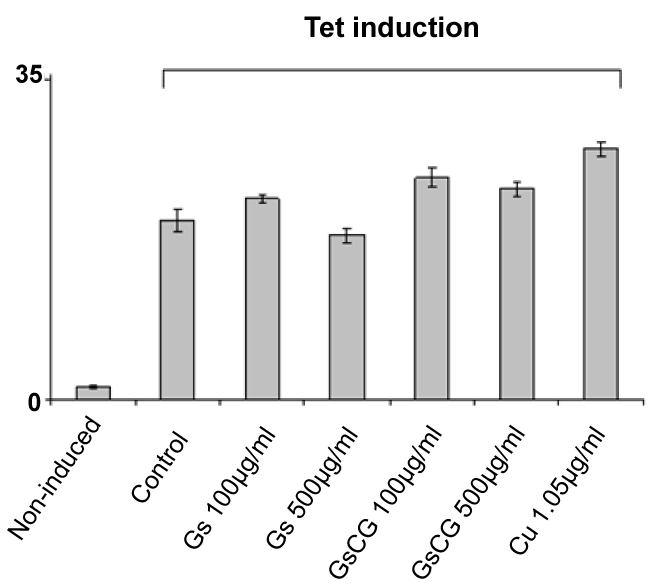
.
Supplementary Figure 1: Impact of GSCG, GS and Cu on NADPH oxidase activity of Nox4 in HEK293 T-REx™ cells.
Nox4 expression in HEK293 Nox4 T-REx™ cells were induced or not induced with 1 μg/ml tetracycline for 24 h. Nox4 activity was assessed by chemiluminescence from 5.105 HEK293 Nox4 T-REx™ alive cells after 48 h pre-incubation with 100 or 500 μg/ml GS or GSCG and Cu 1.05 μg/ml. In all experiments, results are expressed as the sum of all « Relative Light Units » (RLU) measurements from a 20 min kinetic. Values represent the mean +/- S.E. of three determinations obtained the same day on three independent experiments. *p < 0.05 versus control tetracycline-induced cells alone.
View Supplementary Figure 1
The enzymatic activity of HO-1 results in decreased oxidative stress, attenuated inflammatory response, and a lower rate of apoptosis [26,27]. In addition, HO-1 exerts anti-inflammatory effects by inhibition of tumour necrosis factor (TNF) and interleukin-1 (IL-1), or by upregulation of interleukin-10 (IL-10) [28]. Nuclear related Factor 2 (Nrf2) binds to the antioxidant response element in the promoter region of numerous genes encoding antioxidant and phase 2 enzymes, including HO-1, NADPH: quinone oxidoreductase 1, glutathione reductase and glutathione peroxidase. Phase 2 enzymes play a major role in the detoxification of ROS during ischemia reperfusion events [29,30]. In primary human chondrocyte cells it was found that the incubation with IL-1 causes an inhibition of HO-1 gene expression [18,31]. Moreover, treatment with 10 mmol/l of GS restores the expression of HO-1 gene with a maximal effect after 48 h. These results are in agreement with our data showing an increase of the expression of HO-1 upon GS and slightly upon Cu incubations. The additive effect of GS and Cu may explain the greater efficacy of the GSCG compared to GS alone on HO-1 expression. HO-1 was also shown to protect chondrocyte cells form apoptosis induced by IL-1 [4,32]. Therefore, GS and Cu may reverse the deleterious effect of IL-1 on caspase 3 activation via HO-1 activation in the chondrocyte cells lines C-20/A4.
In summary, our data confirm in vitro the potency of glucosamine to prevent pro-catabolic action of IL-1. In addition Copper sulphate and Ginger root extract, could contribute to restore the redox balance by inhibiting Nox4 activity via an upregulation of HO-1 expression in the human chondrocyte cell line C20/A4. In conclusion, GSCG displays supplementary beneficial effect compared to GS and addition of Copper sulphate and Ginger root extract could improve GS therapeutic efficiency in osteoarthritic patients.
Acknowledgments
The authors thank Pr. Mary Goldring for the generous gift of the C-20/A4 cell line. The authors thank also Pr Xavier Ronot, Pr Bertrand Toussaint, Pr. François Rannou, Dr. Corinne Dupuy, Dr Marie Hélène Paclet, Pr Philippe Gaudin and Pr Françoise Morel for helpful discussion.
This work was funded by LABRHA SAS and supported by grants from the Ministère de l'Enseignement Supérieur de la Recherche et Technologie, Paris, the UFR de Médecine, Université Joseph Fourier, Grenoble the roupement des Entreprises Françaises dans la lutte contre le Cancer, délégation de Grenoble, the Société Française de Rhumatologie and the Délégation à la Recherche Clinique et à l'Innovation, from the university hospital, Grenoble. We tanks the Etablissement français du sang (EFS) for his support. FR is supported by grants from the Foundation Arthritis Courtin. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
-
Goldring MB, Otero M, Tsuchimochi K, Ijiri K, Li Y (2008) Defining the roles of inflammatory and anabolic cytokines in cartilage metabolism. Ann Rheum Dis 67: iii75-82.
-
Henrotin Y, Kurz B, Aigner T (2005) Oxygen and reactive oxygen species in cartilage degradation: friends or foes? Osteoarthritis Cartilage 13: 643-654
-
Grange L, Nguyen MV, Lardy B, Derouazi M, Campion Y, et al. (2006) NAD(P)H oxidase activity of Nox4 in chondrocytes is both inducible and involved in collagenase expression. Antioxid Redox Signal 8: 1485-1496.
-
Rousset F, Nguyen MV, Grange L, Morel F, Lardy B (2013) Heme oxygenase-1 regulates matrix metalloproteinase MMP-1 secretion and chondrocyte cell death via Nox4 NADPH oxidase activity in chondrocytes. PLoS One 8: e66478.
-
Bedard K, Krause KH (2007) The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev 87: 245-313
-
Rousset F, Hazane-Puch F, Pinosa C, Nguyen MV, Grange L, et al. (2015) IL-1beta mediates MMP secretion and IL-1beta neosynthesis via upregulation of p22 (phox) and NOX4 activity in human articular chondrocytes. Osteoarthritis Cartilage 23: 1972-1980.
-
Hunter DJ (2011) Osteoarthritis. Best Pract Res Clin Rheumatol 25: 801-814.
-
Conrozier T (2016) Optimizing the effectiveness of viscosupplementation in non-knee osteoarthritis. Joint Bone Spine 83: 1-2.
-
Henrotin Y, Mobasheri A, Marty M (2012) Is there any scientific evidence for the use of glucosamine in the management of human osteoarthritis? Arthritis Res Ther 30: 201.
-
Gouze JN, Gouze E, Popp MP, Bush ML, Dacanay EA, et al. (2006) Exogenous glucosamine globally protects chondrocytes from the arthritogenic effects of IL-1beta. Arthritis Res Ther 8: R173.
-
McCulloch DR, Wylie JD, Longpre JM, Leduc R, Apte SS (2010) 10mM glucosamine prevents activation of proADAMTS5 (aggrecanase-2) in transfected cells by interference with post-translational modification of furin. Osteoarthritis Cartilage 18: 455-463.
-
Shen CL, Hong KJ, Kim SW (2005) Comparative effects of ginger root (Zingiber officinale Rosc.) on the production of inflammatory mediators in normal and osteoarthrotic sow chondrocytes. J Med Food 8: 149-153.
-
Héraud F, Savineau C, Harmand MF (2002) Copper modulation of extracellular matrix synthesis by human articular chondrocytes. Scand J Rheumatol 31: 279-284.
-
Serrander L, Cartier L, Bedard K, Banfi B, Lardy B, et al. (2007) NOX4 activity is determined by mRNA levels and reveals a unique pattern of ROS generation. Biochem J 406: 105-114.
-
Nguyen MV, Lardy B, Rousset F, Hazane-Puch F, Zhang L, et al. (2013) Quinone compounds regulate the level of ROS production by the NADPH oxidase Nox4. Biochem Pharmacol 85: 1644-1654.
-
Zhang L, Nguyen MV, Lardy B, Jesaitis AJ, Grichine A, et al. (2011) New insight into the Nox4 subcellular localization in HEK293 cells: first monoclonal antibodies against Nox4. Biochimie 93: 457-468.
-
Dozin B, Malpeli M, Camardella L, Cancedda R, Pietrangelo A (2002) Response of young, aged and osteoarthritic human articular chondrocytes to inflammatory cytokines: molecular and cellular aspects. Matrix Biol 21: 449-459.
-
Valvason C, Musacchio E, Pozzuoli A, Ramonda R, Aldegheri R, et al. (2008) Influence of glucosamine sulphate on oxidative stress in human osteoarthritic chondrocytes: effects on HO-1, p22(Phox) and iNOS expression. Rheumatology (Oxford) 47: 31-35.
-
Tiraloche G, Girard C, Chouinard L, Sampalis J, Moquin L, et al. (2005) Effect of oral glucosamine on cartilage degradation in a rabbit model of osteoarthritis. Arthritis Rheum 52: 1118-1128.
-
Vo NV, Hartman RA, Yurube T, Jacobs LJ, Sowa GA, et al. (2013) Expression and regulation of metalloproteinases and their inhibitors in intervertebral disc aging and degeneration. Spine J 13: 331-341.
-
Martin G, Andriamanalijaona R, Mathy-Hartert M, Henrotin Y, Pujol JP (2005) Comparative effects of IL-1beta and hydrogen peroxide (H2O2) on catabolic and anabolic gene expression in juvenile bovine chondrocytes. Osteoarthritis Cartilage 13: 915-924.
-
Mendes AF, Caramona MM, Carvalho AP, Lopes MC (2003) Hydrogen peroxide mediates interleukin-1beta-induced AP-1 activation in articular chondrocytes: implications for the regulation of iNOS expression. Cell Biol Toxicol 19: 203-214.
-
Yu YS, Hsu CL, Yen GC (2009) Anti-inflammatory effects of the roots of Alpinia pricei Hayata and its phenolic compounds. J Agric Food Chem 57: 7673-7680
-
Sampaio FG, Boijink Cde L, dos Santos LR, Oba ET, Kalinin AL, et al. (2012) Antioxidant defences and biochemical changes in the neotropical fish pacu, Piaractus mesopotamicus: responses to single and combined copper and hypercarbia exposure. Comp Biochem Physiol C Toxicol Pharmacol 156: 178-186
-
Dautremepuits C, Betoulle S, Vernet G (2002) Antioxidant response modulated by copper in healthy or parasitized carp (Cyprinus carpio L.) by Ptychobothrium sp. (Cestoda). Biochim Biophys Acta 1573: 4-8.
-
Ryter SW, Choi AM (2009) Heme oxygenase-1/carbon monoxide: from metabolism to molecular therapy. Am J Respir Cell Mol Biol 41: 251-260.
-
Basuroy S, Tcheranova D, Bhattacharya S, Leffler CW, Parfenova H (2011) Nox4 NADPH oxidase-derived reactive oxygen species, via endogenous carbon monoxide, promote survival of brain endothelial cells during TNF-a-induced apoptosis. Am J Physiol Cell Physiol 300: C256-265.
-
Otterbein LE, Bach FH, Alam J, Soares M, Tao Lu H, et al. (2000) Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nat Med 6: 422-428.
-
Leonard MO, Kieran NE, Howell K (2006) Reoxygenation-specific activation of the antioxidant transcription factor Nrf2 mediates cytoprotective gene expression in ischemia-reperfusion injury. FASEB J 20: 2624-2626.
-
Datla SR, Dusting GJ, Mori TA, Taylor CJ, Croft KD, et al. (2007) Induction of heme oxygenase-1 in vivo suppresses NADPH oxidase derived oxidative stress. Hypertension 50: 636-642.
-
Guillén MI, Megías J, Clérigues V, Gomar F, Alcaraz MJ (2008) The CO-releasing molecule CORM-2 is a novel regulator of the inflammatory process in osteoarthritic chondrocytes. Rheumatology (Oxford) 47: 1323-1328.
-
Huser CAM, Davies ME (2008) Effect of a glucosamine derivative on impact-induced chondrocyte apoptosis in vitro. A preliminary report. Osteoarthritis Cartilage 16: 125-128.





